Abstract
Study Design:
Animal experimental model.
Objective:
To study the clinical behavior and histological changes in the spinal cord, nerve roots and perivertebral muscles of the spine after induced leakage of polymethylmethacrylate (PMMA) loaded with antiblastic drugs during vertebroplasty in an animal model of pigs.
Methods:
We performed vertebroplasty on 25 pigs. The animals were divided into 3 groups: vertebroplasty with PMMA alone (control group), vertebroplasty with PMMA loaded with methotrexate (MTX) and vertebroplasty with PMMA loaded with cisplatin (CYS). At 2 vertebral levels, epidural and prevertebral, massive cement leaks were induced. Animals were evaluated daily. Two weeks later, the pigs were sacrificed, and the tissues that came in contact with the cement were analyzed.
Results:
The clinical results for each of the groups were reported. The control group had no clinical alterations. In the MTX group, 2 pigs died before 1 week due to pneumonitis. In the CYS group, 4 animals had motor impairment, and 3 of the 4 had paraplegia. The histological results were as follows: the control and MTX groups showed synovial metaplasia, inflammatory reaction, crystal deposits, and giant cell reaction in the dura mater and muscle and all the animals in the CYS group had spinal cord and muscular necrosis.
Conclusions:
Massive cement leak after vertebroplasty with PMMA loaded with cisplatin is extremely toxic to the spinal cord and muscles around the spine. Therefore, its use cannot be recommended for the treatment of vertebral metastases. Using PMMA loaded with methotrexate seems to be a safe procedure, but further research is needed.
Introduction
Early diagnosis and treatment improvements have resulted in longer survival of patients with cancer but also in an increase in the incidence of vertebral metastases. 1 Vertebral metastasis can cause pain or neurological impairment. Pain is the most common first symptom and can be caused by fracture, inflammation or nerve root or spinal cord compression. 2 Treatment of spinal metastases requires a multidisciplinary approach and mainly depends on the expected survival, spinal stability and involvement extent. There are tools that help guide clinicians through the process of treatment choice, such as the Bartels’ life expectancy model 3 or the SINS score. 4 Treatment of spinal metastases is generally palliative, seeking to alleviate pain. Radiotherapy is the cornerstone of vertebral metastasis treatment, and in the past few years, stereotactic radiotherapy has emerged as an alternative to classic external beam radiotherapy, achieving better pain relief and local control. 5 However, it has a higher incidence of vertebral fractures.
Surgery for spinal metastases can improve pain, deformity, neurologic function and quality of life. 6 There has been a trend in recent years to use minimally invasive techniques such as percutaneous instrumentation, cement augmentation techniques and decompression techniques. Although currently the most frequent indication for vertebroplasty is osteoporotic vertebral compression fractures, this technique was initially developed in the 1980s to treat tumors in the spine. 7 Cement augmentation achieves pain relief in spine metastases by reinforcement and stabilization of the fracture. 8 Both vertebroplasty and kyphoplasty techniques achieve rapid pain control in those patients without response to conservative treatment, disabilities related to metastasis improve and decrease opioid consumption. 9 In addition in vitro studies have demonstrated that polymethylmethacrylate (PMMA) has some cytotoxic effects. 10 Recently a clinical study has shown that after vertebroplasty there was not anatomopathological evidence of cement-induced tumor necrosis. 11
One characteristic of PMMA is the capability of drug diffusion. Since the 1970s, antibiotics have been added to PMMA to treat bone infections. In the late 1980s, the first studies using PMMA with methotrexate were published by Hernigou. 12 In vitro studies have proved that methotrexate has antitumoral effect in 9 different cancer types. 13 In a previous experimental study, we found that after vertebroplasty, methotrexate was released, with plasma levels peaking early within 30 minutes and reaching maximal concentration at 1 week; cisplatin showed similar kinetics with plasma levels peaking at 24 hours. 14 By adding antineoplastic drugs to cement the beneficial effect of augmentation techniques in the treatment of spinal metastasis could be improved.
The main complication of vertebroplasty is cement leakage. For cancer-related compression fractures, cement leakage can occur in 22-70% of cases treated with vertebroplasty and 9-14% of cases treated with kyphoplasty. 9 In cases where antiblastic drugs are added to PMMA when treating spine metastases, cement leakage could be more harmful. The adverse effects of cement migrating to the canal or to other locations are compounded by the toxicity of the drugs. The purpose of this study was to evaluate the clinical and organic effects of massive leakage of cement containing antineoplastic drugs, specifically in myeloradicular tissues and prevertebral musculature after vertebroplasty.
Materials and Methods
Study Design: An Experimental Study in Pigs
We obtained approval for this study from the local institutional review board committee for animal investigation (CIFA 039/05). We followed all the rules for animal comfort, like free delivery of food and water, we took care on alleviation of preoperative and postoperative pain and distress and we followed all the endpoint or euthanasia indications by the Animal Welfare Information Center.
Animals
Twenty-five Large White-Landrace adult female pigs weighing 30-35 kg were provided by a professional stockbreeder and housed in stalls. The animals were divided into 3 groups. Five animals were included in the control group in which vertebroplasty using PMMA without any additives was performed; the rest of the animals were divided into 2 groups: the first group underwent vertebroplasty using cisplatin-containing cement, and the second group underwent vertebroplasty using methotrexate-containing cement.
Surgical Procedure
We based our technique on a previously published model of epidural and prevertebral leakage of polymethylmethacrylate for vertebroplasty in pigs.14,15
We administered preanesthetic sedation in the stall (intramuscular ketamine 10 mg/kg). After that, we transported the animals to the operating room where general anesthesia was administered (intramuscular azaperone 2 mg/kg, atropine 1 mg intramuscularly, intravenous etomidate 3 mg/kg and pancuronium 0.2 mg/kg) followed by orotracheal intubation. Anesthesia was maintained with isoflurane 0.8-1% plus oxygen, intravenous pancuronium 0.2 mg/kg and fentanyl 0.05 mg/kg every 30 minutes.
We used a radiolucent table on which the animals were placed in a prone position. The skin of the back was shaved and cleaned with soap, disinfected with chlorhexidine, and draped with sterile surgical fields. Ampicillin 1 g was administered as an antibiotic prophylaxis before incision. For guidance, we used a radioscopic C-arm (Powermobil® Siemens, Erlagen, Germany) in the anteroposterior and lateral views. Two nonconsecutive vertebrae were selected between L1-L4.
13G trocars (Osteo-Site, Cook, IN, USA) were used to perform cement injection. To obtain cement contact with prevertebral and myeloradicular tissues, 2 different types of cement leaks were created. In the cranial vertebra, anterior leak was made by introducing the trocar through the left pedicle at a near 90° angle trajectory in the lateral view until the anterior cortex of the vertebra was penetrated. Then, in the caudal vertebra, the trocar was introduced through the right pedicle from the lateral to the medial direction, seeking to penetrate the medial cortex of the vertebral body at a 30° angle trajectory to produce an epidural leak. The location of the trocar tip was confirmed in the AP and lateral views with injection of radiological contrast agent (Radialar®) (Figure 1).
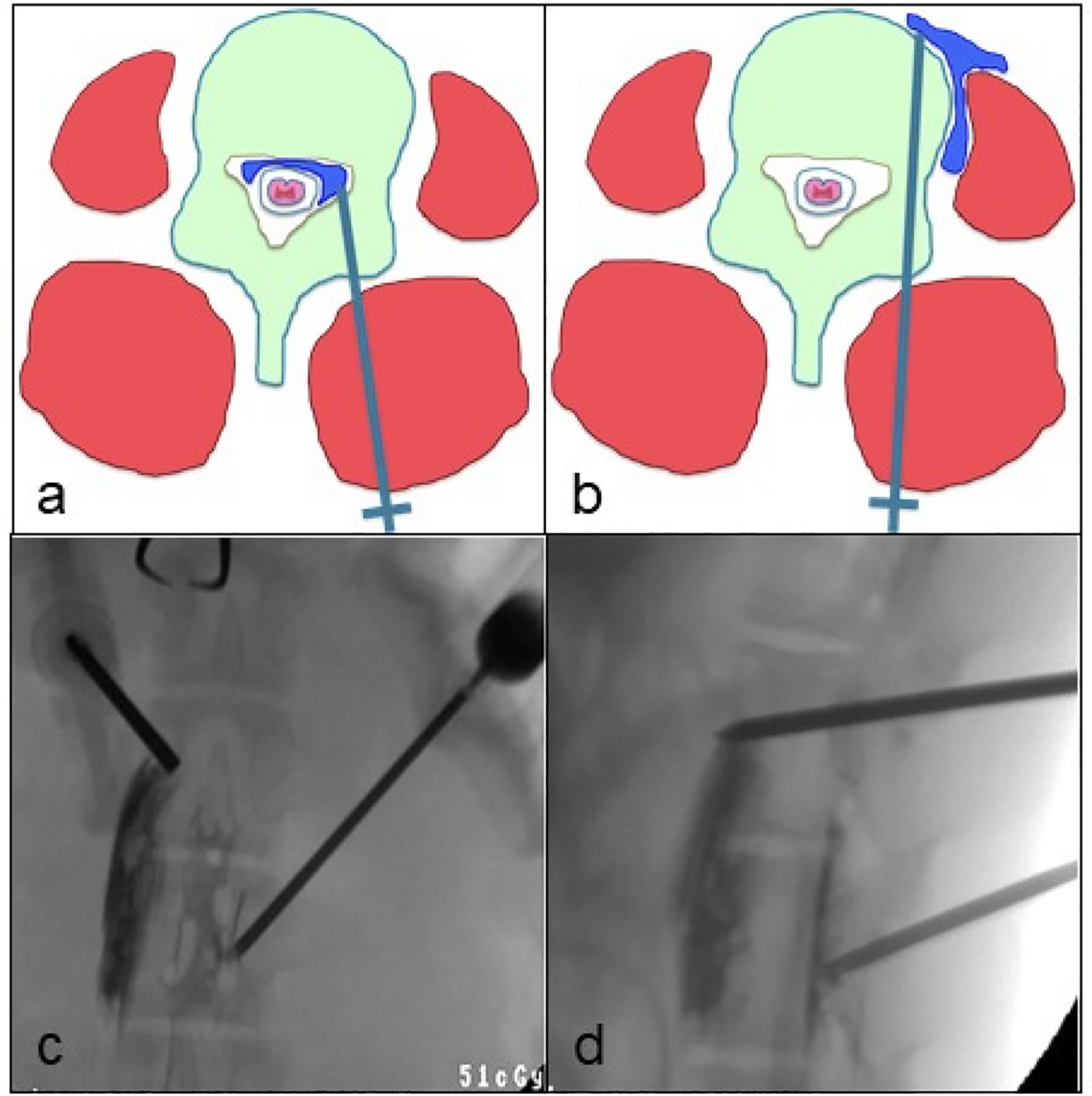
A. Example of epidural injection of PMMA. b. Example of prevertebral, perimuscular injection of PMMA. c and d. AP and lateral views showing epidural cement placement and prevertebral muscle cement location.
Cement was manually prepared using 22.5 g acrylic cement powder polymer (polymethylmethacrylate, PMMA) and 9 mL of liquid monomer. Three different mixtures were made (Vertebroplastic, De Puy Spine, Raynham, MA, USA). The first mixture, for the control group, was cement without any additions. For the second mixture, 500 mg of powdered cisplatin was added to the powder polymer. For the third mixture, 1000 mg of powdered methotrexate was used. With a luer-lock syringe, 1.5 cc of acrylic cement (1 mg of cisplatin or 2 mg of methotrexate) was injected into each pedicle under fluoroscopic guidance. Finally, the skin was disinfected with chlorhexidine and sutured with surgical silk 3/0. Buprenorphine 0.005 mg/kg was administered postoperatively as analgesia.
Clinical Study
We returned the animals to the stall and observed them for general status. We measured time to return to consciousness and time to stand up. Limb motor function was evaluated using the Tarlov scale 16 (Table 1). The animals were observed during the following days for any motor function impairment or any other clinical signs. We followed the rules for assessment and alleviation of postoperative pain and endpoint or euthanasia criteria for animals exhibiting signs of pain behavior, paraplegia, and dying.17,18 If any of these criteria appeared, then the animals were sacrificed.
Tarlov Motor Scale.

Euthanasia
Animals were sacrificed 2 weeks after surgery with an intravenous injection of sodium pentobarbital 18% 200 mg/kg. We utilized a posterior approach to dissect the lumbar spine of each animal. After location of the trocar trajectories, the spine, including the anterior muscles, was resected. We performed an extensive laminectomy and carefully extracted the cement in contact with the dural sac. The anterior cement leaks were located by manual palpation. Areas of muscle and dural sac in contact with cement were marked with Chinese ink (Figure 2).
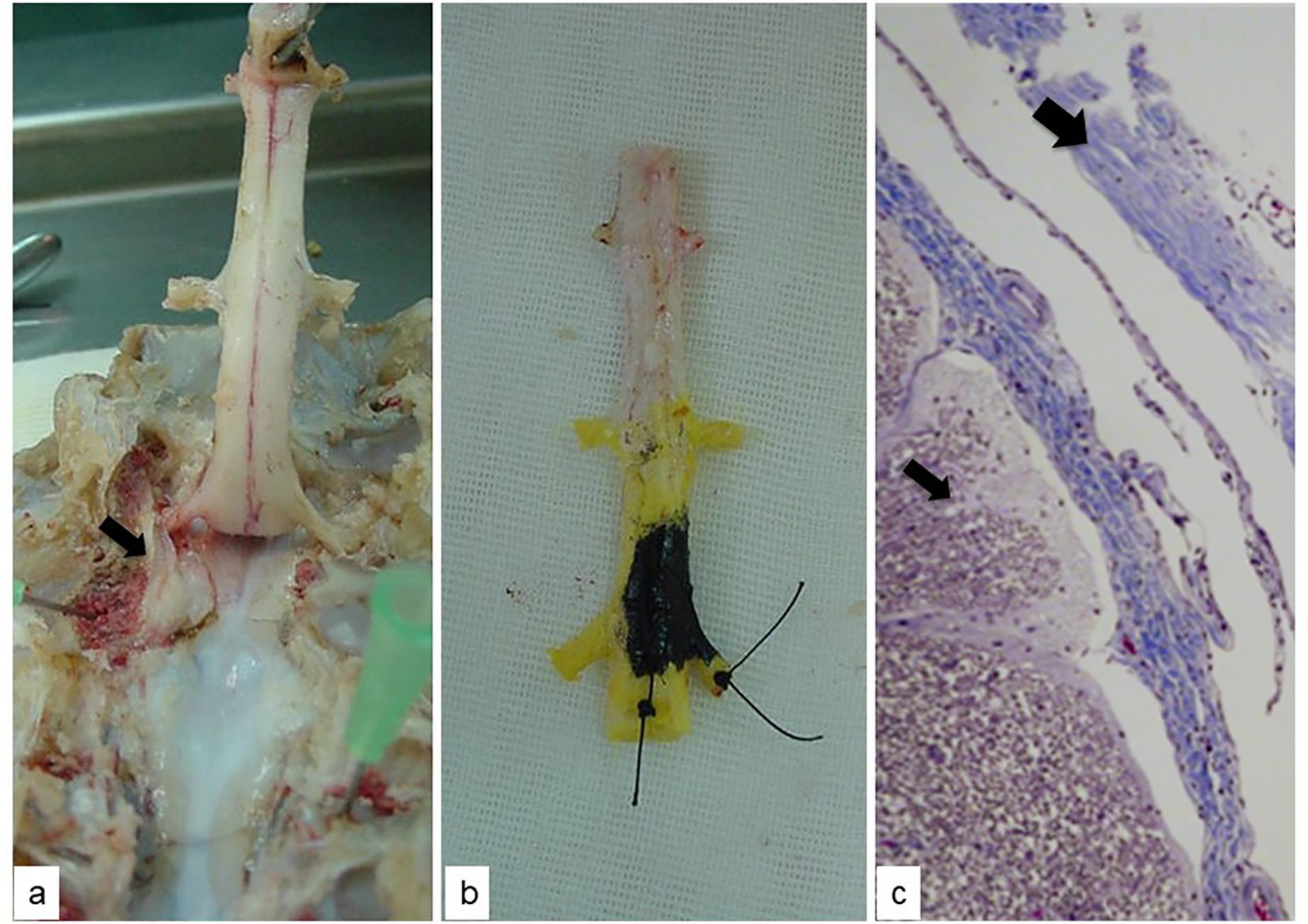
A. Release of the spinal cord after laminectomy. The arrow shows the PMMA in the epidural space near a pedicle. b. Spinal cord surrounded by dura mater marked with Chinese ink over the areas that contact the PMMA. c. Histological HE study of the spinal cord (thin arrow) and dura (wide arrow) that came into contact with the cement 4x magnification.
Histologic Study
All tissue samples were placed in formaldehyde. Then, each tissue sample was dehydrated with ethanol, fixed in wax, cut with a microtome and stained with hematoxylin–eosin (HE). An experienced pathologist inspected all tissue samples using a light microscope (Carl Zeiss; AG, Oberkochen, Germany). Samples were evaluated, and in case abnormal histology was found, the lesion was graded as mild, moderate, or severe. Digital photographs were taken of each sample.
Statistical Analysis
Differences between groups were compared using nonparametric statistics. A p value of <0.05 was considered statistically significant. Statistical analyses were conducted with SPSS® v. 23
Results
Surgical Technique and Anesthesia
All the animals showed an excellent tolerance to anesthesia and the surgical procedure. There were no major complications during the procedure. The mean time to return to consciousness was 43 minutes (10-65 minutes), and the mean time to stand up after the procedure was 83 minutes (30-240 minutes). No differences between groups were observed.
Clinical Results
General status
None of the animals in the control or the cisplatin group showed symptoms that could indicate any organic pathology due to adverse effects of the treatment with vertebroplasty.
Five animals in the methotrexate group presented symptoms that indicated pulmonary complications. Animal n° 95 had a cyanotic episode 24 hours after surgery that resolved spontaneously, but 48 hours later, the animal died. Necropsy showed pneumonic infiltrate in the right lung. Animal n° 97 had an episode of motor agitation 72 hours after surgery that resolved spontaneously. Animal n° 99 had a fever 2 weeks after surgery followed by breathing difficulty, anorexia and petechia on ears, limbs, and thorax. This animal was sacrificed, and necropsy showed pneumonia in the right lung. Animal n° 100 also had fever and breathing difficulty 2 weeks after surgery, so the animal was sacrificed, and necropsy confirmed pneumonia. Animal n° 79 had fatigue, fever, diarrhea and breathing difficulty 7 days after surgery. The animal was also sacrificed, and necropsy also showed pneumonia.
Limb motor function
None of the animals in the control group showed limb motor impairment. All animals had the maximum score on the Tarlov scale during follow-up.
In the methotrexate group, only 8 animals completed a follow-up of 2 weeks. Of these, 7 animals obtained the maximum score on the Tarlov scale. Only 1 animal presented a score of 4/5 24 hours after surgery and then recovered completely. The 2 animals that were sacrificed due to pneumonia did not show any motor impairment after surgery.
The cisplatin group presented the highest number of animals with motor dysfunction. Four animals had motor impairment in the immediate postoperative period. One animal showed an unsteady gait during all follow-up periods. The other 3 animals had progressive motor dysfunction, all of them presenting with paraplegia 3 days after surgery.
Comparing the motor function changes between the 3 groups, we found the worst outcomes in the cisplatin group, which were statistically significant (p = 0,028 by Pearson’s χ2).
Macroscopic Changes in Muscle and Dural Sac
Control group
In all animals, cement was found in a laminar distribution around the dural sac and with a fusiform distribution inside the prevertebral muscle. After removing the cement, no macroscopic changes were visible in the dural sac compared to segments not in contact with cement. In the prevertebral muscle, cement created a cavity with smooth walls.
Methotrexate group
In all animals, cement was found around the dural sac and inside the prevertebral muscle. The distribution was the same as in the control group; the only appreciable change was that the cement had a yellowish color, while in the control group, the cement was white. No macroscopic changes in the dural sac were found, and the same type of cavity as in the control group was seen in the muscle.
Cisplatin group
In this group, cement was found in the peridural space in 8 of the 10 pigs. The cement distribution was the same as in the other 2 groups. In 2 animals, the dural sac showed a purple coloration in the area in contact with cement.
Histological Changes
Control group
The analysis of the dura mater showed crystal deposits in 4 of 5 animals. Additionally, an inflammatory reaction with the presence of lymphocytes and polymorphonuclear cells was observed. Some samples showed thickening and synovial metaplasia of the meninges in the areas in contact with cement. The spinal cord did not show any histological changes. All the samples of muscular tissue in contact with cement presented well organized and thick synovial metaplasia with atrophy of muscular cells.
Methotrexate group
Eight pigs completed the 2 weeks of follow-up, and we observed the same histological changes as in the control group. In 6 animals, crystal deposits were found in the dura mater, and 4 animals presented an inflammatory reaction with polymorphonuclear cells. Six animals had synovial metaplasia. The spinal cord was normal in all animals. All the animals had synovial metaplasia and crystal deposits in muscular tissue. Additionally, muscular fibrosis was observed in the 8 animals.
Cisplatin group
The study of the dura mater showed a moderate inflammation reaction in the meninges. Only 1 animal presented crystal deposits and other giant cell reactions. None of the animals had synovial metaplasia. Seven out of 8 animals with PMMA within the epidural space had variable grades of necrosis of the spinal cord, 3 had mild necrosis, one had moderate necrosis and 3 had severe necrosis. Necrosis was described as lysis of cellular membranes, loss of normal medullar architecture, presence of spongious cells (liquefactive necrosis) and presence of hyaline microthrombi (Figure 3).
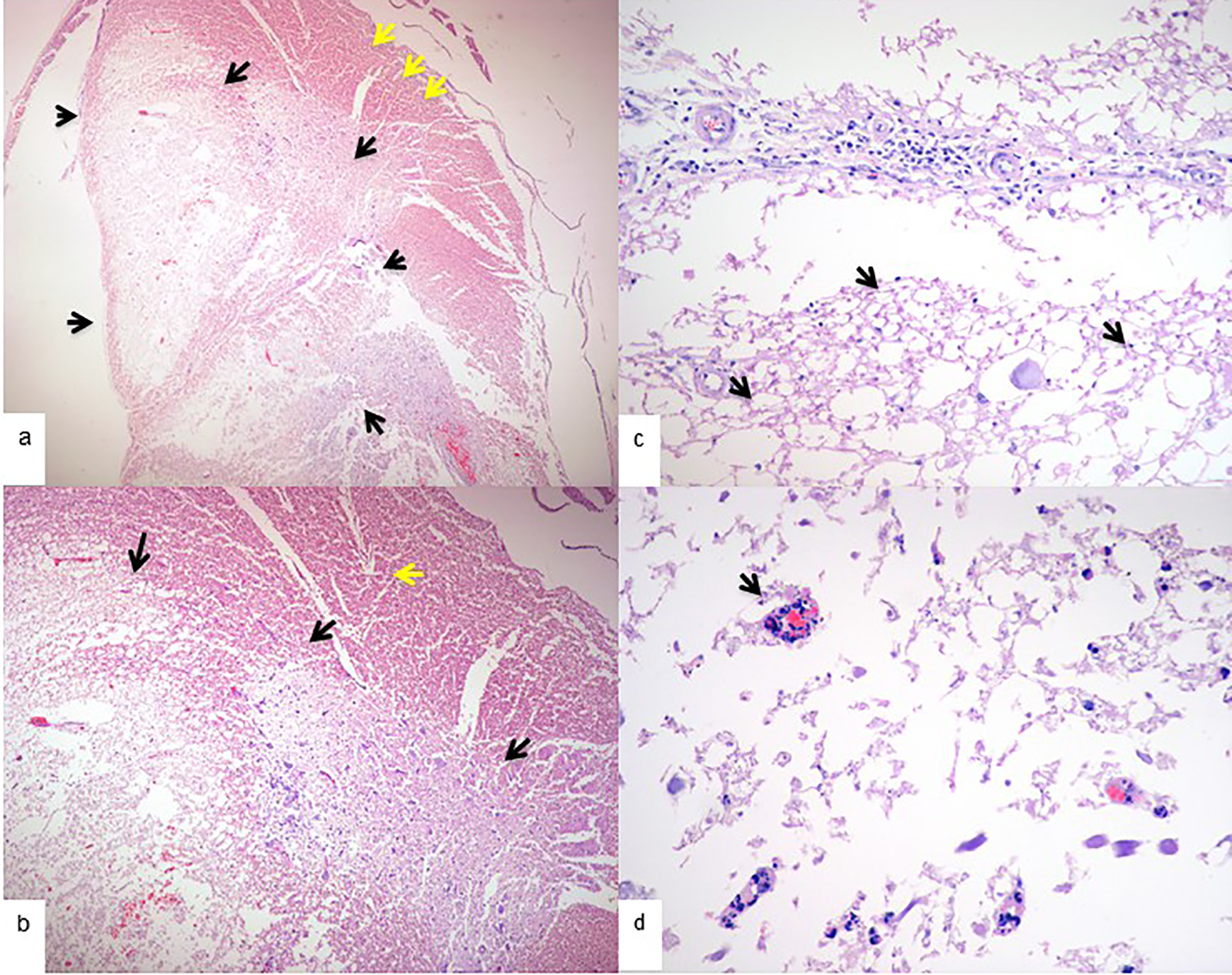
Hematoxylin and eosin microphotography of the spinal cord of a cisplatin group animal. a. 2x magnification, black arrows show lysis of cellular membranes, loss of normal spinal cord architecture, presence of spongious cells (liquefactive necrosis) and presence of hyaline microthrombi. b. 4x magnification, black arrows show spinal cord necrosis, yellow arrow shows normal spinal cord tissue. c. 20x magnification, arrows show spongious cells and membrane lysis. d. 40x magnification, the arrow shows hyaline microthrombi.
In muscle tissue, crystal deposits were found in 8 animals, while synovial metaplasia was found in only 4 animals. In all animals, muscular necrosis was mild to severe (Figure 4). Additionally, wide areas of dystrophic calcifications and granular tissue were observed. In 1 animal, regenerative muscle fibers were observed.
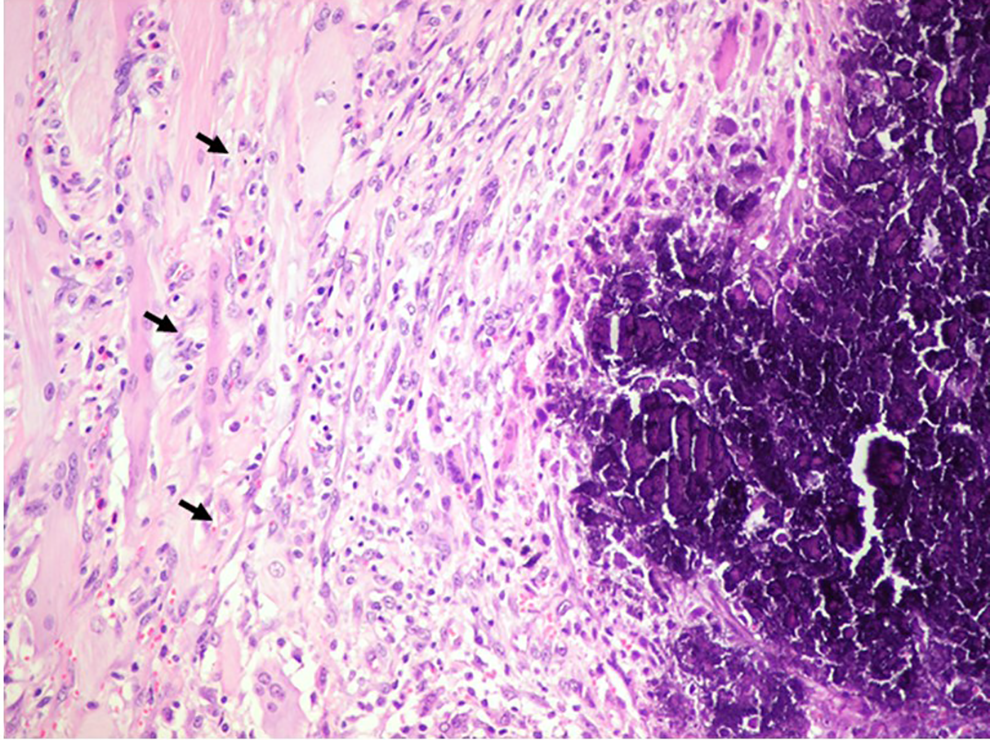
a. 20x magnification of HE microphotograph of paravertebral muscle of a cisplatin group animal. the arrows show wide areas of muscular necrosis.
Correlation Between Clinical Behavior and Spinal Cord Histological Findings
The control and methotrexate groups did not show any histological changes and had no motor function impairment. In the cisplatin group, 4 out of 8 animals with intracanal cement leakage had neurologic deterioration. These animals had spinal cord necrosis, 1 moderate and 3 severe. The animals without neurologic deficits had mild necrosis. When we compared spinal cord necrosis in the animals with worse motor impairment, we found that those with moderate and severe necrosis had worse motor dysfunction (paraplegia), which was highly significant (P < 0.001 Pearson’s χ2).
Discussion
To our knowledge, this is the first study to evaluate the possible consequences of cement leakage after vertebroplasty using cement enhanced with antineoplastic drugs. We found large differences when using either cisplatin or methotrexate. Cisplatin-containing cement can lead to severe sequelae such as paraplegia when it reaches the myeloradicular tissues. On the other hand, the use of methotrexate has been shown to be locally secure when cement leaks from the vertebral body into the canal.
None of the animals in the control group had motor impairment after surgery. We injected a high volume of cement into the peridural space, but it was distributed in a laminar fashion and did not cause spinal cord compression. Histological changes observed in the dura mater of the animals in the control group were inflammatory changes characterized by the presence of numerous lymphocytes and multinuclear giant cells. These changes are similar to those described by Urrutia 19 in rabbits and by Togawa 20 in human vertebrae. The results of the histologic study also revealed synovial metaplasia in the dura mater and muscle tissues; to the best of our knowledge, synovial metaplasia of the dura and muscle has not been described in the literature. This finding is characteristic of interfaces between tissues and mobile implants. 21 In our previous study, 15 we found similar results, and we concluded then that the laminar distribution summed to the pulsed compression effect by the dural sac could have avoided the focal accumulation of cement so that there was not enough volume to compress the spinal cord. In 2011, the temperatures of bone cement in the spine canal were measured after vertebroplasty with cement inside the vertebral body and leaking into the canal. 22 The authors concluded that cement leakage might result in thermal injury, but this was an ex vivo model using only the vertebral body without the surrounding soft tissues. Our results are contrary to that hypothesis, as a large volume of cement injected into the canal did not result in clinical complications, and only mild histological changes were found in the dura mater. Most of the previously published literature focuses on the thermal effect on bone. 23 It is likely that the dura mater and cerebrospinal fluid are sufficient to insulate neural structures from the cement.
Methotrexate and cisplatin are the most commonly studied drugs for elution from PMMA. 24 Their properties and kinematics when mixed with PMMA have been well studied in vitro and in vivo. Methotrexate is used to treat patients with leukemia, lymphoma, and osteosarcoma, among others. Although neurotoxicity is one possible adverse effect of MTX, it is low, and MTX is safe enough to be administered intrathecally. 25 Methotrexate can produce leukoencephalopathy, different demyelination degrees, white matter necrosis, oligodendroglia loss, axonal swelling, microcystic encephalomalacia, and deep cerebral white matter atrophy. 26 Our results showed that cement with methotrexate leaks in the canal did not affect the motor function of the animals. Only 1 animal had transient mild motor impairment. The histological changes we found were similar to those in the control group. Therefore, considering our results, adding MTX to PMMA does not increase the risk of spinal cord damage and could be locally safe in the treatment of pathological fractures due to vertebral metastases. The high rate of pulmonary complications observed in this group, makes it necessary to demonstrate the potential advantage of the substance in the palliative therapy of spinal metastases. Methotrexate antitumor therapy plays an important role in the treatment of chemosensitive tumors such as Ewing’s sarcoma, osteogenic sarcoma, and lymphoma. 27 However, it has a limited effect on bone marrow in the spine because its bone distribution is limited due to low blood flow and the blood-bone marrow barrier that prevents the entry of MTX. This is why new forms of MTX delivery systems are being studied. 28 In vitro studies have shown that MTX has antitumoral effect by by tumor inhibition, stasis, or even regression. 13 More experimental studies are needed to establish the real effect of local administered MTX into vertebral metastasis and to establish a secure dose of MTX with local action but without systemic damage.
In contrast, in the cisplatin group, we observed wide areas of spinal cord and muscle necrosis. Clinically, 4 pigs presented neurological compromise that can be related to local toxicity rather than cement compression. Cisplatin has poor penetration through the blood-brain barrier; for this reason, the drug crosses the blood-brain barrier in small amounts, and peripherical neuropathy is more commonly produced. 29 Namikawa found that cisplatin caused neural and vascular lesions shown as hemorrhagic foci accompanied by necrosis and edema in the spinal cord in rabbits. 30 Our study of the spinal cord showed necrosis in all animals with lysis of cellular membranes, loss of normal medullar architecture, presence of spongious cells (liquefactive necrosis) and presence of hyaline microthrombi. Previously, in our diffusion study, we had one cement leak into the canal in the cisplatin group, and a histological study was performed. 14 Spongiosis and nuclear pyknosis, but not necrosis, were observed. This may be explained because the leak was unintended, and therefore, the concentration of cisplatin around the spinal cord was not enough to produce necrosis.
We decided to use this porcine experimental model because the anatomy and size of the pigs’ vertebrae are very similar to human vertebrae. This allows us to perform the exact vertebroplasty technique and use the same cannula as we use in human patients. This model can easily be translated into clinical applications. We deliberately provoked malposition of trocars to produce cement leakage. As we do in our clinical practice we used fluoroscopy to guide trocar insertion. In pathological vertebral fractures anatomy can be altered and the pedicles are not always easy to identify. New navigation technologies can help the surgeon to avoid trocar malplacement in those cases 31 and also reduce surgery time and radiation dose. 32
Our study has some limitations. In our model of massive cement leak into the spinal canal and into the prevertebral muscles, the possibility of causing embolism is higher than in normal practice. Four animals in the methotrexate group had respiratory problems. Necropsy showed pneumonic pulmonary infiltration. In those animals, cement emboli could not be detected in the lungs, and pulmonary infiltration might be due to a decrease in immunity due to a high dose of MTX.
Conclusion
Leakage of PMMA loaded with cisplatin during vertebroplasty is extremely toxic to the spinal cord and paravertebral muscle. Therefore, its use cannot be recommended for the treatment of vertebral metastases.
Using PMMA loaded with methotrexate seems to be a locally secure procedure, but further research is needed to establish a safe systemic dose.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
