Abstract
Study Design
Systematic Review and Meta Analysis
Objective
This study sought to compare patient-reported outcomes, success, complications, and radiographic outcomes directly and indirectly between different cervical total disc arthroplasty (TDA) devices and anterior cervical discectomy and fusion (ACDF).
Methods
Patients of prospective randomized controlled trials of 1-level cervical TDA with a minimum of 2 years follow up were identified in the literature. A frequentist network meta-analysis model was used to compare each outcome across the different TDA devices included and ACDF using the mixed effect sizes.
Results
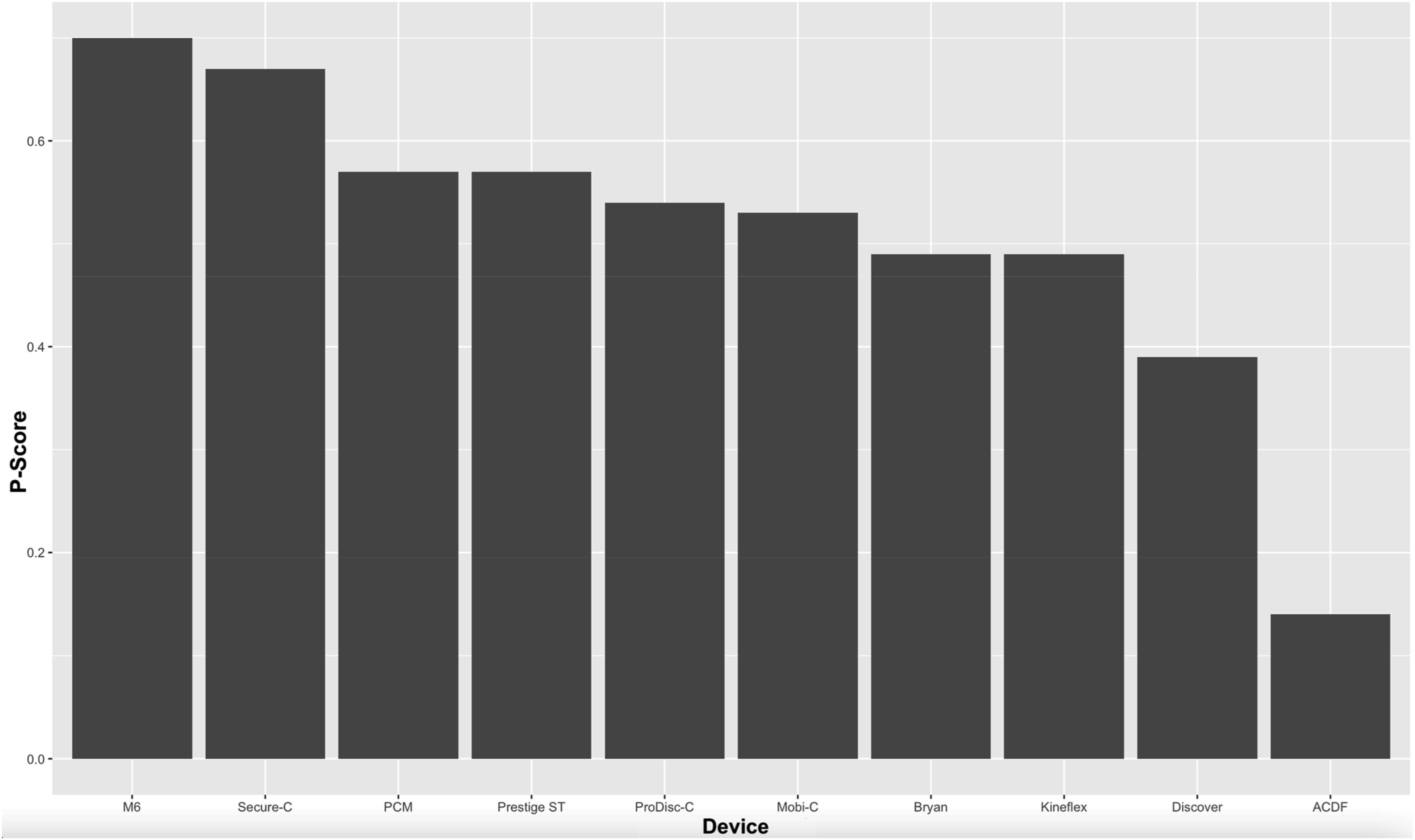
15 studies were included for quantitative analysis, reporting the outcomes of 2643 patients with an average follow-up was 67.3 months (range: 24-120 months), 1417 of whom underwent TDA and 1226 of whom underwent ACDF. Nine TDA devices were compared to ACDF, including the Bryan, Discover, Kineflex, M6, Mobi-C, PCM, Prestige ST, ProDisc-C, and Secure-C cervical prostheses. Several devices outperformed ACDF for certain outcomes, including Visual Analog Scale (VAS) Arm, Physical Component Score of the Short-Form Health Survey (SF PCS), neurological success, satisfaction, index-level secondary surgical interventions (SSI), and adjacent level surgeries. Cumulative ranking of each intervention assessed demonstrated the highest performance with the M6 prosthesis (P = .70), followed by Secure-C (P = .67), PCM (P = .57), Prestige ST (P = .57), ProDisc-C (P = .54), Mobi-C (P = .53), Bryan (P = .49), Kineflex (P = .49), Discover (P = .39), and ACDF (P = .14).
Conclusion
Cervical TDA was found to be superior on most outcomes assessed in the literature of high-quality clinical trials. While most devices demonstrated similar outcomes, certain prostheses such as the M6 were found to outperform others across several outcomes assessed. These findings suggest that the restoration of near-normal cervical kinematics may lead to improved outcomes.
Introduction
Cervical total disc arthroplasty (TDA) has proven to be an efficacious treatment option for patients with radiculopathy and/or myelopathy who have failed conservative management.1,2 Additionally, it has been associated with lower complication rates as compared to anterior cervical discectomy and fusion (ACDF).3,4 Given its motion-preserving qualities and preferable complication profile, its use has been increasing over several years in patients meeting criteria for arthroplasty.5,6
As TDA aims to restore physiologic motion and load sharing of the diseased level, its effectiveness can be reliant on both patient and device-specific factors. As each device has a unique design, they offer distinct mechanical properties that may impact clinical outcomes. 7 Randomized controlled trials (RCT) of multiple prostheses have demonstrated significant improvement in patient-reported outcome metrics (PROMs) with long-term follow up.1,8-16 However, inter-device performance comparisons are currently rare in the literature.
Given the increasing incidence of TDA use in patients with cervical pathology, along with the abundance of devices available on the market, performance analysis between devices has become of increased interest and importance. This study aims to assess the impact of prosthesis design on clinical outcomes relative to ACDF, based on the current body of high-quality RCTs. Improved understanding of device design and performance will empower surgeons to navigate the myriad factors that may currently influence surgical indications and device selection.
Methods
Search Strategy
The methodology of this network meta-analysis adhered strictly to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. A search of the PubMed and MEDLINE databases was performed in 2020 to identify all published English language studies that pertained to cervical TDA. The terms utilized for the search were “total disc arthroplasty”, “total disc replacement”, “cervical disc arthroplasty”, and “cervical disc replacement”. The reference lists of all relevant studies were also reviewed to identify any additional studies that may not have been captured by the initial query.
Study Eligibility and Data Extraction
Studies were included in the network meta-analysis if they met the following inclusion criteria: (1) the study design was a RCT comparing cervical TDA to another intervention, (2) included patients who underwent index surgery at a single motion segment, (3) included TDA patients who all underwent surgery with a single device, (4) allocated a minimum of 45 patients to each treatment arm, (5) obtained a minimum average follow-up of 2 years, and (6) reported 1 or more outcome of interest to this network meta-analysis determined a priori. Exclusion criteria were: (1) early results of clinical trials that later reported further follow-up, and (2) studies reporting the same outcomes on the same patient cohort also described elsewhere (ie, results from a single institution participating in a multicenter trial). The same inclusion and exclusion criteria were maintained for all studies and for all interventions assessed.
Following the initial search, the titles and abstracts of all identified studies were reviewed independently by 2 authors for relevancy to the subject of interest. The full texts of potential studies were then screened by the same 2 authors and studies for which a consensus on eligibility was achieved were then included for data extraction. If a consensus was not reached, a third author reviewed the full text to make the final decision on inclusion. Once the included studies were identified, the same 2 authors extracted data on predetermined variables of interest.
Outcomes Assessment
The outcomes of interest were selected based on common variables frequently reported in the device-specific Food and Drug Administration (FDA) Investigational Device Exemption (IDE) trials. PROMs included the Neck Disability Index (NDI), Visual Analog Scale (VAS) for neck and arm pain, and Physical Component Score of the Short-Form Health Survey (SF PCS). Additionally, data on overall procedural success, neurological success, patient satisfaction, postoperative dysphagia, device or procedure-related adverse events, index-level secondary surgical interventions (SSI), and adjacent segment surgeries were collected. Radiographic data included were postoperative segmental range of motion (ROM), cervical (C2-C7) ROM, and heterotopic ossification (HO) in TDA or successful fusion following ACDF. The summary of safety and effectiveness data (SSED) reports for the device FDA IDE trials were also searched to supplement data points missing from the manuscripts.
Statistical Analysis
Unlike a standard pairwise meta-analysis, which compares 2 competing treatments directly compared by the pooled studies, the network meta-analysis statistical method allows for the comparison of multiple treatments if there is a common comparator, which in this case was the control group (ACDF). A frequentist approach to network meta-analysis was performed to directly and indirectly compare the performance of individual TDA devices and ACDF with respect to the previously specified outcome measures. All statistical methods for the network meta-analysis were performed utilizing the netmeta package for R Studio version 2021.09.2 build 382 (R Core Team, Vienna Austria). The effect size (ES) and standard error (SE) of each outcome was computed for each of the included studies. For continuous variables, the ES was the mean difference (MD) between the 2 treatments of interest, while the log odds ratio (OR) was used for categorical data. Network meta-analysis was utilized to compute effect size estimates for treatment comparisons by pooling the effect sizes under a random-effects model. A separate analysis was performed for each outcome assessed, and effect sizes were reported with the corresponding 95% confidence interval (95% CI). For both MD and log OR effect size comparisons, a statistically significant difference was established if the corresponding 95% CI did not include 0. The Q statistic was used to assess the total heterogeneity within the network.
Forest plots were constructed to graphically represent the comparison of TDA devices across all the outcomes assessed with ACDF as the reference intervention. The efficacy of the competing treatments in the network was ranked by computing the P-Score, a representative linear score of the certainty that the given intervention is superior to others in the network. 17 A P-Score was generated for each outcome assessed, then the average P-Score was calculated to create a cumulative rank of the efficacy of each treatment under investigation in the network while weighting each variable equally. When calculating the average P-Score for ACDF, segmental ROM and the presence of bridging bone were excluded from the equation since, unlike TDA, the goal of the procedure is to create a robust fusion mass, thus obviating ROM at the motion segment.
Risk of Bias Assessment
Risk of bias was assessed in accordance with the Cochrane Back and Neck Group guidelines for included studies Figure 1.
18
Overall, the RCTs included for quantitative analysis demonstrated a low risk of bias in most criteria analyzed. High-risk for bias was primarily attributed to the lack of blinding of the care provider or the absence of an intention-to-treat analysis. Risk of bias assessment for the included studies.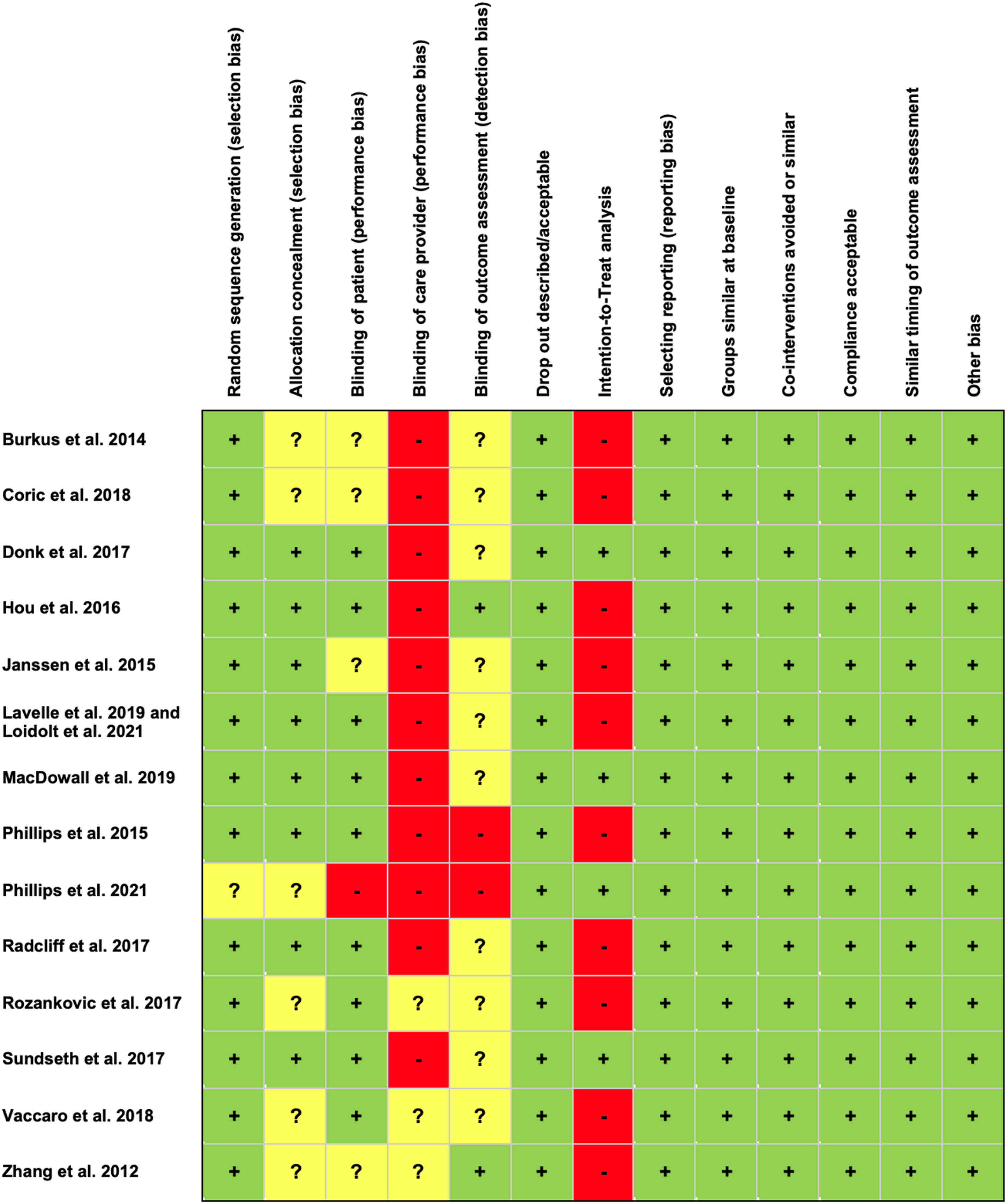
Results
Search Results and Study Characteristics
A total of 2310 studies were identified, which after filtering for duplicates, resulted in a total of 2307 studies for screening. After the title and abstract screen, 186 studies were identified for full-text review. A total of 15 studies satisfied the inclusion criteria and were selected for data extraction.1,8-11,13–16,19-24 The search strategy is summarized by Figure 2 PRISMA flow chart summarizing the search strategy.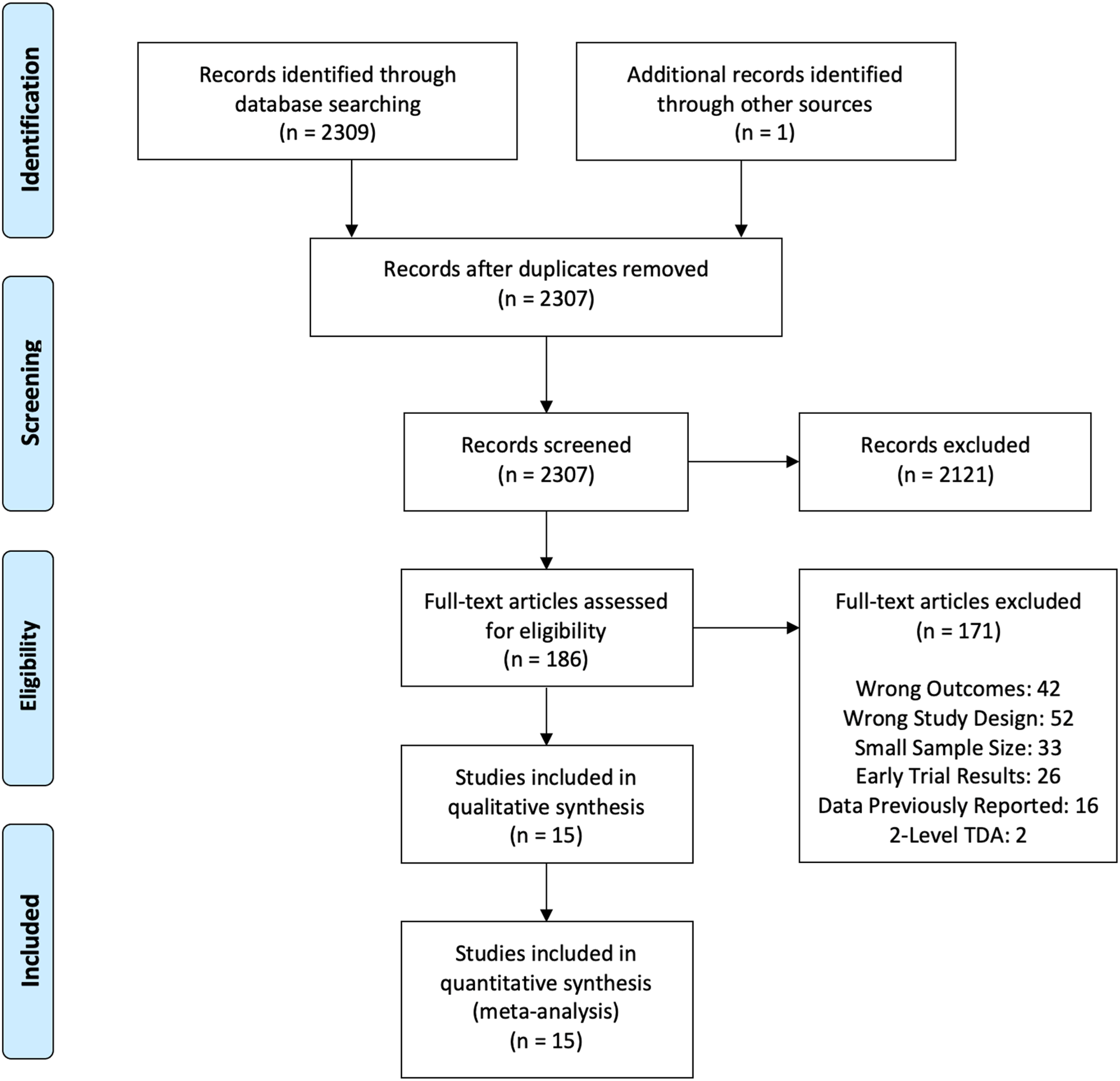
Summary Data of Studies Included for Quantitative Analysis.
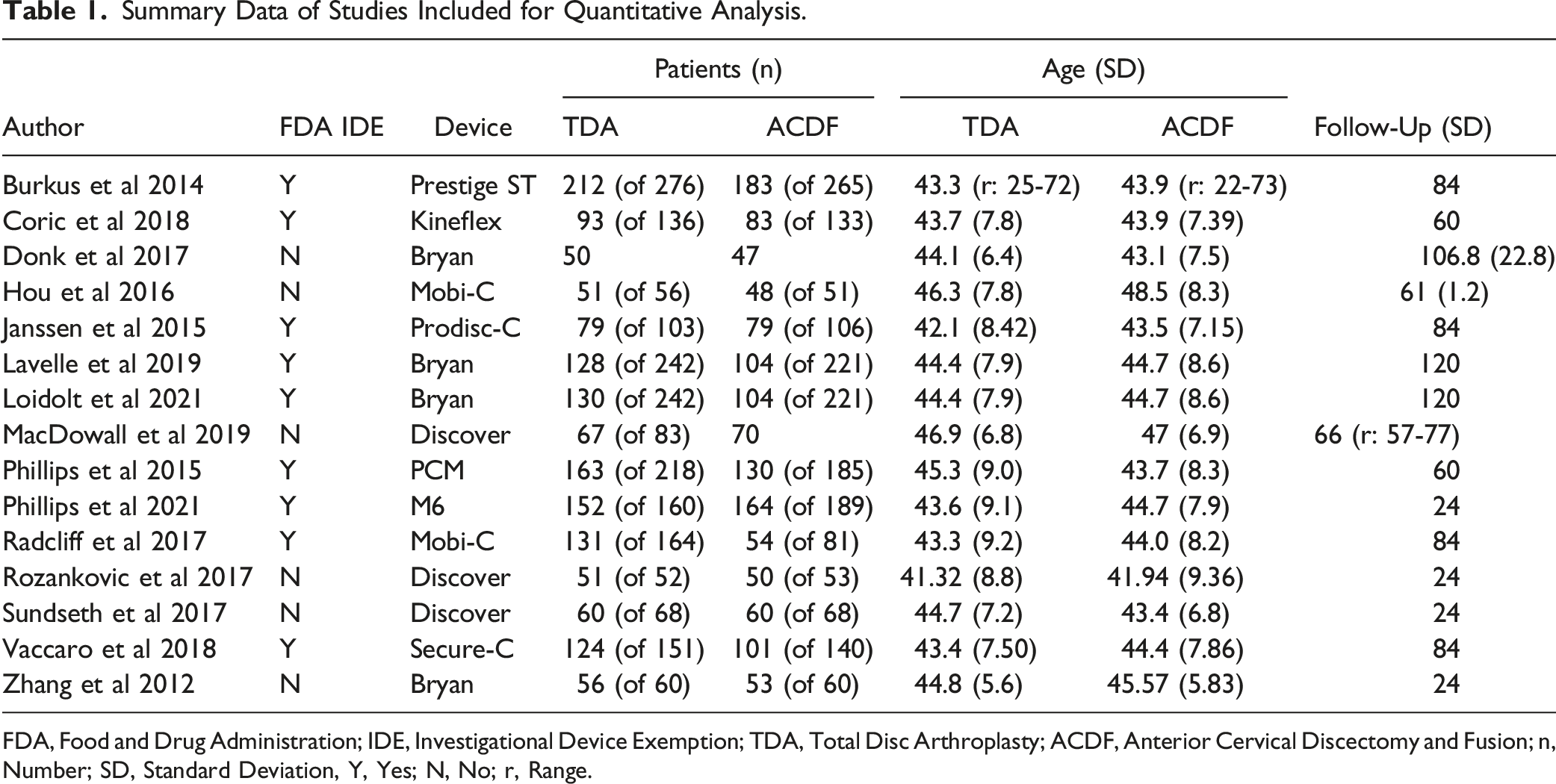
FDA, Food and Drug Administration; IDE, Investigational Device Exemption; TDA, Total Disc Arthroplasty; ACDF, Anterior Cervical Discectomy and Fusion; n, Number; SD, Standard Deviation, Y, Yes; N, No; r, Range.
Summary of Inclusion and Exclusion Criteria Upheld by All Included Trials and Definitions of Overall and Neurological Success.

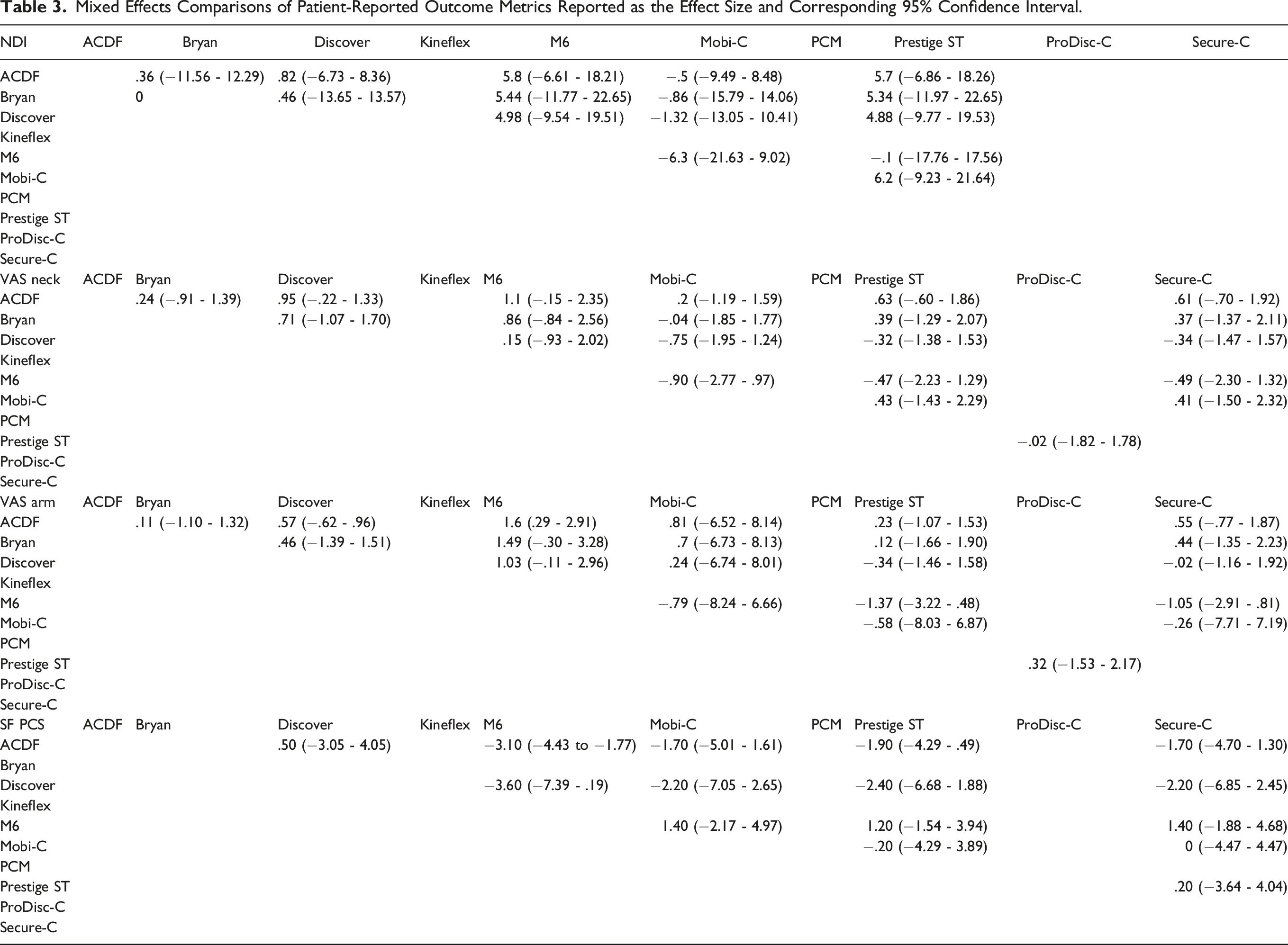
Mixed Effects Comparisons of Patient-Reported Outcome Metrics Reported as the Effect Size and Corresponding 95% Confidence Interval.
Patient-Reported Outcome Metrics
Neck Disability Index
Eight studies1,8,11,19-22,24 comparing 5 cervical TDA devices (Bryan, Discover, M6, Mobi-C, Prestige ST) to ACDF contained sufficient data for analysis (Figure 3). Direct comparison of the effect size estimates (Table 4) demonstrated that no single TDA device significantly outperformed ACDF for reducing neck disability. Indirect comparison between devices found similar results across all paired comparisons. Forest plots demonstrating the MD and 95% CI of each device compared to ACDF as the reference for (A) NDI, (B) VAS Neck, (C) VAS Arm, and (D) SF PCS. Mixed Effects Comparisons of Patient Success and Satisfaction Reported as the Effect Size and Corresponding 95% Confidence Interval.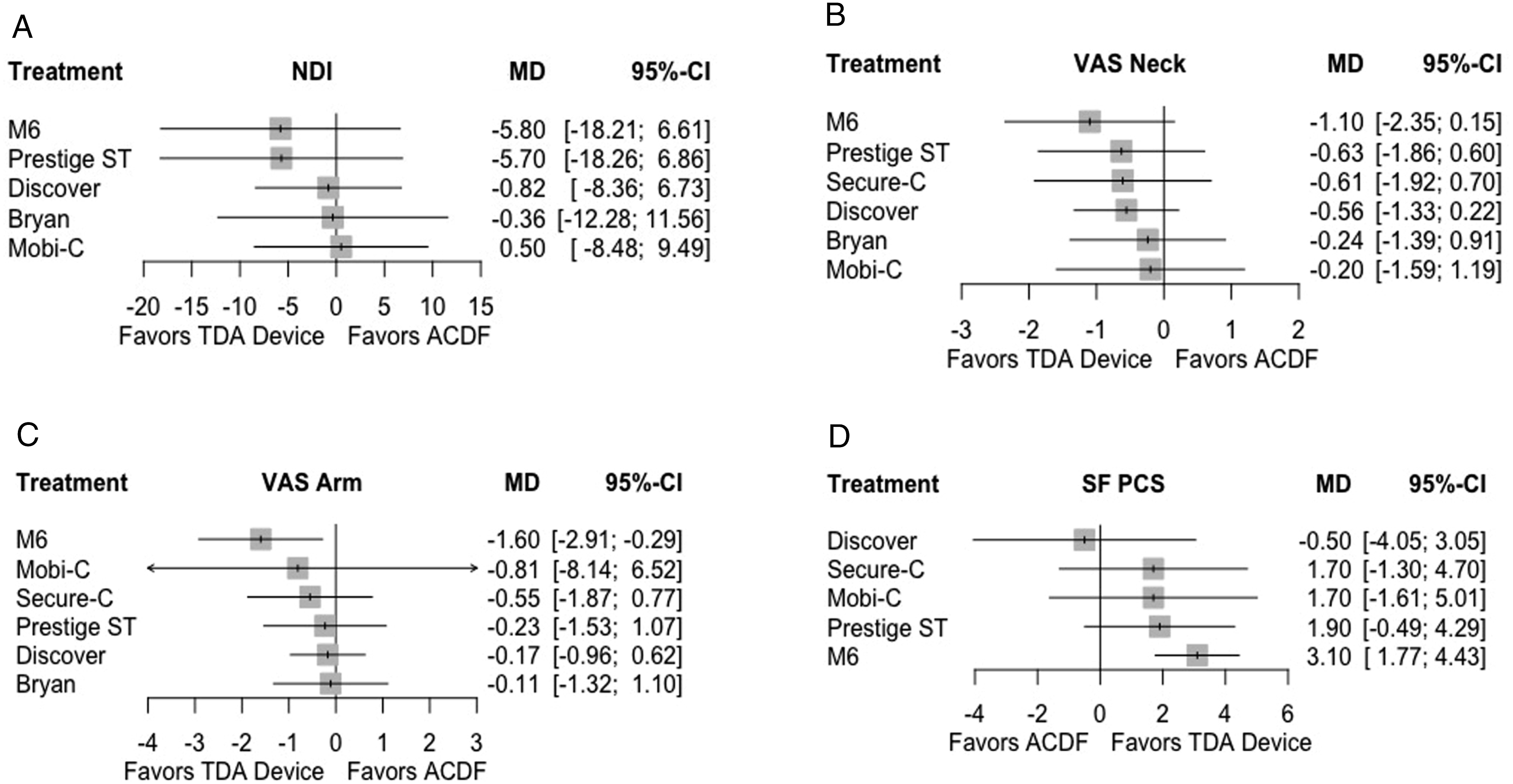

Visual Analog Scale Neck and Arm Pain
Eight studies1,8,19–24 comparing 6 TDA devices (Bryan, Discover, M6, Mobi-C, Prestige ST, Secure-C) to ACDF contained sufficient data for analysis of neck and arm pain. For VAS Neck, direct and indirect comparisons demonstrated similar outcomes between all TDA devices and ACDF. For VAS Arm, the M6 device performed significantly better than ACDF in reducing arm pain (MD: 1.6, 95% CI: .29 - 2.91). No other significant differences were noted in direct or indirect comparisons.
Physical Component Score of the Short-Form Health Survey
Five studies1,8,20,22,23 comparing 5 TDA devices (Discover, M6, Mobi-C, Prestige ST, Secure-C) to ACDF contained sufficient data for analysis. The M6 device performed significantly better than ACDF in improving physical health status (MD: −3.10, 95% CI: −4.43 to −1.77). No other significant differences were noted in direct or indirect comparisons.
Success and Satisfaction
Overall Success
Six studies1,8,9,14,20,23 comparing 6 TDA devices (Bryan, Kineflex, M6, Mobi-C, Prestige ST, Secure-C) to ACDF were included for analysis (Figure 4). Both direct and indirect comparisons demonstrated similar achievement of overall success between all TDA devices and ACDF (Table 5). Forest plots demonstrating the MD or log OR and 95% CI of each device compared to ACDF as the reference for (A) overall success, (B) neurological success, (C) categorical satisfaction, and (D) VAS satisfaction. Mixed Effects Comparisons of Adverse Events and Subsequent Surgeries Reported as the Effect Size and Corresponding 95% Confidence Interval.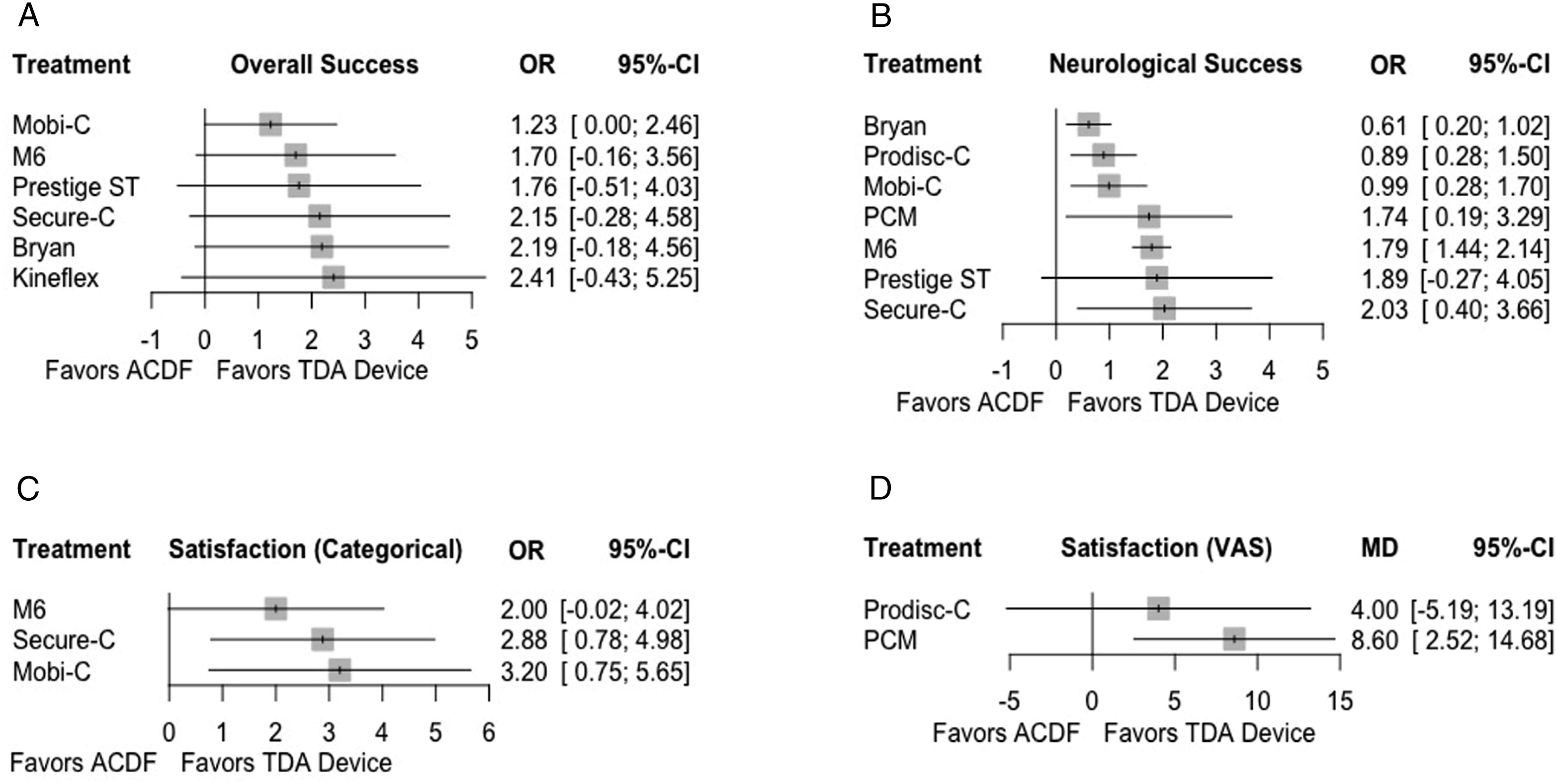
Neurological Success
Seven studies1,8,13,14,16,20,23 comparing 7 TDA devices (Bryan, M6, Mobi-C, PCM, Prestige ST, ProDisc-C, Secure-C) to ACDF were included for analysis. Direct comparison demonstrated that all devices except for the Prestige ST performed significantly better than ACDF in achieving neurological success. Additionally, indirect comparison between devices found that the M6 outperformed the Bryan, Mobi-C, and ProDisc-C prostheses.
Satisfaction
Patient satisfaction was compared against ACDF categorically by 3 studies1,20,23 for 3 devices (M6, Mobi-C, Secure-C) and with a VAS Satisfaction scale by 2 studies13,16 for 2 devices (PCM, ProDisc-C). For categorical satisfaction, both Mobi-C and Secure-C significantly outperformed ACDF. For VAS satisfaction, only PCM performed significantly better than ACDF. Indirect comparisons between devices found similar results across all paired comparisons for both categorical and VAS satisfaction.
Adverse Events and Subsequent Surgery
Dysphagia
Four studies1,8,15,22 comparing categorical dysphagia for 3 TDA devices (Bryan, M6, Prestige ST) to ACDF were included for analysis (Figure 5). Direct comparison demonstrated that the M6 device had a significantly lower association with dysphagia when compared to ACDF (Table 6). Additionally, comparison between devices found that the M6 device performed significantly better than the Bryan prosthesis. Forest plots demonstrating the log OR and 95% CI of each device compared to ACDF as the reference for (A) dysphagia, (B) adverse events, (C) index level SSI, and (D) adjacent segment surgeries. Mixed Effects Comparisons of Radiographic Outcomes Reported as the Effect Size and Corresponding 95% Confidence Interval.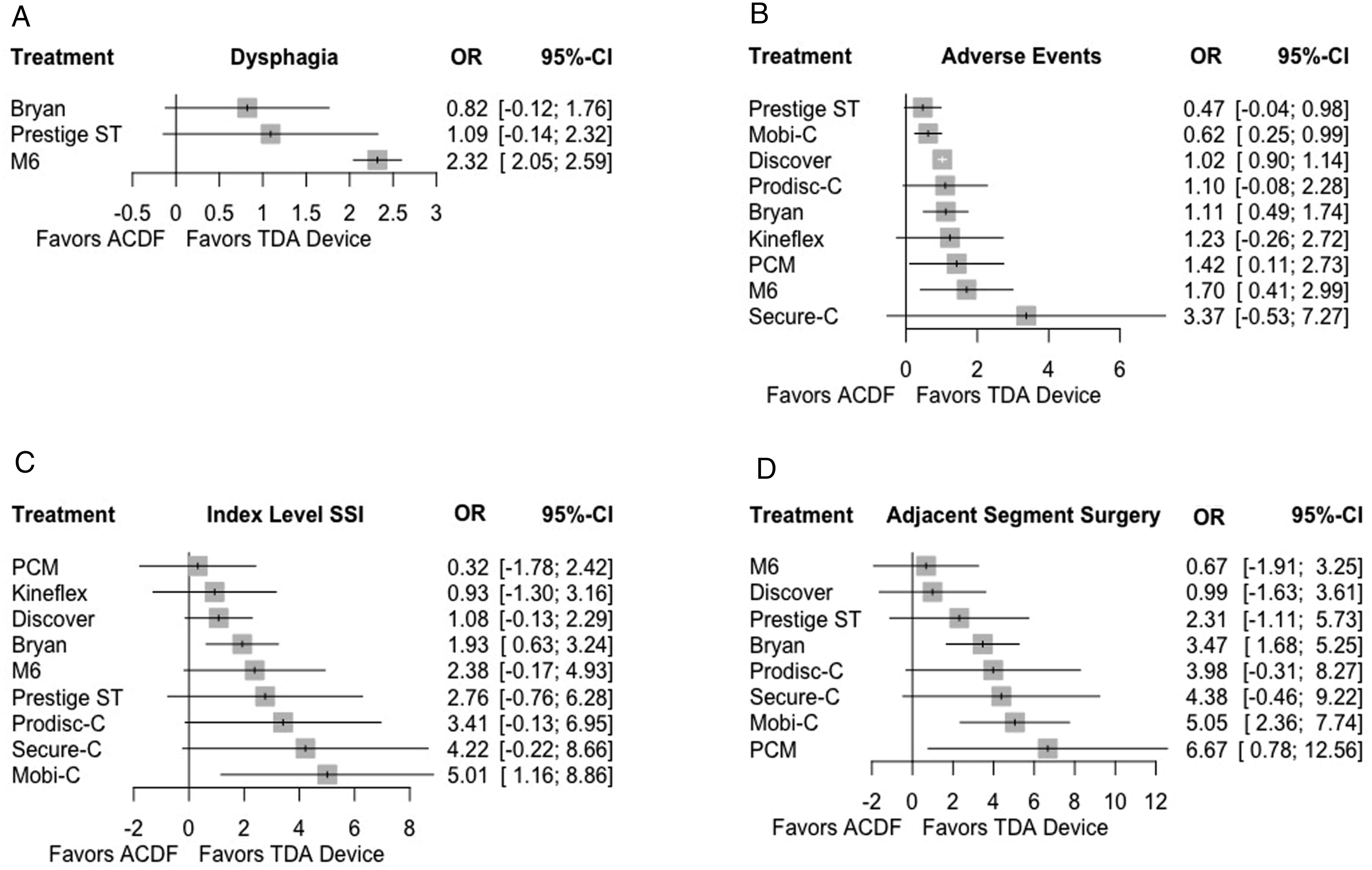
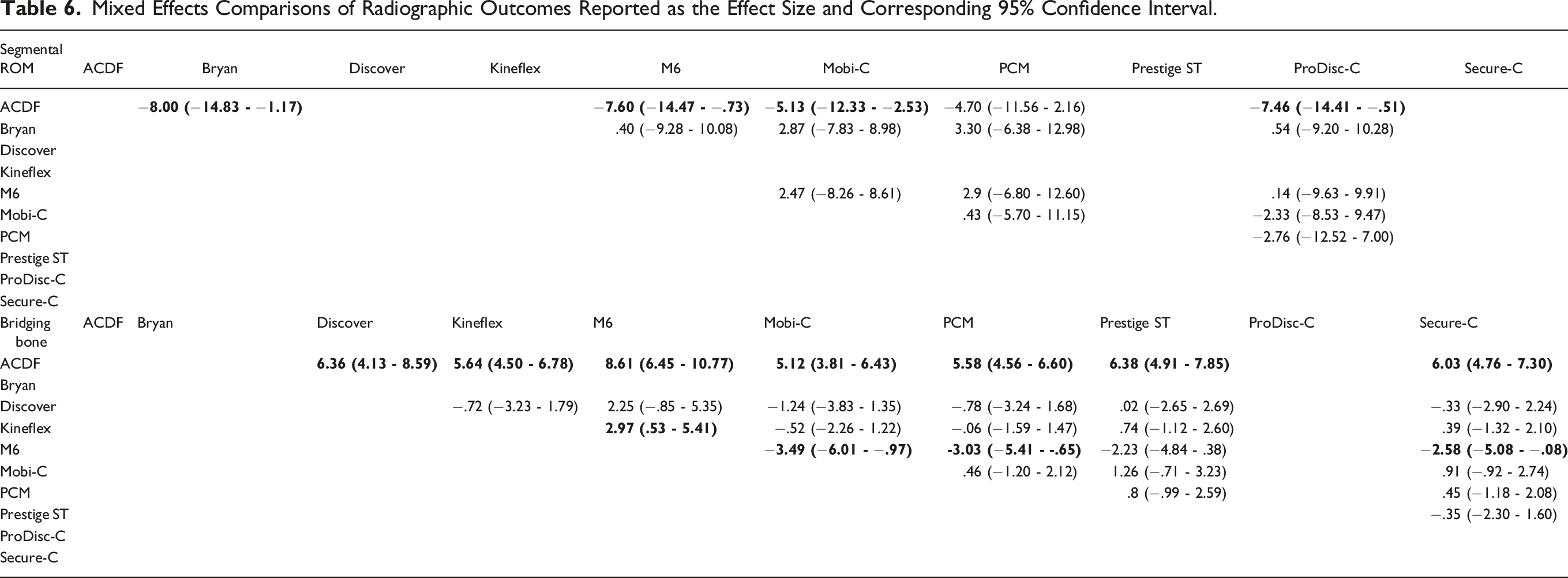
Adverse Events
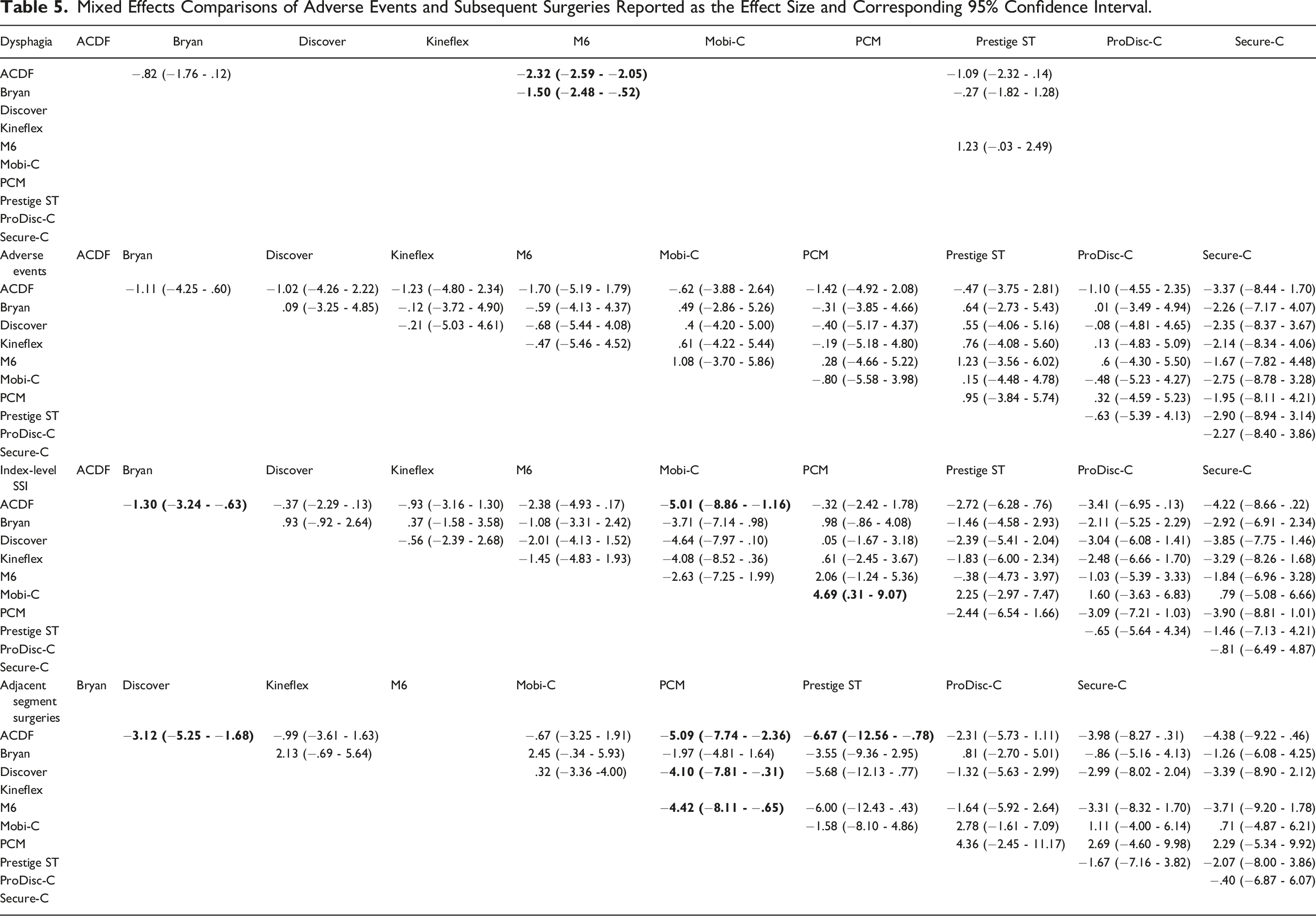
Ten studies1,8-10,13,15,16,20,21,23 compared device or surgery related adverse event profiles for 9 TDA devices (Bryan, Discover, Kineflex, M6, Mobi-C, PCM, Prestige ST, ProDisc-C, Secure-C) to ACDF. Both direct and indirect comparisons between all TDA devices and ACDF demonstrated similar associations with adverse events.
Index-Level Secondary Surgical Intervention
Fourteen studies1,8–11,13,15,16,19-24 compared the incidence of index-level SSI in 9 TDA devices (Bryan, Discover, Kineflex, M6, Mobi-C, PCM, Prestige ST, ProDisc-C, Secure-C) to ACDF. Direct comparison to ACDF demonstrated that the Bryan and Mobi-C devices were associated with significantly fewer surgeries at the index operative level. Additionally, when indirectly comparing between TDA devices, Mobi-C was significantly associated with fewer subsequent index-level surgeries than the PCM disc.
Adjacent Segment Surgery
Thirteen studies1,8,10,11,13,15,16,19-24 compared the incidence of adjacent level surgery in 9 TDA devices (Bryan, Discover, M6, Mobi-C, PCM, Prestige ST, ProDisc-C, Secure-C) to ACDF and were included for analysis. When compared to ACDF, a significantly lower association with adjacent segment surgery was seen with the Bryan, Mobi-C, and PCM devices. Additionally, indirect comparison between devices demonstrated that the Mobi-C device was associated with fewer adjacent segment surgeries than both the Discover and M6 prostheses.
Radiographic Outcomes
Segmental Range of Motion
Six studies1,11,13,16,20,24 provided sufficient data when comparing segmental ROM in 5 TDA devices (Bryan, M6, Mobi-C, PCM, ProDisc-C) to ACDF (Figure 6). The direct comparison between TDA devices and ACDF was performed as a precursor for indirect analysis between devices. Direct comparison between devices and ACDF demonstrated greater segmental ROM for all devices assessed (Table 7). This difference was significant for all devices except PCM. Indirect comparison between devices found similar results across all paired comparisons. Forest plots demonstrating the MD or log OR and 95% CI of each device compared to ACDF as the reference for (A) segmental ROM and (B) bridging bone across the operative segment. Effect Size Estimate Comparisons of Radiographic Outcomes Reported as the Log Odds Ratio (Bridging Bone) or Mean Difference (Segmental ROM) and Corresponding 95% Confidence Interval. aReported as the Mean Difference (95% Confidence Interval). bReported as the Log Odds Ratio (95% Confidence Interval).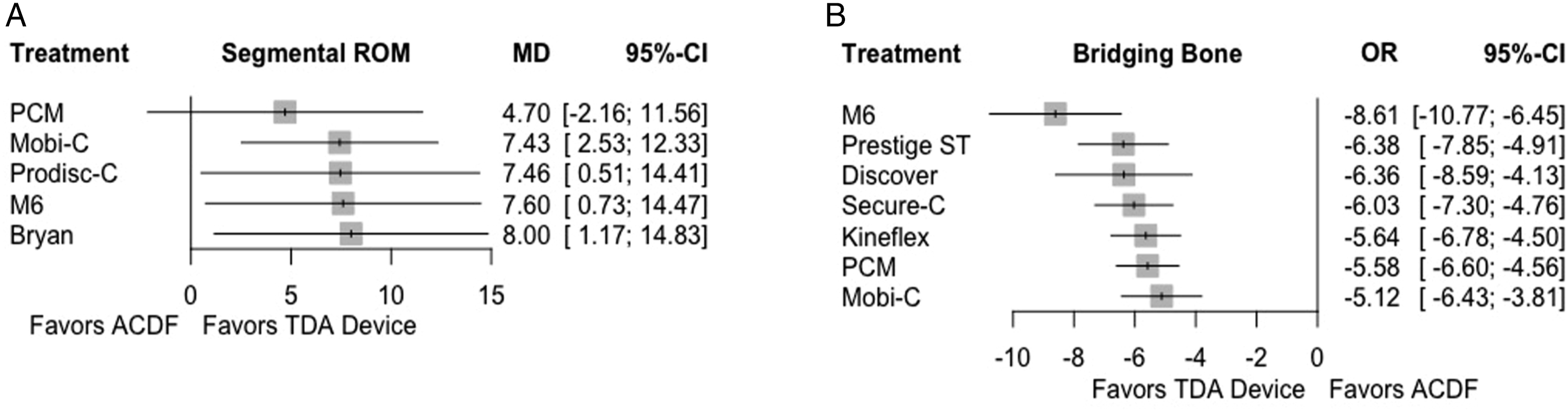
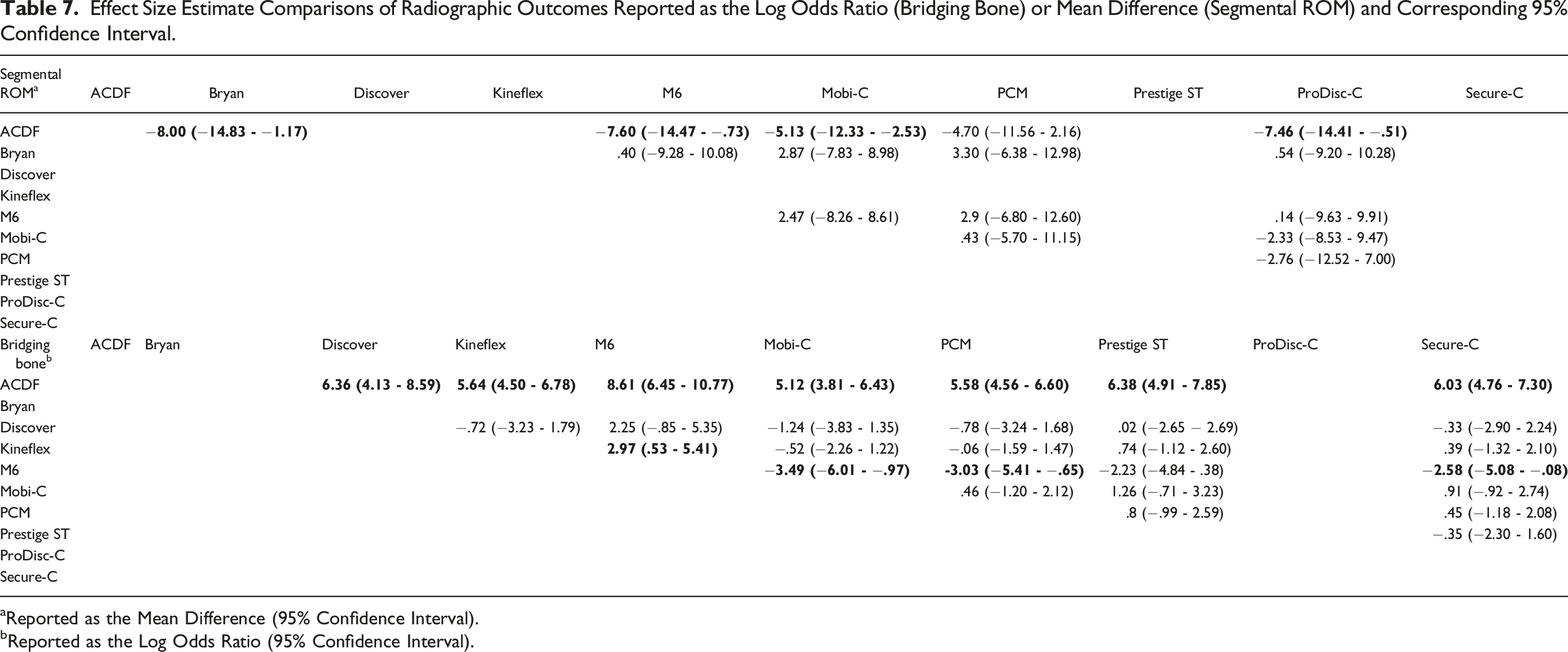
Bridging Bone
Seven studies1,8,9,16,20,21,23 reported the incidence of bridging bone secondary to HO in 7 TDA devices (Discover, Kineflex, M6, Mobi-C, PCM, Prestige ST, Secure-C) and successful fusion in ACDF. The direct comparison between TDA devices and ACDF was performed as a precursor for indirect analysis between devices. As expected, ACDF was associated with a significantly higher incidence of bridging bone than all TDA devices assessed. Indirect comparison of HO with bridging bone between TDA devices demonstrated a significantly lower association with M6 than the Kineflex, Mobi-C, PCM, and Secure-C devices.
Treatment Ranking
The P-scores for each treatment across all outcomes assessed are reported in Figure 7. Cumulative ranking of the average P-Score weighting each outcome assessed equally for ACDF and each TDA devices showed that the M6 prosthesis earned the highest overall rank (P = .70), followed by Secure-C (P = .67, Figure 8). The Discover device was the lowest ranked TDA device with a P-Score of .39, while ACDF was the lowest overall ranked treatment (P = .14). There was minimal variation in the performance of the remaining 6 devices, which had P-scores ranging from .49 to .57. Heat map of computed P-Scores assigned to each treatment modality for all outcomes assessed. Green denotes good performance, while red denotes poor performance. Cumulative ranking of each treatment based on average P-Score.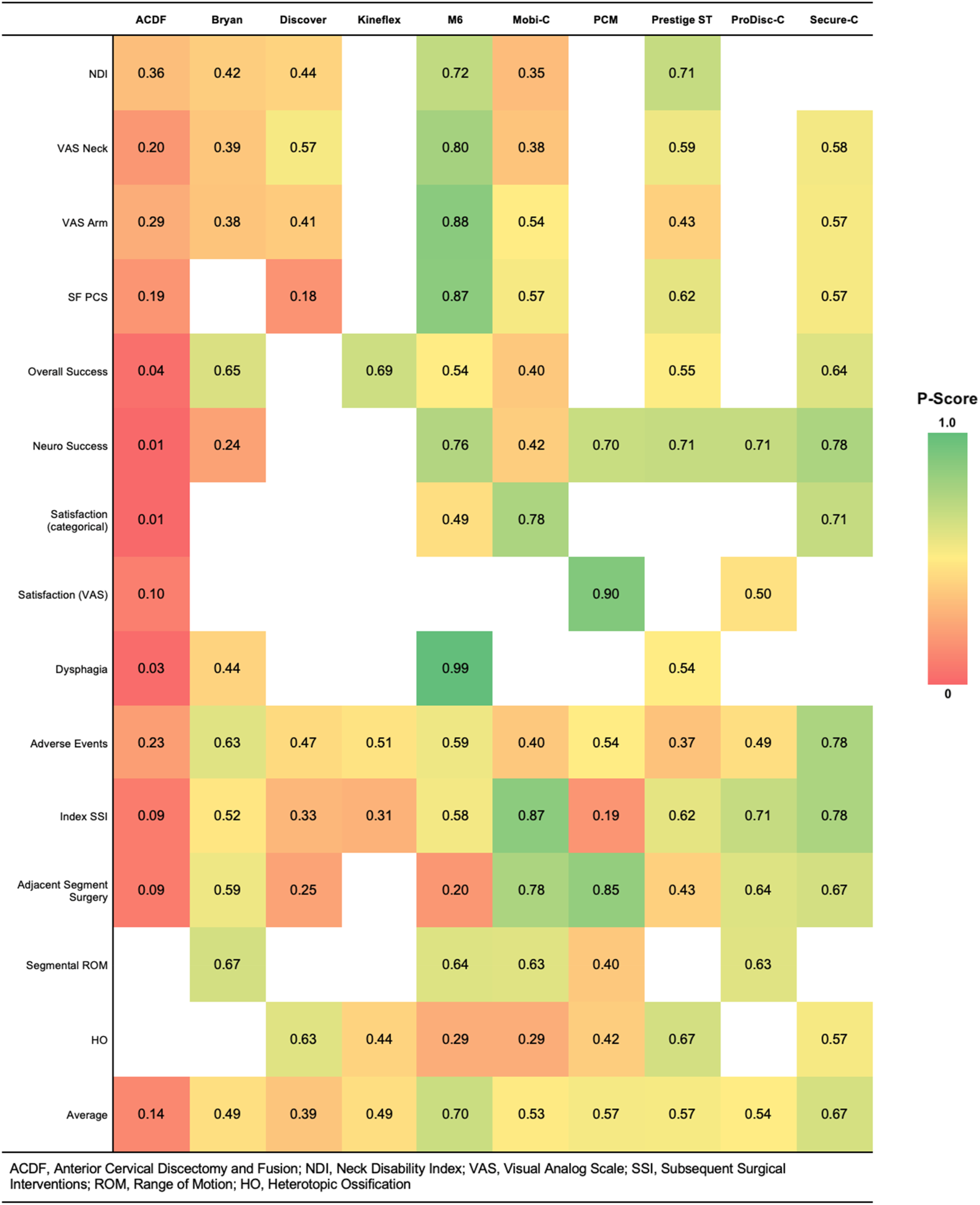
Discussion
This study quantitatively compared PROMs, patient success and satisfaction, complications, and radiographic outcomes between ACDF and TDA with 1 of 9 different devices, including the Bryan, Discover, Kineflex, M6, Mobi-C, PCM, ProDisc-C, Prestige ST, and Secure-C prostheses. When compared directly to ACDF, several devices were associated with superior outcomes for VAS Arm, SF PCS, neurological success, satisfaction, index-level SSI, and adjacent level surgeries. Few differences were noted in paired comparisons between individual devices. Cumulative ranking of treatments based on paired comparisons demonstrated the highest overall performance with the M6 device, followed by Secure-C. The remaining devices performed at a similar level, except for the Discover prosthesis which had the lowest performance among TDA devices. However, ACDF was the lowest performing treatment among all assessed.
The current body of high-quality literature supports the use of cervical TDA for the treatment of cervical radiculopathy and myelopathy, with most studies demonstrating non-inferiority to ACDF.1,8,9,12-14,16,23,25 The primary proposed benefit of the procedure includes the maintenance of motion at the operative segment, thus potentially decreasing abnormal strain at adjacent segments known to accelerate degeneration.26-29 Further, the current evidence suggests that preservation of cervical kinematics may translate to superior pain and disability outcomes both in the short and longer term.13,16,30 Findlay et al. conducted a systematic review of RCTs, finding that 6 out of the 8 trials assessed reported significantly less disability 3 months after the index surgery, as assessed by the NDI, while 5 of 7 trials reported lower neck pain. 31 The same review identified similar outcomes between TDA and ACDF at 2 years postoperatively among the included trials, however, at longer follow up between 4 to 7 years, more studies favored TDA. 31 Our quantitative analysis identified similar outcomes between all treatments in the network for neck disability and pain at an average follow up of nearly 6 years, while only the M6 device demonstrated superior outcomes to ACDF for arm pain and general physical health. The superior ability of the M6 design with 6 degrees of freedom to biomechanically mimic the kinematics of a native cervical motion segment has been described.32,33 Thus, the better performance of the M6 disc noted in our study may be reflective of more physiologic kinematics.
The current study found similar rates of device or surgery-related adverse events between all TDA devices and ACDF, although subjective dysphagia was less frequently associated with the M6 device compared to both ACDF and TDA with the Bryan prosthesis. This difference in dysphagia between TDA and ACDF may be attributed to the lower profile of the TDA, contrasted to ACDF with an anterior plate, as used in the IDE trials, which is known to contribute to irritation and edema of prevertebral structures.34-36 Anderson et al. compared the adverse event profiles reported in the Bryan FDA IDE trial at 2 years follow-up, finding a higher incidence of dysphagia and dysphonia among the investigational cohort, which the authors attributed to longer operative times, the use of a special frame, heightened sensitivity among researchers, or a potential true difference. 37 Additionally, it is possible that the milling process required for the proper placement of the Bryan disc may release more proinflammatory cytokines contributing to soft tissue edema and transient dysphagia relative to placement of the M6 device, which relies on small fins for immediate stability until osseointegration is achieved.
Subsequent index-level revision or reoperation following ACDF is most commonly secondary to pseudarthrosis, subsidence/fracture, or instrumentation failure.38,39 While subsidence and fracture may be seen following TDA, recent literature has identified a wide range of device-related complications that may necessitate further surgical intervention, including recurrent stenosis, device migration, motion loss, and even wear particle-induced osteolysis.40-42 Index-level SSI were less frequently associated with the Mobi-C and Bryan devices than ACDF, and less frequently with Mobi-C than PCM. Device migration has been associated with the PCM disc, whose press-fit design and less optimal immediate fixation leaves it prone to migration in the early in the postoperative period. 40 Further, the PCM disc achieves motion via gliding movement that is limited by the facet joints and soft tissues, rather than intrinsic device core limitations, increasing the strain on the joints which may accelerate the progression of facet arthropathy.43,44
Although not typically revised surgically, HO with bridging bone results in restricted or lost ROM, in effect leading to a nearly fused or ankylosed motion segment. While the underlying cause remains poorly understood, some have suggested that the development may be related to device-induced biomechanical stress, constitutional factors such as osteophytes and preoperative calcification, or iatrogenic factors including endplate milling and over-distraction of the disc space via placement of an oversized device.45-47 Conversely, gentle handling of soft-tissue structures, meticulous hemostasis, and the use of non-steroidal anti-inflammatory drugs (NSAIDs) early in the postoperative period may have prophylactic value in attenuating HO formation.48,49 This analysis identified that the M6 device was less likely to be associated with motion-restricting HO than the Kineflex, Mobi-C, PCM, and Secure-C prostheses. While the shorter follow up reported by Phillips et al. for M6 may have likely contributed, device-specific design elements may also predispose them to HO formation. For example, both the Kineflex and Secure-C discs contain a keel for immediate fixation, potentially promoting HO.50,51 Additionally, alteration in the segmental center of rotation (COR) has been correlated with a higher rate of HO. 52 For most devices, the COR is fixed, thus TDA implantation leads to a change from the physiologic axis, which is typically inferior to the intervertebral disc and posterior to the center of the endplate at C1 and gradually moves more superiorly and anteriorly at more caudal segments.53-55 Patwardhan et al. compared cervical kinematics before and after implantation with either the M6 or Mobi-C discs, finding that the M6 restored native cervical kinematics, while significant intra-specimen differences were observed following Mobi-C placement. 33
This network meta-analysis is not without limitations. All studies included in the quantitative analysis compared a single TDA device to ACDF, thus all between-device analyses in the network relied on indirect comparisons. Furthermore, due to the stringent inclusion and exclusion criteria maintained by the systematic review, a small number of studies were included in the final analysis, which can primarily be attributed to the few randomized trials currently in the literature for cervical TDA. Although all trials included in the analysis were of high quality, there were minor differences in the inclusion and exclusion criteria for enrollment and randomization for each trial, meaning that the patient population across all studies was not entirely homogenous. Finally, the studies included in the analysis reported outcomes at follow-ups ranging from 2 to 10 years. While the IDE trials that have spanned 10 years follow up have largely demonstrated maintenance of comparable outcomes from the short to long-term, certain variables assessed by this analysis have a temporal relationship, including HO and subsequent surgeries at both the index and adjacent segments. Despite these limitations, this is the first study to compare outcomes quantitatively and comprehensively across a wide range of cervical TDA devices using a network meta-analysis model. Our findings suggest that TDA may be superior to ACDF in the appropriately indicated patient population. Moreover, restoration of normal cervical kinematics with newer generation devices may improve patient outcomes, although future studies directly comparing device performance are necessary.
Conclusion
Cervical TDA was found to be superior on most outcomes assessed in the literature of high-quality clinical trials. When compared directly to ACDF, several devices were associated with superior outcomes for VAS Arm, SF PCS, neurological success, satisfaction, index-level SSI, and adjacent level surgeries. While most devices demonstrated similar outcomes, newer generation devices were found to outperform others across several outcomes assessed. This may be related to improved restoration of cervical kinematics, although future longitudinal studies directly comparing different TDA designs are necessary.
Footnotes
Disclosures
All co-authors have seen and agree with the contents of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
