Abstract
Study Design
Prospective comparative study.
Objective
To quantify the educational benefit to surgical trainees of using a high-fidelity simulator to perform minimally invasive (MIS) unilateral laminotomy for bilateral decompression (ULBD) for lumbar stenosis.
Methods
Twelve orthopedic and neurologic surgery residents performed three MIS ULBD procedures over 2 weeks on a simulator guided by established AO Spine metrics. Video recording of each surgery was rated by three blinded, independent experts using a global rating scale. The learning curve was evaluated with attention to technical skills, skipped steps, occurrence of errors, and timing. A knowledge gap analysis evaluating participants’ current vs desired ability was performed after each trial.
Results
From trial 1 to 3, there was a decrease in average procedural time by 31.7 minutes. The cumulative number of skipped steps and surgical errors decreased from 25 to 6 and 24 to 6, respectively. Overall surgical proficiency improved as indicated by video rating of efficiency and smoothness of surgical maneuvers, most notably with knowledge and handling of instruments. The greatest changes were noted in junior rather than senior residents. Average knowledge gap analysis significantly decreased by 30% from the first to last trial (P = .001), signifying trainees performed closer to their desired technical goal.
Conclusion
Procedural metrics for minimally invasive ULBD in combination with a realistic surgical simulator can be used to improve the skills and confidence of trainees. Surgical simulation may offer an important educational complement to traditional methods of skill acquisition and should be explored further with other MIS techniques.
Keywords
Introduction
Minimally invasive surgery (MIS) training as a resident or fellow in spine surgery is dependent on anatomic knowledge in tandem with manual ability to perform each step of a procedure skillfully, reproducibly, and safely. The learning curve for acquiring new techniques is associated with an increased risk of complications and morbidity, even when considering experienced surgeons trying to adopt a newer technique in their practice.1-5 For teaching surgeons, there is a fine balance between allowing a trainee autonomy to thoughtfully and carefully work through a procedure and protecting patient safety, which is the first and foremost priority.
Surgical simulation can complement traditional methods of skill acquisition by providing realistic anatomy, tactile feedback, and discovery of complication management situations that can be practiced on and addressed prior to surgery on a live patient. While simulators have been used in other fields including thoracic surgery, 6 oral maxillofacial surgery, 7 and anesthesia8-10 for many years, there has been a paucity of functional spine models that can realistically simulate the soft tissue, bone, bleeding, and dura routinely encountered during standard lumbar spine surgeries. 11
Our group has recently reported expert consensus regarding the performance of key steps for MIS unilateral laminotomy for bilateral decompression (ULBD) for training purposes (Figure 1).12,13 In this study, our objective was to assess the trainee educational benefit of performing MIS ULBD on a validated, high-fidelity lumbar stenosis model.14,15 We prospectively evaluated their performances in specific surgical tasks and assessed for a change over the course of multiple procedures. Schematic and intraoperative views from the high-fidelity surgical simulator depicting examples of key procedural steps as defined in the AO consensus guidelines for unilateral laminectomy for bilateral decompression (ULBD). (A) Starting point depicting the inferior edge of the superior level lamina and ligamentum flavum interface prior to drilling. (B) Exposure of the ipsilateral ligamentum flavum up to the insertion after hemilaminotomy. (C) “Airplaning” the table and angling the retractor medially to view the contralateral lamina in preparation for the over-the-top drilling. (D) Contralateral ligamentum visualization following contralateral laminotomy in preparation for flavectomy.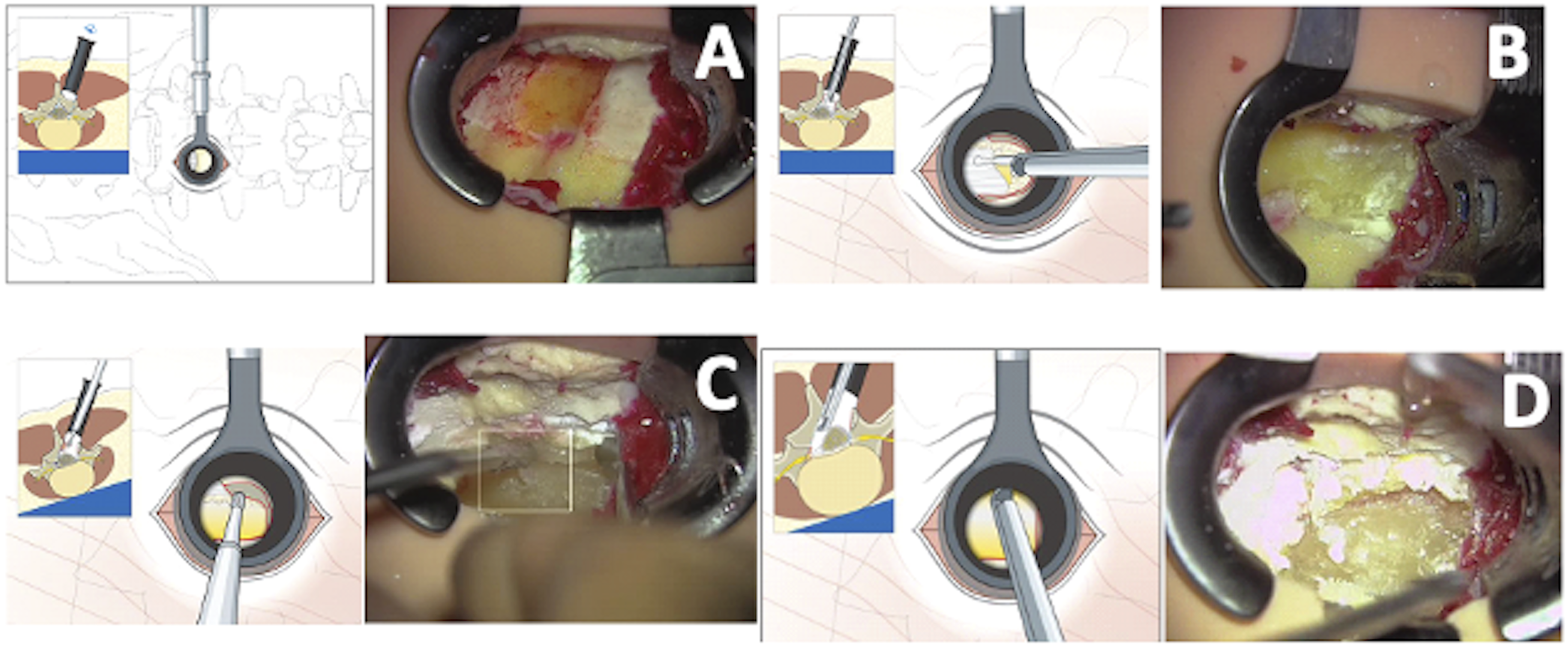
Material and Methods
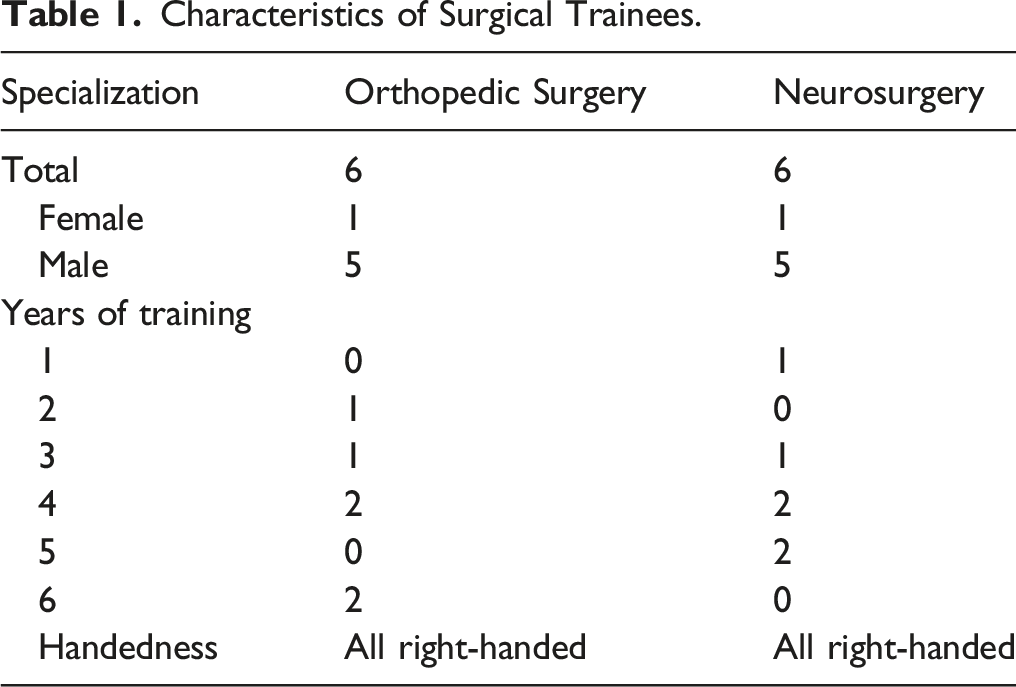
Cohort
Characteristics of Surgical Trainees.
Lumbar Stenosis Simulator
A high-fidelity degenerative lumbar stenosis model (Real Spine, Realists Training Technologies GmbH, Leipzig, Germany) was used for each procedure. We acknowledge a conflict of interest in that the senior author is an investor in this company. Funding for the study was solely provided by the AO Foundation (AO TK 310.04). These models were derived from an MRI scan of an actual 75-year-old female with L4/5 stenosis (Figure 2A-B). The model is entirely synthetic and includes the anatomic structures encountered during MIS decompression including skin, paraspinal muscles, lumbar vertebrae, ligamentum flavum, epidural fat, and dura (Figures 2C-E). The dura contains the cauda equina nerve roots and is filled with water simulating cerebrospinal fluid (CSF). Additionally, the model can simulate bleeding specifically from the muscle layers and epidural bleeding which can be controlled to increase or decrease based on the simulation environment desired. High-fidelity lumbar stenosis surgical simulator. (A) Sagittal and (B) axial T2-weight MRI of a patient with high-grade lumbar stenosis at the L4/5 level due to a combination of disc bulge, facet arthropathy, and ligamentum flavum hypertrophy. The anatomical features of this MRI were used to develop the lumbar stenosis model. Trainees are able to review imaging prior to the operative trials. (C) Photograph of a surgical trainee performing ULBD on the complete simulator (Real Spine, Realists Training Technologies GmbH, Leipzig, Germany) with external housing consisting of skin and subdermal layers to create a more realistic simulation of operating on a human lumbar spine. (D) This simulator consists of a core base (skin cover removed in this picture) with power system and space for bags of simulated blood and cerebrospinal fluid (CSF) which can be manually controlled (for more bleeding or higher-pressure CSF flow). (E) Intraoperative microscopic view of the trainee performing the decompression while managing epidural and bony bleeding.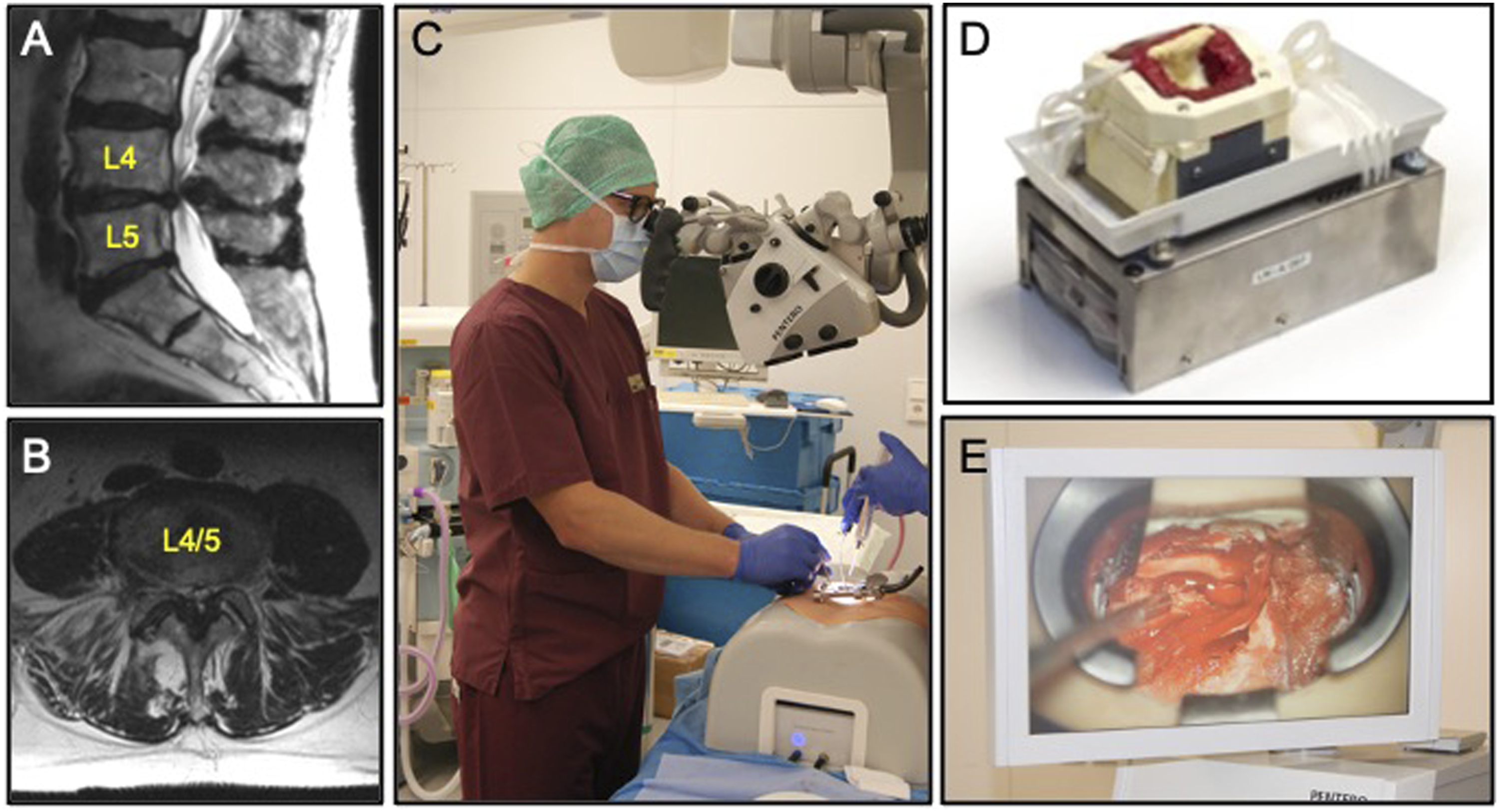
Procedure Set up
Each resident was briefed on the procedure and the evaluation metrics, followed by a practical demonstration by an experienced expert who followed the AO consensus guidelines regarding the standard surgical steps to perform ULBD.12,13 The practical demonstration was scheduled to occur within 24 hours of the trainees first surgical simulation. Over the course of 14 days, each trainee performed the procedure three times. The model was placed on a table with the surgeon facing the defect and the approach side that had been verified by the instructor beforehand with the proper OR surgical setup. Initial trocar, dilators, and retractor were docked and secured by each trainee prior to initiation of the microscopic portion of the case. The instructor verified that the retractor and the specimen were in the ideal position before the trainee began to manually set the microscope according to his or her preferences. The procedure ended when complete decompression was achieved per instructor assessment or when 2.5 hours of operative time passed (whichever came first). The whole operation was recorded through the microscope video feature and the duration of each procedure was noted. After every procedure, trainees were debriefed with individual strengths and areas to target for improvement.
Performance Metrics
Each resident was scored individually by the instructor using the metrics sheet (Supplemental Table 1). Within the metrics, a step was defined as a component task, which constitutes the completion of a specific procedure, while an error was defined as a deviation from optimal performance. A sentinel error on the other hand constitutes an occurrence involving a serious deviation from optimal performance of a procedure that exposes the patient to serious risk but falls short of being a critical error (i.e., life threatening). Within the metric sheet the duration of the different steps was noted as well as the skipped steps, errors, and sentinel errors that occurred during the procedure.
Overall Surgical Proficiency Score
Global Rating Scale Used by the Blinded, Independent Experts to Assess Surgical Proficiency Through Video Analysis of Trainees Performing ULBD on the Surgical Simulator.

Knowledge Gap Analysis
After each procedural trial, participants were surveyed regarding their current ability to perform the procedure and their desired ability using a numerical grading scale. Knowledge gap was calculated as a percentage using the following equation:
Knowledge gap = (current ability score/desired ability score)*100
Wilcoxon matched-pairs signed rank test with two-tailed P-value was used to compare overall knowledge gap scores for the first and last procedures. P-values less than .05 were considered statistically significant. All analyses were performed in SAS v9.4 (SAS Institute Inc, Cary, NC).
Results
Procedure Duration
Surgical time decreased during each of the three procedures. Mean duration of the first procedure was 72.1 (43–130) minutes, while the second and third procedures lasted a mean of 68.6 (35–140) minutes and 40.4 (27–70) minutes, respectively (Figure 3A). There was no significant decrease in operative time between the first and second trials. However, Trial 3 was statistically significantly faster than Trial 1 or Trial 2 (P < .005). Decrease in mean (A) procedure duration, (B) total skipped steps, and (C) errors over the course of successive trials. While the differences between trials 1 and 2 were mild to moderate, there was substantial improvement in all 3 areas when comparing the first and third trials.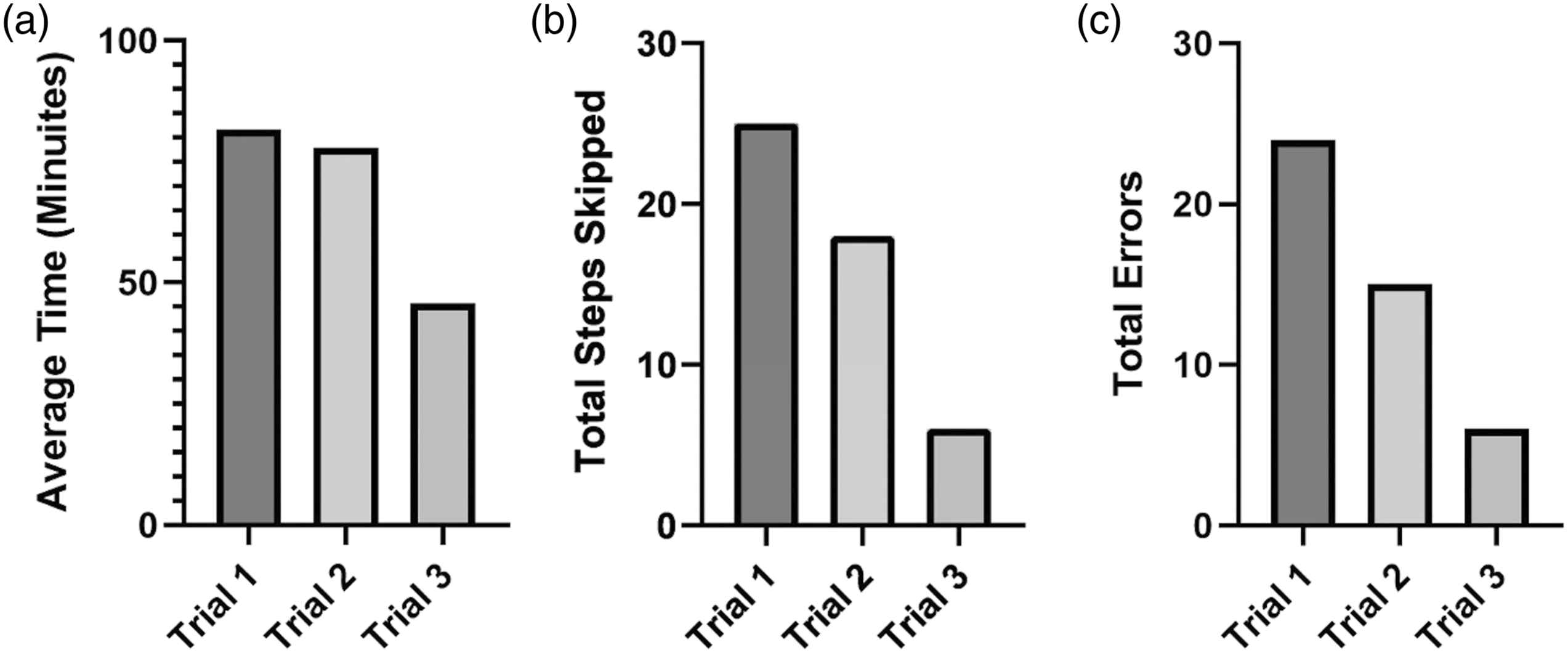
Skipped Steps
While scoring each of the 42 steps on the metrics sheet, skipped or missed steps were noted (Supplemental Table 2). Evaluation of all skipped steps showed recurrence of the same steps being missed intra- and inter-individually with reduction within the three procedures. During the first procedure, a grand total of 25 steps were missed by the twelve residents, in the second the total number of errors were reduced to 18 and even further reduced to 6 in the third procedure (Figure 3B). Of the overall 25 steps missed, the main steps were to “Rotate table away from surgeon” or “sit down to eliminate need to tilt table” (N = 8) and “Inspect disc by medializing dura with ball tip or dissector and determine if discectomy should be performed” (N = 7). Interestingly, these skipped steps were made by almost all residents once and repeated by 2 within the other procedures.
Errors
Overall, 45 errors were recorded across the 36 procedures. During metrics development, 21 errors were defined within the six phases. Of these 21 errors, 10 occurred repeatedly within the procedures of almost all residents, with “Failure to keep microscope centered” and “Failure to change sizes of Kerrison during procedure depending on situation” being the most common (Supplemental Table 3). A decrease in errors was noted with subsequent procedures, with a decrease from 24 errors in the first procedure, to 15 errors in the second, and just 6 errors in the last procedure (Figure 3C).
Details of Sentinel Errors for Trial 1–3 Based on the AO Spine Procedural Metrics.

Procedural Metric Component Scores and Overall Surgical Proficiency
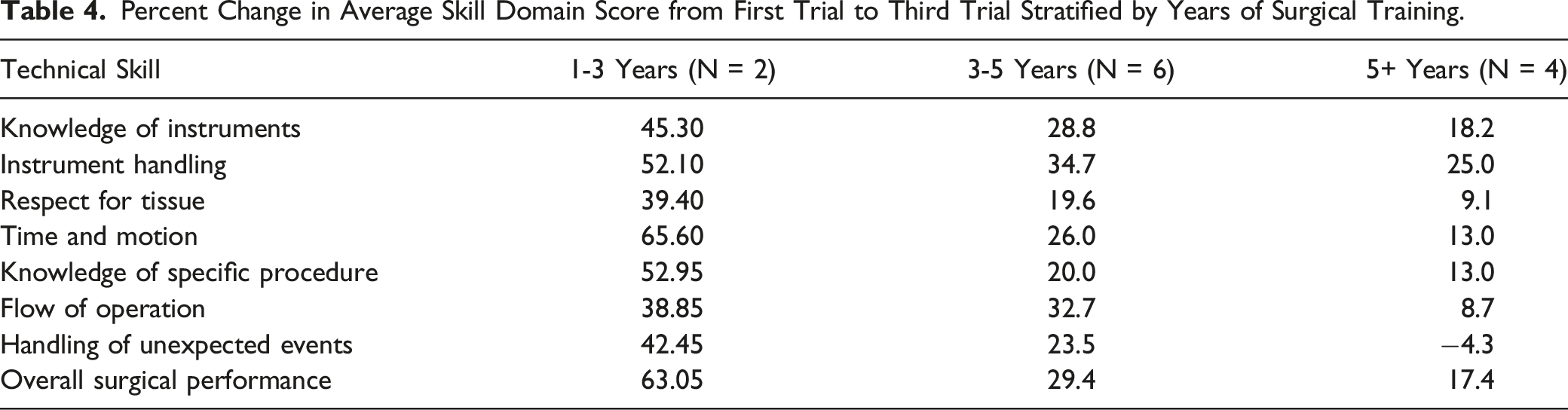
Over the course of the three trials, improvements were noted in each of the aforementioned eight skill domains aforementioned (Figure 4). Taking the mean of the three ratings for each procedure, an improvement of 29.7% (7.1%–71.4%) was seen when comparing the first and third procedure. The increase was the highest within the group of the very unexperienced ( < 1 year) and unexperienced (1–3 years), but still seen in the group of the very experienced participants (Table 4). Furthermore, overall surgical proficiency improved over the course of three trials. Surgical proficiency from first and third trials based on the eight skill domains from the Global Rating Scale. There was improvement in every category, with greatest improvement seen with knowledge of instruments, instrument handling, and overall surgical proficiency. Percent Change in Average Skill Domain Score from First Trial to Third Trial Stratified by Years of Surgical Training.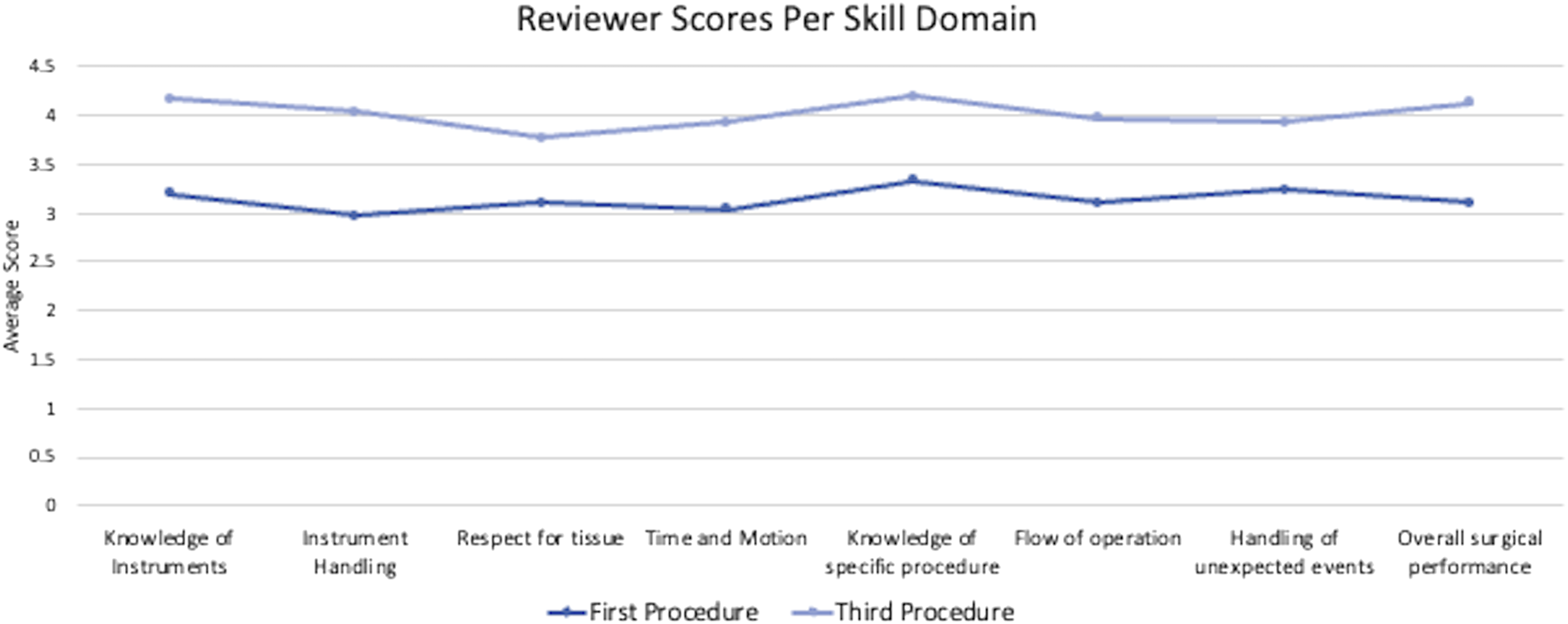
Knowledge Gap Analysis
The overall knowledge gap among participants after the first trial was 60%. This gap decreased by a mean of 30 percentage points after three trials. The difference between the medians of trial 1 and trial 3 was significantly different (P = .001) (Figure 5). The lower the initial performance was rated by the participant, the bigger a decrease was noted. The overall knowledge gap of all participants significantly decreased in the third surgical trial compared to the first trial. Knowledge gap = (current skill level/desired skill level)*100. Two tailed P-value was calculated via Wilcoxon matched-pairs signed rank test. *** indicates P = .001.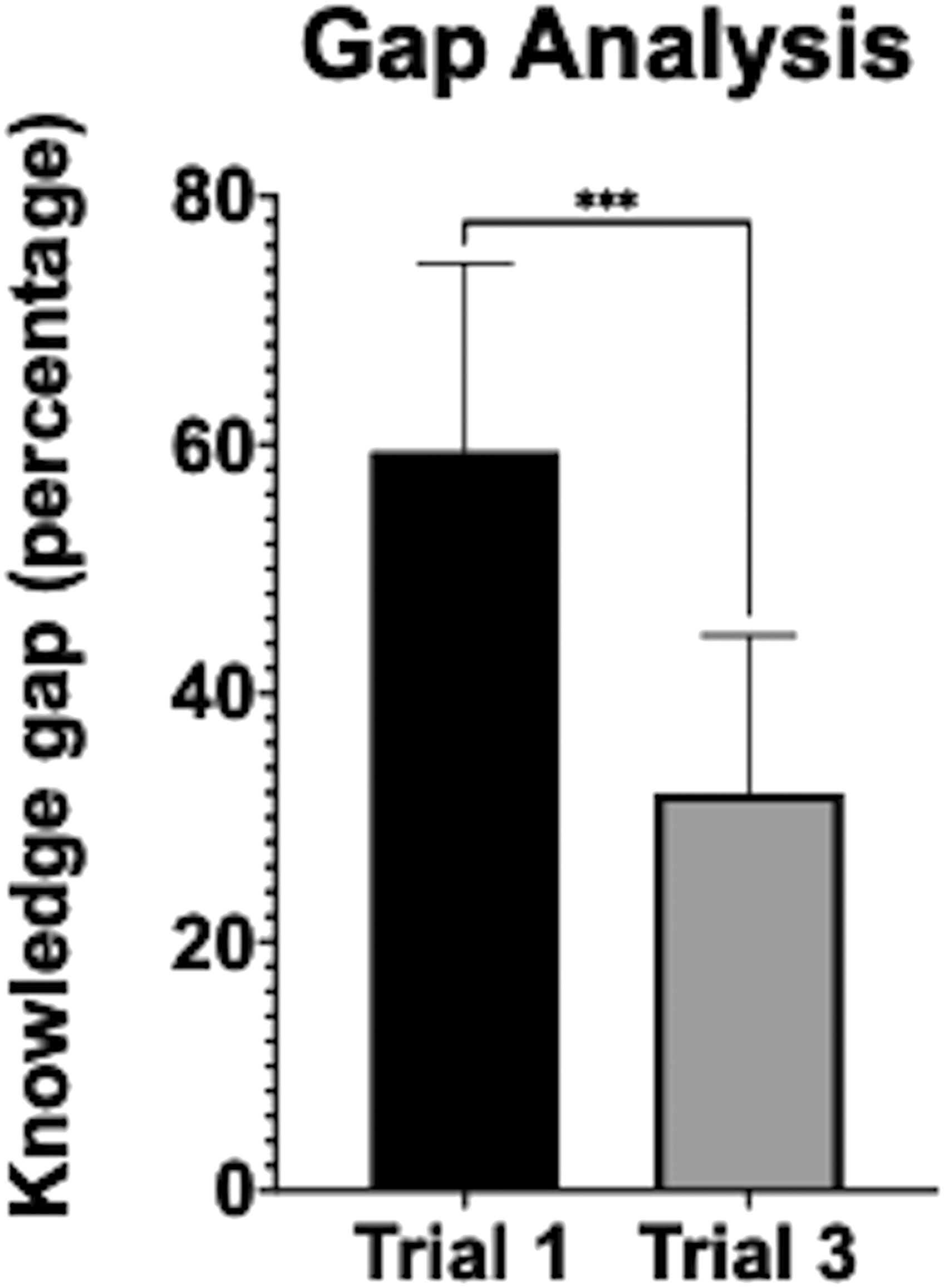
Discussion
Lumbar spinal stenosis is one of the most prevalent causes of disability in the elderly, with over 37 500 surgeries performed annually for treatment at an overall healthcare cost exceeding $1.6 billion. 16 Multiple studies, including retrospective and prospective trials, have demonstrated that surgical intervention is superior to conservative management in short-term and long-term functional improvement and pain relief.17-21 However, these results come at the expense of higher complication rates, chronic low back pain, and the potential for more invasive procedures due to iatrogenic instabilty. 22 Minimally invasive surgery leveraging the intermuscular Wiltse approach was first described in the late 1990s for the treatment of lumbar herniated discs.23,24 From this, grew the technical development of MIS ULBD, which proliferated throughout the 2000s and 2010s.12,23,25-28 Compared with traditional open approaches, MIS ULBD has thoroughly demonstrated lower blood loss, infection risk, postoperative pain, opioid usage, complication rates shorter hospital stays, and faster return to work while maintaining non-inferior clinical outcomes.29-33 Furthermore, preservation of the posterior tension band and paraspinal musculature helps mitigate the risk of iatrogenic instability of the decompressed level, which can lead to revision surgeries and the need for instrumented fusion to adequately address. 30
Despite the reduction in invasiveness of this procedure, the acquisition of skills to perform ULBD through a tubular retractor (ranging in typical use from 18 to 26 mm) or speculum-type retractor requires mastery of certain skills that are outside those typically leveraged for conventional open decompression. Visualization through these narrow corridors constrains orientation due to the lack of adjacent landmarks that can be easily identified. The working corridor is vastly diminished making bimanual maneuvers difficult to perform. Proficiency is required with extended, bayonetted instruments, including drill, cautery, Kerrison , and dissectors. The use of these in conjunction with microscopic magnification at various angles relative to the patient must be mastered in order to perform each key step of the procedure safely and efficiently. Lastly, certain complications can be difficult to address, such as muscular bleeding, epidural bleeding, and most notoriously, dural tears. Despite techniques and alternative instrumentation developed for these situations, 12 they can be daunting for the novice surgeon.
Due to these challenges, the learning curve for MIS can be time-consuming; in some cases taking years to master in appropriately selected patients.1,2,4,5,34-36 Simulations for performing MIS can accelerate learning and provide a safe environment for exploration and practice prior to intervention on live patients. In principle, 3 different types of simulators exist in surgical education: human/animal cadaveric models, virtual reality (VR) (haptic/computerized) applications, and synthetic (physical) models. Apart from ethical aspects, cadavers differ in many characteristics when compared to a living patient, as the simulation of bleeding is not possible and haptic feedback of the tissue is altered. 37 Virtual reality (VR) simulators have more recently been developed, which have partial haptic feedback. 38 While extremely useful for understanding anatomic relationships, these simulators still possess limitations of software-based scenarios and accurate proprioception for the purposes of technical skill acquisition.
Synthetic anatomical models have mostly been used for teaching screw placement as a bone substitute since they are simplified and lack soft tissue and vascular features. 39 However, models that can more precisely recapitulate the true experience of the lumbar spine surgical approach while maintaining flexibility in situational awareness and proprioception have become increasingly sophisticated over the past several years. Adermann et al described their device made of specially designed synthetics which replicated all critical structures encountered during a lumbar discectomy, including the vertebrae, ligamentum flavum, dura, spinal nerves, CSF, intervertebral disc, and disc prolapse. Their simulator had a two-point bleeding system for simulating muscular and laminar bone bleeding. Furthermore sensors were implanted to provide feedback on CSF and neural pressure during retraction of the dura and nerves. 14 A newer generation of this model was recently validated by Mehren et al, who were able to discern experienced vs inexperienced surgeons based on the differences in nerve root traction pressure during microdiscectomy. This simulator has now been adapted for simulations of lumbar spinal stenosis. 15
While the authenticity of a model is an important aspect of training for a particular procedure, costs for state-of-the-art simulators can be prohibitive. High-fidelity simulators can cost between $950 and 1100, whereas partial cadaver torsos with complete lumbar spines roughly cost around $1500 for medical training purposes (excluding storage and preservative expenses). However, simulators, such as the one described in this study, recapitulate pathology at only 1 level. Therefore, accounting for five lumbar levels, the cost per-level for cadavers is roughly 3–3.5x cheaper, and affords the opportunity to deal with variations in anatomy (e.g., narrower laminae in the upper vs lower lumbar levels) and greater versatility for surgical techniques (e.g., anterolateral interbody approaches, pedicle screw insertion) from the same specimen. While it is only a matter of time before high-fidelity simulators overcome the economic drawbacks compared to cadavers, cost-effectiveness still limits their current use in training at many medical universities and low- and middle-income countries (LMICs).
Besides the actual medium used for simulation, defining metrics for evaluation is the other critical half of optimally utilizing models for assessment and determination of technical expertise and understanding. Medical and surgical specialties outside of spine, including orthopedics, neurosurgery, general surgery, and gastroenterology, have devised validated metrics for various procedures they perform in order to enhance education for trainees.40-46 Most of these have been devised by expert consensus and Delphi methodology, and furthermore establish a proficiency benchmark.41,47 However, as Guyer et al. noted, the lack of formal parameters for defining success in minimally invasive spine surgery due to the absence of universally accepted outcome measures was a hindrance to furthering education. 48
To formally address this, the significant steps of procedural metrics for a minimal invasive ULBD were defined by our group in 2020. 13 These metrics were developed through multinational expert consensus but had yet to be validated in a real teaching situation. To see if procedural metrics enhance performance of trainees of different levels and of different training backgrounds, a double-armed study was initiated using the simulator for analogous OR simulation. We also assessed if differences in the training programs in Europe and the US were comparable. Our data demonstrated significant improvement in the overall surgical proficiency of spine surgery trainees performing MIS ULBD.
Equally important to learning curves, retention of what has been learned is needed when evaluating learning strategies and design interventions. Retention of skills is a better indicator of actual learning than performance during practice because the goal of training is consolidated skills and a consistent performance. That said, skill consolidation and a consistent improved performance was shown by all participants regardless of their experience, specialty, and teaching/geographical background. Though our study is small, these results are encouraging and suggest a possible complementary role for simulation in the broader scheme of MIS training.
Determining which types of residents will most benefit from simulation of this nature will take further study. In the past, different simulators have been rated differently by surgeons based upon the surgeon’s personal level of experience. Junior residents in postgraduate years 1–3 reportedly benefit the most from simulation, while senior residents benefit from computerized simulators, followed by cadavers and physical models.49,50 In our study, our sample number was too low to draw robust conclusions regarding the utility of simulation among trainees of differing levels. However, we did find a trend in greater improvement in total scores with decreasing experience. These data suggests that the use of high-fidelity models may have the most impact early in training. It also raises the possibility that more senior trainees benefitted less due to higher baseline scores with a corresponding decrease in the range of improvement achievable by our metrics, or that they have already established habits which are more resistant to modification. Nonetheless, the significant overall improvement in surgical proficiency among our diverse population sample demonstrate that all trainees could stand to benefit from simulation.
Over the years, neurosurgical training in the US has evolved from a sheer volume of exposure to one of structured curricula. Subsequently, additional aspects were considered important and the Accreditation Council for Graduate Medical Education (ACGME) implemented the Milestone program, a measurable competency-based matrix in which mastery is based on completion of agreed-upon milestones and not presupposed by year. 51 Still, differences in training programs were prevalent, which was further addressed by implementation of a Neurosurgical “boot camp” and transitioning all programs nationwide to a mandatory 7-year track.51,52 Considering the differences between training programs as well as specialties in spine surgery (neurosurgery and orthopedic surgery), exposure to different techniques including MIS is anticipated
Data from the American Academy of Orthopedic Surgeons and the American Association of Neurological Surgeons reveals that spinal procedures comprise approximately 14% of orthopedic, but up to 60% of neurosurgical practice. 53 In 2019 these numbers differed even more as neurosurgical residents performed 3.6 times more total spine procedures than orthopedic surgery residents on average, a number that increased from 2.8-fold in 2009 to 6.0-fold in 2018. 54 Even though no tangible numbers exist, exposure to spine is rare for orthopedic residents in Europe. Differences in technical abilities of the decompression procedure between residents from the US, who were neurosurgeons, and the ones from Europe, who were orthopedic surgeons, were expected for these reasons.
However, all participants regardless of their training background showed improvement in every measure and every test from the first procedure onwards with reduction of errors, skipped steps and OR time, as well as superior results in the instructor survey, gap analysis and global rating scale. Accordingly, the universal applicability of the metrics combined with the realistic RealSpine simulator demonstrates an acceptable teaching method for a standardized minimally invasive lumbar stenosis decompression. Another aspect of experience level and simulation that was not addressed by this study is the potential benefit for trained surgeons seeking to gain expertise with minimally invasive techniques. A systematic review by Scalfani et al demonstrated that even in experienced “open” spine surgeons, the learning curve for adoption of MIS techniques in terms of operative time and complication reduction was 20–30 cases. 1 We can only postulate that simulation using this model could potentially accelerate the learning curve, given that fellows and experienced spine surgeons are already acquainted with the anatomy, instruments, and their optimal usage during specific portions of the case. Future studies using this study design in advanced spine surgeons naïve to MIS techniques will be explored.
Earlier studies on simulation-based learning postulated that task complexity might be an important factor that may mediate the relationship between instructional elements and simulation‐based learning outcomes. Haji et al. used a simple vs complex lumbar puncture simulation in medical students and found that reduced task complexity was associated with superior performance and lower cognitive load during skill acquisition and retention. 55 Although novices seemingly plateau early during simulation-based training, there is some evidence that continued practice increases automaticity of the procedure and reduces the cognitive load of the learner. In general, theoretical knowledge seems to be retained better than practical skills, and complex tasks abate more easily over time than simpler ones.56-58 In our study, even though the cognitive load seemed high in the beginning, especially for novices, the participants were found to perform superiorly over time. Improvement was seen using the metrics in combination with the simulator from the second procedure. The parameters were mostly technical, but a significant decrease in OR time and errors was seen that can be correlated to the clinical findings.
Limitations
Our study is limited in several ways. First, while findings were consistent across study participants, our limited sample size limits the generalizability of these findings. Second, while trainees performed the dilation and retractor placement, this step was checked by the instructor prior to initiating the microscopic portion of the case and therefore was not included in the scoring. This initial step of any MIS tubular surgery is key, and poor technique here can cause undue damage to the soft tissue and position the surgeon in the wrong anatomical location when initiating the decompression. There is also some limitation of the model in that dilating through the paraspinal musculature is not as realistic as one would expect in a live patient, with less density and tension in the model. Third, the extent of decompression was not objectively measured. The models are not imaging compatible due to the hypodense nature of the material used for the “bony” anatomy and opening the model for visual inspection of the lamina was not performed. Fourth, while this simulator is high-fidelity and appears to be an optimal model system, we did not track whether participants were able to translate these skills into the operating room. Finally, participants were assessed over the course of 14 days. Ideally, we would assess applicants over a longer period of time to assess whether the benefits of simulation are durable. These will be key issues to address in a future study.
Supplemental Material
Supplemental Material – Use of a High-Fidelity Training Simulator for Minimally Invasive Lumbar Decompression Increases Working Knowledge and Technical Skills Among Orthopedic and Neurosurgical Trainees
Supplemental Material for Use of a High-Fidelity Training Simulator for Minimally Invasive Lumbar Decompression Increases Working Knowledge and Technical Skills Among Orthopedic and Neurosurgical Trainees by Carolin Melcher, Ibrahim Hussain, Sertac, Kirnaz, Jacob L. Goldberg, Fabian, Sommer, Rodrigo Navarro-Ramirez, Branden Medary and Roger Härtl in Global Spine Journal
Supplemental Material
Supplemental Material – Use of a High-Fidelity Training Simulator for Minimally Invasive Lumbar Decompression Increases Working Knowledge and Technical Skills Among Orthopedic and Neurosurgical Trainees
Supplemental Material for Use of a High-Fidelity Training Simulator for Minimally Invasive Lumbar Decompression Increases Working Knowledge and Technical Skills Among Orthopedic and Neurosurgical Trainees by Carolin Melcher, Ibrahim Hussain, Sertac, Kirnaz, Jacob L. Goldberg, Fabian, Sommer, Rodrigo Navarro-Ramirez, Branden Medary and Roger Härtl in Global Spine Journal
Supplemental Material
Supplemental Material – Use of a High-Fidelity Training Simulator for Minimally Invasive Lumbar Decompression Increases Working Knowledge and Technical Skills Among Orthopedic and Neurosurgical Trainees
Supplemental Material for Use of a High-Fidelity Training Simulator for Minimally Invasive Lumbar Decompression Increases Working Knowledge and Technical Skills Among Orthopedic and Neurosurgical Trainees by Carolin Melcher, Ibrahim Hussain, Sertac, Kirnaz, Jacob L. Goldberg, Fabian, Sommer, Rodrigo Navarro-Ramirez, Branden Medary and Roger Härtl in Global Spine Journal
Footnotes
Author Contributions
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Roger Härtl is an investor in Realists.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by a grant from the AO Foundation (AOTK 310.04).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
