Abstract
Study Design:
Multicenter retrospective study.
Objectives:
To investigate adverse events (AEs) in patients with neuropathic pain related to lumbar disease who switched to mirogabalin from pregabalin.
Methods:
This study surveyed the records of 82 patients with peripheral neuropathic leg pain related to lumbar disease who switched to mirogabalin from pregabalin. We evaluated AEs associated with pregabalin and mirogabalin, the continuation rate of mirogabalin, and the pain-relieving effect at 4 weeks after switching from pregabalin to mirogabalin. We compared patients who switched due to lack of efficacy (LoE group) and patients who switched due to AEs (AE group).
Results:
The incidence rates of somnolence and dizziness with pregabalin were 12.2% and 14.6%, respectively, while the incidence rates with mirogabalin were reduced to 7.3% for somnolence and 4.9% for dizziness. The incidence of AEs with pregabalin was significantly higher in the AE group (LoE group: 11.1%, AE group 100%), especially for somnolence (LoE group: 3.2%, AE group: 47.1%) and dizziness (LoE group: 4.8%, AE: 52.9%). After switching, the incidences of AEs with mirogabalin were not significantly different between the 2 groups (LoE group: 15.9%, AE group: 23.5%), including for somnolence (LoE group: 7.9%, AE group: 5.9%) and dizziness (LoE group: 4.8%, AE group: 5.9%). There were no significant differences in continuation rate of mirogabalin or the pain-relieving effect between groups.
Conclusions:
The patients who experience somnolence and dizziness with pregabalin might be able to continue safely receiving treatment for their pain by switching to mirogabalin.
Introduction
The number of people with chronic pain, including low back pain and neuropathic pain, is increasing due to our aging society. Treatments for neuropathic pain are reported to have the potential to improve physical and mental quality of life in middle-aged and older people. 1 The prevalence of neuropathic pain for chronic low back pain has been reported to be as high as 36.6%. 2
Pregabalin was developed as a treatment for neuropathic pain, exhibiting rapid peak blood levels and better bioavailability, and has been widely used for the treatment of neuropathic pain. However, pregabalin causes adverse events (AEs), including somnolence (22.8%), weight gain (22.0%), and dizziness (20.3%). 3 Somnolence and dizziness are particularly problematic because they increase the risk of falling, which is an issue for older people who already have frailty and/or locomotive syndrome.
Mirogabalin is an orally administered gabapentinoid for the treatment of peripheral neuropathic pain, including diabetic peripheral neuropathic pain and post-herpetic neuralgia.4,5 Mirogabalin induces persistent pain relief due to its high affinity and slow dissociation from the dorsal root ganglion–expressed α2δ-1 subunit. In addition, mirogabalin has a low affinity for the α2δ-2 subunit, which is found primarily in the cerebellum, and it is rapidly dissociated from the subunit, resulting in lower levels of central nervous system–specific AEs such as somnolence and dizziness. 6 The drug is approved in Japan for the treatment of peripheral neuropathic pain and is in clinical development for this indication elsewhere in Asia.6-9 Although the incidence of AEs due to mirogabalin was expected to be lower than that of pregabalin, 7 there have been few reports on the safety of mirogabalin in individuals with lumbar disease in clinical practice. Therefore, the purpose of this study was to investigate AEs in patients with neuropathic pain related to lumbar disease who switched to mirogabalin from pregabalin.
Materials and Methods
This was a retrospective observational study conducted at 6 institutions. We conducted this study in compliance with the principles of the Declaration of Helsinki. The study’s protocol was reviewed and approved by our institutional review board. The opt-out method was adopted to obtain informed consent from the subjects.
This study surveyed the records of patients with peripheral neuropathic leg pain who were treated from April 2019 to June 2020. The inclusion criteria were (1) magnetic resonance imaging or computed tomography scans of lumbar spine that explain the neuropathic pain, (2) sensory impairment in the area related to the impaired nerve, (3) switching from pregabalin to mirogabalin, and (4) no change in concomitant painkillers other than pregabalin. The exclusion criteria were (1) patients who underwent surgery within 4 weeks of switching from pregabalin to mirogabalin, (2) patients who underwent nerve block, including epidural block, caudal block, or nerve root block within 4 weeks of switching, or (3) patients with spinal tumors, infections, inflammatory diseases, or fractures.
The initial dose of mirogabalin was 10 mg daily, and the dose was gradually increased to 20 mg daily at intervals of 1 week or longer to 30 mg daily. The doses were increased or decreased by the attending physicians depending on each patient’s age and symptoms.
We evaluated AEs associated with pregabalin and mirogabalin, the continuation rate of mirogabalin (whether mirogabalin was continued) at 4 weeks after switching from pregabalin to mirogabalin, and the pain-relieving effect at 4 weeks after switching from pregabalin to mirogabalin. The pain-relieving effect was noted by the attending physician during an interview 4 weeks after the patient switched from pregabalin to mirogabalin, and the effect was assigned as “improvement,” “no change,” or “deterioration” compared to before switching.
For statistical analyses, SPSS Statistics version 26.0 (IBM Corp., Armonk, NY, USA) was used. Values were expressed as average ± standard deviation. For comparisons between 2 groups, we used Student t test or Fisher’s exact test. The level of significance was set at less than 5%.
Results
Patient Demographics
Among the 85 patients who met the study criteria, 3 were excluded due to insufficient data. Thus, the total number of study subjects was 82 patients (41 males and 41 females), with an average age of 65.3 years (range, 23-92). The specific lumbar disease diagnoses were lumbar spinal stenosis (n = 62) and lumbar disc herniation (n = 20). The reasons for switching from pregabalin to mirogabalin were lack of efficacy in 63 patients (LoE group), AEs with pregabalin in 17 patients (AE group), and unknown in 2 patients (Figure 1).
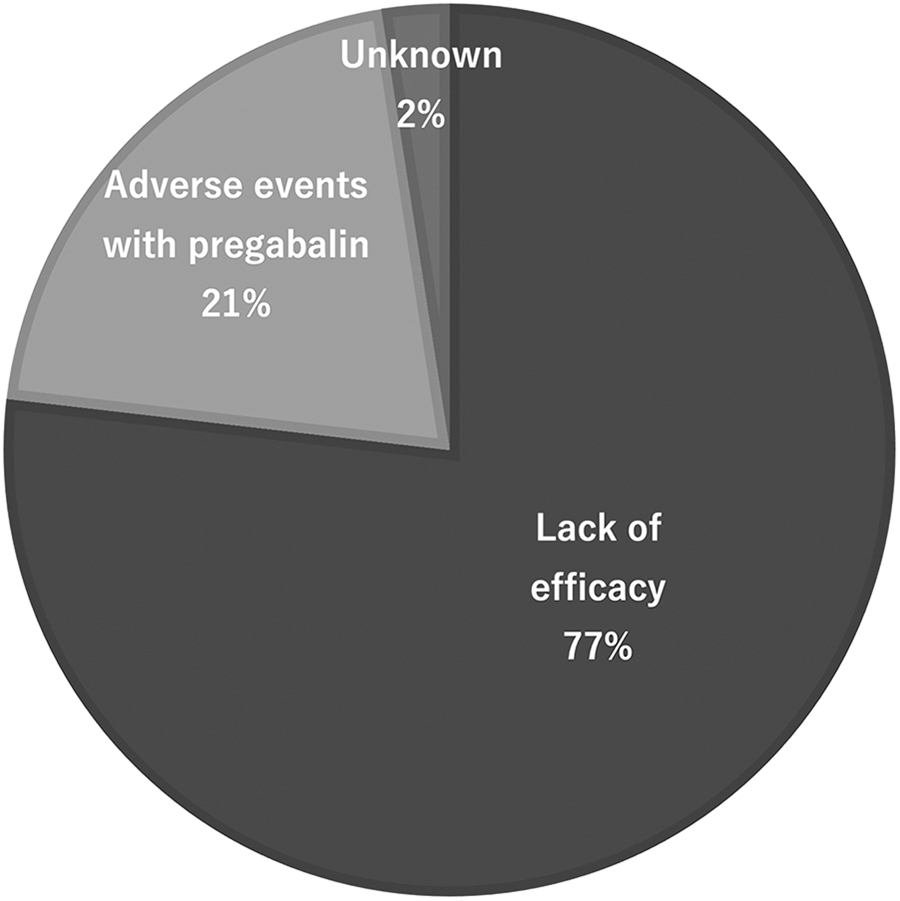
The reasons for switching from pregabalin to mirogabalin were lack of efficacy in 63 patients (77%), AEs with pregabalin in 17 patients (21%), and unknown in 2 patients (2%).
The average pregabalin dose before switching was 154.0 ± 100.3 mg/day (25-525 mg/day). Seventy patients received concomitant medication. The concomitant medications were loxoprofen (n = 11; dose, 60-180 mg/day), celecoxib (n = 9; 200 mg/day), diclofenac sodium (n = 2; 50-75 mg/day), acetaminophen (n = 31; 325-3600 mg/day), tramadol (n = 29; 37.5-300 mg/day), duloxetine (n = 8; 20-60 mg/day), an extract from inflamed cutaneous tissue of rabbits inoculated with vaccinia virus (n = 18; 16 units/day), limaprost alfadex (n = 12; 10-15 μg/day), mecobalamin (n = 4; 1500 μg/day), buprenorphine tape (n = 3; 5-10 mg/week), and fentanyl tape (n = 1; 4.2 mg every 3 days) (Table 1).
Patient Demographics.a
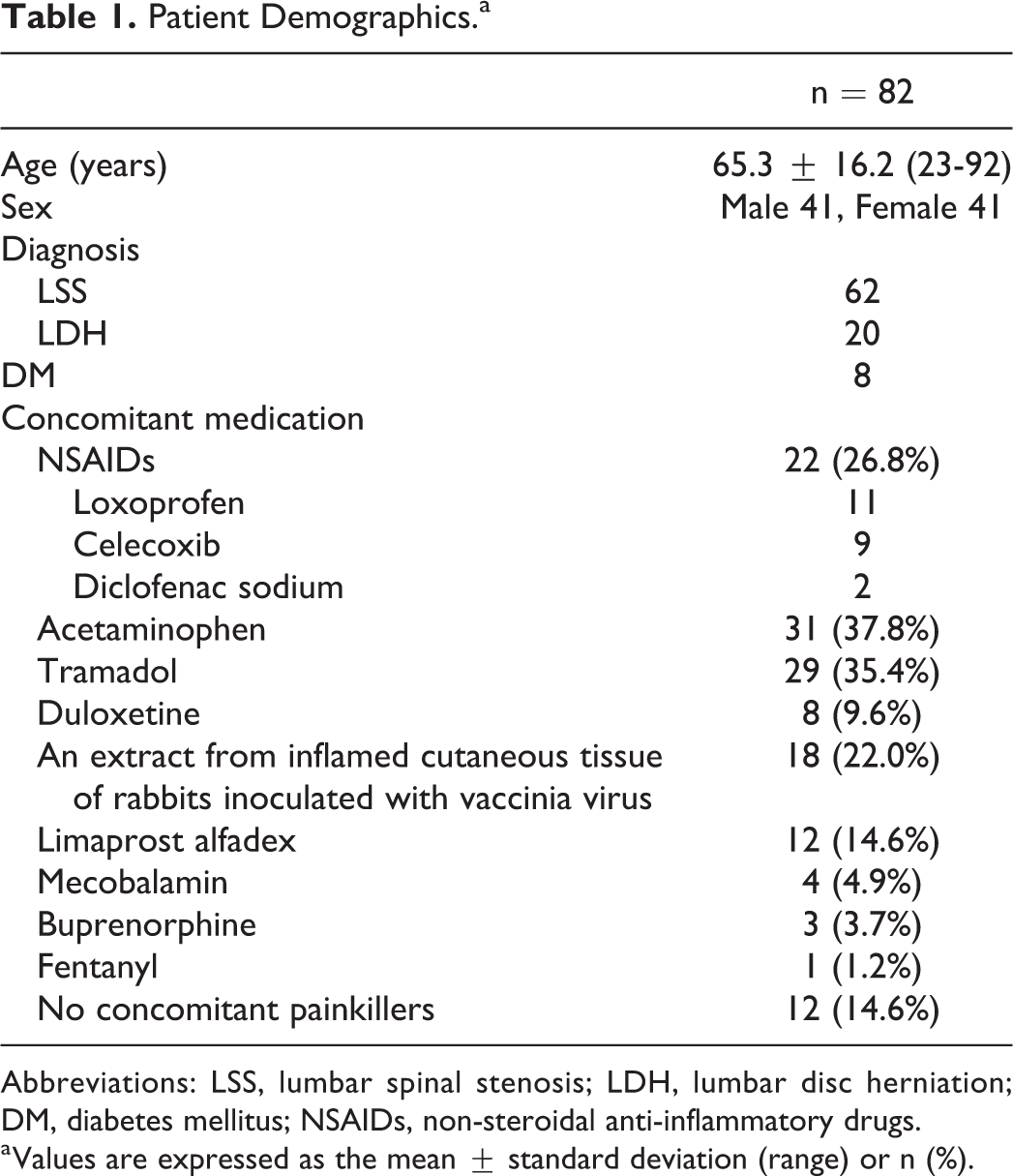
Abbreviations: LSS, lumbar spinal stenosis; LDH, lumbar disc herniation; DM, diabetes mellitus; NSAIDs, non-steroidal anti-inflammatory drugs.
a Values are expressed as the mean ± standard deviation (range) or n (%).
Treatment of Mirogabalin
The initial dose of mirogabalin was 10 mg/day in all patients. The average maximum dose was 14.9 ± 6.5 mg/day (range, 10-30 mg/day). Seventy-three patients (89.0%) continued to take mirogabalin for 4 weeks after switching from pregabalin. The pain-relieving effect based on the interview was improvement in 42 patients (51.2%), no change in 39 (47.6%), and deterioration in 1 (1.2%).
Adverse Events
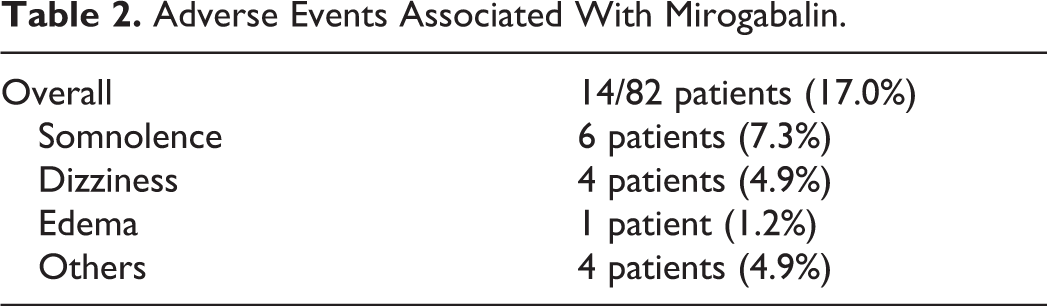
AEs before switching from pregabalin to mirogabalin were observed in 24 patients (29.3%): 10 patients experienced somnolence (12.2%), 12 patients experienced dizziness (14.6%), 1 patient experienced edema (1.2%), 2 patients experienced nausea (2.4%), and hyperamylasemia, numbness, heartburn, and diarrhea were each experienced by 1 patient.
AEs associated with mirogabalin were observed in 14 patients (17.0%): 6 patients experienced somnolence (7.3%), 4 patients experienced dizziness (4.9%), 1 patient experienced edema (1.2%), and nausea, numbness, heartburn, and tachycardia were each experienced by 1 patient (Table 2).
Adverse Events Associated With Mirogabalin.
Comparisons Between Groups That Switched for Different Reasons
We compared 63 patients who switched due to lack of efficacy (LoE group) and 17 patients who switched due to AEs (AE group). Two patients whose reasons were unknown were excluded from these comparisons.
There were no significant differences in age, sex, diagnosis, or history of diabetes mellitus (DM) between the LoE and AE groups (Table 3).
Comparisons of Demographics Between Groups That Switched for Different Reasons.a
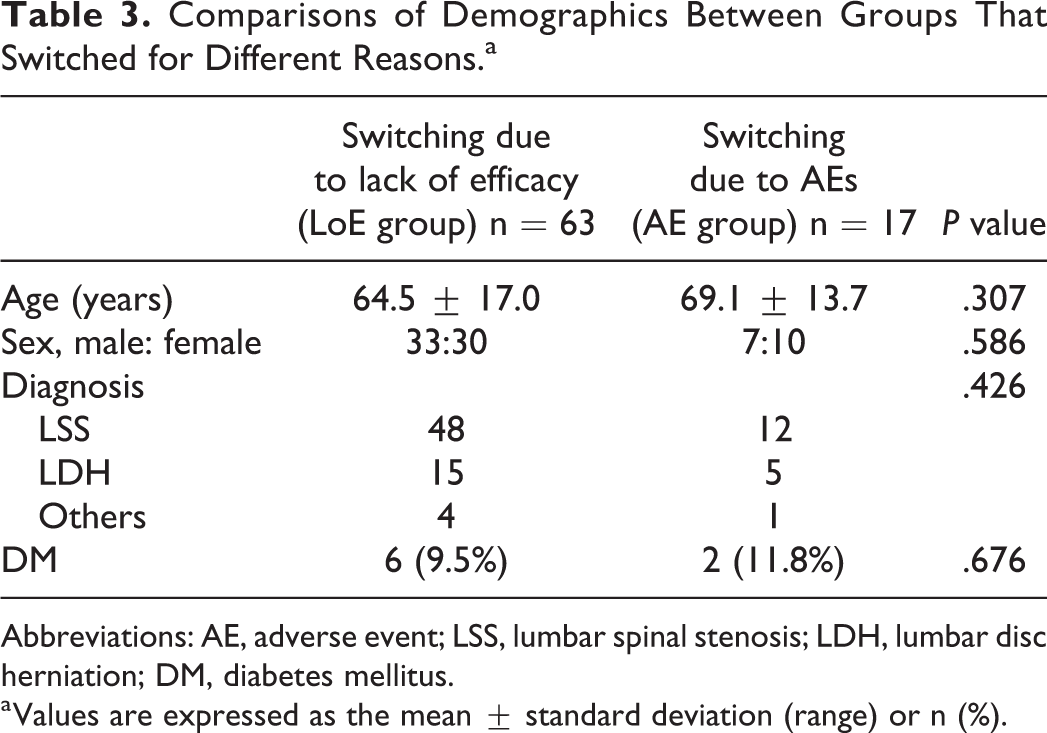
Abbreviations: AE, adverse event; LSS, lumbar spinal stenosis; LDH, lumbar disc herniation; DM, diabetes mellitus.
a Values are expressed as the mean ± standard deviation (range) or n (%).
The dose of pregabalin was lower in the AE group, but it was not significantly different from the LoE group. The incidence of AEs associated with pregabalin was significantly higher in the AE group (LoE group: 11.1%, AE group 100%; P < .001), especially for somnolence (LoE group: 3.2%, AE group: 47.1%; P < .001) and dizziness (LoE group: 4.8%, AE: 52.9%; P < .001). After switching, the maximum doses of mirogabalin were 15.8 ± 6.8 mg/day in LoE group and 11.8 ± 3.9 mg/day in AE group. The incidences of AEs associated with mirogabalin were not significantly different between the 2 groups (LoE group: 15.9%, AE group: 23.5%; P = .482), including for somnolence (LoE group: 7.9%, AE group: 5.9%; P = 1.00) and dizziness (LoE group: 4.8%, AE group: 5.9%; P = 1.00). There was no significant difference between the incidence of AEs with pregabalin and the incidence of AEs with mirogabalin in the LoE group. There were no significant differences in continuation rate of mirogabalin or the pain-relieving effect between groups (Table 4).
Comparisons of Dose, AEs, Continuation of Mirogabalin, and Pain-Relieving Effect Between Groups That Switched for Different Reasons.a
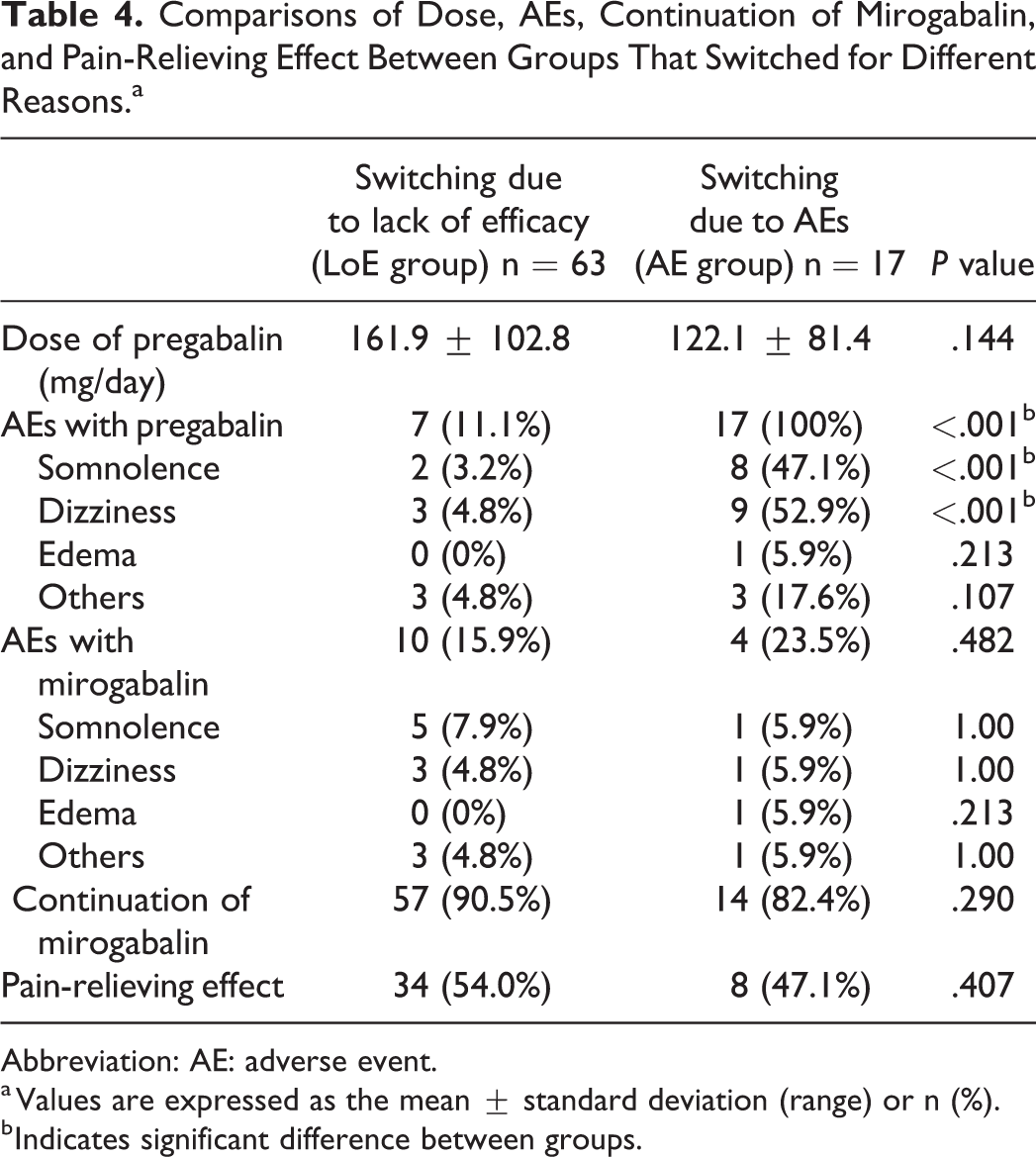
Abbreviation: AE: adverse event.
a Values are expressed as the mean ± standard deviation (range) or n (%).
b Indicates significant difference between groups.
The overlap of AEs was examined. There were 1 patient (1.6%) in the LoE group and 1 patient (5.9%) in the AE group who had somnolence with both drugs. There were 2 patients (3.2%) in the LoE group and 1 patient (5.9%) in the AE group who had dizziness with both drugs. There were no patients (0%) in the LoE group and 1 patient (5.9%) in the AE group who had edema with both drugs.
There were no significant differences in concomitant medication rates of nonsteroidal anti-inflammatory drugs (NSAIDs; including loxoprofen, celecoxib, or diclofenac sodium), acetaminophen, tramadol, duloxetine, an extract from inflamed cutaneous tissue of rabbits inoculated with vaccinia virus, or limaprost alfadex between groups (Table 5).
Comparisons of Concomitant Medication Between Groups That Switched for Different Reasons.a
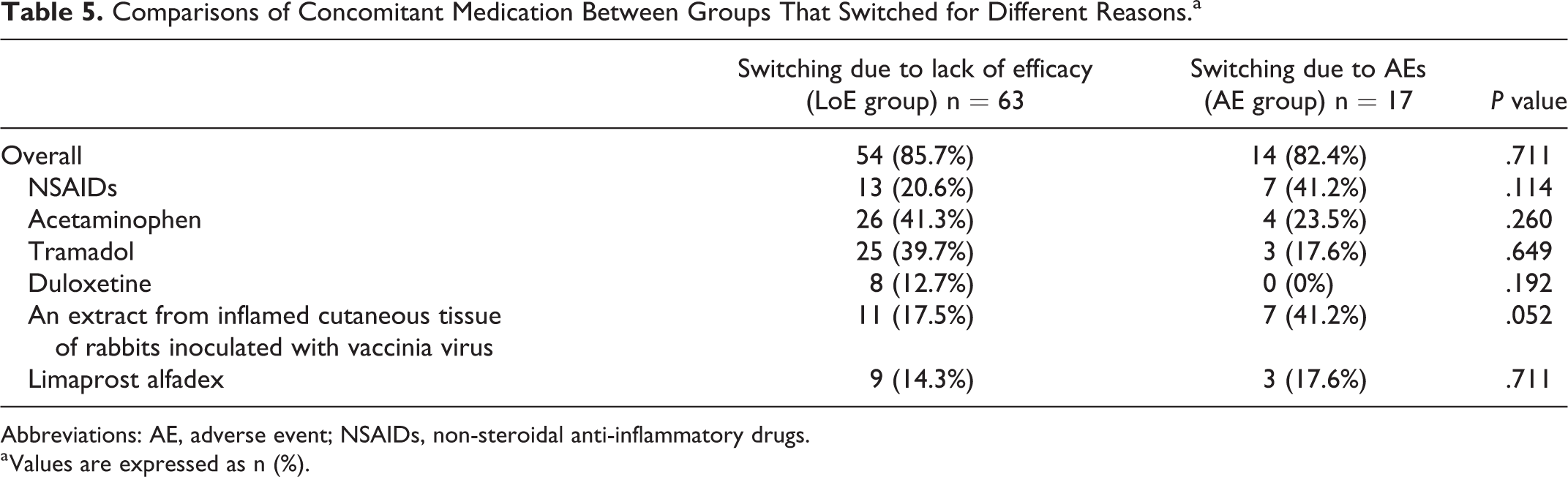
Abbreviations: AE, adverse event; NSAIDs, non-steroidal anti-inflammatory drugs.
a Values are expressed as n (%).
Discussion
In this study, we investigated patients with lumbar disease presenting with neuropathic pain who switched from pregabalin to mirogabalin. We divided patients into 2 groups based on reason for switching and found that those patients who switched due to AEs (AE group) had significantly higher incidences of somnolence and dizziness, which are AEs associated with pregabalin, compared to the incidence rates of these AEs in patients who switched due to lack of efficacy (LoE group). When we compared the incidence of AEs with mirogabalin, the continuation rate of mirogabalin, or the pain-relieving effect of mirogabalin between these 2 groups (LoE group and AE group), we found that they did not significantly differ. In other words, patients suffering from somnolence and dizziness with pregabalin treatment might be able to continue receiving treatment for their pain safely by switching to mirogabalin.
During treatment with pregabalin and mirogabalin, somnolence and dizziness are serious issues as they increase the risk of falling in older people with frailty and/or locomotive syndrome. The incidence rates of AEs with pregabalin in patients with diabetic neuropathic pain have been reported to be 22.8% for somnolence and 20.3% for dizziness. 3 The incidence rates of AEs with pregabalin in patients with neuropathic leg pain derived from lumbar disease have been reported to be 12.6% to 46.9% for somnolence and 2.1% to 30.5% for dizziness.10-12 Somnolence and dizziness have also been reported as the main AEs with mirogabalin. Baba et al found that incidence rates were lower with mirogabalin 30 mg/day versus pregabalin 300 mg/day for somnolence (14.5% vs 20.9%) and dizziness (10.9% vs 19.4%) in patients with diabetic peripheral neuropathic pain. 4 In patients treated with mirogabalin for neuropathic leg pain derived from lumbar disease, the incidences of somnolence and dizziness were reported to be 8.1% to 26.7% and 5.4% to 12.3%, respectively.7-9 These reports suggested that mirogabalin was safer in terms of the AEs of somnolence and dizziness. In the current study, we also found that the incidence of somnolence was 12.2% and that of dizziness was 14.6% with pregabalin, while it was reduced to 7.3% for somnolence and 4.9% for dizziness with mirogabalin. Although the current study was not a direct comparison of AEs between pregabalin and mirogabalin, we showed that patients experiencing somnolence and dizziness during pregabalin treatment could continue to be treated for their pain while suppressing the AEs by switching to mirogabalin.
Multiple drugs are often prescribed for the treatment of neuropathic pain.10,13,14 It was reported that a combination of pregabalin and NSAIDs was effective for patients with lumbar disc herniation. 15 It was also reported that a combination of mirogabalin (20 mg/day or higher) and an extract from inflamed cutaneous tissue of rabbits inoculated with vaccinia virus was effective for patients with neuropathic pain. 16 In the current study, we found that 11 kinds of painkillers were used in combination with pregabalin or mirogabalin. Although concomitant medication may influence the pain-relieving effect of pregabalin or mirogabalin, there was no significant difference in the concomitant medication rate between the patients who switched due to AEs (AE group) and the patients who switched due to lack of efficacy (LoE group).
Although previous studies have reported clinical use of mirogabalin,4,5,7-9,16,17 most reports did not focus on predictors of AEs, continuation, or pain-relieving effect for mirogabalin. Kanbayashi et al reported that maintenance dose (≤ 20 mg), concomitant use of opioids and an extract from inflamed cutaneous tissue of rabbits inoculated with vaccinia virus were identified as predictors for the utility of mirogabalin. 16 In our study, concomitant medication did not affect AEs, continuation, or pain-relieving effect for mirogabalin. We did not analyze the effects of dose of mirogabalin on incidence of AEs and pain-relieving effects because of the small number of cases. This will be a future research.
There were several limitations in this study. First, this was a retrospective observational study of patients who switched from pregabalin to mirogabalin, not a prospective study comparing patients treated with pregabalin to those treated with mirogabalin. Therefore, it was not a study that directly compared the AEs of these 2 drugs. Second, we did not use scales such as the numeric rating scale or the visual analog scale for the therapeutic effect of pain because this was a retrospective study. Third, we did not analyze the effects of dose of pregabalin and mirogabalin on incidence of AEs and pain-relieving effects because of the small number of cases. Fourth, 11 kinds of drugs that are effective in reducing pain were used in combination with pregabalin/mirogabalin. Although the concomitant medication may affect the pain-relieving effect of mirogabalin or pregabalin, there was no significant difference in the concomitant medication rate between the patients who switched due to AEs (AE group) and the patients who switched due to lack of efficacy (LoE group). Concomitant medications are often prescribed for the treatment of neuropathic pain and this study may reflect clinical practice.10,13,14 Fifth, we did not examine the severity of adverse effects because this was retrospective study. Sixth, it was likely that we were only looking at better controlled patients. Patients who had surgery or epidural injections within 4 weeks after the switching were excluded because we thought the effects of surgery and injections helped reduce pain. The possibility of selection bias could not be ruled out.
Conclusion
We investigated patients with lumbar disease presenting with neuropathic pain who switched from pregabalin to mirogabalin. The incidence rates of somnolence and dizziness with pregabalin were 12.2% and 14.6%, respectively, while the incidence rates with mirogabalin were reduced to 7.3% for somnolence and 4.9% for dizziness. The patients who experience somnolence and dizziness with pregabalin might be able to continue safely receiving treatment for their pain by switching to mirogabalin.
Footnotes
Authors’ Note
Tsutomu Akazawa wrote and prepared the manuscript, and all of the authors participated in the study design. All authors have read, reviewed, and approved the article. Ethical Approval: No. 5019, Institutional Review Board of St. Marianna University School of Medicine. Informed Consent: The opt-out method was adopted to obtain informed consent from the subjects.
Acknowledgments
The authors thank Ms. Natsumi Horikita for an assistance of data collection. The authors also thank Dr. Kentaro Fukuda, Dr. Yasuhito Kaneko, Dr. Daisuke Matsuyama, Dr. Yoshiaki Torii, Dr. Atsushi Kojima, Dr. Jun Ueno, Dr. Shingo Kuroya, Dr. Koichi Ishimori, Dr. Kota Asano, Dr. Yoshimichi Onose, and Dr. Ken Tomochika for useful discussions and advice on study design.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Tsutomu Akazawa received honoraria from Daiichi Sankyo Co., Ltd. Hisateru Niki received scholarship donations provided by Asahi Kasei Pharma Corporation, Mochida Pharmaceutical Co., Ltd., and Taisho Pharmaceutical Co., Ltd. as a representative of Department of Orthopaedic Surgery, St. Marianna University School of Medicine.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
