Abstract
Study Design
: Retrospective Chart Review.
Objectives
Incidental durotomies (IDs) are common spine surgery complications. In this study, we present a review on the most commonly utilized management strategies, report our institutional experience with case examples, and describe a stepwise management algorithm.
Methods
A retrospective review was performed of the electronic medical records of all patients who underwent a thoracolumbar or lumbar spine surgery between March 2017 and September 2019. Additionally, a literature review of the current management approaches to treat IDs and persistent postoperative CSF leaks following lumbar spine surgeries was performed.
Results
We looked at 1133 patients that underwent posterior thoracolumbar spine surgery. There was intraoperative evidence of ID in 116 cases. Based on our cohort and the current literature, we developed a progressive treatment algorithm for IDs that begins with a primary repair, which can be bolstered by dural sealants or a muscle patch. If this fails, the primary repair can be followed by a paraspinal muscle flap, as well as a lumbar drain. If the patient cannot be weaned from temporary CSF diversion, the final step in controlling postoperative leak is longterm CSF diversion via a lumboperitoneal shunt. In our experience, these shunts can be weaned once the patient has no further clinical or radiographic signs of CSF leak.
Conclusions
There is no standardized management approach of IDs and CSF leaks in the literature. This article intends to provide a progressive treatment algorithm and contribute to the development process of a treatment consensus.
Keywords
Introduction
Cerebrospinal fluid (CSF) leaks due to incidental durotomy (ID) are among the most common complications of spine surgery. The estimated incidence ranges from 2% to greater than 20%.1-7 The current literature has identified several risk factors for intraoperative ID, such as advanced age, the female gender, history of spine surgery, high BMI, patients undergoing either laminectomy, osteotomy or interbody procedures and high risk anatomical zones, including the caudal margin of the cranial lamina, the cranial margin of the caudal lamina, herniated disc levels, and the medial aspect of facet joints.4,7-9
With a larger number of patients undergoing lumbar spinal surgery and a stable rate of ID, there will inevitably be an increase in the number of patients experiencing these complications. Postoperatively, patients with ID may experience significant short and long-term effects that need to be taken into consideration by both the patient and provider, including but not limited to a significant increase in operative time (OT), estimated blood loss (EBL), and length of stay (LOS), patient dissatisfaction, reoperation rate, time to return to work and overall back pain with associated functional limitations. CSF leaks resulting from ID in lumbar spine surgery are also further associated with pseudomeningocele, external CSF fistula, severe positional headaches, meningitis, arachnoiditis, and intracranial hemorrhage.10-18
IDs are inevitable, however, to avoid further complications, proper management is crucial. Despite the current literature, there remains significant discordance among spine surgeons on the optimal strategy for intra- and postoperative ID management. Strategies differ vastly between surgeons, even within the same institution, especially in the absence of high-quality evidence-based recommendations. 19
In this article, we present a review of literature on the most commonly utilized strategies to manage IDs intraoperatively and postoperatively and report our institutional experience with variations of ID identified during lumbar spine surgeries, with case examples. This aims to develop a stepwise approach to the management of intraoperative IDs and persistent postoperative CSF leaks.
Methods
This study is a retrospective chart review of all lumbar spine surgery cases performed at the authors’ institution between the years: March 2017 to September 2019 by 4 spine neurosurgeons with a minimum follow-up of 1 year. Ethical approval was obtained from our Institutional Review Board (IRB approval number 834405). A waiver of informed consent was requested and obtained as this research project involved no more than minimal risk to the subjects. Cases that acquired an ID, or presented with a postoperative CSF leak were identified via electronic medical records (EMR) documentation with special attention to operative notes and reports of return to the operating room (OR). Patients’ characteristics, demographics, surgical details, postoperative complications, resource allocation, incidence and symptoms of CSF leaks, and variations of management strategies were recorded. This was followed by a literature review and outline of the current methods to treat IDs and persistent postoperative CSF leaks following lumbar spine surgeries.
Results
Overview of Patient Characteristics and Surgical Details.
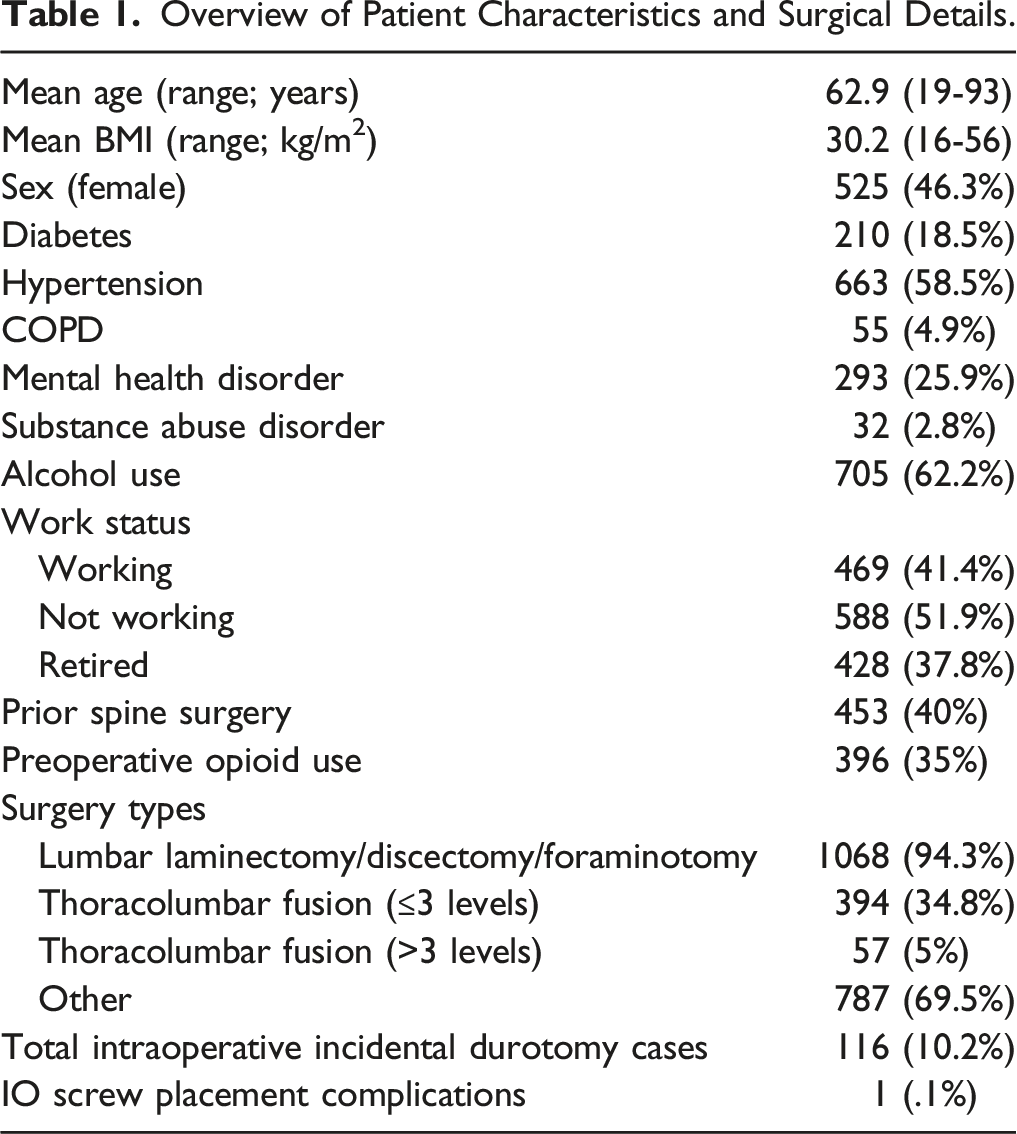
Overview of Postoperative Complications.

Overview of Postoperative CSF-Related Complications.

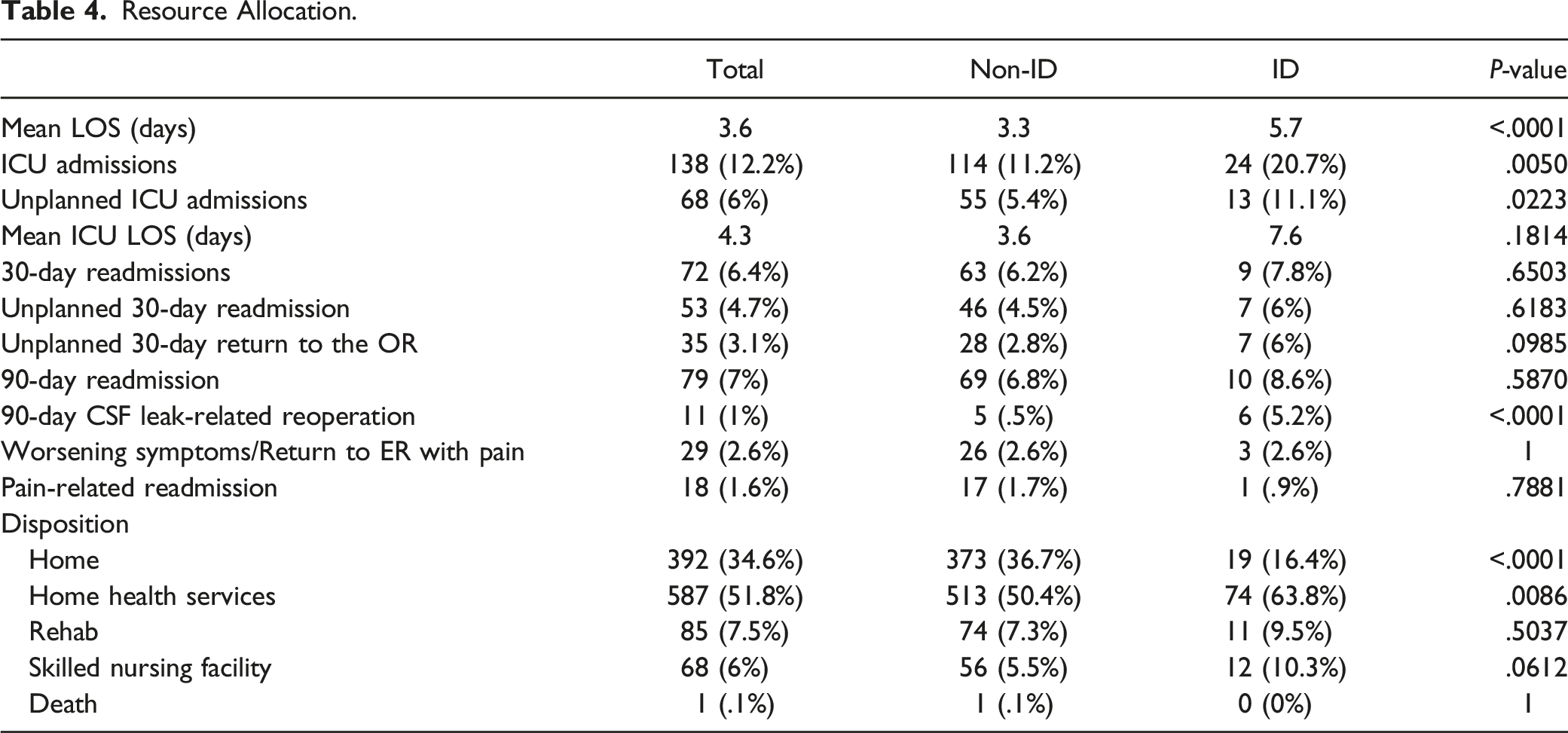
Resource Allocation.
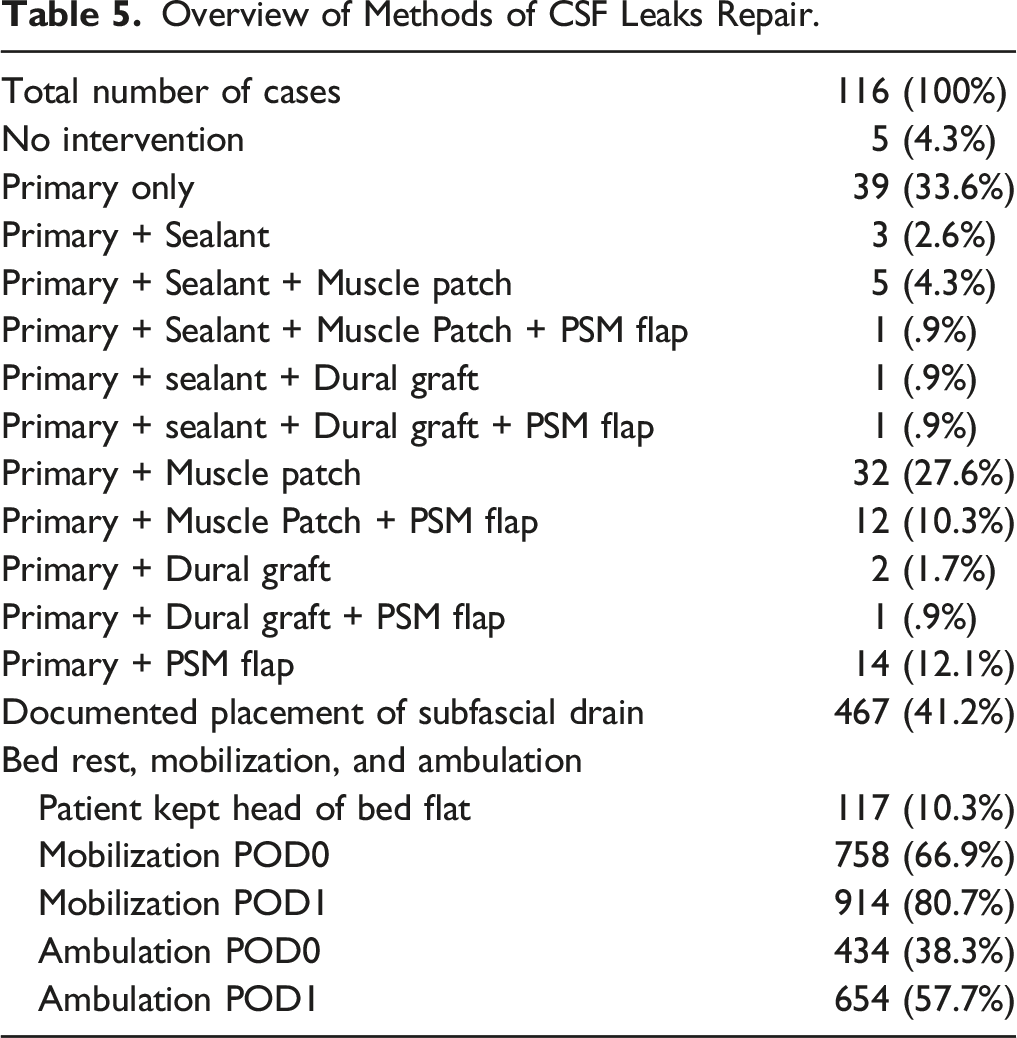
Overview of Methods of CSF Leaks Repair.
Discussion
Suggested Management of Intraoperative Incidental Durotomy
The treatment algorithm begins after the identification of an ID with a direct dural repair and subfascial drain that can be monitored postoperatively. Direct dural repair can be augmented in several ways, including an autologous muscle patch or epidural blood patch. If the patient develops a persistent CSF leak or any of its associated complications, such as a pseudomeningocele, the next step in the algorithm is re-exploration and repair of durotomy, again with a subfascial drain, except the revision surgery should be now completed with a paraspinal muscle flap, as well as a subarachnoid spinal drain placed a few levels above the durotomy site. If the revision surgery fails to successfully manage the persistent CSF leak, then the algorithm moves to a more permanent CSF diversion in order to lower the intrathecal CSF pressure and give extensive time for the durotomy site to heal (Figure 1). Below, we discuss each step in further detail. Algorithm outlining the authors’ institutional experience managing intraoperative incidental durotomy (ID) and persistent postoperative CSF leaks.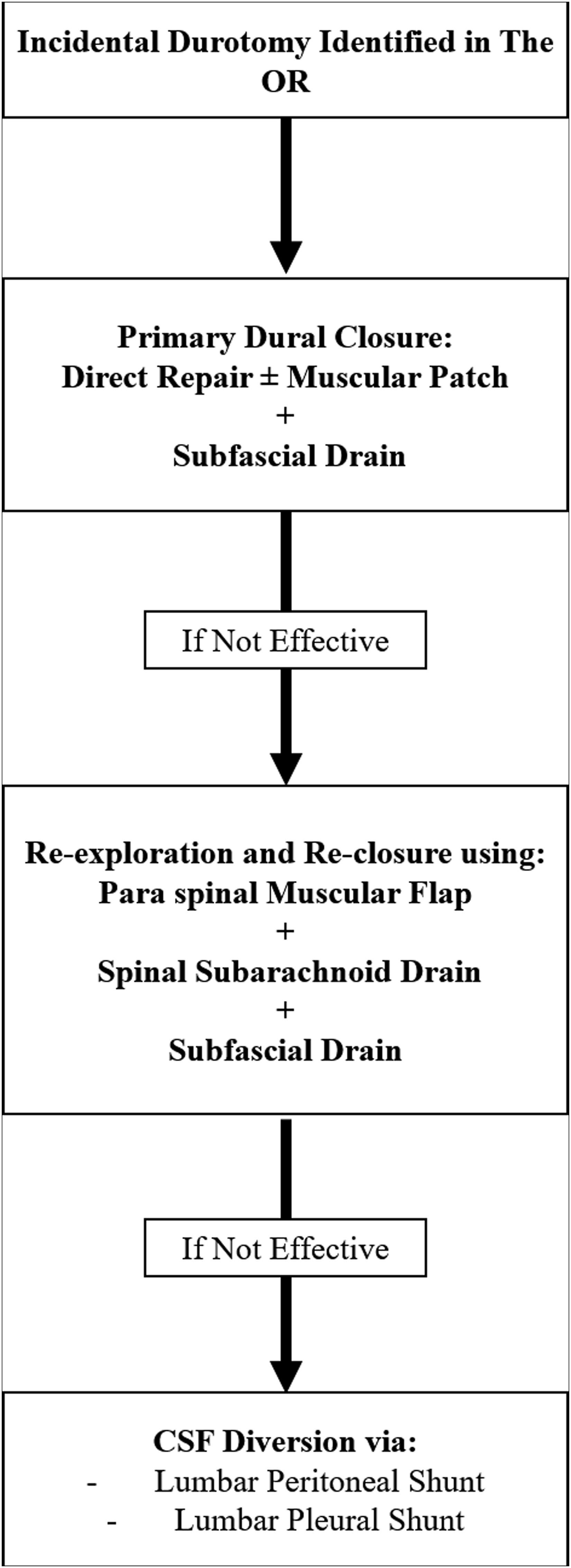
Direct Dural Repair
When an intraoperative ID is recognized, the initial step in management is to directly repair the tear, provided that the defect is amenable to repair. There were 111 out of 116 patients (96%) that underwent a direct primary repair. The 5 patients that did not undergo primary repair initially showed intraoperative dural “bleb” without frank CSF and did not require further intervention. Typical reparable dural tears are often linear along the medial aspect of the thecal sac, rather than the lateral edge, with sufficient surrounding tissue to close over the defect. Irreparable tears typically have irregular borders or tears within the nerve root sleeve or an inaccessible lateral aspect of the thecal sac. In our institution, direct repair of dural tears is usually done using 4-0 silk sutures in a simple running or figure-of-eight fashion. In the case of irreparable dural rents or if there is insufficient tissue to close over the defect, a dural or autologous muscle graft may be used to augment the primary closure. Muscle patches are particularly useful when both sides of the dura cannot be approximated to achieve direct closure due to dural retraction, which may occur after the coagulation of the bleeding point in the dural defect. 20 We note that augmentation of closure using a muscle patch may benefit patients with known rates of persistent CSF leak in the literature such as increased age, history of prior spine surgeries, and increased BMI.
After direct repair is complete, the surgeon can request anesthesia to elicit a Valsalva maneuver to test the integrity of the repair. The majority of current literature favors the direct repair of dural tears with non-absorbable sutures when possible, as opposed to simple patching alone. In one case series that included 3183 cases of whom 238 cases sustained dural tears, only 6 patients (2.5%) resulted in persistent leak after direct repair using 4-0 silk sutures. 21 Additionally, a case-control study compared direct repair using sutures only (n = 12, 19%), patching only using TachoSil ± muscle or fat flaps (n = 22, 32%), and combined sutures and patching (n = 34, 49%) in a total of 69 cases of ID. While there was no statistical difference for either persistent CSF leaks or clinical outcomes noted between the three groups, isolated patching showed twice the rate of revision surgeries compared to that of suture alone (7.7%) or combined suture and patching (8.8%). 8 Lastly, in a recent meta-analysis studying outcomes of different methods of dural tears management, direct repair of dural tears with suture with or without accompanying use of sealants or flaps (.037, 95% CI (.024–.053)) was significantly more successful to repair with patching or sealants alone (.047, 95% CI (.026–.074)). 19 Therefore, the initial step in our management algorithm of intraoperative ID favors the use of non-absorbable sutures, with or without augmentation with muscle patches depending on patient- and durotomy-specific characteristics, to achieve direct repair when possible.
Fibrin Glue and Sealants
Fibrin glue utilization has become increasingly popular to manage ID since its introduction about two decades ago. Fibrin glue is thought to achieve a more “water-tight” seal of the dural defects when combined with direct suture repair, as well as stimulate the fibroblastic activity in an effort to enhance dural scarring postoperatively. However, in a previous retrospective analysis that included 4835 procedures in the lumbar spine that resulted in 547 ID cases (11.3%), the use of fibrin glue did not decrease the rate of persistent CSF leaks or improve postoperative clinical outcomes when compared to primary dural repair. 22 Additionally, while no significant complications have been reported with the use of fibrin glue and similar sealants, there is a paucity of literature proving their efficacy against physiological CSF burst pressures, as well as its potential effects on dural or surgical wound healing. In light of the issues and with the current data on cost-effectiveness, a 2017 survey showed a significant decrease in the number of surgeons who use fibrin glue in their primary repair of dural tears compared to a 2007 study.21-26 In our series, dural sealant was used on only 11 patients. We, therefore, do not include sealants or glue as a critical step within our algorithm, as our data and the current literature do not support its efficacy.
Subfascial Drain
The subfascial drain is a simple and common, yet crucial, aspect of our management of ID postoperatively. Even after a layered closure, there often remains a significant dead space between the paraspinal muscle and the thecal sac. In the case of CSF leaks, this dead space tends to fill with CSF coming from the dural defect, even after its repair. The continued pooling of CSF predisposes patients to a persistent CSF leak in the form of a pseudomeningocele or potential cutaneous fistula, further increasing the risk for postoperative infection and potential meningitis. To avoid these complications, a subfascial drain with mild negative pressure (ie compressed bulb suction) to remove the fluid that builds up postoperatively is employed. The drain is removed when the total output is less than 30 milliliters in a 24-hour period, which is typically around 5-7 days postoperatively. If a patient continues to have exceedingly high output from their subfascial drain after a prolonged period (ie at least >7 days), the decision is then made to pull the drain and monitor the patient for signs and symptoms that would indicate failure of a primary durotomy closure and the need for revision surgery, such as symptomatic pseudomeningocele or cutaneous CSF fistula.
A number of recent studies support these management steps. In one prospective study, the use of a subfascial drain for 3-4 days, 5-6 days, or 7-10 days after ID during posterior spine surgery was compared. The results showed that patients using subfascial drains for more than 7 days displayed the shortest wound healing times, as well as the lowest incidence of postoperative cutaneous CSF fistula or pseudomeningocele. 27 The authors attributed the improved outcomes to the ability of the subfascial drain to create low subfascial pressure through the prevention of CSF accumulation during the early postoperative period, a time when wound resistance is still low and prone to CSF accumulation. Once the subfascial tissue achieves sufficient healing strength and continuity within the dead space, enough pressure develops on top of the dura to counter the subarachnoid pressure at the durotomy site. Therefore, the authors believe that the drain can promote wound healing by redirecting leaking CSF until the pressure difference normalizes. In similar studies, prolonged use of subfascial drainage did not correlate with postoperative complications and patients did not have different rates of persistent CSF leak after drain removal. 28 Our algorithm calls for the standard use of subfascial drainage in all patients with ID during both the index and all subsequent surgeries.
Bed Rest
Postoperatively, the majority (81%) of patients in our cohort maintained a degree of bed rest for 24 hours after their surgery.29-32 Furthermore, we found that patients with CSF leaks had significantly longer total LOS (5.7 days vs 3.3 days, P < .0001) as compared to those who did not which may be attributed partially to this practice pattern. Bedrest is a standard practice at our institution, however, there remains controversy within the literature about the efficacy of this intervention. A number of studies show no significant difference in outcomes or CSF leak-related complications when comparing patients who undergo bed rest vs those that do not. These studies also show no significant adverse events related to short-term postoperative bed rest, however, patients on bed rest tend to display increased average LOS, which can affect hospital costs and overall patient satisfaction.29-31,33 Although these reports advocate that bed rest may be unnecessary for patient recovery, postural headaches are a common complication after durotomy and subsequent CSF leaks. In these instances, maintaining a flat posture is evidently an effective treatment until CSF production can overcome the amount lost. To prevent the worsening of postoperative postural headaches, we prefer, especially when using a subfascial drain, to maintain a degree of flatbed rest.29-31,33 Therefore, the standard initial management of patients with ID in our algorithm includes flatbed rest for the first 24 hours, followed by gradual mobilization through elevating the head 30 degrees every 6 hours, ultimately leading to mobilization with assistance.30,32
Indications for Re-Operation
If a patient fails primary durotomy closure, revision surgery may be the next step. The decision to take a patient back to the OR can be difficult, especially when dealing with CSF leaks and the paucity of literature surrounding proper management. Similar to the overall management techniques, the indications for re-operation in persistent CSF leaks vary between surgeons and are heavily influenced by experience, as well as the patient’s overall health status.
Unfortunately, there is a critical lack of evidence describing the natural history of ID-associated CSF leaks and an even greater lack of data clarifying which risk factors or common comorbidities in this patient population predispose patients to require revision surgery. In our cohort, we found that presence of CSF leak was significantly associated with durotomy-related return to the OR within 90 days (5.2% vs .5%, P < .0001) compared to those without a dural rent. Poor nutritional status is associated with decreased rates of wound healing and can increase the rate of pseudomeningoceles and the need for operative revision.5,34 However, most pseudomeningoceles are less than 5 cm and tend to be asymptomatic, obviating the need to rush for revision surgery. Although they require close monitoring, even larger pseudomeningocele collections larger than 8 cm have been shown to resolve spontaneously over time. 35 For patients with postoperative pseudomeningocele, especially in those with poor nutritional status or even uncontrolled diabetes, it may be beneficial to monitor the patient while also optimizing their overall health status, prior to initiating further surgical revision. Alternatively, if a patient becomes symptomatic or develops a cutaneous CSF fistula in the setting of a persistent CSF leak, we advocate for revision surgery. This is especially true, even in the absence of any symptoms, due to the high risk of associated infections. 13 Other symptoms that warrant further investigation and potentially re-operation include postural headaches, infections such as epidural abscesses or meningitis, and the development of a new-onset neurologic deficit, which on rare occasions can be due to cauda equina nerve roots settling into the dural defect causing nerve entrapment. In these instances, the symptoms warrant wound exploration and revision.36,37 Revision surgery involves re-exploration and identification of any durotomy sites that can be primarily repaired in the previously described fashion. After this, the closure should then include bolstering via a paraspinal muscle flap, a lumbar subarachnoid drain placed above the level of the primary surgery, and finally a new subfascial drain.
Paraspinal Muscle Flaps
A critical step in revision surgery is to eliminate the surgical cavity dead space and create sufficient barriers to CSF drainage, both of which can be done with a paraspinal muscle flap. To efficiently repair the surgical wound, initially, the skin incision should be ellipsed in order to expose healthy tissue for improved wound healing. In cases of pseudomeningocele, careful dissection around the pseudomeningocele should be done to preserve the external pseudomeningocele capsule, which can be used as an extra layer of tissue to repair the durotomy. If the source of a leak is identified, the next step is incision of the pseudomeningocele sac and the use of the external walls of the capsule to achieve a tight dural closure. In a recent report on pseudomeningocele repair, Banerjee et al described a Z-shaped flapping technique utilizing the bilateral paraspinal musculature. In this small series, they describe a similar wound exploration technique as above, followed by wide mobilization of the paraspinal muscles by creating a plane between the fascia and muscle layers. The muscles are then brought together in an overlapping manner and closed using 3-0 polydioxanone sutures medially through both flaps simultaneously. 38 This technique allows the bulk of the paraspinal musculature to layer within the surgical cavity, reducing the dead space that typically follows a lumbar spine surgery. 39 Of note, during the paraspinal muscle flap mobilization, the lateral lumbar perforators should be preserved and the flaps should be loosely sutured together to avoid muscle necrosis.38,39 This increases the resistance against CSF drainage and decreases the rate of CSF tracking out through the incision and ultimately wound breakdown. Next, a subfascial drain is placed underneath the muscle repair layers. 38 The fascial layer is then closed tightly above the muscle flap. Another drain, such as a JP drain, may be placed superficially above the fascial layer to further decrease any fluid, seroma, or CSF, that could accumulate at this layer. In some reports, a flap from the tensor fascia lata muscle could be used to augment the paraspinal musculature during revision surgery for dural tears that resulted in pseudomeningocele.40-42 Paraspinal muscle flaps may be used during the primary durotomy repair, however, with the degree of dissection and tissue manipulation, we maintain these flaps for revision surgery within our algorithm.
Lumbar Subarachnoid Drain
Generally, lumbar subarachnoid drains, commonly referred to as lumbar drains, are used along with revision surgery to treat persistent CSF leaks. These drains divert CSF away from the durotomy site, therefore lowering the pressure difference between the subfascial space and the subarachnoid space. This, combined with subfascial drainage, minimizes the accumulation of CSF in the surgical site, allowing for proper wound healing and ultimately preventing recurrence.3,43-45 The drain is usually inserted into the subarachnoid space intraoperatively 1 or 2 levels above the repair site. The drain is then tunneled to the skin away from the incision and connected to a drainage system that can be titrated based on flow or pressure gradients. Typical use involves draining 10 mL in a single hour rather than basing drainage off of pressure gradients. This prevents the potentially devastating complications, such as hindbrain herniation, associated with over-drainage. Usually, the drain will continue at this setting until weaned off 7-10 days postoperatively, once there is a clear absence of progressive CSF leak.
In one case series, 11 patients with symptomatic (>8 cm) pseudomeningocele after spinal surgery were treated with a combination of open revision for the extirpation of the pseudomeningocele, dural repair, and implantation of a subarachnoid drain. All the patients were successfully treated with no revision-related complications observed or recurrence through a mean follow-up of 16.5 months. 45 In a similar report, 12 patients with either pseudomeningocele or cutaneous CSF fistulas were treated using lumbar subarachnoid drainage and wound over-sewing. After treatment, there was no notable fistula or pseudomeningocele recurrence, as well as no neurologic or infection-related complications observed in any of the patients through the follow-up period. 46 These drains are not without risk. Numerous studies cite drain-related complications including infections, over-drainage of CSF leading to herniation, nausea, vomiting, and development of transient radiculopathy.44,46-48 Therefore, we prefer to utilize this option as a subsequent step during revision surgery, after the failure of primary durotomy repair.47,48
CSF Diversion
The last resort in our strategy to manage persistent CSF leaks is continuous CSF diversion via a lumbo-peritoneal (LP) shunt. The use of shunts is described as a bailout maneuver in a number of intracranial pathologies, as well as in previous reports of refractory CSF fistulas or pseudomeningocele after spinal surgery.49-51 One small case series included 4 patients with refractory CSF leaks that failed revision surgeries, temporary external drainage via subarachnoid spinal drains, and epidural blood patches. The utilization of a LP shunt successfully treated the persistent CSF leaks, as well as avoided complications related to external drain use, and even shortened the average postoperative hospital LOS. 52
CSF diversion via a LP shunt carries similar risks as a temporary lumbar drain, including infections and over drainage, but is also at risk for catheter occlusions and the development of acquired Chiari malformation.53,54 Once there is a complete resolution of the CSF leak, these drains can be removed or tied off. However, this requires another surgery and has the potential to lead to a separate CSF fistula. For these reasons, we consider CSF diversion a last resort. It must be approached with careful consideration and lengthy patient conversations. Nonetheless, the use of LP shunts must remain another option in a neurosurgeon’s armamentarium.
Example Case #1
The patient is a 57-year-old male with bilateral lower extremity radiculopathy and neurogenic claudication with MRI findings consistent with severe canal narrowing and foraminal stenosis. The patient underwent L3-5 laminectomies with foraminotomies complicated by a small dural tear at the L4-5 level that was repaired primarily with sutures. A subfascial drain was left in place. Postoperatively, the patient maintained bed rest for 24 hours. The patient’s positioning restrictions were gradually liberalized and he tolerated mobilization well. At the end of postoperative day 1 (POD1), his drain displayed 150 cc of serosanguinous fluid consistent with normal postoperative drainage. After discussions with the patient, he was discharged home the same evening. However, the patient represented on POD3 in the ED with severe postural headaches that were refractory to typical alleviating maneuvers, including lying flat, and clear drainage from his incision. Due to the overwhelming symptoms and persistent incisional drainage, an MRI was completed that showed evidence of a CSF leak (Figure 2). The patient was taken back to the OR on POD3 for re-exploration. A persistent leak was noted from the lateral edge durotomy at the L4-5 level. Another primary repair with sutures was performed. A Valsalva maneuver did not elicit further CSF drainage. At this point, due to the need for revision surgery, the decision was made to perform a paraspinal muscle flap from L2 to the sacrum and a drain was left within the paraspinal muscle. At the same time, due to the clear control of the durotomy site, a lumbar drain was deferred. Postoperatively, the patient remained on flatbed rest for 24 hours. His drain output was less than 40 mL on POD1 and only 10 mL on POD2, even after having his positioning restrictions liberalized and gradual mobilization. His drain output remained low (<20 cc/day) and his incision remained dry, so the decision was made to remove the patient’s drain and discharge the patient on POD5. He did not experience any postoperative complications or require further intervention after more than 1 year of follow-up. MRI at 6 months showed resolution of CSF leak (Figure 2). Postoperative T2 MRI studies of lumbar spine. (A) immediate post-operative MRI after index surgery with evidence of CSF tracking towards the incision surface. (B) resolution of CSF leak at 6-month follow-up MRI.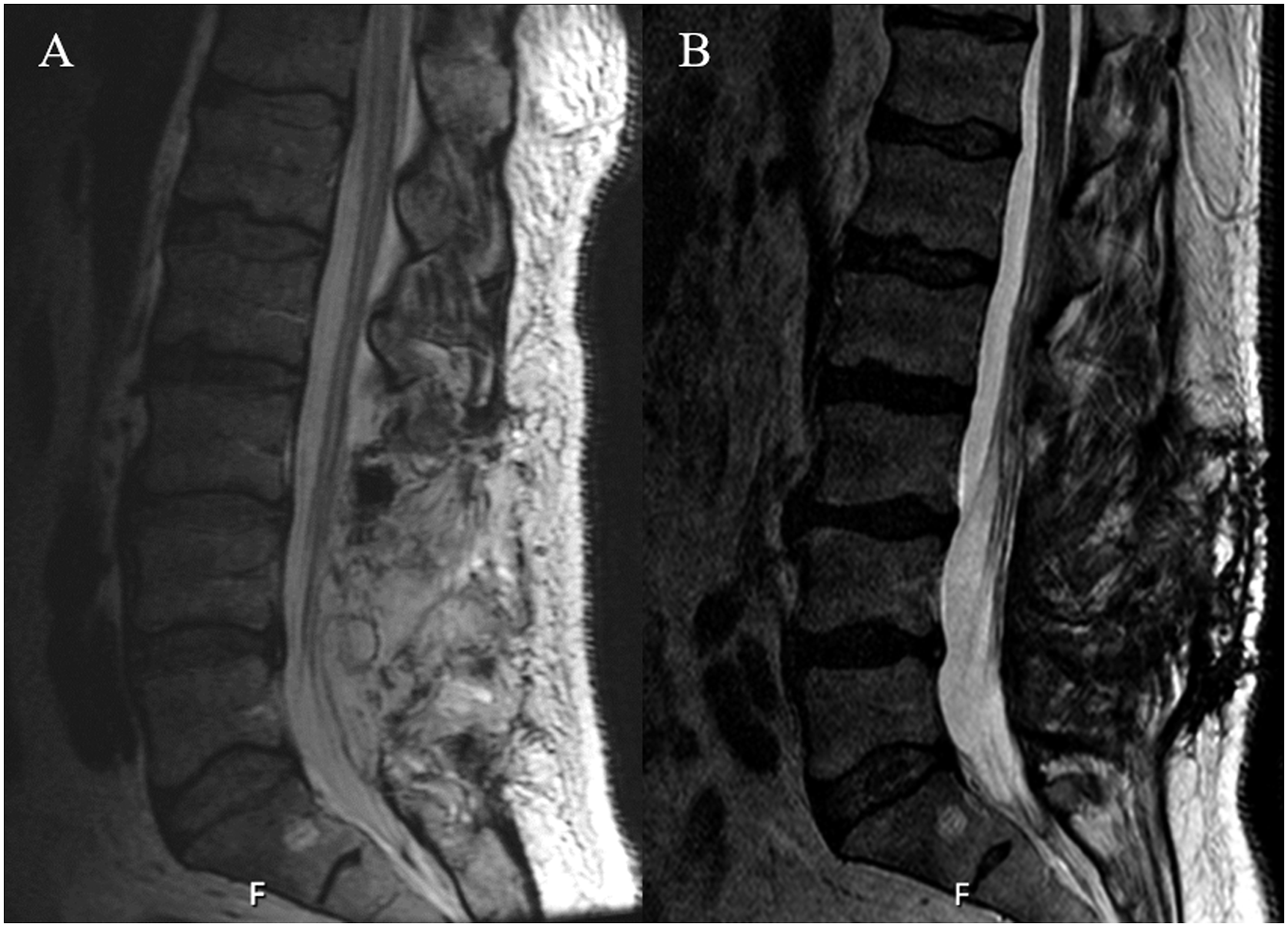
Example Case #2
A 30-year-old female with a history of scoliosis and multiple prior lumbar surgeries, including a prior decompression and non-instrumented fusion, followed by an instrumented fusion with multiple revisions, ultimately resulting in a L2-pelvis fusion. She presented to the clinic with significant lower back pain and progressive lower extremity weakness. Imaging displayed a grade 5 spondylolisthesis at L5-S1 with hardware insufficiency. She underwent staged surgeries that began with the removal of prior hardware and decompression with transpedicular corpectomy at S1. A durotomy was encountered at the S1 transpedicular dissection site and was closed primarily. Postoperatively, the patient was maintained on bed rest with the HOB flat for 24 hours with low output from her drain. On POD2, she underwent the second stage of her surgery which involved vertebral column resection for L5-S1 spondylolisthesis reduction and re-instrumentation of L2-pelvis. CSF was again noted during this procedure and the durotomy was closed primarily. Two subfascial drains were left in place due to the large incision size and the patient’s HOB remained flat for another 24 hours. On POD2 from her second surgery, her HOB was slowly liberalized. However, as she began to sit upright and mobilize, clear drainage was noted around one of the JP sites, consistent with a CSF leak. This drain was removed and a stitch was placed to close the JP exit site. No further drainage through the drain site was noted. She continued gradual mobilization, however, she began to complain of a persistent headache on POD6 that was associated with higher outputs of clear drainage through her remaining drain. She was laid flat again and her drain was taken off of mild bulb suction. Her headache improved when flat but would recur, along with increased drain output, when sitting up past 30 degrees. Due to the resistant symptoms and continued signs of a persistent leak, an MRI was completed that showed evidence of a CSF leak, including a pseudomeningocele (Figure 3). She was taken back to the OR for re-exploration. Due to her multiple prior surgeries, plastic surgery was consulted to assist with the revision. A dural rent was noted at the previous site of durotomy and was closed primarily with sutures. With assistance from plastic surgery, a paraspinal muscle flap was mobilized with a deep muscular drain, as well as a suprafascial drain, left in place. Due to the extensive nature of this leak and multiple prior surgeries, a lumbar drain was also placed intraoperatively. Postoperatively, she was kept on bed rest with her HOB flat and 10 cc per hour was drained from her lumbar drain. She gradually liberalized her HOB restrictions but maintained her lumbar drain for a total of 7 days. Her subfascial and suprafascial drains continued to put out low volumes of serosanguinous fluid. On POD7 from the revision, her lumbar drain was removed. She continued to mobilize and her incision and drain sites remained dry without evidence of a pseudomeningocele. On POD8, her remaining deep drain was removed and she was discharged home with the superficial drain to be managed by plastic surgery as an outpatient. She has not experienced any CSF-related complications or underwent further revision after 3 years of follow-up. Postoperative T2 MRI studies of lumbar spine showing evidence of CSF leak with an ex-tension of pseudomeningocele superficially towards incision site.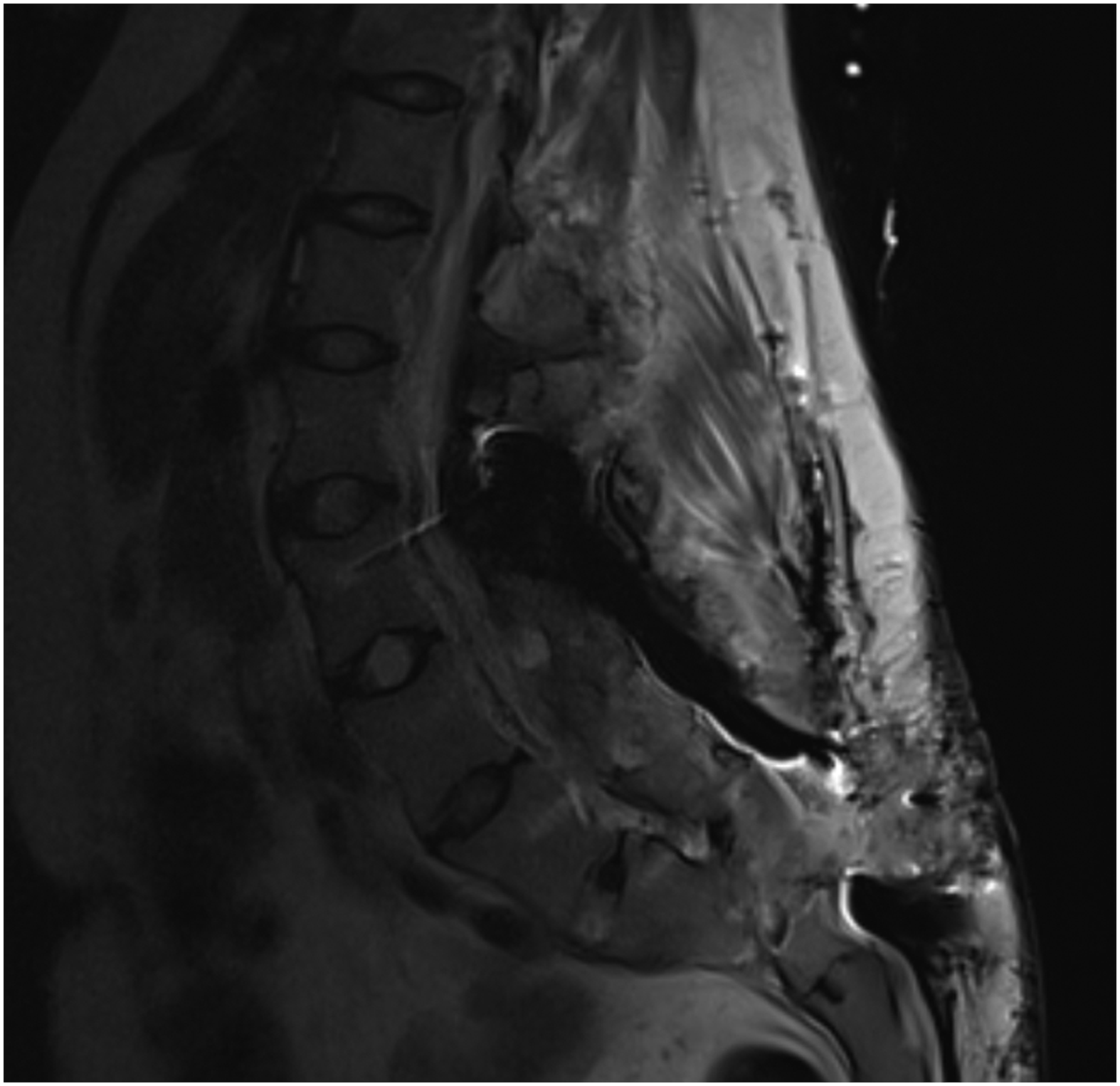
Example Case #3
A 49-year-old male presented with subacute onset of persistent bilateral perineal numbness and pain when sitting. He has reduced sensation during intercourse and defecation. An MRI pelvis demonstrated subarticular zone bilateral sacral nerve root cysts at S2, corresponding with his symptoms (Figure 4). He was taken to the operating room for a sacral laminectomy for bilateral cyst resection. Intraoperatively, CSF was encountered during cyst removal and was primarily repaired and reinforced with a dural graft. A paraspinal muscle flap was also used during closure during the index surgery, which is the standard of care at our institution when performing sacral cyst resections. Postoperatively, the patient remained on bedrest with his HOB flat for 24 hours. On POD1, the patient's HOB restrictions were liberalized. His drain began to put out thin, serosanguinous fluid consistent with CSF, and a pseudomeningocele at the surgery site developed. On POD2, he began to complain of positional headaches. He was placed back on bedrest with his HOB flat and his drain was placed to gravity. However, his drain continued to have high output consistent with CSF, so a lumbar drain was placed at the L4-5 level, above the site of the surgery. He remained on bedrest while using the lumbar drain to drain 5 cc of CSF per hour. After 5 days of drainage via the lumbar drain, the lumbar drain was clamped and the patient’s HOB was liberalized to 30 degrees. However, he continued to report positional headaches with elevation and displayed worsening of the pseudomeningocele at the surgical site. From POD 9 to 12, the patient remained on bed rest with HOB flat, with the lumbar drain continuing to drain 5 cc/hr. His pseudomeningocele improved and was able to tolerate slow increments of HOB elevation. Due to the persistent CSF leak with resolution with lumbar drain placement, the decision was made to pursue LP shunt placement to allow for long-term CSF diversion and allow the patient to begin physical therapy and rehabilitation. Postoperatively, only a small pseudomeningocele remained and the patient was able to work with physical therapy and ultimately be discharged to home with outpatient physical therapy on POD3 from LP shunt placement. The patient did well after discharge, but at his 2-month follow-up appointment, the patient again complained of positional headaches that were beginning to worsen. His incision was well-healed without significant pseudomeningocele on MRI (Figure 3). The decision was made to take the patient to the operating room to tie off the LP shunt. The patient tolerated the procedure well with resolution of his symptoms, which continued at his most recent 3-month follow-up. Postoperative T2 MRI studies of lumbar spine. (A) preoperative imaging of sacral cysts. (B) resolution of the cysts and post-operative CSF leak 2-months after lumbo-peritoneal shunt placement.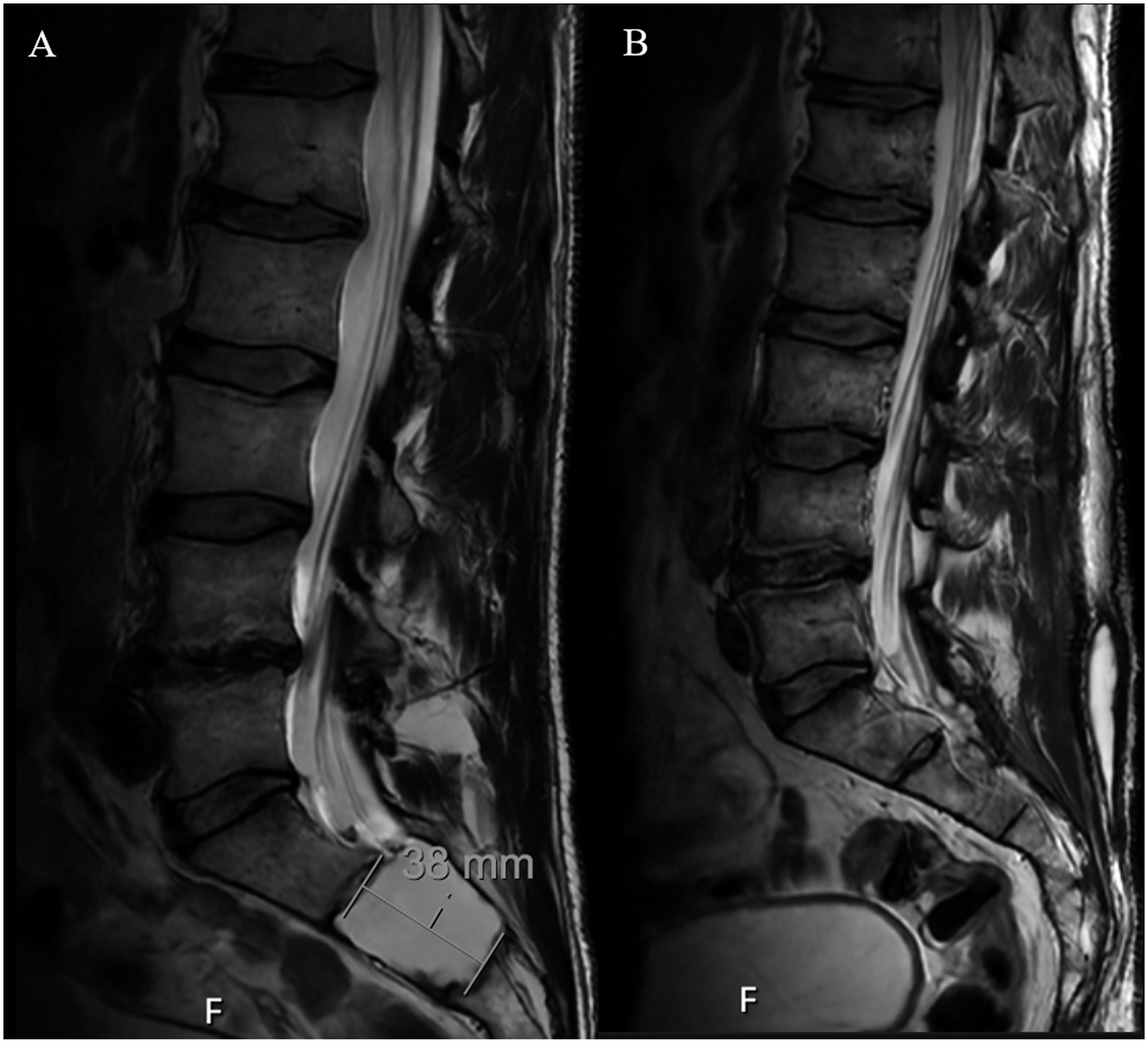
Conclusion
There is a pressing need to adopt a standardized approach for the management of IDs and CSF leaks. We provide a progressive treatment algorithm for the management of intraoperative ID and associated CSF leaks. This guide should improve the quality of patient postoperative care and pave the way for further studies to identify the most efficient methods of CSF leaks management.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Approval was obtained from our Institutional Review Board (IRB approval number 834405).
Informed Consent
A waiver was requested and obtained for this research project.
