Abstract
Study Design
Systematic Review and Meta-analysis.
Objective
The purpose of this study was to evaluate whether transcranial motor evoked potential (TcMEP) alarms can predict postoperative neurologic complications in patients undergoing cervical spine decompression surgery.
Methods
A meta-analysis of the literature was performed using PubMed, Web of Science, and Embase to retrieve published reports on intraoperative TcMEP monitoring for patients undergoing cervical spine decompression surgery. The sensitivity, specificity, and diagnostic odds ratio (DOR), of overall, reversible, and irreversible TcMEP changes for predicting postoperative neurological deficit were calculated. A subgroup analysis was performed to compare anterior vs posterior approaches.
Results
Nineteen studies consisting of 4608 patients were analyzed. The overall incidence of postoperative neurological deficits was 2.58% (119/4608). Overall TcMEP changes had a sensitivity of 56%, specificity of 94%, and DOR of 19.26 for predicting deficit. Reversible and irreversible changes had sensitivities of 16% and 49%, specificities of 95% and 98%, and DORs of 3.54 and 71.74, respectively. In anterior procedures, TcMEP changes had a DOR of 17.57, sensitivity of 49%, and specificity of 94%. In posterior procedures, TcMEP changes had a DOR of 21.01, sensitivity of 55%, and specificity of 94%.
Conclusion
TcMEP monitoring has high specificity but low sensitivity for predicting postoperative neurological deficit in cervical spine decompression surgery. Patients with new postoperative neurological deficits were 19 times more likely to have experienced intraoperative TcMEP changes than those without new deficits, with irreversible TcMEP changes indicating a much higher risk of deficit than reversible TcMEP changes.
Keywords
Introduction
Cervical decompression and fusion surgeries are commonly performed to relieve spinal cord and nerve root impingement.1-4 During these procedures, the spinal cord and nerve roots are placed at risk, which may manifest as new postoperative neurologic deficits.5-7 Studies have reported the incidence of new postoperative motor deficits at rates of .2%-.9%6,8 and radicular symptoms at rates of .2%-3.2%8,9 following spine surgery for cervical myelopathy. Furthermore, these estimates are likely conservative, given that C5 palsies are often underreported in the literature. 10 Although the overall incidence of postoperative deficits may be low, the consequences of these injuries, such as quadriplegia, may be devastating. Studies have shown that postoperative neurological deficits after cervical spine surgery significantly increase length of stay and increase risk of in-hospital morbidity and mortality.11,12
Intraoperative neurophysiological monitoring (IONM) has been widely adopted for real-time feedback of spinal cord function to alert the operative team of potentially reversible neurological deficit.13,14 Specific IONM modalities including somatosensory evoked potentials (SSEPs) and transcranial motor evoked potentials (TcMEPs) may be used to monitor the sensory and motor tracts, respectively.15-18 With regards to cervical spine surgery, a previous meta-analysis has demonstrated that SSEP alarms may indicate increased risk of postoperative neurological deficit with high specificity. 15 However, while previous clinical studies and systematic reviews have demonstrated the utility of TcMEP alarms in idiopathic scoliosis correction,19-22 very few reviews have been conducted regarding the diagnostic utility of TcMEPs in cervical spine surgery, most of which are limited by sample size. Furthermore, the Guidelines for the Best Practices in Intraoperative Neuromonitoring in Spine Deformity Surgery, as established by a Delphi Consensus Report, have recommendations regarding standardized TcMEP monitoring protocols but have limited information regarding the risk of neurologic complications associated with TcMEP changes specifically during cervical spine surgery. 23 Finally, given the various types of TcMEP changes including reversible and irreversible changes, it is important to understand the diagnostic value of each to better direct both intraoperative and postoperative interventions. Due to the low incidence of postoperative neurologic deficits in cervical spine surgery, a meta-analysis combining multiple studies allows for a larger patient cohort to assess the predictive value of TcMEP changes more accurately.
The purpose of the present study was to perform a systematic review and meta-analysis of the relevant literature to evaluate whether significant TcMEP changes can predict the risk of postoperative neurologic complications in patients undergoing cervical spine decompression surgery. As a secondary analysis, two categories of significant TcMEP changes, reversible and irreversible changes, were evaluated. A tertiary subgroup analysis was performed to assess the predictive power of intraoperative TcMEP changes for postoperative neurologic complications in anterior vs posterior cervical spine surgeries. The hypothesis was that intraoperative TcMEPs could serve as a predictive marker for post-operative neurological deficits.
Methods
Protocol and Registration
This systematic review and meta-analysis followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines
24
which outlines the identification, screening, eligibility, and inclusion process (Figure 1). The electronic database search string used in this study is included in the supplemental materials (Supplemental Figure 1). Preferred reporting items for systematic reviews and meta-analyses (PRISMA). Diagram depicting study elimination process. Footnote: This PRISMA diagram depicts the flow of information through the different phases of systematic review, mapping out records included and excluded along with reasons for exclusion.
Eligibility Criteria
A meta-analysis of the literature was performed using PubMed, Web of Science, and Embase to retrieve published reports on intraoperative TcMEP monitoring for patients undergoing cervical spine or spinal cord decompression surgery, not including aneurysm, tumor, or trauma surgeries. Reports utilizing other IONM modalities in addition to TcMEP monitoring were also included although the analyses of this study were limited to TcMEP data alone. Patient outcomes were defined as postoperative neurological deficits, both motor and sensory, as determined by the surgeon during postoperative clinical follow-up. All titles and abstracts were independently screened to identify relevant studies. The following keywords were used to identify relevant publications: surgery, spine/spinal cord, motor evoked potential/MEP/TcMEP, cervical.
Study Selection
Inclusion criteria included: (1) randomized controlled trials or prospective/retrospective cohort studies, (2) studies conducted in patients undergoing elective decompressive cervical spine decompression surgery with intraoperative TcMEP monitoring (excluding surgeries for aneurysm, deformity, trauma, or tumor) (3) studies that reported on all postoperative neurological deficits and correlated them with TcMEP changes, (4) studies conducted with sample size ≥20 patients, (5) studies with only adult patients ≥18 years of age, (6) studies published in English, (7) studies with an abstract.
Two authors (ASV and RC) independently screened all titles and abstracts against the inclusion criteria, rejecting those that did not meet them. A second round of full text screening was subsequently performed for the accepted abstracts. All accepted and rejected studies were recorded in an Excel spreadsheet with the reason for rejection indicated by the corresponding inclusion criteria (1-7). A final list of papers was assembled after reconciliation of any disagreements between the two authors by a third author (RR).
Data Extraction
For each study, the collected data included study design, study demographics, procedure type, procedure approach, procedure indication, IONM modalities used, TcMEP alarm criteria, intraoperative TcMEP changes (including classification of changes into reversible and irreversible changes), and postoperative neurological outcomes. Both motor and sensory deficits were considered in the analysis, as TcMEP alarms can indicate nerve root injury, which may manifest as sensory or motor deficit.25,26 Data regarding intraoperative TcMEP changes were collected using the criterion for significance established by each respective study. Irreversible changes were regarded as significant changes that were unable to be reversed by intraoperative interventions. This data was used to make 2 × 2 tables of true-positives (intraoperative TcMEP change and neurological deficit), false-positives (intraoperative TcMEP change and no neurological deficit), false-negatives (no intraoperative TcMEP change and neurological deficit), and true-negatives (no intraoperative TcMEP change and no neurological deficit). For the studies that also reported on the reversibility of TcMEP changes, the authors further delineated the number of patients with reversible or irreversible TcMEP changes into 2 × 2 tables.
Assessment of Methodological Quality
The authors used the QUADAS-2 tool to assess the susceptibility to bias of the included studies (Supplemental Figure 2). The methodological quality of the included studies was assessed independently by two authors and disagreement was resolved by reexamination of the literature. 27 The four domains assessed by the QUADAS 2 tool were patient selection, index test, reference standard, and flow and timing. If the answers to all signaling questions in a domain are “yes” then the “low” risk grade is given. If the answer to any signaling question is “no” then a “high” risk grade is given. The “unclear” category was only used where the reported data was insufficient to permit a judgment.
Statistical Analysis
The statistical analyses were carried out in R using the MADA package (accessed in November 2022). The 2 × 2 tables were used to calculate the sensitivity and specificity of TcMEPs for detecting postoperative neurological deficit. If a zero-cell occurred in the study, calculations were performed by adding .5 to all cells to allow for statistical inference. Analyses was conducted using a bivariate normal model for the logit-transformed pairs of sensitivities and false-positive rates before the fitting of a linear mixed model. This model preserved the bivariate nature of the data by considering any correlation between sensitivity and specificity. The mean logit sensitivity, specificity, and covariance were estimated from this model. Forest plots and a summary ROC with a 95% confidence ellipsoid were constructed.
As a secondary analysis, fixed and random effects models for log-diagnostic odds ratios, of not only overall TcMEP change, but also for each TcMEP change subgroup (reversible vs irreversible) were fitted. Unlike the bivariate normal model, this converted the bivariate nature of the data to a univariate problem. Q and I2 statistics were also calculated to describe the percentage of variation across studies that is due to heterogeneity rather than chance. Q values show presence of heterogeneity but I2 values were used as a true indicator of heterogeneity because they show the extent of heterogeneity in the sample. An I2 value of greater than .2 was interpreted as significant heterogeneity.
Finally, the positive and negative likelihood ratios were calculated for total TcMEP changes in predicting postoperative neurologic deficit. The likelihood ratios were used to create a Fagan nomogram with the pre-test probability assumed to be the total incidence of postoperative neurologic deficit in the study cohort.
Internal and external validity of individual studies were assessed with the QUADAS-2 tool (Supplemental Figure 2). Publication bias and heterogeneity across all studies were assessed with a funnel plot (Supplemental Figure 3).
Results
Study Characteristics
Study Characteristics and Deficit Details.
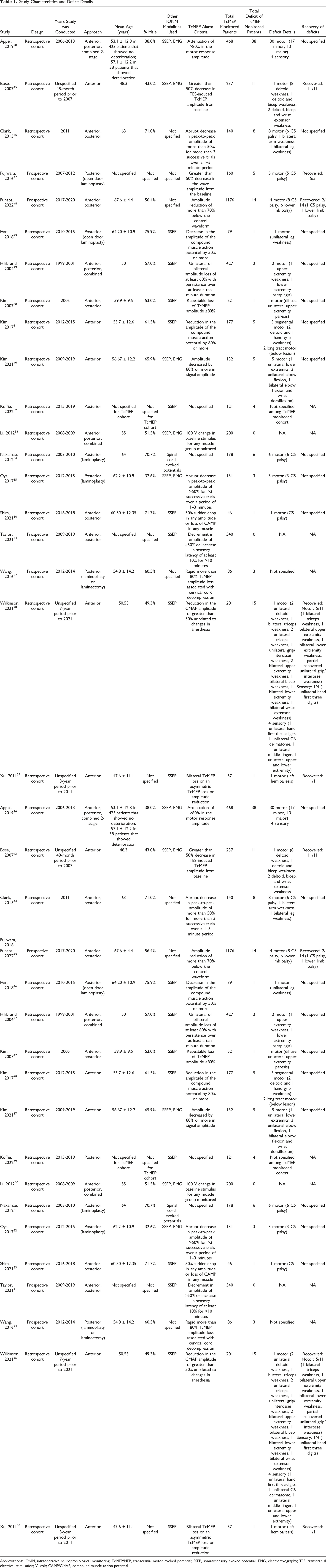
Abbreviations: IONM, intraoperative neurophysiological monitoring; TcMEP/MEP, transcranial motor evoked potential; SSEP, somatosensory evoked potential; EMG, electromyography; TES, transcranial electrical stimulation; V, volt; CAMP/CMAP, compound muscle action potential
The total cohort consisted of 4608 patients and the incidence of postoperative neurological deficits was 2.58% (119/4608). Of the total cohort, 7.68% (354/4608) had a significant intraoperative TcMEP change. The incidence of postoperative neurological deficits in patients with intraoperative TcMEP changes was 17.80% (63/354), while the incidence in those without was 1.32% (56/4254). Thus, the deficit rate was 13.5 times greater in patients with TcMEP changes than patients without. When subdivided into reversible and irreversible TcMEP changes, 8.86% (7/79) of patients with reversible TcMEP changes had postoperative neurological deficit and 55.32% (26/47) of patients with irreversible TcMEP changes had postoperative neurological deficit.
Data Analysis
All postoperative neurological deficits and all significant TcMEP changes
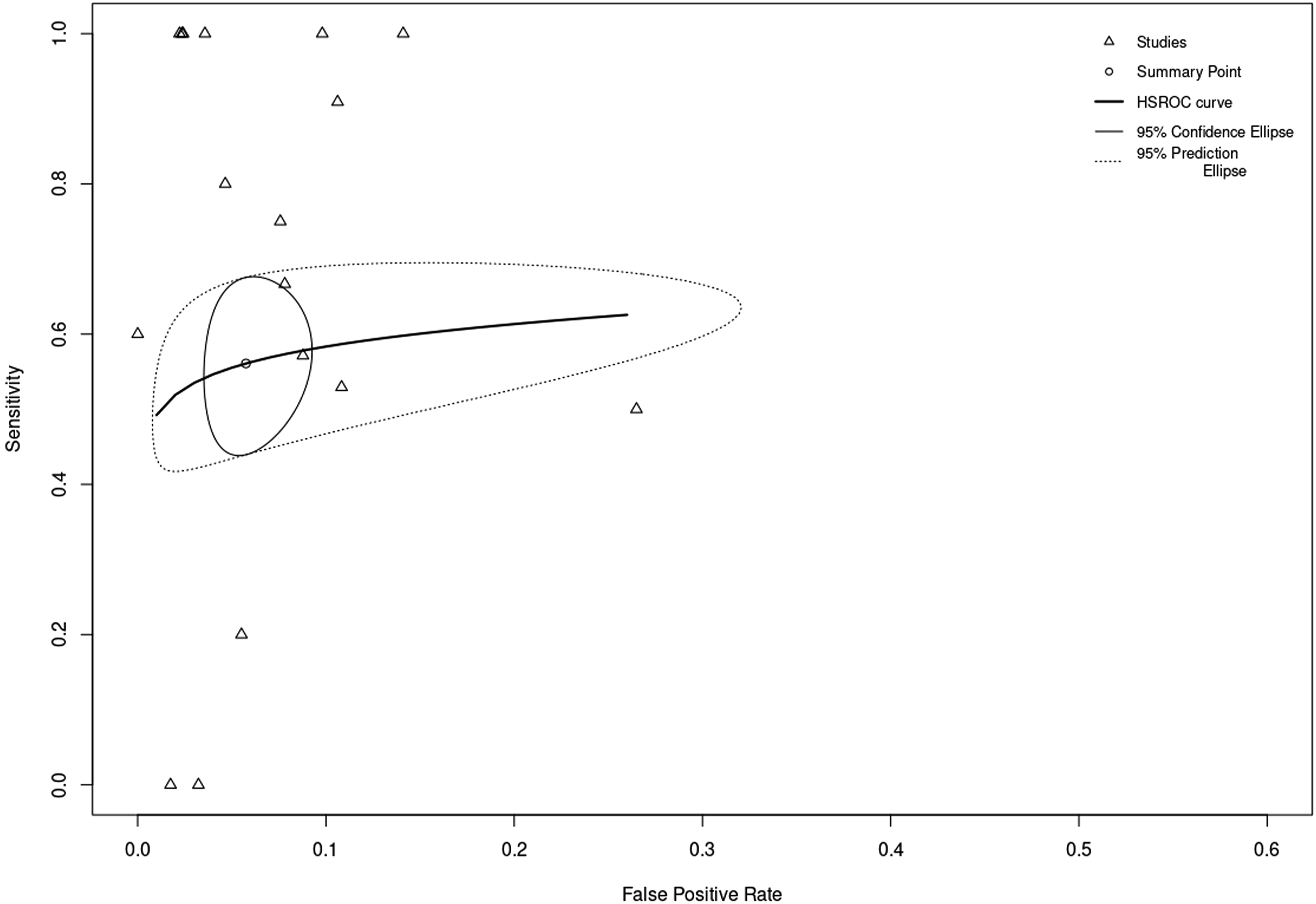
Study sensitivities ranged from 3% to 88%, while specificities ranged from 73% to 99%. The combined sensitivity and specificity across all studies using a bivariate mixed-effects model were 56% (95% CI, 46%–65%) and 94% (95% CI, 92%–96%), respectively (Figure 2). The model-based pooled area under the ROC curve was approximately .65 (Figure 3). Forest plot of the sensitivities and specificities for all significant intraoperative TcMEP changes in predicting postoperative neurologic deficit. Abbreviations: TcMEP: transcranial motor evoked potential. Hierarchical Summary Receiver Operating Characteristic (HSROC) curve for sensitivity and specificity of intraoperative TcMEPs in predicting postoperative neurological deficit. Abbreviations: TcMEP: transcranial motor evoked potential.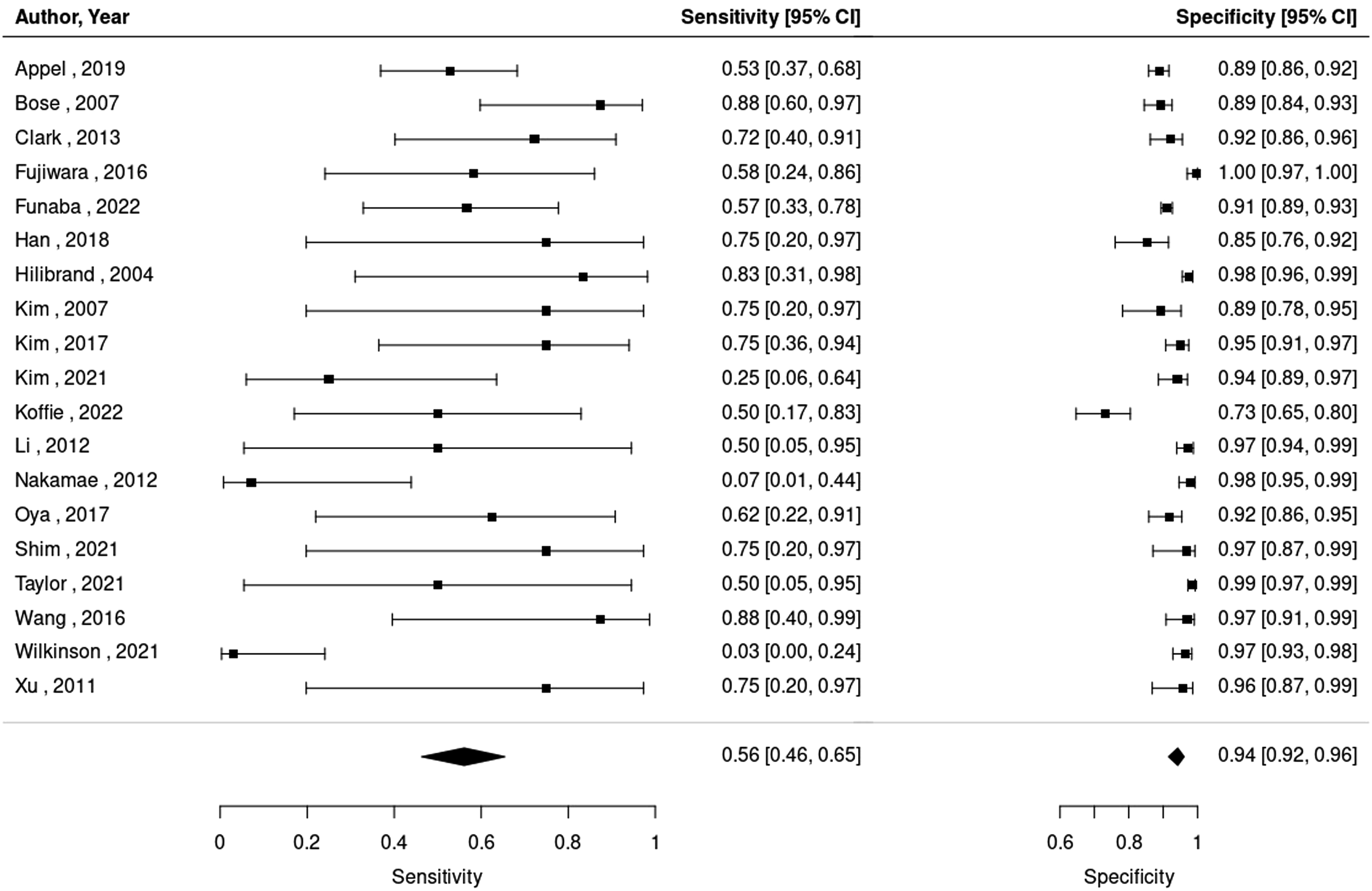
Using a pooled random effects model, the diagnostic odds ratio (DOR) for any significant intraoperative TcMEP change predicting neurological deficit was 19.26 (95% CI, 10.56–36.31, I2 = 5.8%), indicating that patients who developed new postoperative neurologic deficit were 19 times more likely to have had a significant intraoperative TcMEP change than those who did not have a postoperative neurologic deficit. The pooled log-diagnostic odds ratios (log-DOR) were calculated to minimize variations in the confidence intervals of individual studies (Figure 4A). (A) Forest plot of log-diagnostic odds ratios (log-DOR) for all significant TcMEP changes in predicting postoperative neurological deficit. (B) Forest plot of log-diagnostic odds ratios (log-DOR) for all reversible TcMEP changes in predicting postoperative neurological deficit. (C) Forest plot of log-diagnostic odds ratios (log-DOR) for all irreversible TcMEP changes in predicting postoperative neurological deficit. Abbreviations: TcMEP: transcranial motor evoked potential.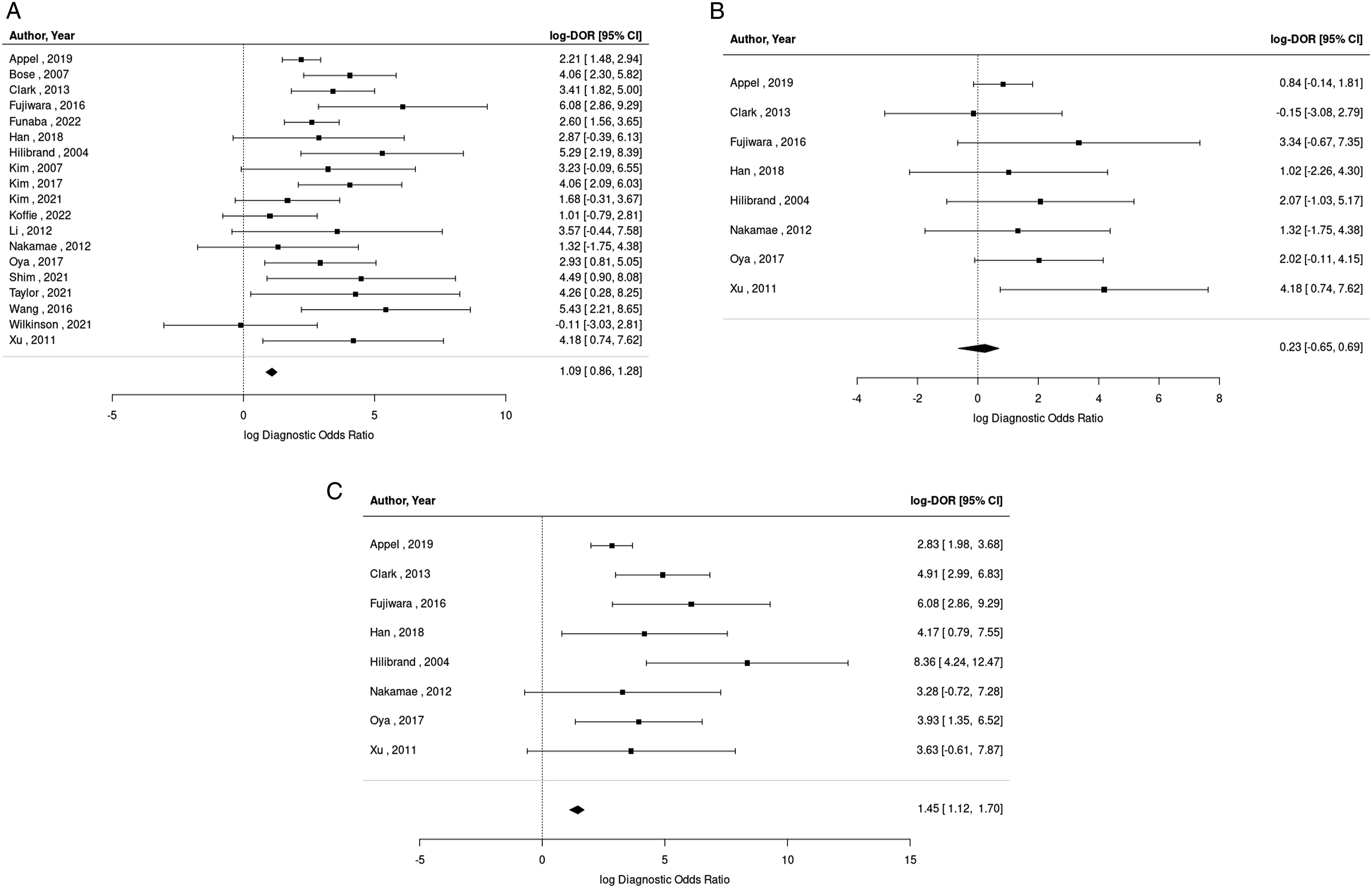
The positive and negative likelihood ratios depicted in the Fagan nomogram were 9.33 and .47, respectively (Figure 5). The pre-test probability was assumed to be 2.58%, the total incidence of postoperative neurologic deficits in the study cohort. For patients with a significant intraoperative TcMEP change, the estimated posterior probability of developing a new postoperative neurologic deficit increased to 20.0%. For patients without a significant intraoperative TcMEP change, the estimated posterior probability of developing a new postoperative neurologic deficit decreased to 1.0%. Fagan nomogram depicting pre- and post-test probability of developing postoperative neurologic deficit with TcMEP monitoring. Abbreviations: TcMEP: transcranial motor evoked potential.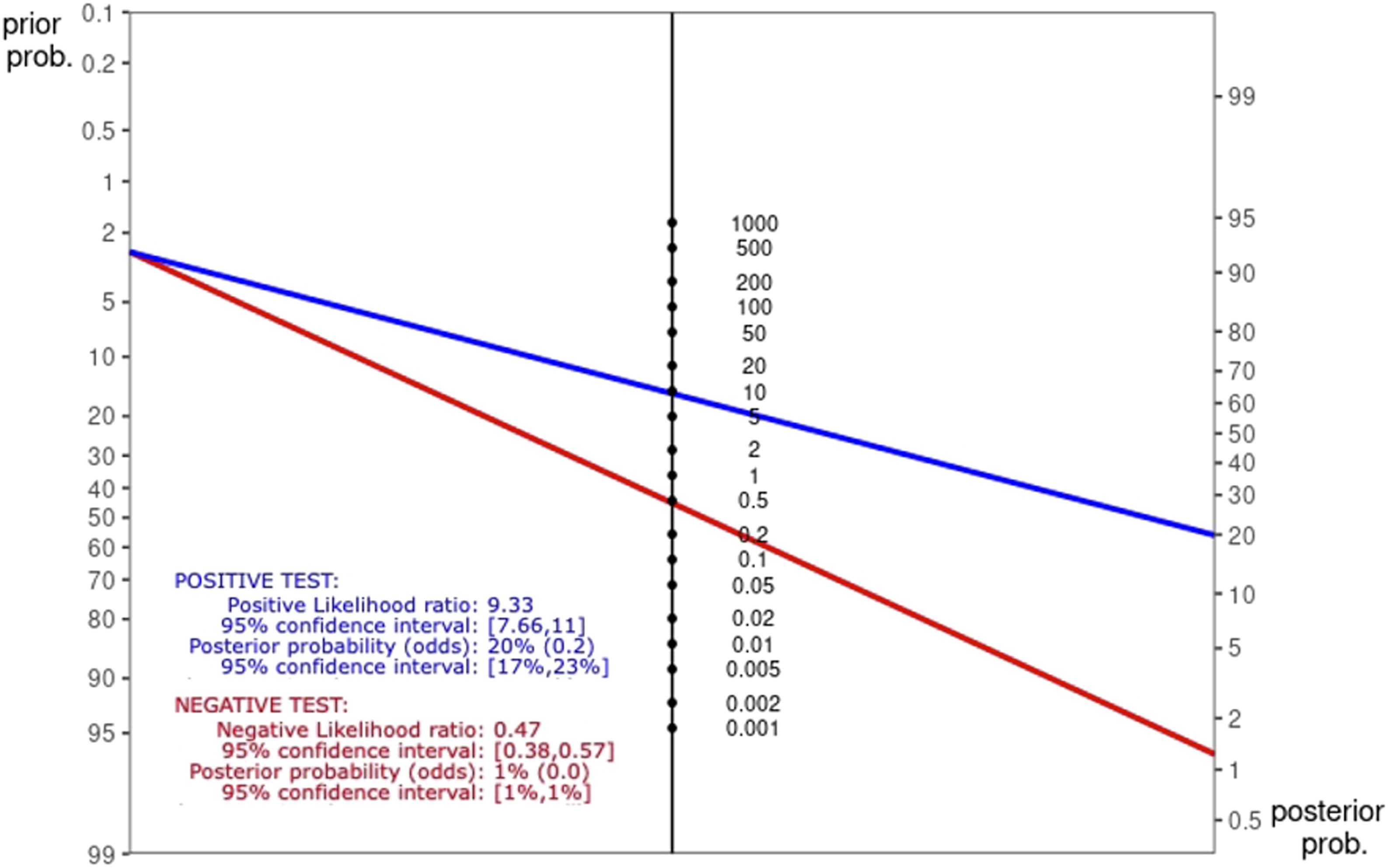
All postoperative neurological deficits and reversible TcMEP changes
Of the 19 total studies, 8 evaluated reversible TcMEP changes, with a cohort of 1640 patients. The incidence of reversible TcMEP changes in this cohort was 4.82% (79/1640). The DOR for reversible TcMEP changes in predicting postoperative neurologic deficit was 3.54 (95% CI, 1.69-7.40, I2 = 0%) (Figure 4B). The sensitivity and specificity of reversible TcMEP changes across these studies was 16% (95% CI, 9%–29%) and 95% (95% CI, 92%–97%), respectively, with an area under the ROC curve of .38.
All postoperative neurological deficits and irreversible TcMEP changes
Of the 19 total studies, 8 evaluated irreversible TcMEP changes, with a cohort of 1640 patients. The incidence of irreversible TcMEP changes in this cohort was 2.87% (47/1640). The DOR for irreversible TcMEP changes in predicting postoperative neurologic deficit was 71.74 (95% CI, 21.20–242.7, I2 = 0%) (Figure 4C). The sensitivity and specificity of irreversible TcMEP changes across these studies was 49% (95% CI, 35%–63%) and 98% (95% CI, 97%–99%), respectively, with an area under the ROC curve of .81.
Anterior vs posterior cervical spine procedures
Among the 19 studies in this study, 5 reported data from anterior cervical spine procedures while another 6 reported data from posterior cervical spine procedures. The remaining 8 studies reported data from a mix of anterior and posterior procedures. Subgroup analysis was performed to compare the diagnostic accuracy of intraoperative TcMEPs in anterior and posterior procedures.
The 5 studies conducted in anterior cervical spine procedures had a cohort of 804 patients, of whom 7.84% (63/804) had a significant intraoperative TcMEP change and 4.60% (37/804) experienced a new postoperative neurologic deficit. The DOR for all TcMEP changes in predicting postoperative neurologic deficit during anterior procedures was 17.57 (95% CI, 3.77–81.79, I2 = 6.76%) (Figure 6A). The sensitivity and specificity of TcMEP changes across the anterior cervical studies was 49% (95% CI, 14%–85%) and 94% (95% CI, 91%–96%), respectively. (A) Forest plot of log-diagnostic odds ratios (log-DOR) for all significant TcMEP changes in predicting postoperative neurological deficit in anterior spine surgeries. (B) Forest plot of log-diagnostic odds ratios (log-DOR) for all significant TcMEP changes in predicting postoperative neurological deficit in anterior spine surgeries. Abbreviations: MEP: transcranial motor evoked potential.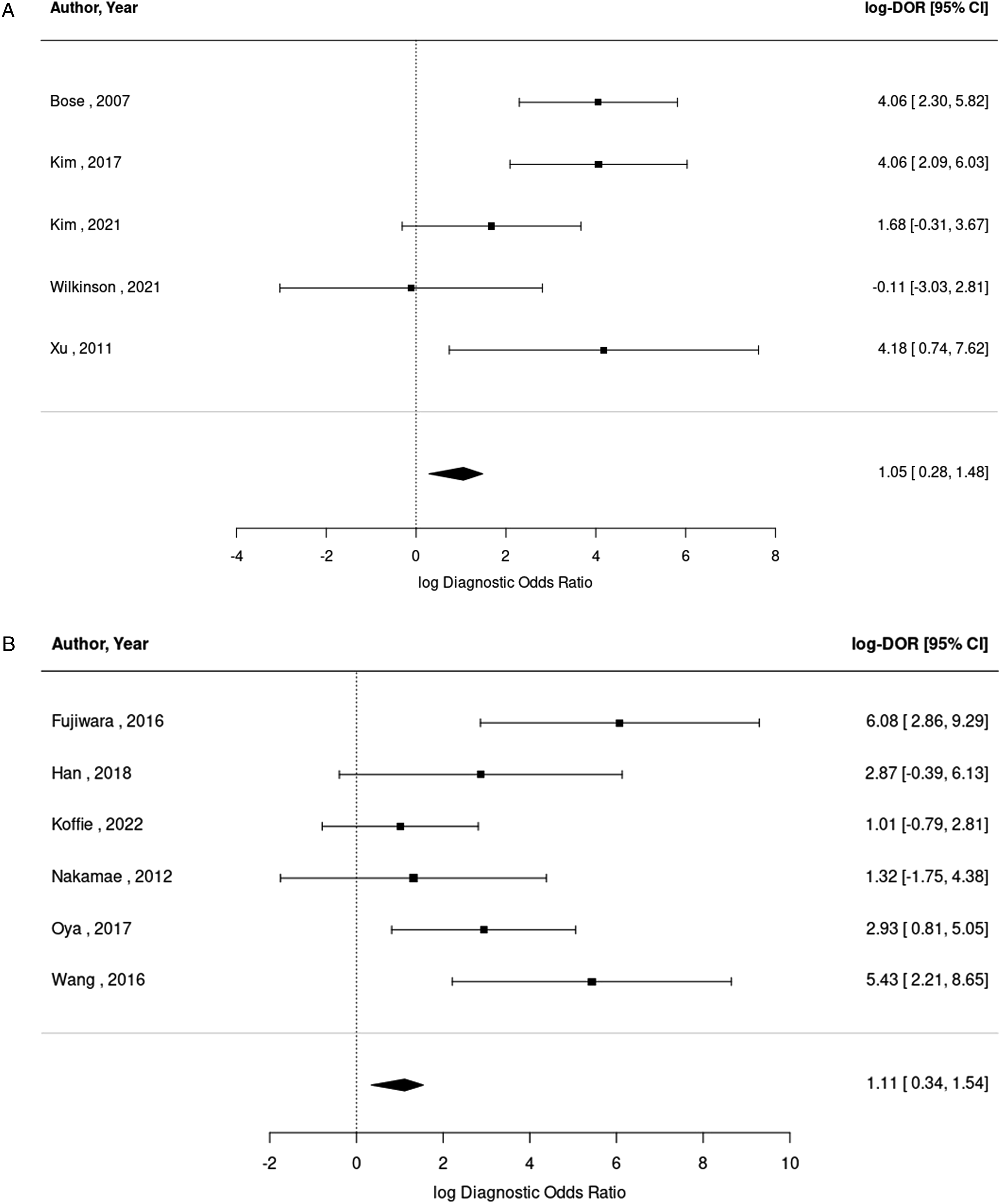
The 6 studies conducted in posterior cervical spine procedures had a cohort of 755 patients, of whom 9.00% (68/755) had a significant intraoperative TcMEP change and 2.91% (22/755) experienced a new postoperative neurologic deficit. The DOR for all TcMEP changes in predicting postoperative neurologic deficit during posterior procedures was 21.01 (95% CI, 4.10–107.64, I2 = .33%) (Figure 6B). The sensitivity and specificity of TcMEP changes across the anterior cervical studies was 55% (95% CI, 34%–75%) and 94% (95% CI, 83%–98%), respectively.
Discussion
The results of this meta-analysis demonstrate that IONM with TcMEP may be useful in predicting the risk of postoperative neurological deficit in cervical spine decompression surgery. The study found that patients who awoke with new postoperative neurological deficits were approximately 19 times more likely to have experienced intraoperative TcMEP changes than those without new deficits. Additionally, according to the Fagan nomogram, the probability of postoperative deficit in those with TcMEP changes increases from a baseline of 2.58% to 20.00%. The risk of postoperative neurological deficit also appears to vary among the different subcategories of TcMEP changes, with irreversible TcMEP changes indicating a much higher risk than reversible changes. Patients with new postoperative neurological deficits were only 3.5 times more likely to have had reversible TcMEP changes but nearly 72 times more likely to have exhibited irreversible TcMEP changes intraoperatively compared to patients without deficits. This may be because changes that were reversible improved with intraoperative interventions, but changes that were irreversible were either unable to be reversed with interventions or were not heeded. Thus, while all TcMEP alarms must be paid attention, those that do not reverse with intraoperative intervention portend higher risk of postoperative deficit and should be monitored/rehabilitated as such.
Overall, intraoperative TcMEP changes had a high specificity but relatively low sensitivity in predicting postoperative neurological deficits in patients undergoing cervical spine decompression surgery. Thus, while TcMEP changes may not predict all deficits and falsely predicts complications at a high rate, the nature of each TcMEP alarm (irreversible vs reversible, bilateral vs unilateral) should be scrutinized, as more severe TcMEP changes are more likely to be indicative of imminent neural deficits. TcMEPs monitor the integrity of the motor pathway originating from the motor cortex, including the descending corticospinal tracts as well as specific lower motor neuron pathways depending on the muscles tested.28-30 Thus, an injury to any portion of this motor pathway, including a spinal cord injury or a nerve root injury during surgery may result in significant TcMEP alarms. 31 Injury in this context may include both iatrogenic physical insults and neuronal ischemia from compromised blood supply.30,31 In this study, both motor and sensory deficits were grouped in the sensitivity and specificity analysis, as TcMEPs have been shown to have utility in monitoring for nerve root injury which can manifest as both sensory and motor deficit.25,26,32 TcMEPs may therefore play a minor role in the detection of specific sensory deficits secondary to nerve root injury. In this study, TcMEP monitoring in posterior procedures, where the lateral corticospinal tract is theoretically at higher risk, had a higher DOR, sensitivity, and number of significant changes. However, because TcMEPs only directly monitor motor pathways, pure sensory deficits from injury to the spinothalamic and dorsal column medial lemniscus pathways may go undetected, potentially contributing to their low sensitivity. In contrast, somatosensory evoked potentials (SSEPs) directly monitor ascending sensory tracts and may be therapeutically useful in combination with TcMEPs, although institutional standard of care may dictate use of monitoring modalities.33,34
A Delphi consensus report published in 2014 outlines a standardized checklist protocol in response to IONM alarms such as TcMEP changes. 23 The protocol involves alerting the neurophysiologist and surgeon, optimizing hemodynamic parameters and other vital signs, examining the status of anesthetic and paralytic agents, evaluating the surgical field for spinal cord/nerve root compression, and employing surgical maneuvers to reverse the TcMEP change to baseline values.35,36 If this protocol is not performed or if the TcMEP change does not revert to baseline despite this protocol, this may be considered an irreversible change, which portends a higher risk of postoperative neurological deficit per this study. However, it is important to note that TcMEPs are also sensitive to non-surgical factors including anesthetic agents, hypoxemia, hypotension, hypercapnia, and electrolyte abnormalities, although these TcMEP changes may manifest globally rather than focally.17,30,37,38 For this reason, there may be a nontrivial rate of false-positive TcMEP alarms that do not actually indicate neural deficit. In this study, out of the 354 TcMEP changes, only 63 (17.80%) of them resulted in postoperative deficit. In addition to TcMEP sensitivity to non-surgical factors, other reasons for this high false-positive rate include prevention of imminent neural deficits with intraoperative maneuvers and inconsistent/improper alarm criteria. Unlike SSEPs, TcMEP alarm criteria often varies by institution due to lack of standardization, especially in studies conducted before 2015. Nearly all the studies in this analysis had their own criteria. Thus, a further refinement of TcMEP alarm criteria may be necessary to reduce the high false-positive rate associated with significant changes.
Due to their anesthetic complexity, required workflow changes, and resultant increase in procedure time, TcMEPs remain underutilized. 39 Furthermore, although TcMEPs may be used to predict risk of neurological deficit, there is a lack of reliable evidence regarding whether IONM with TcMEPs is able to reduce the incidence of postoperative deficit. Certain clinical studies including Kim et al. compared outcomes from an IONM cohort to a historic control cohort without IONM and found a deficit rate of 3.79% in the monitored group compared to 14.06% in the control group. 40 However, it is difficult to attribute this deficit reduction to IONM alone, as improved surgical techniques and technology may have played a role. Regardless, IONM is the only tool that can serve as an indication for neural injury intraoperatively and allows for targeted therapeutic intervention before irreversible damage.
The overall incidence of postoperative neurological deficits in this study was 2.58%, which is consistent with rates found in the literature. C5 palsy was the most documented deficit, often presenting as deltoid weakness without associated sensory deficits. Various mechanisms have been proposed for this complication including mechanical trauma from surgical instrumentation, 41 intraoperative ischemia, 42 and reperfusion injury, 43 but they remain hypotheses due to lack of reliable evidence. 44 It is also important to note that C5 palsy is often underreported due to its diagnostic difficulty and unknown pathophysiology. 10
Although this meta-analysis is comprehensive in its analysis of the available literature, with quality assessment via the QUADAS-2 tool, several limitations exist. First is the possibility of search bias due to the difficulty of screening every possible study assessing TcMEP use in cervical spine surgery. Second is the possibility of publication bias due to significant results being published over non-significant results, although the funnel plot (Supplemental Figure 3) does not show evidence of such. Third, as an inherent limitation of systematic reviews, the studies included within the analysis may lack uniformity with variability in follow-up times, postoperative evaluations, surgical/anesthetic techniques, TcMEP response protocols, TcMEP alarm criteria and postoperative rehabilitation. Fourth, many of the included studies do not give detailed reports of preoperative neurological status or neuropathic comorbidities including diabetes mellitus. Fifth, many studies do not report details of each TcMEP change including muscle tested, length of time for reversal to baseline, and intraoperative interventions in response to TcMEP changes. Furthermore, very few studies reported on the correlation of TcMEP change location to the specific postoperative deficit and the severity of deficit. As a result, this study was unable to perform a subgroup analysis of deficits related to spinal cord vs nerve root insults, sensory vs motor deficits, and paralysis vs palsy. Additionally, many of the studies did not report on the timing of the deficits and whether the deficits were permanent or resolved, making analysis of this information infeasible. Finally, many of the studies in the analysis utilized other IONM modalities, which likely altered the predictive power of TcMEPs. If surgeons responded to alarms from other monitoring modalities before a significant TcMEP change was observed, this could perhaps inflate the false negative values. A prospective multicenter study combining SSEP and TcMEP monitoring with detailed recording of all significant changes and corresponding postoperative deficits may best allow for further evaluation of IONM in cervical spine surgery.
Conclusion
TcMEP monitoring has high specificity but low sensitivity for predicting postoperative neurological deficit in cervical spine decompression surgery. Patients with new postoperative neurological deficits were approximately 19 times more likely to have experienced intraoperative TcMEP changes than those without new deficits, with irreversible TcMEP changes indicating a much higher risk of deficit than reversible TcMEP changes. Thus, the utilization of IONM, specifically TcMEPs, may serve as a marker for impending neural deficit thereby permitting a chance for therapeutic intervention to be performed. Without IONM, the surgical team has no indication of an ongoing change in neurological function and therefore, any chance to intervene is lost.
Supplemental Material
Supplemental Material - Transcranial Motor Evoked Potentials as a Predictive Modality for Postoperative Deficit in Cervical Spine Decompression Surgery – A Systematic Review and Meta-Analysis
Supplemental Material for Transcranial Motor Evoked Potentials as a Predictive Modality for Postoperative Deficit in Cervical Spine Decompression Surgery – A Systematic Review and Meta-Analysis by Rajiv P. Reddy, Anya Singh-Varma, Robert Chang, Abhinav Vedire, Katherine M. Anetakis, Jeffrey R. Balzer, Donald J. Crammond, Varun Shandal, Joon Y. Lee, Jeremy D. Shaw, and Parthasarathy D. Thirumala in Global Spine Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
