Abstract
Study Design
Cross-sectional international survey with literature review.
Objectives
To evaluate the evidence for these strategies and to understand the current trends in prophylactic antibiotic use during ASD surgery through an international survey.
Methods
An online survey was conducted among international AO Spine members regarding the peri-operative management of patients with ASD. The details of perioperative systemic and topical antibiotic use were solicited. Descriptive data were summarized for the responding surgeons who perform at least 10 long-segment fusions of >5 levels extending to the pelvis annually.
Results
The literature supports the use of prophylactic antibiotic effective against gram positive organisms. The use of topical vancomycin remains debated, and there is limited evidence for topical tobramycin use. A total of 116 responses were received. 74 (64%) surgeons use topical vancomycin, most usually deep to the fascia only 45 (61%). The most usual dose used is 1-2 g. 4 (3%) surgeons use topical tobramycin deep to fascia. Following surgery, 90 (78%) surgeons use prophylactic cephalosporin with 3 (3%) using cloxacillin, 5 (4%) using ciprofloxacin and 9 (8%) using vancomycin and 6 (5%) using clindamycin either in addition or alone.
Conclusions
The present survey identifies a trend towards systemic and topical antibiotic prophylaxis primarily targeted at gram positive pathogens. The use of topical tobramycin, proposedly effective against gram negative infections, remains uncommon. There is a lack of consensus in the selection of perioperative antibiotic prophylaxis, thus a prospective study of the rates of infection with each strategy would be useful to inform guidelines.
Introduction
The use of prophylactic antibiotics in complex spine surgery is ubiquitous, with the evidence supporting its efficacy in reducing surgical site infection (SSI) being largely above debate.1-10 Their use in adult spine deformity surgery is particularly essential, given the complex, extensive and high-risk nature of the surgery, and the multiple comorbidities common to the patient cohort, such as age, sarcopenia and frailty rendering patients vulnerable to SSI. This is reflected in guidelines offered to surgeons tackling these challenging procedures to mitigate the risks of infection in their practice. 11 Given that the majority of SSI are caused by commensal Gram-positive organisms, the evidence suggests that a parenteral first-generation cephalosporin (such as cefazolin) should be administered within 30 minutes of incision.7,11 These antibiotics show broad efficacy and good bone penetration, rendering them particularly suitable for use in musculoskeletal surgery. 12 Over recent years however, a debate regarding the use of antibiotic prophylaxis effective against gram negative bacteria has arisen, with some evidence showing that this strategy can reduce SSI by up to 50%; however the infrequent nature of these infections and the scarcity of gram negative infections has excluded them from guidelines. 13
The use of postoperative antibiotics is more controversial. The discussion is clouded by differing criteria for the diagnosis of SSI, the procedures carried out and the risk factors for the patients included in analyses. Nonetheless, as recently as 2021, Macki et al used registry data regarding 37,161 patients to show that there was no reduction in superficial SSI with the use of postoperative antibiotics, but perhaps a 45% reduction in deep SSI after 24 hours of postoperative antibiotic use, though only in patients with additional risk factors for infection. 14 In contrast, Orenday-Barraza et al conducted a meta-analysis of 13 studies encompassing both instrumented and uninstrumented procedures, showing that postoperative antibiotics fail to reduce the incidence of SSI regardless of their duration of use. 15 Sub-analysis showed that this relationship persisted when instrumented cases were assessed in isolation. These conclusions are supported elsewhere.16,17
A more recent development in antibiotic strategy has been the use of topical vancomycin and tobramycin in combatting gram negative bacteria. Vancomycin reaches peak concentration within a wound at 6 hours after use, and the theory goes that this will tackle any gram- positive contamination within the surgical bed. 18 Its use is supported by several meta-analyses, which show that it is efficacious and cost effective in reducing SSI.19-21 However, topical antibiotic use is not without complication, including delays to fusion and anaphylaxis.22,23 Tobramycin use has received far less attention, with animal studies and a case series suggesting that it may be effective in reducing deep SSI.24,25 As a result, the use of topical antibiotics remains varied across the spine surgery community.
The objective of the present study was to capture the current trend in surgeon preference for postoperative and topical antibiotic use during ASD surgery, and to review the current body of literature relevant to both, to inform ongoing practice and identify directions for subsequent inquiry.
Methods
A peri-operative spine survey was formulated by a study group within AO Spine. Institutional Review Board approval was not required. Each respondent granted informed consent within the survey questions. The study group included experts in the AO knowledge forum degenerative and AO knowledge forum deformity spine. The questionnaire included demographic information and was designed to collect data regarding the aspects of peri-operative care including wound management, antibiotic prophylaxis, bracing and activity instructions. Formulated as an online survey, the questionnaire was distributed via email to AO Spine users and members between March 3 and March 22, 2022. The survey was targeted at surgeons performing at least 10 cases per year of one or more of the following procedures: A. Long fusion (>5 levels) for adult spine deformity patients extending to pelvis B. Long fusion (>5 levels) for adult spine deformity patients NOT extending to pelvis C. Open 1 or 2 level fusion for adult lumbar degenerative pathologies D. MIS 1 or 2 level fusion for adult lumbar degenerative pathologies E. Open 3 to 5 level fusion for adult lumbar degenerative pathologies
It was estimated that over 6000 AO Spine users and members received the email. 354 responses were received, with 280 completing the survey. Of the surgeons who completed the survey, 164 performed adult spine deformity surgeries (procedures A and/or B) and 261 performed adult spinal degenerative surgeries (procedures C, D and/or E). Only data regarding procedures A or B were included in this analysis.
The data were analysed by underlying specialty (orthopaedic or neurosurgery), geographic location, intensity of and years in practice. Proportions were evaluated using Chi2 or Fisher’s exact tests. Median values were compared using the k-sample equality of median test. Data were recorded in Microsoft Excel v 16.32 (Microsoft, Redmont, USA) and analysed using Stata v.14.2 (StataCorp, College Station, USA). Significance was set at .05.
Results
Demographics of Survey Respondents.
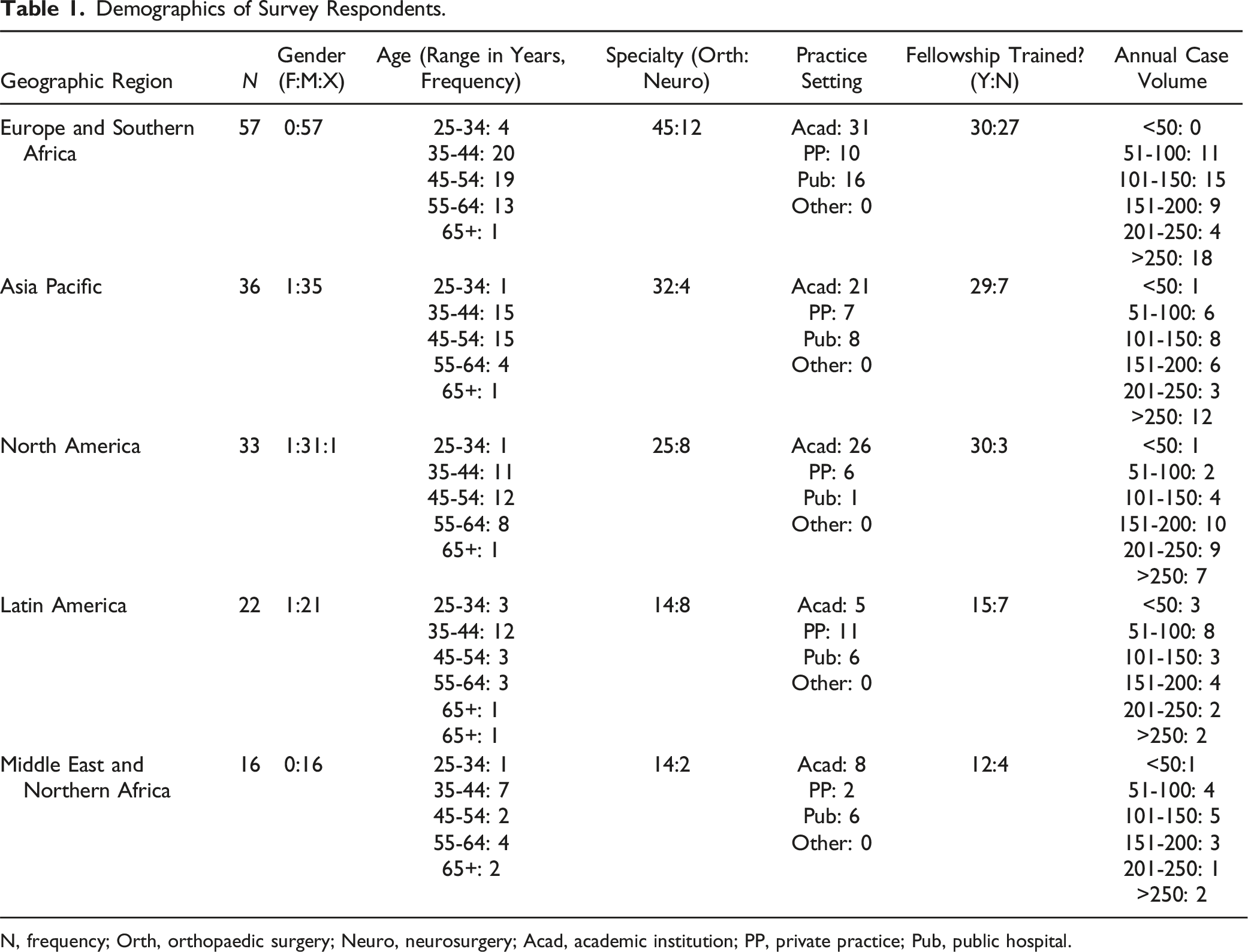
N, frequency; Orth, orthopaedic surgery; Neuro, neurosurgery; Acad, academic institution; PP, private practice; Pub, public hospital.
There was no regional variation in the selection of prophylactic parenteral antibiotic or the use of topical antibiotic. The Asia Pacific regional uses parenteral prophylactic antibiotics for a longer period than North America, Europe and the Middle East regions.
The Duration of Postoperative Antibiotic Use by Procedure.

The 48 surgeons carrying out procedure B, 1 did not use any postoperative parenteral antibiotics. Of the remaining 47, 44 use a cephalosporin, 1 reported cefotaxime (a third generation cephalosporin) use, 1 vancomycin and 1 cefazolin. Of the 44 using cephalosporin postoperatively, 5 use additional aminoglycosides, 4 additional clindamycin, 3 additional ciprofloxacin and 2 additional vancomycin.
The duration of post-operative antibiotics did not significantly differ by the number of procedures performed per year (.56), by the number of years in practice (P = .34) or by specialty (P = .45).
A greater proportion of neurosurgeons (91%) use topical vancomycin following adult spine deformity surgery than orthopaedic surgeons (70%) (P = .01). 74 (64%) surgeons who carry out procedure A use topical vancomycin, most usually deep to the fascia only (59%), with the remaining 41% using it superficial to the fascia alone. The most usual dose used is 1-2 g. In contrast, of the 48 surgeons carrying out procedure B, 19 (40%) use topical vancomycin, 15 (79%) of which use it deep to the fascia. The remainder use topical vancomycin superficial to the fascia. Ten surgeons use 1 g, the remaining 9 using between 1 and 2 g. There is no difference in the use of vancomycin by years in practice, however a greater number of cases per year correlates with a greater use of topical vancomycin (P = .014).
No surgeons carrying out procedure B use topical tobramycin during their procedures. Four (3%) surgeons who carry out procedure A use topical tobramycin. A single surgeon uses 1 g, 2 surgeons use 1-2 g and a single surgeon uses more than 2 g deep to the fascia. Two of these surgeons practice in North America, 1 in Latin America and 1 in Europe and Southern Africa.
Discussion
These data describe the current antibiotic practices of adult spine deformity surgeons across the globe and show that there is a lack of consensus in the duration and selection of postoperative parenteral antibiotics. Furthermore, there is substantial variation in the use of topical vancomycin, and a paucity of centers using topical tobramycin. The primary specialty correlates with the use of topical vancomycin.
These findings are reflected in the literature. Whilst preoperative prophylactic antibiotics are a fundamental component of reducing the risk of surgical site infection, particularly in the high risk adult spinal deformity patient, the evidence supporting postoperative antibiotic use is varied, reflected in the North American Spine Society guidelines published in 2013, which recommends the use of preoperative antibiotic prophylaxis effective against gram positive contamination, but offers no recommendations regarding postoperative use.2,11 More recent evidence has continued the debate. In 2020, Tan et al carried out a systematic review of 41 studies, showing that postoperative antibiotics does not lead to a reduction in SSI after uninstrumented spinal surgery, and notes that there is insufficient data to draw a similar conclusion after instrumented fusion. 16 These findings are supported by Orenday-Barraza et al, who in their 2022 meta-analysis of 13 reports were able to show that postoperative antibiotics were not useful in reducing SSI, regardless of the presence of instrumentation, and Abola et al who showed no benefit to the addition of 24 hours of postoperative antibiotics in over 4000 cases, 79% of which were instrumented.15,17
In contrast is work from Macki et al, who found some benefit to postoperative antibiotics in their analysis of a statewide database evaluating data regarding all procedures carried out for 37,161 cases of degenerative lumbar disease. 14 The authors found that deep SSI was reduced by between 40 and 45% with 24 h or more of post-operative antibiotic prophylaxis in patients with risk factors. Unfortunately, the weight of evidence seems to be contrary to this finding, and so a recommendation along these lines is yet to appear in any practice guidelines. Regardless, the duration of postoperative antibiotic use seems to be trending towards 24 h of cephalosporin. This is perhaps an extrapolation of the guidelines from the North American Spine Society, which recommends preoperative prophylaxis effective against gram positive bacteria. 11 Both Sasso et al in 2008 and Parchi et al in 2015 made similar recommendations, with their reviews suggesting that post-operative antibiotics not be continued beyond 24 hours if used at all; findings confirmed by Orenday-Barraza et al in 2022.12,15,26 These data here suggest that despite the weight of evidence being against their use, most surgeons are using postoperative antibiotics, with over 80% using it for 24 h or more.
Although increasingly recognized as being an effective strategy to reduce perioperative infection, the use of topical vancomycin has attracted controversy since its introduction in 2011.27,28 The hypothesized adverse effects include an increase in gram negative and polymicrobial infection, delayed union, and systemic reaction. Despite these risks, it would be reasonable to consider them as potentially valuable tool in the adult spine deformity patient, where the duration of the surgery, the extent of exposure and soft tissue injury, as well as comorbidities renders them at particularly high risk for infection.
The characteristics of infection after vancomycin use have been investigated in several studies. 29 Adogwa et al showed that in 34 infections from over 1200 cases treated with vancomycin powder to the surgical bed, the majority were found to be gram negative or polymicrobial in nature; far more than expected. 30 Xie et al expanded on this in their 2018 study of 2394 patients treated with either 1 or 2 g of topical vancomycin prior to wound closure. 31 The results of this study showed that the use of 1 g of vancomycin predisposed patients to a higher risk of Gram-negative infection, in contrast to 2 g where Gram-positive infection was more often seen.
These ideas were furthered by Gande et al, who showed in their 2019 meta-analysis that gram negative and polymicrobial infections occurred more than twice as often in patients treated with topical vancomycin than in those who did not. 32 Data from Grabel et al supports this, showing that over 23% of deep infections after the application of topical vancomycin are gram negative or polymicrobial in nature, and Li et al show that its use confers no advantage in the prevention of gram negative infection.33,34 The mechanism for this finding is not clear given the effectiveness of vancomycin against gram negative infection, and remains to be convincingly elucidated. 35 Recent study by Rocos et al implicates local contamination of the wound from fecal and urine sources in 65% of deep infections following lumbosacral and lumbo-pelvic fusions, negating any potential benefit offered by local or systemic antibiotics, placing the focus on prevention through barrier dressings and appropriate wound care. 36 The potential for devastating consequences of SSI in the adult spine deformity patient lends weight to the criticality of determining the value of topical antibiotics for this at-risk cohort. Prospective studies evaluating their effectiveness and adverse effects, particularly in terms of long term consequences and acute adverse events will be essential in determining its usefulness for adult spine deformity surgery into the next decade.
The addition of topical tobramycin was initially hypothesized to reduce the incidence of Gram-negative infection. Unfortunately, its use remains incompletely evidenced, and initial reports seem to show no additional benefit to topical vancomycin; though prospective work remains absent. 24
The effect of topical vancomycin on union is poorly investigated. 37 Guimbard-Pérez et al suggested that the effect was real in a study of rabbit spines, however this was refuted by Kim et al.22,38 Given that its use across adult spine deformity surgery is extensive, it appears that surgeons remain unconvinced by this hypothesized adverse effect. At present, robust evidence in a human population after spinal surgery remains to be produced.
Adverse reactions to topical vancomycin are uncommon. First described in 2013, the incidence of allergy to topical vancomycin is undefined, but it appears to be an infrequent occurrence.23,39 Other adverse effects described have included seroma formation, nephrotoxicity and ototoxicity, at an incidence of less than .3% in a series of over 9200 patients, suggesting that its use is generally safe.40,41
Neurosurgeons and those with a higher number of cases per year appear to use vancomycin more than orthopaedic surgeons or those surgeons carrying out fewer procedures. At this stage, it is unfeasible to correlate these results to outcomes, however it may be that there are cultural or regulatory differences that have a significant effect on patient recovery.
This study is limited by the number of respondents to the survey, and the varied nature of their practice, the national guidelines that each may be adhering to and the formulary available to them. Though a response rate to the survey cannot be calculated, as the number of surgeons who perform eligible procedures is unknown, only 354 of 6000 who received the survey responded, which may have influenced the results and the inferences that can be drawn. Despite this limitation, the results are consistent with clinical practice and demonstrate that the controversy over antibiotic use in spinal deformity exists throughout the world, highlighting the need for more robust prospective multicenter studies. In addition, the survey did not collect data regarding the transition from parenteral to oral postoperative antibiotics, including the dosage and duration of any used. Later studies should consider this in collecting data used to draw clinical recommendations.
Conclusion
The evidence regarding the use of postoperative antibiotics remains of insufficient quality to make a definitive recommendation regarding their selection and duration. The results of this survey describe level 5 evidence for 24 h of postoperative antibiotics accompanied with the use of topical vancomycin. To date this is the first consensus statement for antibiotic prophylaxis within this complex surgery, and thus could be considered a moderate strength recommendation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BR is a Consultant for Silony Medical GmbH. SK declares no conflict of interest. SJL is a Consultant for Stryker, Medtronic and AOSpine, and receives payment for lectures and travel for meetings from Medtronic, Stryker, AOSpine, and Depuy Synthes.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was organized and funded by AO Spine through the AO Spine Knowledge Forum Deformity, a focused group of international spine deformity experts. AO Spine is a clinical division of the AO Foundation, which is an independent medically guided not-for-profit organization. Study support was provided directly through the AO Spine Research Department. The National Institute for Health and the International Spine Study Group, the Scoliosis Research Society, Broadwater, Scoliosis Research Society and the Seattle Science Foundation.
