Abstract
Study Design
Retrospective Cohort Study.
Objective
The study aimed to analyze anatomical variants of segmental spinal aplasia (SSA) and investigate factors influencing surgical treatment outcomes, with a specific focus on the incidence of complications.
Methods
The study focused on patients with SSA treated at a single medical center, with over two years of follow-up. Neurological function changes were evaluated using the modified Japanese Orthopedic Scale (mJOA). Functional independence was measured using the Functional Independence Measure (FIM/WeeFIM) scale, and complications, well-being, and reoperation instances were documented. Statistical analyses used ANOVA and Kruskal-Wallis test.
Results
The predominant localization of SSA in 36 own cases occurs near or at the level of the thoracolumbar junction, often accompanied by significant spinal cord narrowing and a low position of the conus medullaris. Additionally, it frequently presents with aplasia of the lower ribs. Cervicothoracic SSA was more commonly associated with segmentation disorders (P = .04). The most common early complications were wound problems (17%) and neurological deterioration (17%); the most common late complications were: non-fusion (34%); 38% of patients required one or more revision surgery. The type, age of surgery, level of surgery, and initial neurological deficient did not significantly influence the incidence of complications or neurological and functional outcomes.
Conclusion
SSA, a range of anomalies appearing early in childhood, progresses gradually. Surgery involves vertebrectomy followed by interbody fusion and screw fixation, guided by neurophysiological monitoring. Surgery is recommended for worsening neurological symptoms, but conservative options like bracing can be considered, due to a high risk of complications.
Keywords
Introduction
Within the realm of spinal anomalies, a diverse spectrum of intricate pathologies exists, encompassing combined anomalies of both the spine and spinal cord. One such unique and challenging condition is segmental spinal aplasia (SSA), where a segment of the spine—spanning one, two, or more vertebrae—is either absent (agenesis) or significantly underdeveloped (aplasia).1,2 The etiology of this anomaly remains unclear.3,4 What prompts the sudden interruption of a portion of the spine, and in some cases, the spinal cord, in localized regions?
This condition often presents with irreducible vertebral dislocation, particularly in early childhood, further complicating the clinical landscape. 5 The surgical treatment of SSA poses a formidable challenge, marked by a remarkably high incidence of complications. This raises critical questions: How can these complications be mitigated, and what constitutes the optimal approach to the treatment of patients with this unique set of challenges?
The underlying hypothesis of this study posits that the spectrum of SSA is extensive and not confined to a singular nosological unit. Moreover, in this study, we delve into the outcomes of our own cohort of patients, aiming to shed light on the factors associated with the majority of complications. By analyzing our experiences and findings, we seek to contribute valuable insights that may guide future approaches to the treatment of SSA, paving the way for enhanced patient care and improved clinical outcomes.
Methods
The studied patient cohort comprises individuals diagnosed with SSA. Treatment interventions were administered at a single medical center between 2010 and 2021, with a comprehensive follow-up period extending beyond two years.
The classification of the cases was based on several criteria: 1. Level: cervicothoracic, thoracolumbar, lumbar only; 2. Degree of underdevelopment: agenesis or aplasia; 3. Number of affected vertebrae: one vertebra, two vertebrae (segment), three or more vertebrae; 4. Condition of the spinal cord: moderately narrowed (d > 5 mm), pronounced narrowing (d < 5 mm), and absence (not visualized either on MRI or intraoperatively). 5. Presence of concomitant spinal cord abnormalities, such as myelomeningocele, low-lying conus medullaris and syringomyelia.
Radiological measurements include Cobb angles measurement to assess the degree of correction in spinal deformities. Changes in neurological function were assessed through a combination of clinical examinations and the application of a standardized scale covering upper and lower extremity function, sensitivity, and urological symptoms and chronic pain (modified for pediatric patients Japanese Orthopedist Association Scale/mJOA and Visual Analog Scale/VAS).6 Functional independence was quantified using the Functional Independence Measure (FIM/WeeFIM) scale.7 Early and late complications, subjective well-being, and instances of reoperation were documented.
In this study, we conducted a comprehensive analysis comparing various parameters among groups of patients. To assess the normality of the data distribution, the Kolmogorov-Smirnov test was utilized. Subsequently, data with a normal distribution (age, FIM and mJOA scores, scoliosis and kyphosis severity, duration of observation, blood loss volume, and surgery duration) were analyzed using one-way analysis of variance (ANOVA). Non-normally distributed data were analyzed using the non-parametric Kruskal-Wallis test. All statistical analyses were performed using the open-access platform https://www.socscistatistics.com/.
Results
Preliminary Analysis
Cohort of 36 patients with SSA were analyzed. The majority of these patients were under the age of 10 at the time when they first came to our clinic.
The level of defect in most cases was localized at the level thoracolumbar junction (n = 14, 39%) or near it (n = 10, 28%) (Figure 1). Half of the patients showcased a significant narrowing of the spinal cord (spinal cord diameter less than 5 mm at the level of SSA). Despite spinal cord narrowing, leg functionality varied significantly: 36% (n = 13) could walk independently, 25% (n = 9) needed assistance, and 39% (n = 14) were unable to walk. A low position of the conus medullaris below the L2 level was observed in 44% of cases. Distribution of segmental spinal absence levels in 36 patients.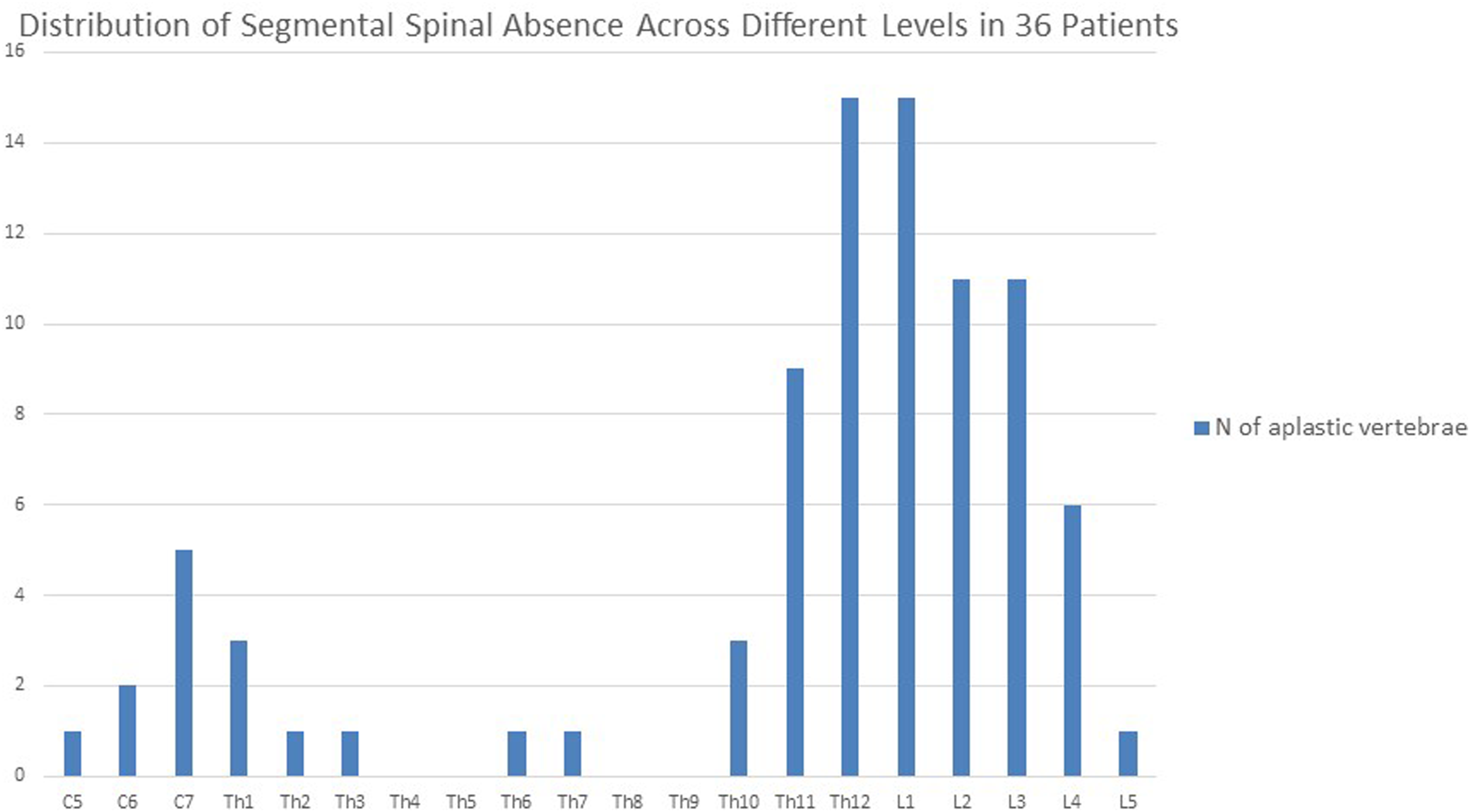
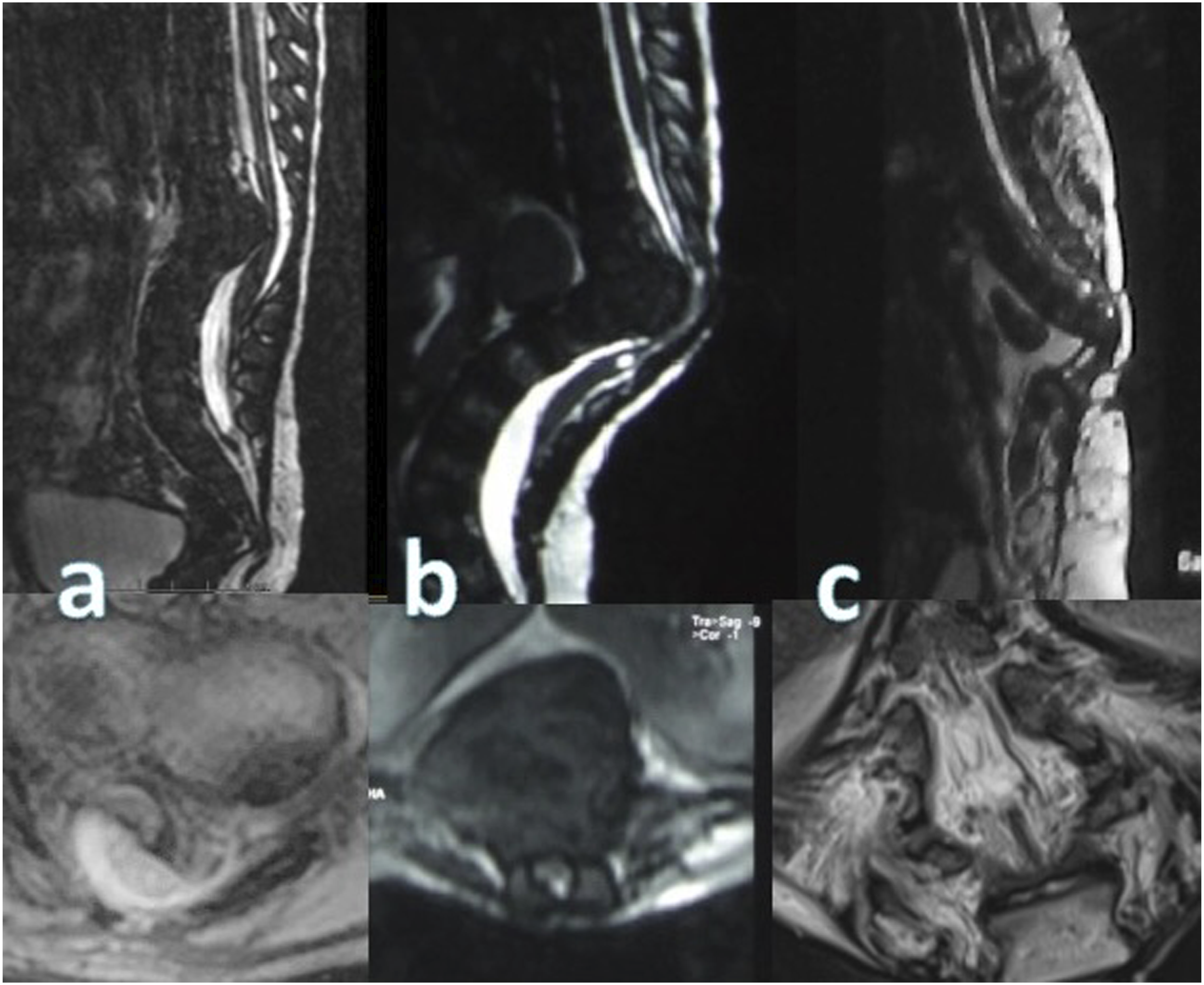
We divided patients into 3 groups depending on the level of anomaly: cervicothoracic SSA (Figure 2A, n = 5), thoracolumbar SSA (Figure 2B, n = 18), and lumbar-only SSA (Figure 2C, n = 13). These groups demonstrated comparability in terms of age, sex, the number of aplastic vertebrae, degree of spinal cord narrowing (Figure 3), and neurological and functional status (Table 1). Notably, in the cervicothoracic SSA group, all patients exhibited segmentation disorders (Klippel-Feil anomaly), distinguishing it significantly from the other groups (Table 4). In contrast, the thoracolumbar SSA group displayed a higher prevalence of lower ribs agenesis. Sagittal CT section of the spine of patients with segmental spinal aplasia at the (A) cervicothoracic, (B) thoracolumbar and (C) lumbar levels. Sagittal and axial MRI section of the spinal cord and spine of patients with (A) moderate narrowing, (B) severe narrowing of the spinal cord, and (C) absence when the caudal portion of the spinal cord is not visualized. Comparative Profile of 36 Patients With Segmental Spinal Absence at Various Levels. FIM - Functional Independence Measure; KFS – Klippel-Feil Syndrome; LLCM – low-lying conus medullaris, MMC – myelomeningocele; mJOA – modified Japanese Orthopedic Scale; N - number.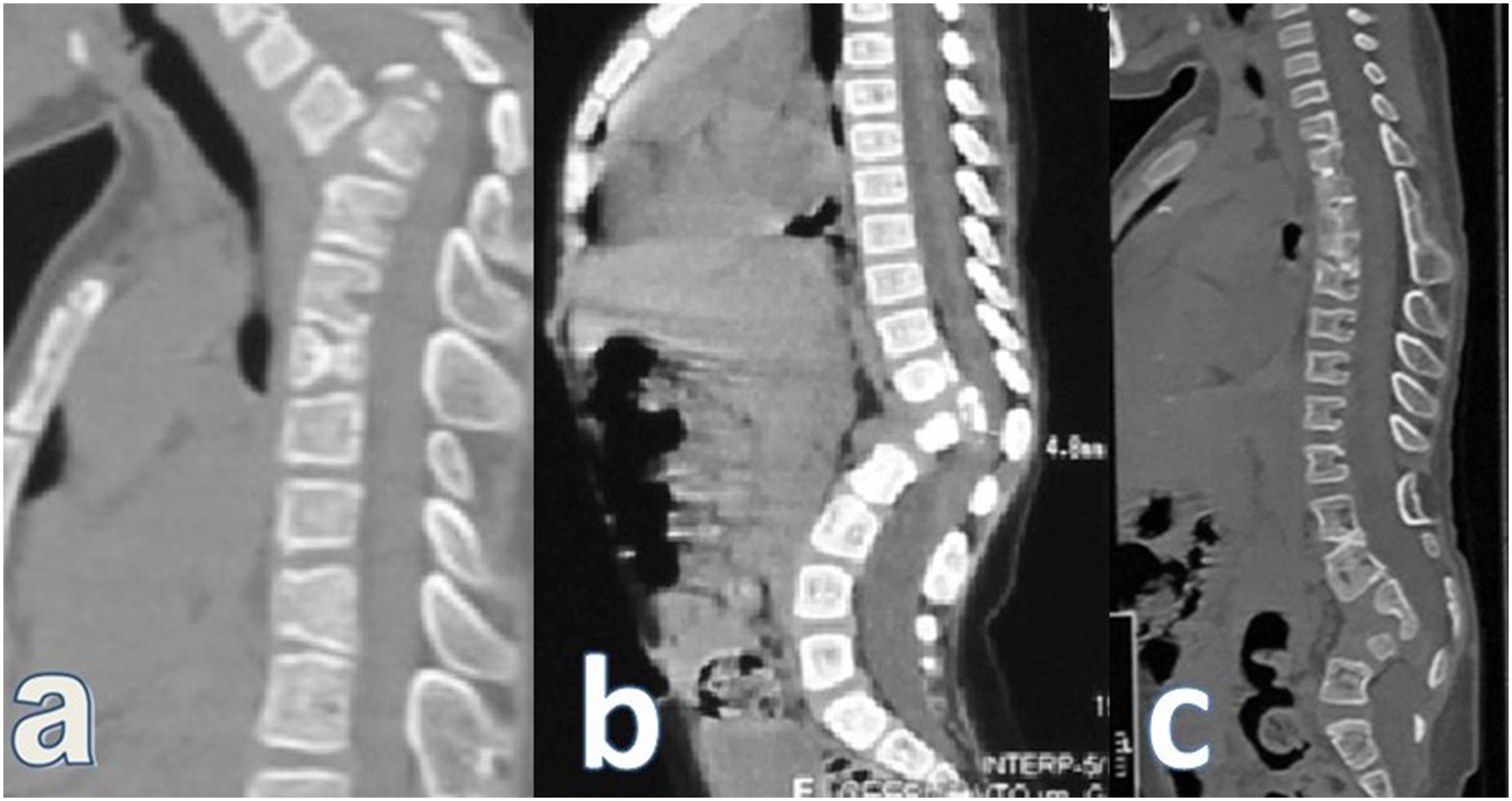
A distinct subset of patients warranting special attention consisted of three individuals with myelomeningocele and SSA (Figure 4). In all cases, a conspicuous kyphotic deformity was observed, accompanied by the absence of the spinal cord at the level and below the point of SSA. This absence correlated with paralysis in the lower limbs (all patients had paraplegia). Notably, these patients exhibited marked cicatricial changes in the soft tissues of the back, characterized by a local deficiency of muscles and adipose tissue, lower extremities hypotrophy and severe orthopedic deformities. Computed tomography (top line) and magnetic resonance (low line) imaging of three patients with open neural tube defects and segmental absence of the spine and spinal cord.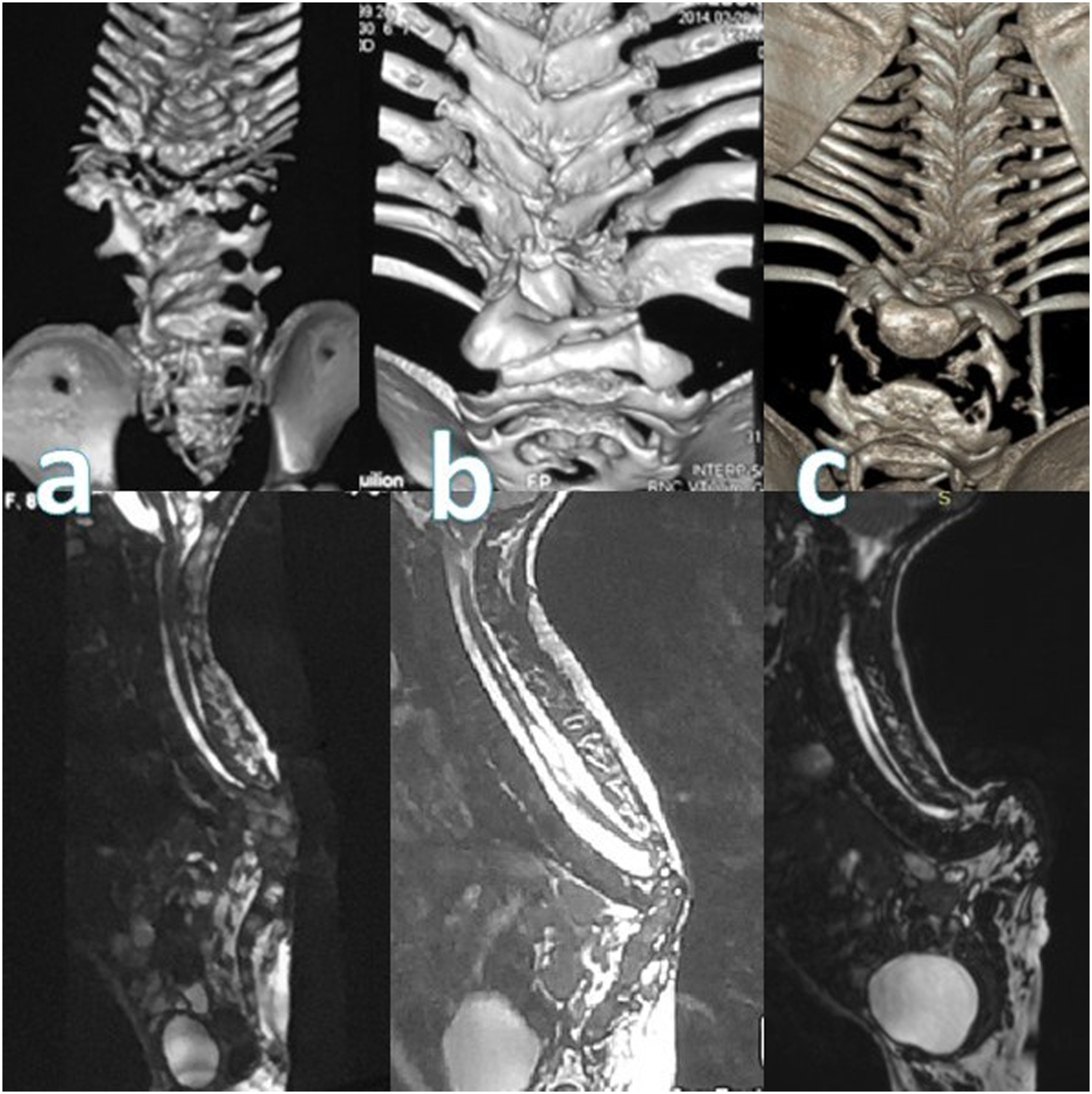
Result of Conservative Treatment
Among patients who did not undergo surgical treatment, there were four males and three females. One had SSA at the cervicothoracic level, three at the thoracolumbar junction, and three at the lower lumbar level. The median follow-up age for these patients was 4.5 years (range 2.3 – 7.2 years). Surgery was declined by parents across all cases due to the perceived high risk of complications.
Of these patients, five received braces, while two did not receive any treatment. In one case, the brace refusal was from the parents of a girl, and in another, wearing a brace was not recommended due to the elevated risk of bedsores attributed to acute-angle post-myelomeningocele lumbar kyphosis and severe scarring.
During the observation period, the clinical condition of three patients remained stable: one girl with stable lower paraplegia, one with mild paresis and pelvic organ dysfunction, and one with moderate paresis and urination disorders. However, four patients experienced a slow, progressive neurological deterioration: mild warsening in stability while walking, onset of impaired sensation in the legs, and new urination problems.
No significant worsening of functional status or chronic back pain was observed in any of the patients during the observation period.
Results of Surgical Treatment
Comparative Analysis of Surgical Outcomes in Three Groups of Patients With Segmental Spinal Aplasia at Different Levels.
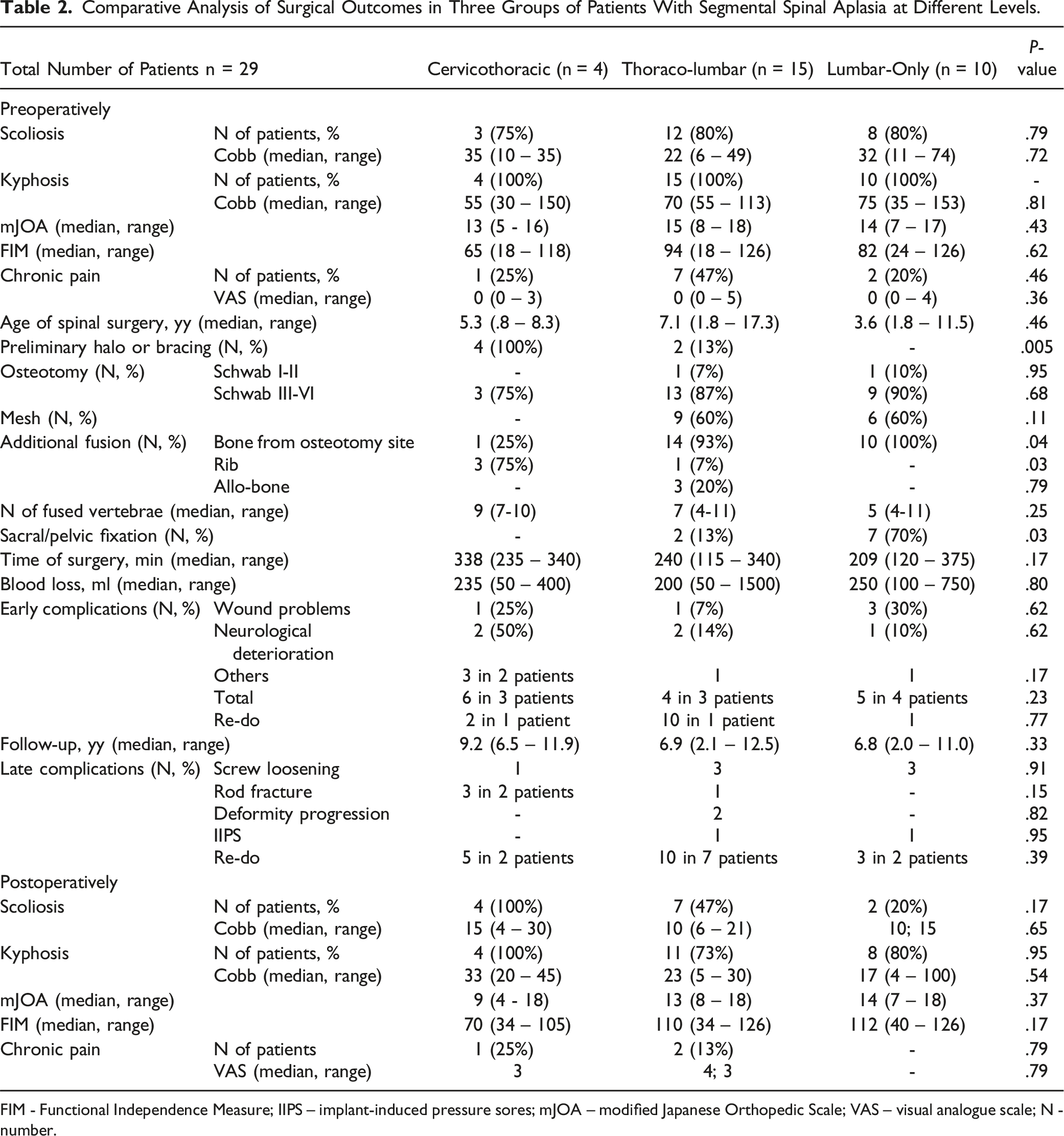
FIM - Functional Independence Measure; IIPS – implant-induced pressure sores; mJOA – modified Japanese Orthopedic Scale; VAS – visual analogue scale; N - number.
Comparative Analysis of Surgical Outcomes in Three Groups of Patients With Segmental Spinal Aplasia, Treated at Different Age.
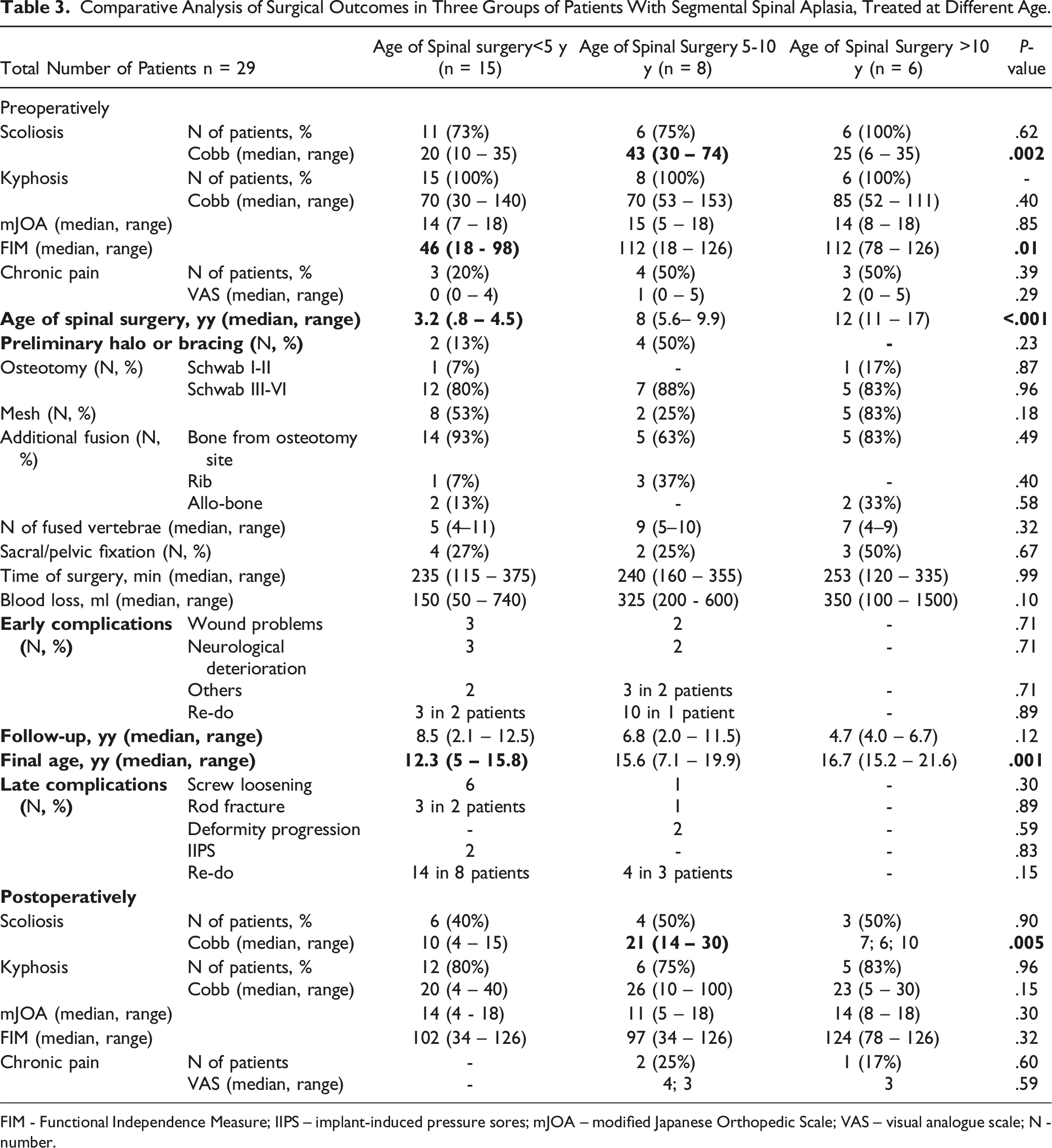
FIM - Functional Independence Measure; IIPS – implant-induced pressure sores; mJOA – modified Japanese Orthopedic Scale; VAS – visual analogue scale; N - number.
Comparative Analysis of Surgical Outcomes in Three Groups of Patients With Segmental Spinal Aplasia and Different Degree of Spinal Cord Narrowing.
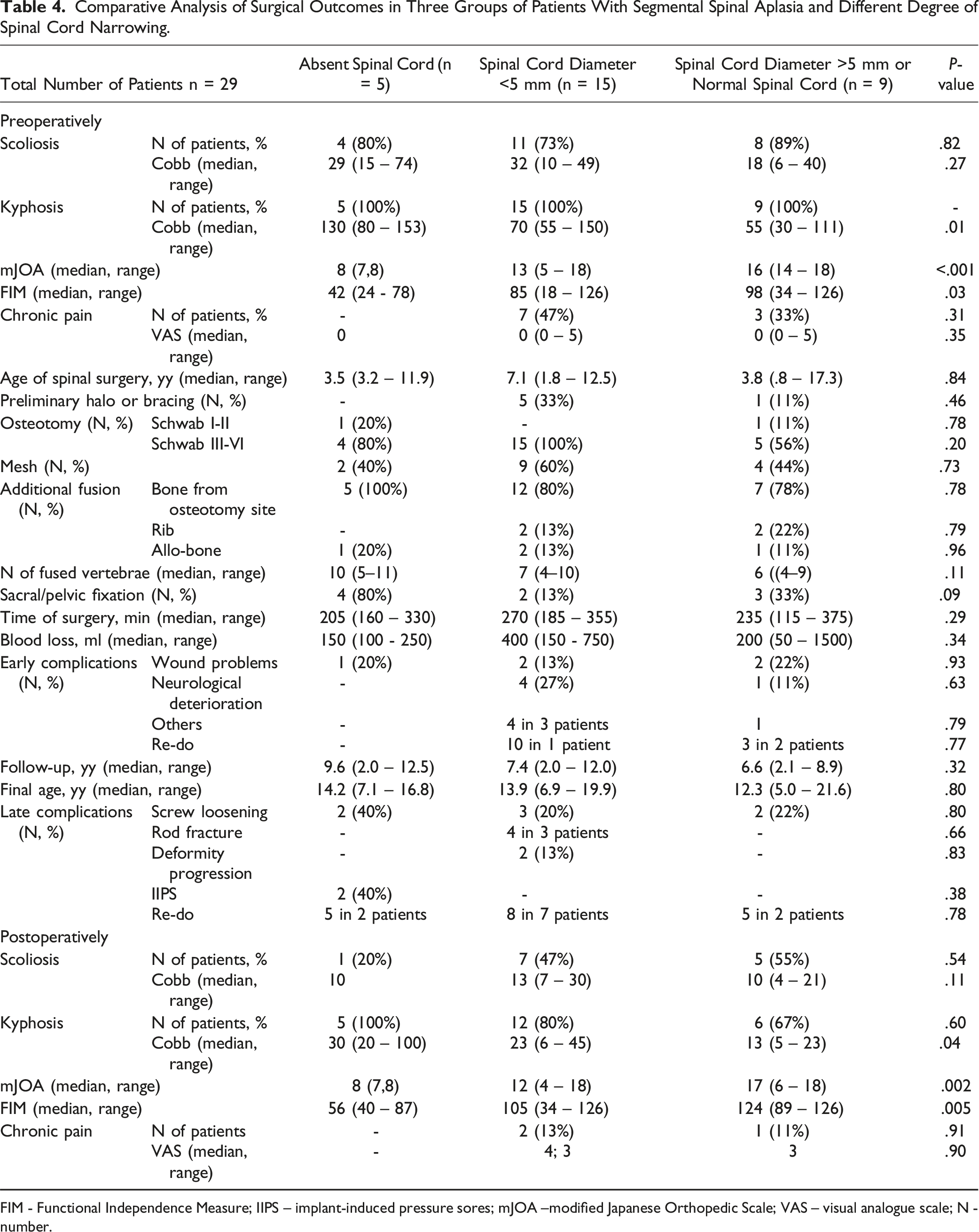
FIM - Functional Independence Measure; IIPS – implant-induced pressure sores; mJOA –modified Japanese Orthopedic Scale; VAS – visual analogue scale; N - number.
Comparative Analysis of Surgical Outcomes in Three Groups of Patients With Segmental Spinal Aplasia, Treated by Different Surgical Techniques.
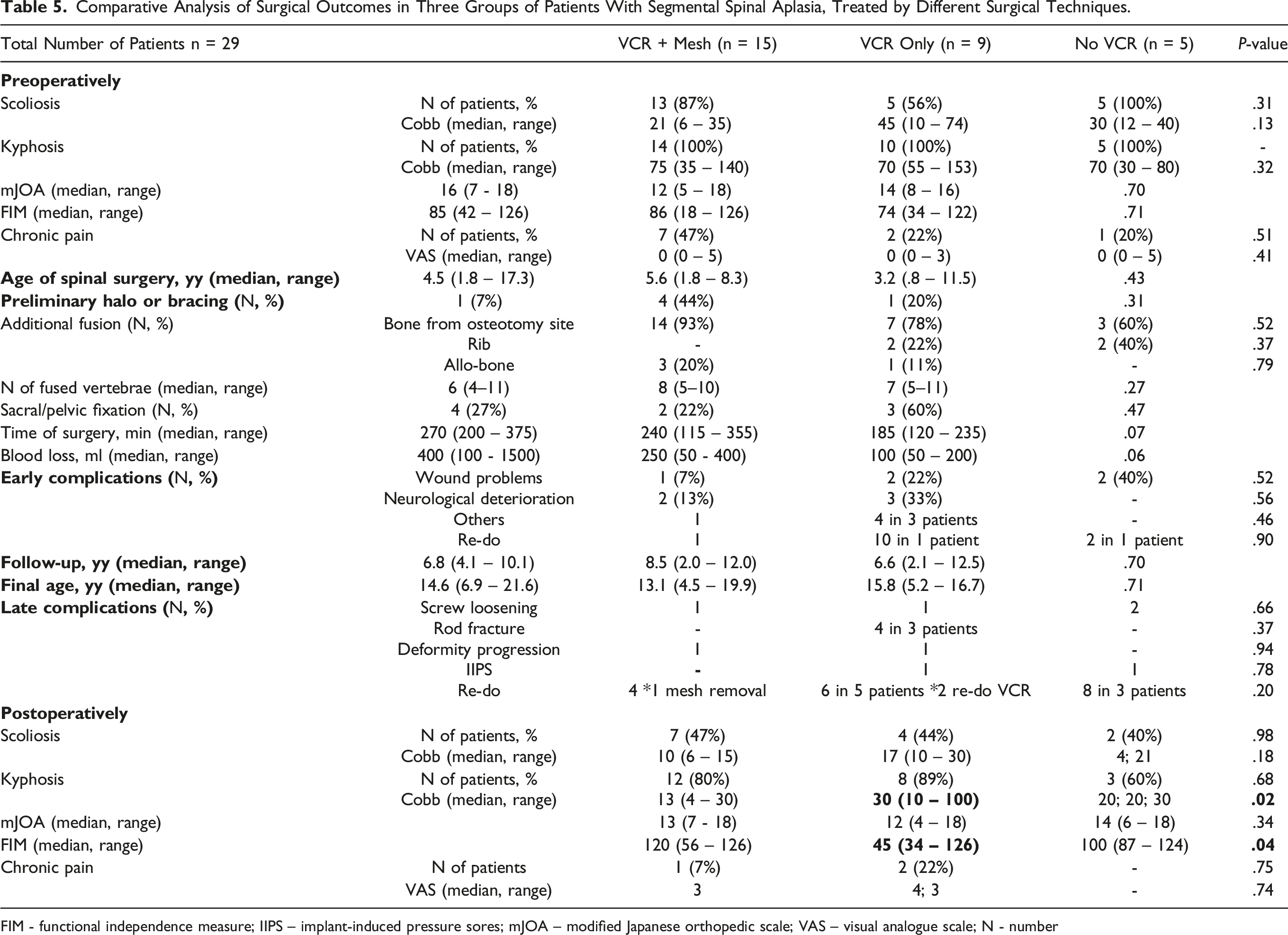
FIM - functional independence measure; IIPS – implant-induced pressure sores; mJOA – modified Japanese orthopedic scale; VAS – visual analogue scale; N - number
Totally, in the early postoperative period, the most common complications were wound problems (5 cases, 17%) and neurological complications (5 cases, 17%). Additionally, there were occurrences of postoperative pneumonia (2 individuals), skull bone fracture due to Mayfield clamp positioning (1 case), intraoperative hemodynamic instability, and subclavian catheter malposition leading to hemothorax (1 case each). Three patients required revision surgeries for early complications, with one of them undergoing a total of 10 surgeries due to significant wound dehiscence, the majority of which were reconstructive procedures.
In the late postoperative period, the most common complication was non-fusion, characterized by screw loosening (7 cases, 29%) and rod fractures (4 cases, 14%), while less frequently, spinal deformities progressed (2 cases, 7%). Overall, 11 patients (38%) underwent one or more revision interventions due to these complications.
When evaluating treatment outcomes among patients with SSA across different spinal levels (cervicothoracic, thoracic-lumbar, and lumbar), no significant disparities emerged (Table 2).
In the analysis of surgical treatment outcomes across various age groups (up to 5 years, 5-10 years, and older than 10 years), no notable disparities were identified (Table 3).
If moderate and severe narrowing of the spinal cord did not significantly correlate with the severity of paraparesis, then extremely severe narrowing (the spinal cord was not visualized by MRI) did correlate significantly with the severity of paresis (Table 4). When evaluating the treatment outcomes among patients with different degrees of spinal cord narrowing, noteworthy distinctions were observed primarily within the cohort characterized by the complete absence of the spinal cord at and below the level of aplasia (Table 4). In this specific group, both initially and at the conclusion of the observation period, there was a notable increase in the magnitude of kyphosis, accompanied by a lower functional and neurological status (paraplegia). Within this subgroup, two patients experienced a distinctive complication — implant-induced pressure sores, necessitating the subsequent removal of the construction.
Given that the primary divergence in our approach to patient treatment centered around the vertebrectomy technique, we categorized our patients into three distinct groups (Table 5): 1. Group 1 (n = 15): This cohort underwent the resection of aplastic vertebrae, accompanied by partial resection of adjacent vertebrae. The treatment protocol included anterior interbody fusion by the mesh filled with autograft (n = 14) or allograft (n = 1), coupled with posterior screw fixation (Figure 5); 2. Group 2 (n = 9): In this group, patients underwent resection of aplastic vertebrae with simultaneous partial resection of adjacent vertebrae. The treatment regimen involved posterior screw fixation and compression maneuver without using of mesh (Figure 6); 3. Group 3 (n = 5): Patients in this category received treatment involving posterior screw fixation with distraction without removing of aplastic vertebrae (Figure 7). Anteroposterior and lateral radiographs documenting the progression of a patient with Th10-12 vertebral aplasia are presented in three key stages: 8(A). At 1 year of age, 8(B). At the age of 2.5 years, and 8(C). Post-surgery following Th9-L1 vertebral resection. Displayed are the preoperative (left) and postoperative (right) anterior-posterior and lateral radiographs capturing the case of a 1.8-year-old female patient with Th11-L1 aplasia. Preoperative (left) and postoperative (right) anterior-posterior and lateral radiographs showcase a patient aged 11.5 years with aplasia of the L3-L4 vertebrae.
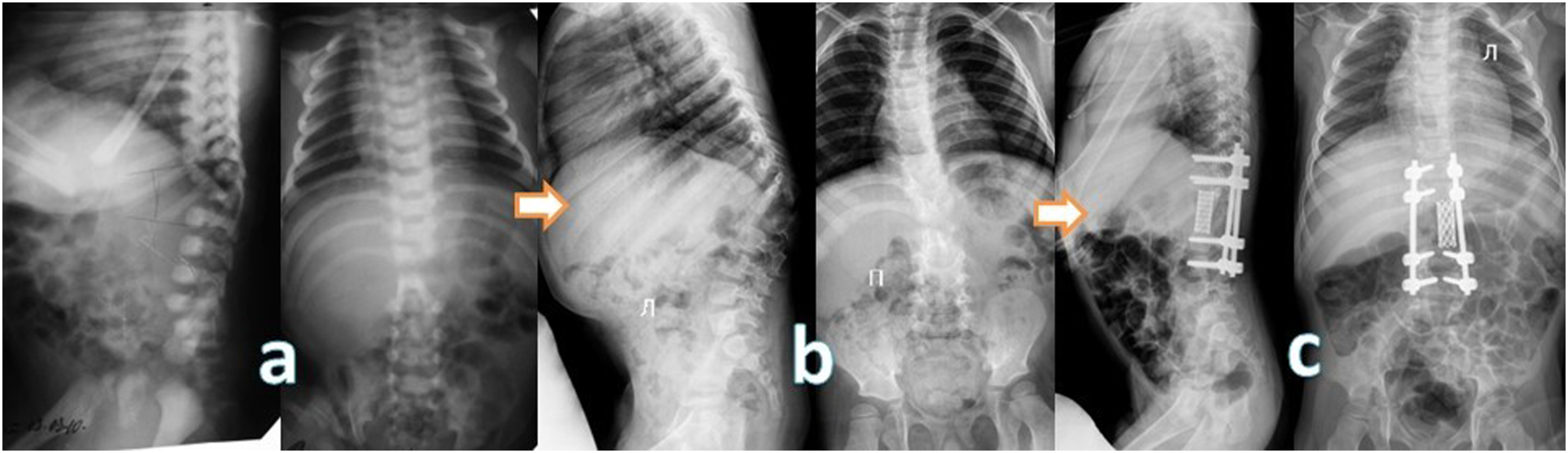
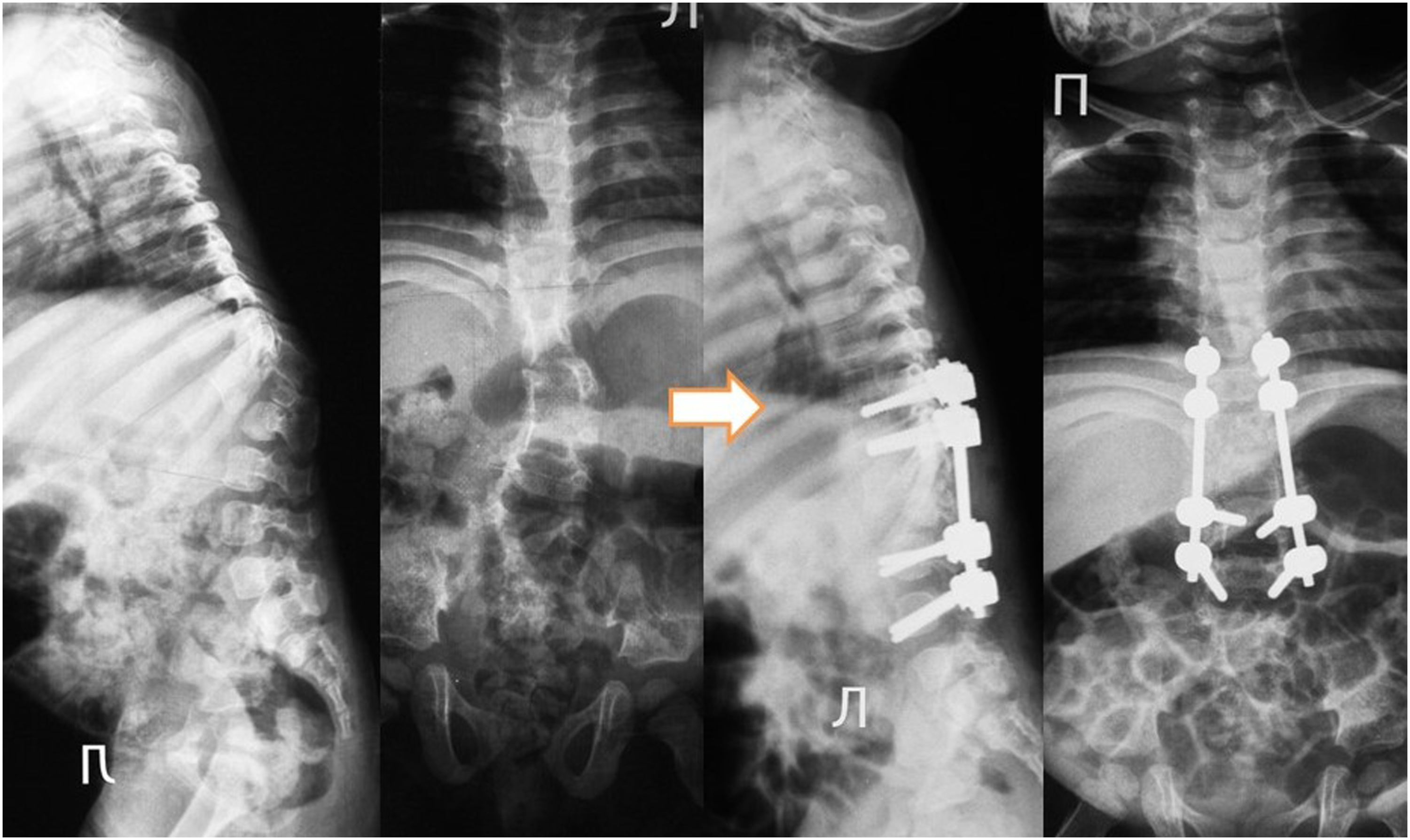
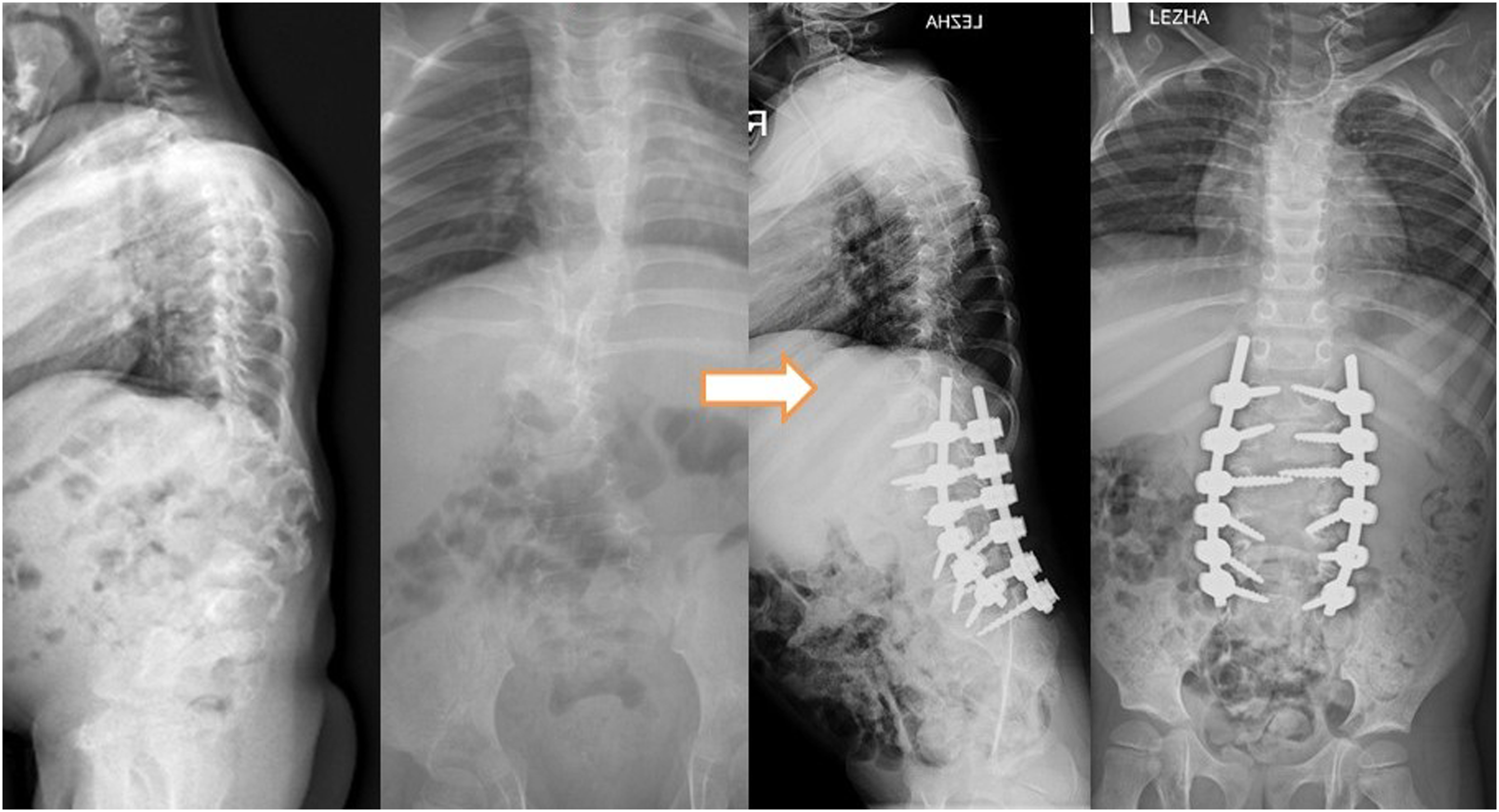
The amount of kyphosis correction in the second group was lower than in the other groups (Table 5).
Discussion
Understanding the Anatomical Variations
Various theories have been postulated to unravel the intricacies of SSA, each providing distinctive insights into the complex etiology of this condition: 1) junctional neurulation disorder4; 2) causal insult and positional apoptosis1 and 3) impaired blood supply. 3
Instances of SSA coexisting with open neural tube defects,8,9 as observed in our selection of patients (three cases), suggests that SSA might occur in conjunction with primary neurulation disorders. Yet, in these instances, the emergence of vertebral aplasia could also be attributed to the severity of the kyphotic deformity or disruptions in both innervation and blood supply.
The comprehensive analysis of SSA development proposes the existence of both mechanisms. Signs pointing to SSA evolving during neurulation or gastrulation include features such as a low-lying conus medullaris, spinal cord lipoma, myelomeningocele, multiple segmentation disorders, and associated malformations in other systems (e.g., tetralogy of Fallot). Conversely, isolated SSA may indicate a later development, potentially triggered by disruptions in segmental blood supply.
In summary, the concentration of SSA at specific junctions sparks an exploration into developmental theories, emphasizing the interplay between junctional neurulation disorders (similar to pathogenesis of open and closed neural tube defects) and ischemic necrosis of spinal segment with segmental resorption (similar to the pathogenesis of small intestinal atresia).
The study identifies two mechanisms leading to neurological symptoms in SSA: primary dysplasia of the spinal cord, nerve roots, and spinal vessels, and myelopathy due to compression of these structures. The presence of a dynamic component (compression) confirms a slowly progressive deterioration in the neurological status of patients. This dual mechanism underscores the multifaceted nature of neurological complications in SSA.
Clinical and Functional Assessment
Our privies studies note that SSA is often, though not necessarily, complicated by progressive worsening of neurological function due to mechanical instability of the spine.5,10-12 This highlights the clinical importance of addressing mechanical stability issues in the management of SSA to mitigate the risk of neurological deterioration. It is plausible to infer that scoliosis among SSA patients tends to advance with age, elucidating the heightened severity observed in patients beyond the age of 5 (Table 2). Conversely, the functional status appears to incrementally improve with age. This correlation suggests that as patients mature, there is a concurrent enhancement in their functional capabilities. Understanding these age-associated nuances is integral for informed treatment planning and underscores the dynamic nature of SSA throughout different developmental stages.
Conservative Treatment
Conservative treatment, such as bracing, presents itself as a viable option, especially for patients and parents who are averse to risks. This approach is particularly relevant given that many patients can walk independently, and surgical intervention poses a considerable risk of neurological deterioration.
Another circumstance where surgical treatment may be reconsidered is in patients with open neural tube defects, such as myelomeningocele and myeloschisis, coupled with paraplegia. In such cases, surgical intervention may not yield significant neurological improvements. However, it is likely to entail complications such as wound problems and a heightened risk of pressure sores around implanted screws, often necessitating subsequent revision surgeries.
Surgical Treatment Strategies
The surgical management of SSA involves a nuanced consideration of the level and extent of the anomaly. In cases where the SSA is localized to the cervicothoracic region, surgeons tend to employ specific approaches tailored to this unique anatomical challenge. Preoperative halo-traction in these cases has obvious advantages. This method could be employed to gradually correct deformities and stabilize the spine, providing a foundation for subsequent surgical interventions. 10
The decision to opt for pelvic fixation is primarily guided by specific clinical considerations, particularly the caudal location of the SSA and the severity of paraparesis. In cases where the defect is positioned caudally or if there is a presence of severe paraparesis, the likelihood of employing pelvic screw fixation increases.
Regarding the decision on whether to proceed with vertebrectomy, it is prudent to carefully weigh its implementation, as refraining from this procedure is associated with an elevated risk of pseudoarthrosis development.5,13
The determination to incorporate a mesh for interbody fusion typically arises intraoperatively, contingent on the identified defect in the soft tissue between the vertebral bodies. If a substantial defect is discerned, opting for a mesh becomes a preferred strategy to fill the void, reduce the risk of spinal cord kinking and achieve more optimal correction of kyphosis. Conversely, in cases where the defect is minor, the decision may lean towards forgoing mesh application.
Complications and Correlations
The substantial number of revision interventions (18 in 11 patients) indicates the complexity of managing late complications. Non-fusion emerged as the most common late complication (30% of patients), emphasizing the challenge of achieving successful fusion in this patient population.
A heightened frequency of complications in the early postoperative period is notably observed, 12 particularly in complex patients with cervicothoracic SSA. 10 This increased incidence is likely linked to the inherent complexities associated with this specific patient subgroup. Several factors contribute to this trend, including a higher level of myelopathy and the proximity of the anomaly to critical neural structures. The intricate nature of cervicothoracic aplasia, often requiring specialized and meticulous surgical approaches, may contribute to the observed complications in the early postoperative period. Our latest publication delves into a more comprehensive exploration of this specific patient subgroup, offering detailed insights into the complexities and nuances associated with cervicothoracic SSA. 10
Limitations of the Study
The study was conducted at a single medical center, potentially limiting the generalizability of the findings to other settings or patient populations. Variability in clinical practices, patient demographics, and treatment approaches across different centers may impact the applicability of the results.
The sample size of the patient cohort, comprising 36 individuals with SSA, may limit the statistical power of the analysis and the ability to detect subtle differences or associations. Larger sample sizes would enhance the robustness of the findings and allow for more comprehensive subgroup analyses. Addressing these limitations through multi-center collaborations, larger sample sizes would strengthen the validity and generalizability of future studies in this field.
Conclusions
SSA encompasses a spectrum of diverse anomalies typically manifesting in early childhood with a slow, progressive course. The preferred surgical approach involves vertebrectomy, followed by interbody fusion and fixation using transpedicular screws, guided by neurophysiological monitoring. Surgical intervention is warranted in cases of progressive neurological deterioration, although conservative management, including bracing, may be considered prior to surgery. However, given the substantial risk of complications conservative management is a viable option when patients and their parents decline surgical treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript. The authors declare no conflict of interest in this work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
