Abstract
Study Design
Retrospective Cohort Study.
Objective
To determine whether 3D-printed porous titanium (3DPT) interbody cages offer any clinical or radiographic advantage over standard solid titanium (ST) interbody cages in transforaminal lumbar interbody fusions (TLIF).
Methods
A consecutive series of adult patients undergoing one- or two-level TLIF with either 3DPT or ST “banana” cages were analyzed for patient reported outcome measures (PROMs), radiographic complications, and clinical complications. Exclusion criteria included clinical or radiographic follow-up less than 1 year.
Results
The final cohort included 90 ST interbody levels from 74 patients, and 73 3DPT interbody levels from 50 patients for a total of 124 patients. Baseline demographic variables and comorbidity rates were similar between groups (P > .05). Subsidence of any grade occurred more frequently in the ST group compared with the 3DPT group (24.4% vs 5.5%, respectively, P = .001). Further, the ST group was more likely to have higher grades of subsidence than the 3DPT group (P = .009). All PROMs improved similarly after surgery and revision rates did not differ between groups (both P > .05). On multivariate analysis, significant positive correlators with increasing subsidence grade included greater age (P = .015), greater body mass index (P = .043), osteoporosis/osteopenia (P < .027), and ST cage type (P = .019).
Conclusions
When considering interbody material for TLIF, both ST and 3DPT cages performed well; however, 3DPT cages were associated with lower rates of subsidence. The clinical relevance of these findings deserves further randomized, prospective investigation.
Introduction
Transforaminal lumbar interbody fusion procedures (TLIFs) are an effective surgery for multiple variants of degenerative lumbar pathology. Studies show that fusion rates are high, and TLIFs may pose certain advantages over other methods of lumbar interbody fusion, including fewer neurological complications, lower blood loss, and greater pain relief.1-4 The surgical technique was initially described by Harms et al. in 1982, 5 and since that time both surgeons and medical device companies (MDCs) have worked to improve patient outcomes and decrease complication rates.
Interbody non-union and cage subsidence are 2 potential complications of TLIFs and destine its recipient for revision surgery. 6 Studies have proposed several risk factors for these complications, and the development of either is likely related to surgical, implant, and patient characteristics. Implicated characteristics include inadequate disc space preparation, improper cage dimension and placement, osteoporosis, patient age and sex, tobacco use, bone morphogenetic protein use, cage material, among others.7-9 Unlike many patient characteristics, interbody cage material is a variable that is easily changed. As such, identifying a cage material that boasts higher fusion rates and lower subsidence rates is important.
Two types of cage materials are in common use today: polyether ether ketone (PEEK) and titanium. PEEK was popularized given its Young’s modulus similar to that of bone, thus leading to decreased stress shielding and improved subsidence rates. 10 However, PEEK implants form a fibrous capsule adjacent to bone which is known to prevent fusion. 11 Titanium, on the other hand, promotes osteointegration when in contact with and fuses well with bone. 12 However, its stiffness leads to greater stress shielding, which can cause bone subsidence. 13 As a solution, titanium-coated PEEK implants were designed to possess the advantages of both materials, but this coating may fail to survive the implantation process. 14
More recently, 3D-printed porous titanium (3DPT) cages were developed as an alternative to the standard solid titanium (ST) cages. This material boasts a Young’s modulus similar to that of PEEK while avoiding the fusion-preventing encapsulation.15,16 Furthermore, MDCs advertise 3D printed titanium’s porous design as an improved lattice for osteointegration and osseous ingrowth.17,18 MDCs cite animal, in vitro, and case report studies when claiming improved subsidence and fusion rates of 3D printed titanium vs standard titanium interbody cages.19-21 However, no large human retrospective analyses have been completed comparing the 2 cage materials. Here we compare complication rates and clinical outcomes related to interbody cage titanium type in short segment TLIFs.
Methods
Study Population and Design
The authors obtained approval from The Ohio State University Wexner Medical Center Institutional Review Board (protocol number 2019H0377). This study was exempt from informed consent as it is a retrospective study imposing minimal risk to study participants aside from loss of confidentiality. Furthermore, it was deemed impracticable to retroactively locate and obtain informed consent for patients that fit the inclusion criteria but were no longer seen at The Ohio State University or were deceased. Retrospective review of a prospectively maintained database from a single tertiary care center identified patients who had undergone spinal fusion procedures with 1 of 3 fellowship-trained spinal neurosurgeons between August 2016 and November 2020. Inclusion criteria was one- or two-level transforaminal lumbar interbody fusion (TLIF) coupled with one- or two-level bilateral posterolateral fusion performed in patients at least 18 years of age for lumbar degenerative disease, with or without spondylolisthesis, without a previous lumbar fusion procedure. Patients were excluded if interbody cage material and type was anything other than 3DPT or ST steerable “banana” cage. For example, patients who received polyether ether ketone (PEEK) or titanium-coated PEEK cages of any morphology were excluded, as were patients who received straight “bullet” titanium cages. Other exclusion criteria included lack of radiographic or clinical follow-up of at least 1 year, or unrelated death within 1 year of surgery. Finally, the indications for TLIF among the analyzed cohort included: grade 1 or 2 spondylolisthesis, radiographic instability, disc space collapse with foraminal stenosis, and lumbar deformity.
Surgical Technique
All surgeries were performed using either a standard open approach or a minimally invasive (MIS) approach. Two neurosurgeons performed all open procedures while the third performed surgeries using both approaches. The latter surgeon chose approach based on patient demographics and spine characteristics. In general, open approaches were performed using a midline incision and subperiosteal dissection to expose corresponding lamina, facet joints, and transverse processes. Care was taken to ensure preservation of the rostral facet joint. Pedicle screws were placed, and then complete bilateral facetectomies and either partial inferior or full laminectomies were performed based on surgeon preference and degree of central stenosis. Disc space preparation included bilateral annulotomy, disc space distraction, and disc removal. Typically, the largest cage that would not cause endplate injury was chosen. Following cage placement, bilateral rods were placed, and dorsal compression was applied prior to final tightening the set caps to optimize lordosis. Autograft was used in all cases after decortication, while allograft and BMP use was per surgeon preference and patient characteristics. Closure was standard with deep and superficial closure, and subfascial drains were utilized.
MIS approach, on the other hand, was performed with 2 paramedian incisions with open dissection stopping at the fascial layer. A series of dilating tubes were then used to perform a unilateral facetectomy on the side of worst stenosis, followed by unilateral disc preparation and cage placement. Similar to the open approach, cage size was maximized while avoiding endplate injury. BMP and autograft were typically placed in the disc space as well. Bilateral pedicle screws were then placed using neuronavigation, and rods were placed without dorsal compression. Closure was standard and drains were not used.
Demographic and Surgical Variables
Demographic variables were collected from retrospective chart review and included age, sex, body mass index (BMI), American Society of Anesthesiology (ASA) preoperative class, and presence of comorbidities. Comorbidities included history of tobacco smoking, history of intravenous drug abuse, alcohol use disorder diagnosis, diabetes mellitus (DM), and osteoporosis or osteopenia. Spondylolisthesis at the level of surgery was determined on preoperative dynamic imaging, and was categorized into grades 1, 2, 3, and 4 corresponding to <25%, 25-49%, 50-74%, and >74% of slippage, respectively.
Operative reports were used to assess the number and levels of interbody fusion, number and levels of posterolateral fusion, surgical approach (minimally invasive vs open), bone morphogenetic protein (BMP) use, and allograft use. Implant records were reviewed to record cage lordosis, width, height, length, and footprint. All cages were supplied by the DePuy Synthes company. 3DPT cage brands included EIT Cellular Titanium® and Nexxt Matrixx®. ST cage brand used was the T-PALTM Interbody Spacer System. Lastly, the transition from ST to 3DPT steerable cages occurred in early 2019 when that material cage became readily available from the regional DePuy spine company.
Radiographic Outcomes
Bridwell Interbody Fusion Grading Classification.

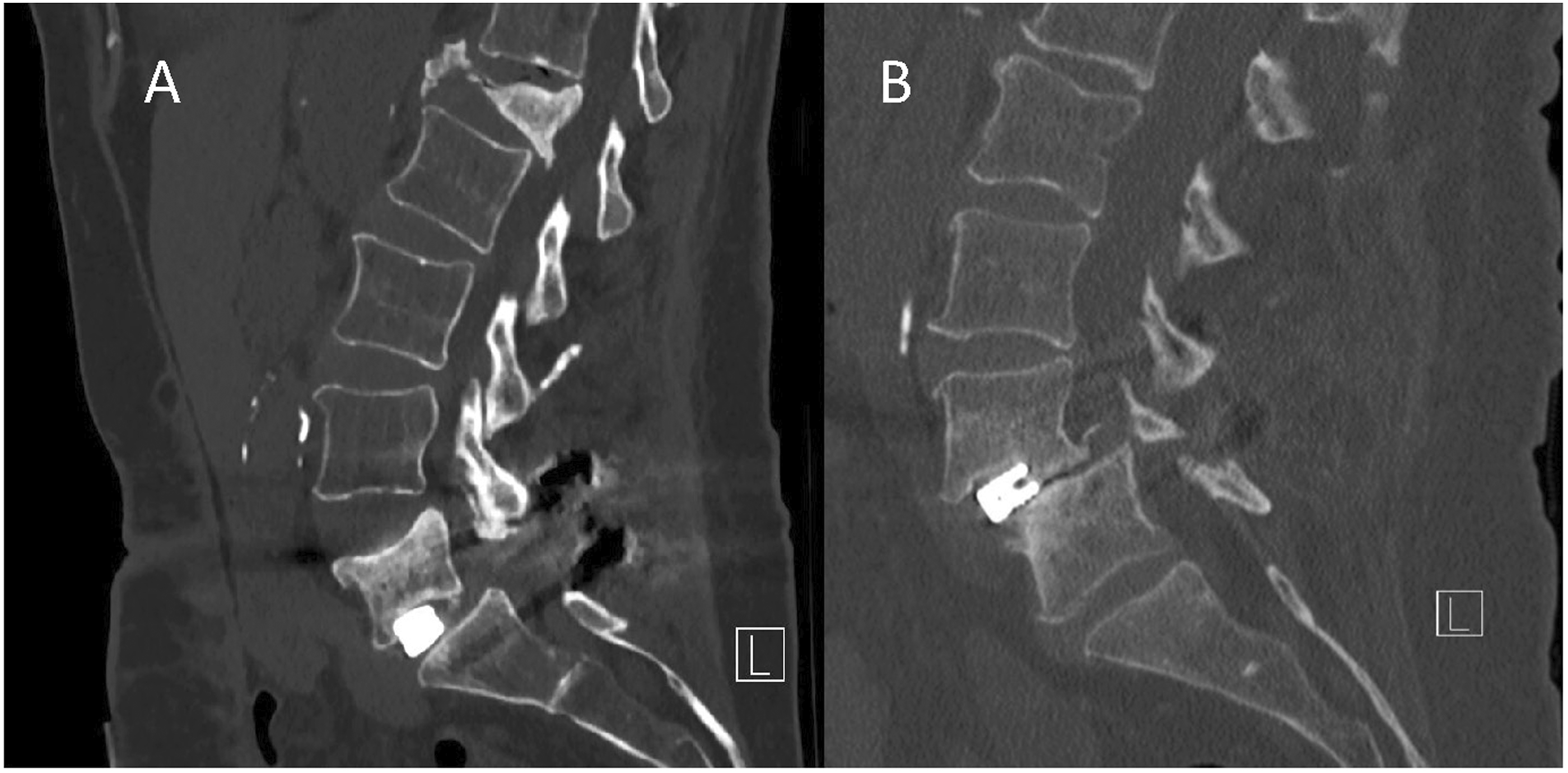
Subsidence grades. Lateral computed tomography demonstrating examples of grade 1 (A) and grade 3 (C) subsidence.
Clinical Outcomes
Patient reported outcome measures (PROMs) were recorded both preoperatively and at least 1 year postoperatively when available. Reported PROMs included visual analog scale (VAS) leg and back pain, Oswestry Disability Index (ODI), and 36-Item short form survey (SF-36) pain and physical functioning scores. Delta scores were only calculated for patients that had both preoperative and > 1-year postoperative PROMs. Reoperation rates and reasons were also recorded up through the time of data collection, specifically November 2021.
Statistical Analysis
Statistical analysis was performed in IBM® SPSS® Version 27. Two main cohorts were created based on type of titanium used (3DPT vs ST). Continuous variables are reported as mean values with standard deviations. Two-tailed Student’s t-test was used for the primary comparison. Paired t-tests were used for comparisons within the same cohort. Unpaired t-tests were for comparisons between cohorts. Categorical variables were analyzed using Pearson’s chi-square test. Categorical variables that included any zero value were analyzed using two-tailed Fisher’s exact test. Multivariate linear regression analysis of subsidence grade was performed by evaluating variables that may demonstrate a causal association according to expert opinion, as is recommended by research literature.25,26 Collinearity was defined as a variance inflation factor (VIF) > 4 among independent variables. A P value of <.05 represented statistical significance.
Results
Demographic and Surgical Variables
Demographic and Surgical Variables.
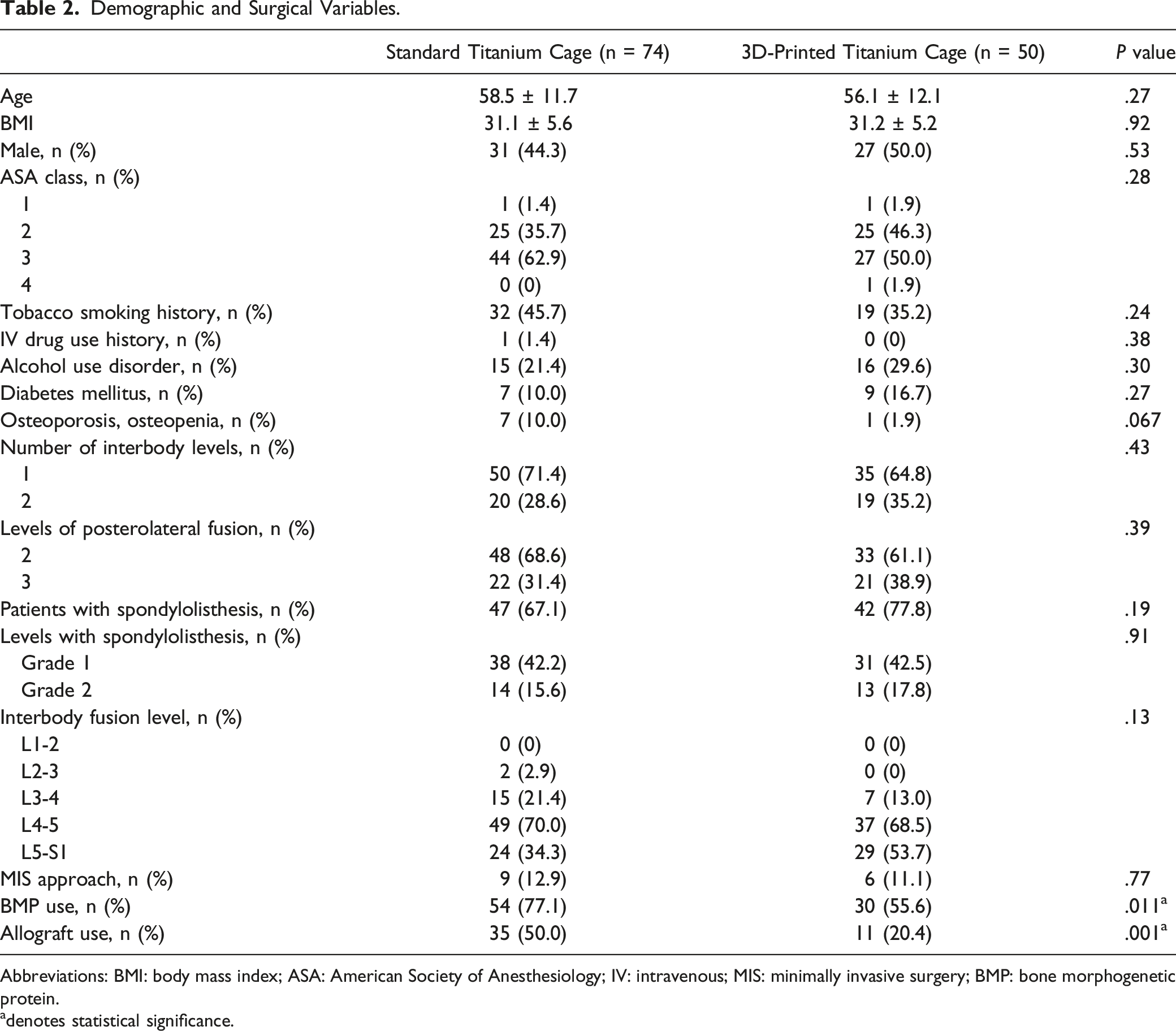
Abbreviations: BMI: body mass index; ASA: American Society of Anesthesiology; IV: intravenous; MIS: minimally invasive surgery; BMP: bone morphogenetic protein.
adenotes statistical significance.
Cage Characteristics.
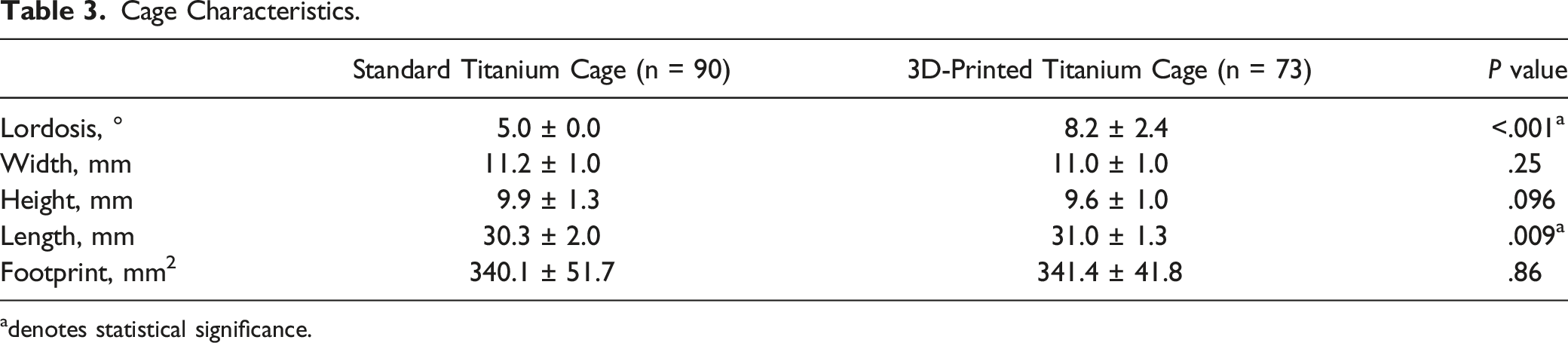
adenotes statistical significance.
Radiographic Outcomes
Radiographic Outcomes.
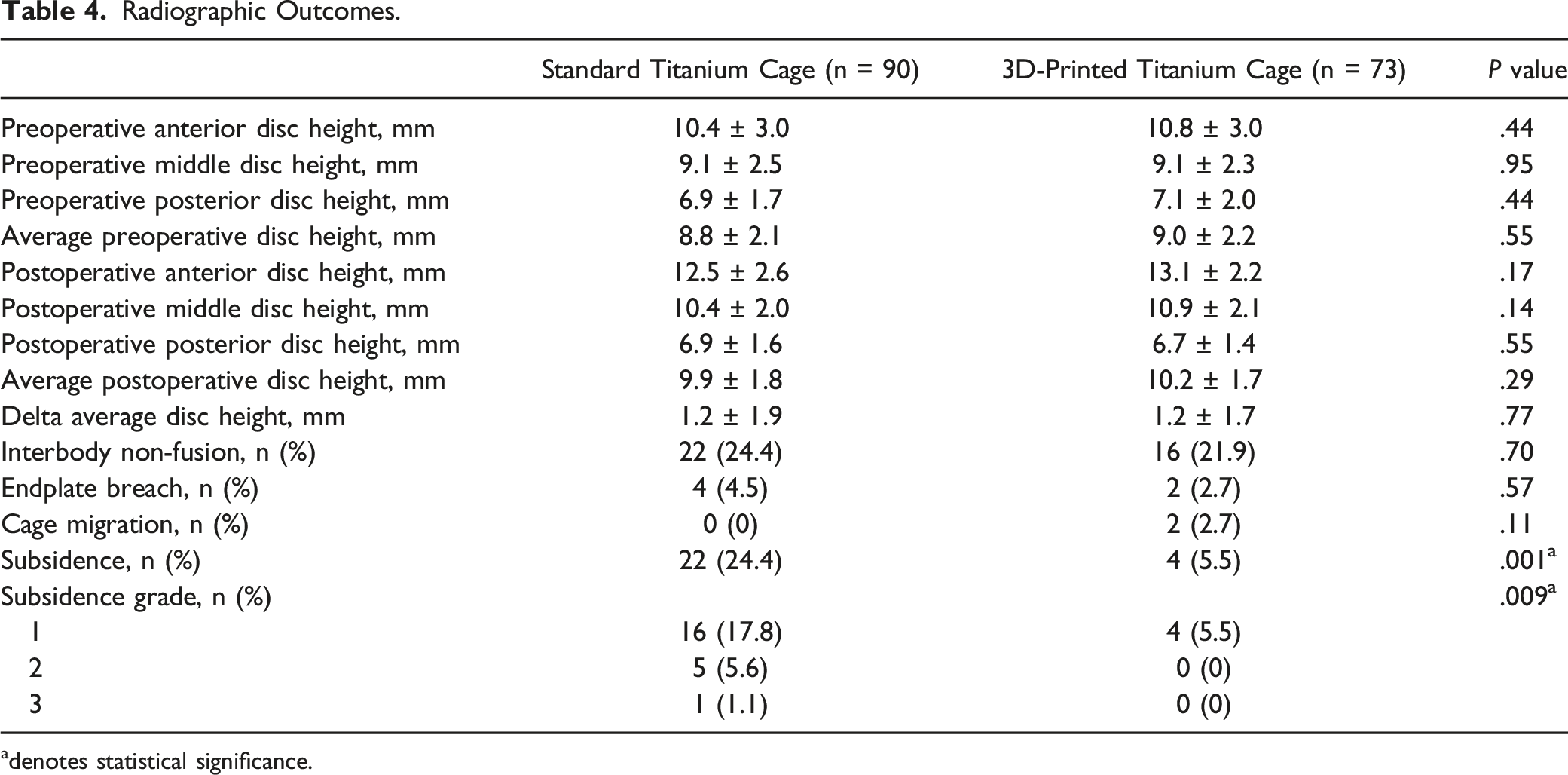
adenotes statistical significance.
Clinical Outcomes
Clinical Outcome.
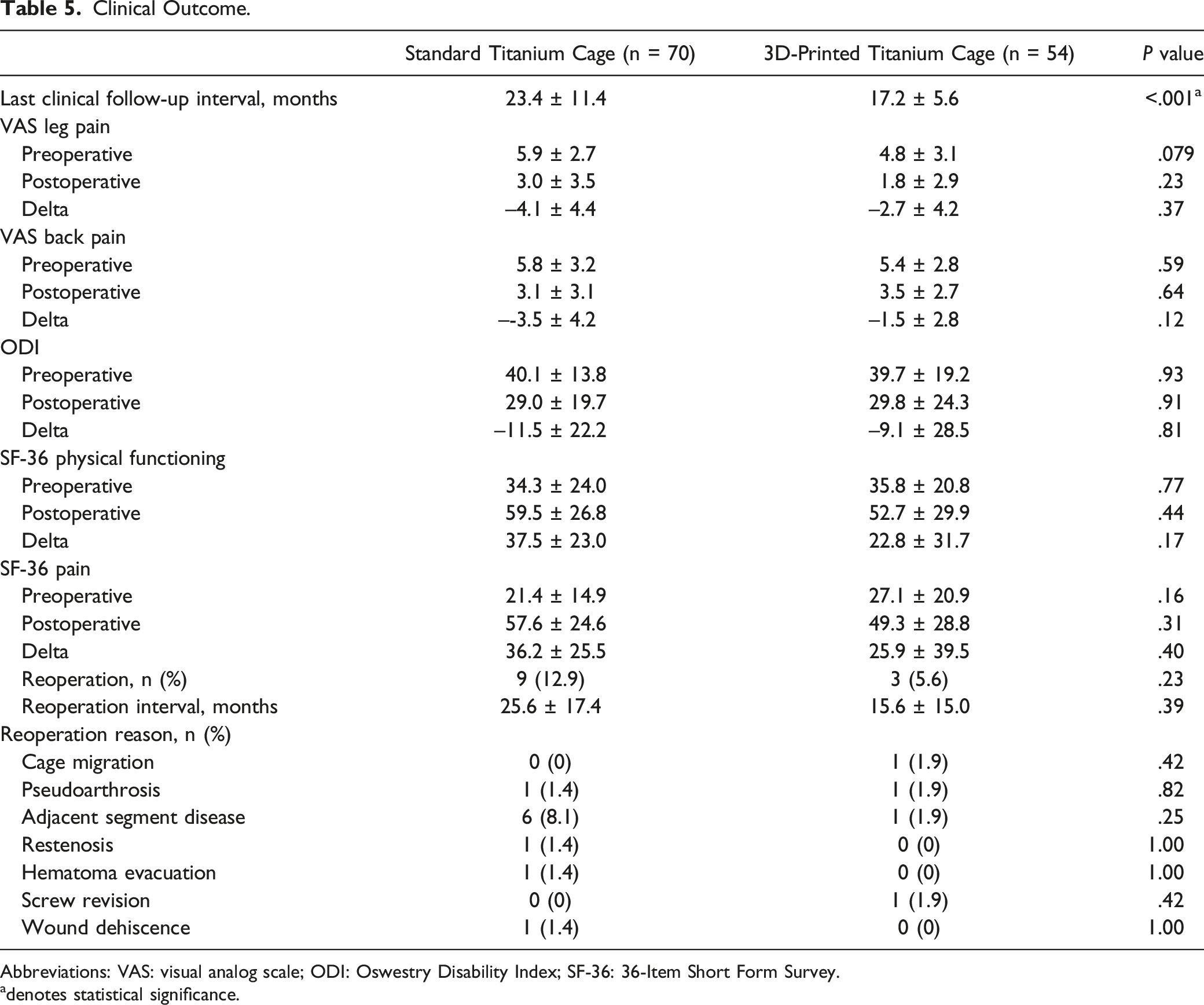
Abbreviations: VAS: visual analog scale; ODI: Oswestry Disability Index; SF-36: 36-Item Short Form Survey.
adenotes statistical significance.
Subsidence Grade Multivariate Analysis
Multivariate Linear Regression Analysis of Possible Predictors of Subsidence Grade.
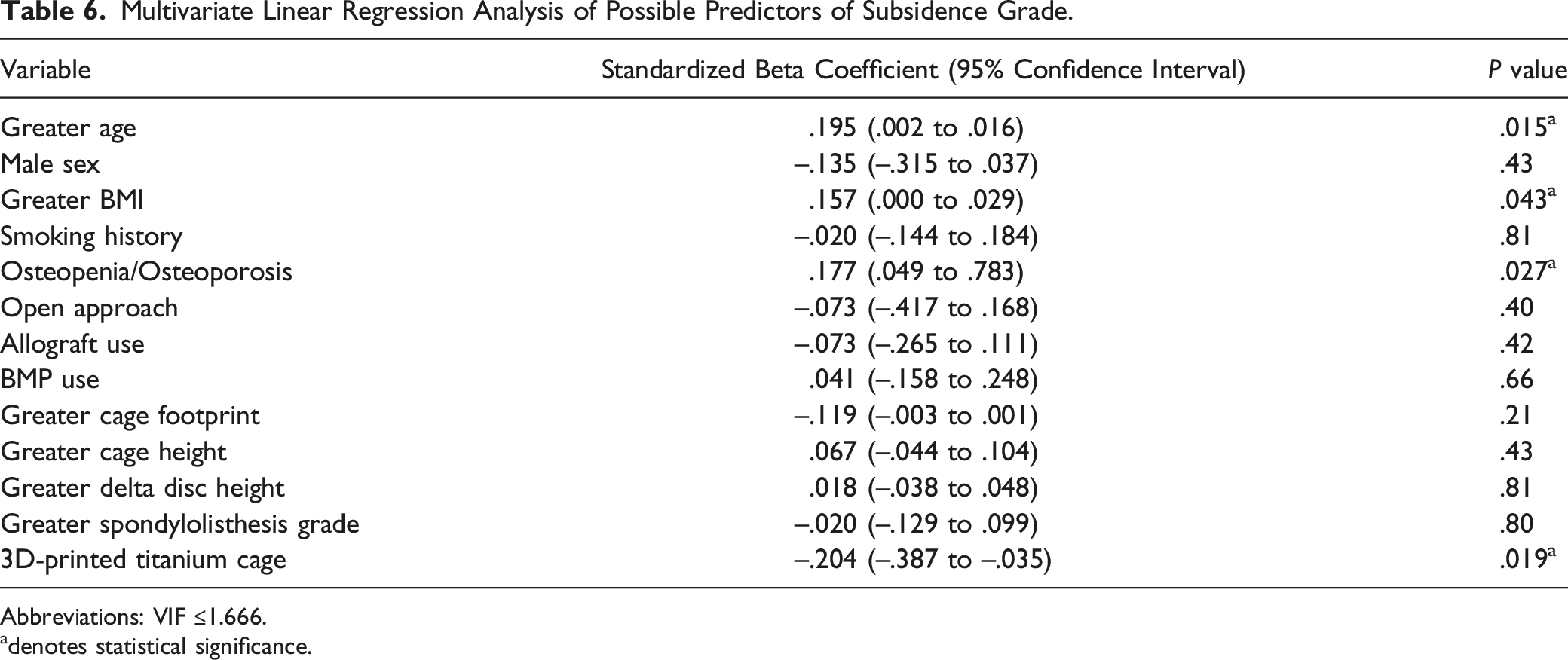
Abbreviations: VIF ≤1.666.
adenotes statistical significance.
Discussion
The search for and development of interbody cage characteristics that improve clinical outcomes and minimize complications such as subsidence is ongoing. Titanium cages are frequently employed as studies have demonstrated higher fusion rates when compared to PEEK in lumbar fusion surgery. 17 This is related to the excellent bone-to-implant contact and osteogenic properties exhibited by titanium implants.27-29 Titanium implants create an osteoconductive surface that provides short-term stability due to friction and long-term stability due to cell adhesion leading to bony ongrowth (direct apposition of bone onto the surface of the cage) and ingrowth (bone formation within the implant). 30 Thus, cells more widely proliferate and differentiate on titanium than on PEEK because of titanium’s osseointegration properties. 24 However, solid titanium features a modulus of elasticity much greater than that of bone, leading to variable rates of subsidence, with previous studies ranging from 16-35%. 31
3DPT has gained popularity in recent years as a superior medium for many types of surgical implants, including interbody cages for TLIFs. Purported advantages of 3DPT include lower subsidence and higher fusion rates when compared to ST cages. These potential advantages over ST cages relate to unique 3DPT characteristics and include optimal porosity and scaffolding for osteoblastic integration and aggregation, and complex macro/micro/nano surface roughness that is ideal for osteoblastic differentiation.16,18,32-34 These qualities, coupled with 3DPT’s modulus of elasticity similar to that of human bone, provides a cage material that theoretically should optimize fusion and subsidence rates. MDCs do not hesitate to advertise these claims. However, current literature comparing the 2 materials is limited. Most existing studies have investigated the efficacy of these cages compared to ST in animal and in vitro models. Human studies more commonly compare PEEK to 3DPT.35,36 As such, the present study fills a need for a large-scale human comparison of clinical outcomes and complication rates related to 3DPT and ST interbody cages.
Even though in vitro work suggests fusion rates may be similar with 3DPT vs ST implants, the present study does not directly support this theory. We identified similar non-fusion rates between the 2 cohorts utilizing the Bridwell classification on lateral radiographs: 24.4 and 21.9%. While these numbers initially appeared high, they are well within the range of previously reported fusion rates after TLIF procedures. Wu et al performed a meta-analysis comparing radiographic fusion between open and MIS approach TLIFs, and reported fusion rates between 46 and 99%. 37 Kim et al compared successful fusion between posterolateral fusion alone vs TLIFs, and found that TLIFs were associated with fusion rates of 58.6% on CT at 1 year. 38 Finally, Seaman et al compared interbody cage material and fusion rates in a meta-analysis, and found that titanium interbody cages were associated with between 46.5 and 100% rate of radiographic fusion. 31 Thus, our findings align with existing literature. Furthermore, radiographic assessment of interbody fusion rates is unreliable when compared to clinical evaluation of successful fusion. 39 This unreliability is likely echoed in the present study, as PROMs were greatly improved in both cohorts, and revision surgery for pseudoarthrosis only occurred in 2 patients. Thus, our reported fusion rates may be underestimates. Furthermore, the different rates of BMP and allograft use between cohorts must be highlighted. The ST cohort was more likely to receive both BMP and allograft than the 3DPT cohort. Importantly, BMP use has been purported to increase fusion rates in TLIFs. 40 So, despite less frequent use of fusion enhancing materials, the 3DPT cohort demonstrated similar radiographic fusion rates to the ST cohort. Thus, our study demonstrates that 3DPT cages offer at least noninferior rates of fusion to ST cages but falls short of supporting claims that fusion rates are improved with 3DPT.
Analysis did, however, identify lower subsidence rates in the cohort of patients that received 3DPT cages. There was a 24.4% rate of subsidence of any grade in the ST cage cohort compared to 5.5% in the 3DPT (P = .001). Further, the ST cohort had higher grades of subsidence as well, with 5.6% of patients demonstrating grade 2 (50-74% disc space collapse and graft subsidence) and 1.1% of patients demonstrating grade 3 (75-100%). This contrast between cohorts stood up on multivariate analyses, with ST cage being a positive predictor for worsening subsidence grade. This degree of subsidence associated with ST cages is mirrored in previous studies. Seaman et al found subsidence rates with ST cages to vary between 16.2 and 35% in their meta-analysis. 31 Similarly, Campbell et al describe a subsidence rate of 20% following lateral lumbar interbody fusion (LLIF) using titanium cages. 41 Further, our rate of subsidence associated with 3DPT is similar to scarce current reviews. Amini et al quote a subsidence rate of 3% following LLIF with 3DPT cages. 42 Despite the difference in approach and cage size between the TLIF and LLIF, comparable subsidence for out study’s ST and 3DPT TLIF cages to the literature of LLIF cages is reassuring due to LLIF cages’ decreased subsidence. Other variables associated with worsening subsidence grade in our analysis included greater age, higher BMI, and the presence of osteoporosis or osteopenia. All these factors have previously been shown to increase the risk for subsidence, which may validate our findings related to cage type and subsidence rates.7,43 Thus, our study supports claims that 3DPT’s modulus of elasticity like that of human bone, as well as its porous architecture promoting early bone ingrowth and structural integrity, decreases subsidence rates when compared to ST cages. 44
Despite the different rates of subsidence between the 3DPT and ST cage cohorts, clinical outcomes were similar and overall favorable with low revision rates and improvement in pain scores. Several reasons likely account for this. While subsidence rates were higher in the ST cohort, most of these cases were grade 1, representing 25-49% disc space collapse. As greater degrees of subsidence are known to correlate with worse clinical outcomes, these low observed grades of subsidence may be clinically insignificant, allowing those patients to still report improved scores. 6 Furthermore, overall rates of subsidence and clinical non-fusion were low. Thus, the few patients that reported worse pain after surgery were likely masked by many patients that experienced pain relief. Interestingly, revision rates were also similar between cohorts despite different subsidence rates. However, there was an overall trend towards higher rates of revision surgery within the ST cage cohort (12.9% vs 5.6%, P = .23). However, this trend may relate to the difference in clinical follow up interval between the 2 cohorts. The ST group had longer overall clinical follow-up, so this trend may represent the known association between time from index surgery and the development of ASD rather than a consequence of cage material. Continued clinical follow-up and larger cohorts will be needed to uncover any significant association between titanium cage type and revision surgery. Lastly, surgical decisions are often not made in a vacuum, and cost efficiency is an important tenet in modern healthcare. There was no cost difference between the ST and 3DPT cages, with both being priced at $9900 by the vendor. Both cages were procured from a single MDC but fashioned by multiple MDC’s through industry partnerships, which increases the overall external validity of our findings.
Conclusion
The current study suggests that certain 3DPT biomechanical properties, namely its elasticity modulus and porosity, may have a protective effect against subsidence when compared to ST interbody cages during TLIF for lumbar degenerative disc disease. This effect was achieved with similar radiographic and clinical outcomes. Longer clinical follow-up will help uncover any difference in clinical outcomes related to the different subsidence rates between cohorts.
Footnotes
Acknowledgments
The authors wish to thank Shukri Ahmed and Joshua Bolender for their work as clinic research coordinators on this study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Andrew Grossbach, MD: Research funding from DePuy Synthes, Medtronic, 3M/KCI; consultation fees from 3M/KCI. Stephanus Viljoen, MD: Research funding from Medtronic. David Xu, MD: NuVasive consultation fees
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Disclaimer
The views expressed in the submitted article are the authors’ own and not the official position of the Ohio State University Medical Wexner Center.
