Abstract
Study Design:
Systematic review.
Objectives:
Postoperative spinal implant infections (PSII) are an increasing challenge in the daily clinical routine. This review summarizes existing knowledge in the field of PSII, including definitions, epidemiology, classifications, risk factors, pathogenesis, symptoms, diagnosis, and treatment.
Methods:
A systematic review was performed using a structured PubMed analysis, based on the PRISMA criteria. The search terminology was set as: “spinal implant associated infection OR spinal implant infection OR spinal instrumentation infection OR peri spinal implant infection.” PubMed search was limited to the categories randomized controlled trials (RCT), clinical trials, meta-analysis and (systematic) reviews, whereas case reports were excluded. Studies from January 2000 to December 2020 were considered eligible. A total of 572 studies were identified, 82 references included for qualitative synthesis, and 19 for detailed sub analysis (12 meta-analysis, 7 prospective RCT).
Results:
Structural problems in the field of PSII were revealed, including (1) limited level of evidence in clinical studies (missing prospective RCT, metanalyzes), (2) small patient numbers, (3) missing standardized definitions, (4) heterogeneity in patient groups, and (5) redundancy in cited literature.
Conclusion:
Evidence-based knowledge about spinal implant-associated infections is lacking. All involved medical fields should come together to define the term PSII and to combine their approaches toward research, training, and patient care.
Introduction
Rationale
While several review articles describe characteristics of general spinal infections like spondylodiscitis or non-specified surgical site infections (SSI), up to date reviews specifically designed for postoperative spinal implant infections (PSII), for example following posterior instrumentation, are limited. The 3 most frequently cited reviews giving an overview over the entire field of spinal implant infections, including epidemiology, pathogenesis, diagnosis, and therapy include Kasliwal (2013), 1 Gerometta (2012) 2 and Quaile et al (2012). 3 However, no review has summarized the latest research (2013-2020) with a focus on all potential level of evidence I and II studies (randomized controlled trials (RCT) and meta-analysis), while describing PSII characteristics in total, and using a systematically analysis following a standardized protocol like the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-analyzes) criteria.
Objectives
This systematic review aims (1) to summarize the clinically most important characteristics of PSII, (2) to include the latest research results in the field, (3) systematically include all RCT and meta-analysis in the field, and (4) to critically challenge existing results and concepts.
Methods
Eligibility Criteria
RCT, clinical trials, meta-analysis, reviews and systematic reviews were eligible study categories, while case reports were excluded. Publications from January 2000 to December 2020 were considered eligible. English and German articles were accepted language categories. English abstracts of studies written in different languages were individually considered eligible based on whether all necessary information were included in the abstract.
Information Sources
The review was performed on the basis of a systemic analysis using the PRISMA criteria and algorithm. PubMed was used as only data base. In addition to the initial search results, studies were included that were found in the reference part of other reviews and papers, but not in the initial search itself.
Search
The PubMed search criteria included: “spinal implant associated infection OR spinal implant infection OR spinal instrumentation infection OR peri spinal implant infection.” Using the PubMed NCBI filters, RCT, clinical trials, meta-analysis, reviews and systematic reviews were accepted study categories, while case reports and undetermined study categories were excluded. The last search date was January 30th of 2021.
Study Selection
The individual article suitability was based on: (1) description of implant associated infections in specific, not of unspecified infections like general SSI or wound healing delay without differentiation between implant and non-implant associated cases (unspecified terms like “infection” were analyzed in the context of the present study and considered equivalent to PSII, if applicable); (2) relevance toward clinical routine, with exclusion of solely experimental studies; (3) narrative (not systematic) reviews covering duplicated/similar research results were compared based on patient numbers, year of publication and/or potential level of evidence, and the best match included accordingly. Exclusion criteria included: (1) primary spondylodiscitis/spondylitis treated with instrumentation, (2) spinal tuberculosis infections, (3) infections of spinal cord stimulators, and (4) non English or non German articles.
Data Collection Process
The search was performed by 2 independent reviewers. Duplicated search results were removed, and the remaining articles analyzed, first based on the abstract, then if considered suitable, as a full text.
Data Items
Data was sought in all fields of PSII, including definition, epidemiology, pathogenesis, risk factors, symptoms, diagnosis, treatment and prophylaxis. Unspecific terms like “deep spinal infection” were considered equivalent to the term PSII depending on the context of the study.
Risk of Bias in Individual Studies and Risk of Bias Across Studies
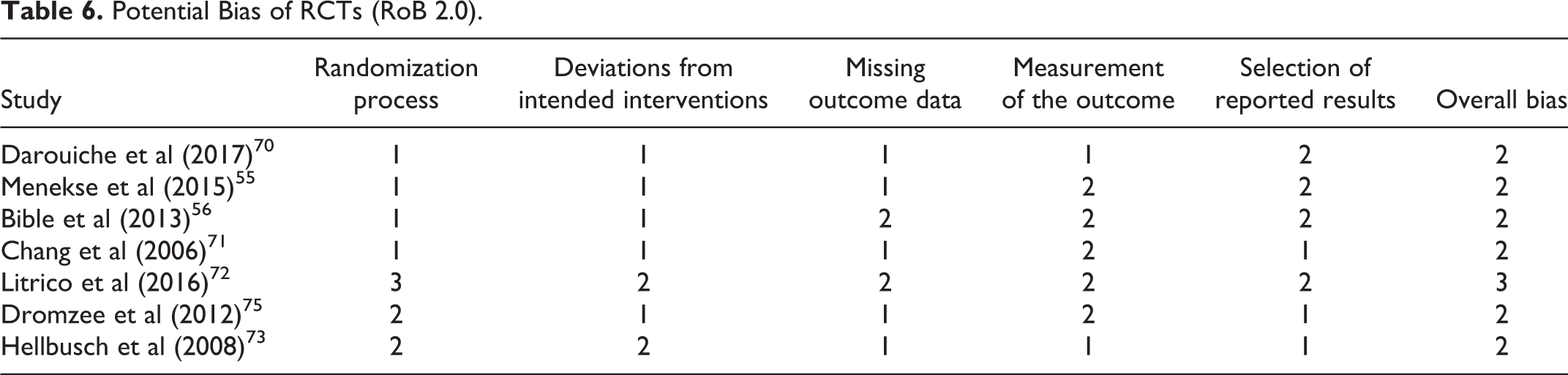
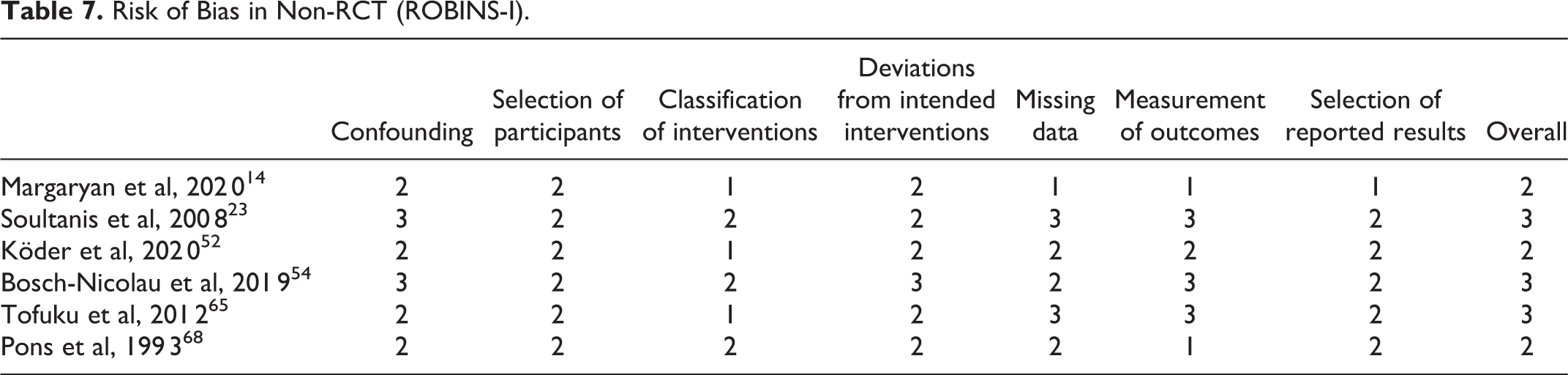
Potential bias of RCTs was evaluated via the revised Cochrane risk of bias tool for randomized trials (RoB 2.0). 4 Five different categories (randomization process, deviations from intended interventions, missing outcome data, measurement of the outcome, selection of reported results) were accessed for risk of bias, and studies graded as “low risk of bias” (1; low risk in all domains), “some concerns” (2; at least 1 domain raising concerns), or “high risk of bias” (3; high risk of bias for at least 1 domain or concerns in multiple domains). Bias analysis was independently performed by 2 reviewers (DK and MP). Non-RCT were evaluated via the “risk of bias in non-randomized studies of interventions” (ROBINS-I) tool. 5 Evaluated bias categories include confounding factors, selection of participants, classification of interventions, deviations from intended intervention, missing data, measurement of outcomes, and selection of the reported results. Categories were classified in no information (0), low bias (1), moderate bias (2), serious bias (3), and critical bias (4).
Additional Analyzes
All prospective RCT and meta-analysis were summarized separately, to give an overview over all potential level of evidence category I and II studies.
Summary Measures and Synthesis of Results
As no meta-analysis was performed no principal summary measures and no synthesis of results were reported.
Results
Study Selection
A total of 572 studies were identified, 528 through the initial search criteria, and an additional 44 records through the reference part of analyzed studies or quotation by analyzed studies (Figure 1).
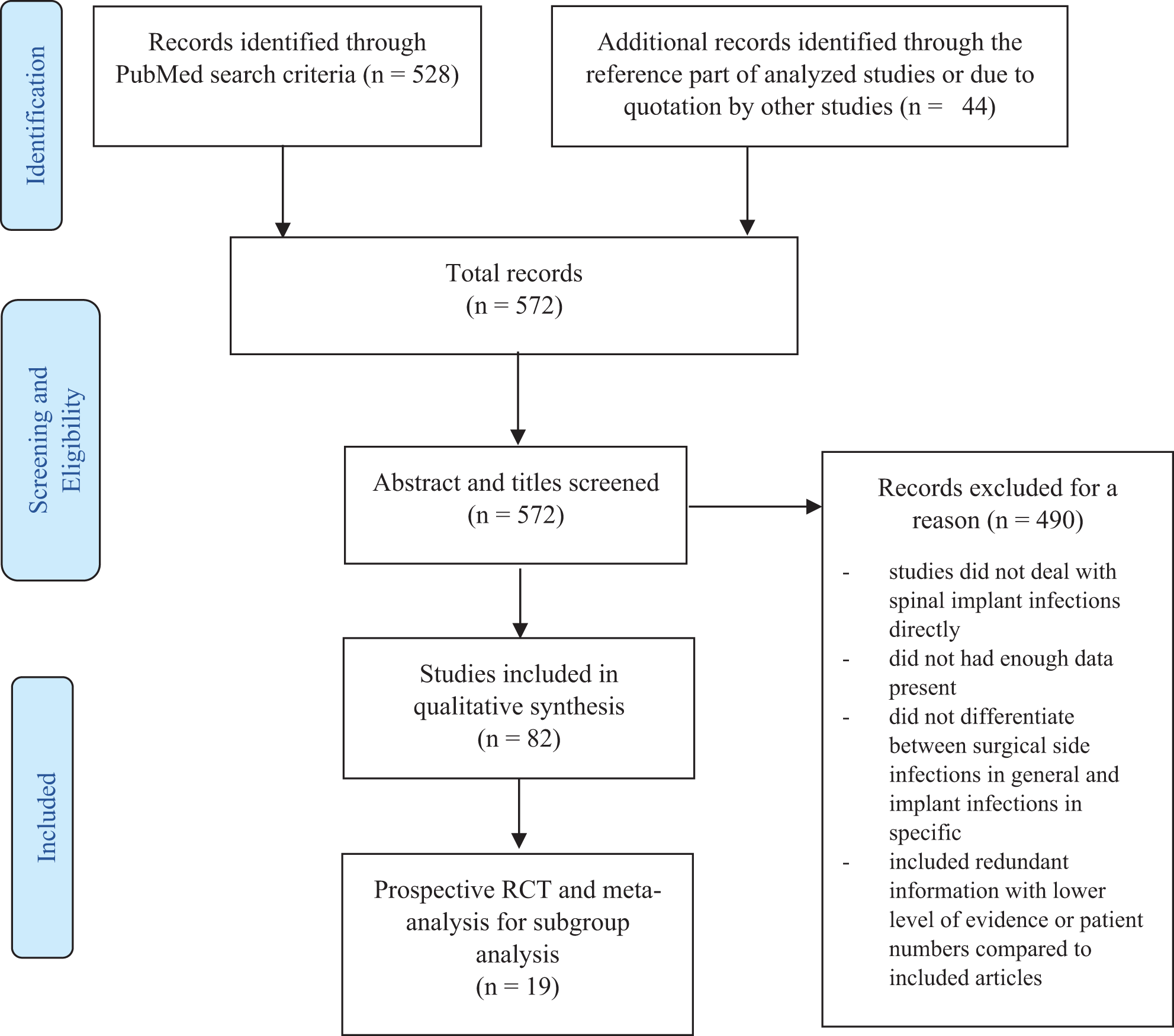
Prisma flow chart.
Study Characteristics
Overall, 82 references, including a detailed analysis of 12 meta-analysis and 7 prospective RCT, were included, as demonstrated by the flow chart (Figure 1).
Results of Individual Studies
Definition and epidemiology
A PSII is not yet defined on an international basis, and is oftentimes not differentiated form the term SSI, especially in older articles.1-3 Trampuz et al (2018) suggested a possible definition based on the EBJIS (European Bone and Joint infection Society) periprosthetic joint infection (PJI) criteria, and thus considers microbiology, histopathology and clinical signs as possible defining criteria (Table 1)6,7 (reference 5 was not included in the systemic PRISMA search). A PSII can either be classified concerning (1) acuity of symptoms or (2) based on the origin of infection. Trampuz et al have suggested a PSII <6 weeks following a surgical intervention an acute, >6 weeks a chronic infection. In cases without a directly prior operation (hematogenous or contiguous), <6 weeks of symptom duration is defined an acute, >6 weeks a chronic PSII. 6 Alternatively, Pawar et al have suggested <3 weeks an acute, >4 weeks a chronic infection based on results used for SSI. 8
PSII Based on PJI Definition.

An infection rate of 0.7 to 20% after spinal instrumentation procedures, based on the review of Kasliwal et al (2013), is cited as a possible range by nearly all authors. 1 In the only systemic analysis present, Patel et al have calculated a pooled average SSI rate of 1.9 (0.1-22.6) % following spinal surgery (196 study cohorts, 425.180 patients), and of 3.8 (0.4-20) % following instrumented spinal fusion (39 study cohorts, 28.628 patients). 9 Jung et al compared patients treated with (1.176) and without spinal implants (699) in the course of open lumbar surgery within a 15-year period and within 1 single institution. The infection rate within the spinal implant group was significantly higher compared to the non-implant group (2.64% vs. 1%; P < .05). 10 Ishii et al reported of a rate of 1.1% of “deep surgical-site infection” within a group of 3.462 instrumented spinal surgeries. 11 To the authors best knowledge, the study with the highest patient numbers in the context of “postoperative spinal site infection” is the 1 of Horn et al (2019) using the 2005 to 2013 results of the American College of Surgeons National Surgical Quality Improvement Program (ACS-NSQIP) database. 90.551 elective spine surgery patients, of whom 1.4% developed SSI, were identified. 12
Pathogenesis and risk factors
A differentiation of PSII types can also be based on the focus of infection as local, hematogenous or contiguous. 13 A prospective study by Margaryan et al (2020) was able to show that 98% of all PSII were acquired during surgery, with the majority of them being acute infections. 14 The most frequently identified microbes include Staphylococcus aureus, Coagulase negative Staphylococci and Cutibacterium spp.14-16 However, in up to 50% of the cases no microbe can be detected. 17 Following an operation, a local focus should always be suspected first. 14 In chronic cases a hematogenous focus should be considered, with genitourinary as its leading cause. 18 Per continuitatem infections are extremely rare and primarily described in the context of an infected aorta graft.19,20 The pathogenesis is based on biofilm formation, with subsequent implant loosening and fistula formation, especially in chronic cases. 6
Based on the risk factors for SSI described by Kasliwal et al (2013) 1 and Blood et al, (2017), 21 a simplified classification of risk factors is possible (Table 2). In the beginning, any foreign spinal material is a risk factor for a spinal infection itself, as compared to non-instrumented cases. 15 In this context, Olsen et al were able to show that a posterior approach was associated with a significantly higher SSI rate in a combined group of patients undergoing laminectomy and/or spinal fusion (OR 8.2, 95% CI 2-33.5). 22 In contrast, an anterior approach is associated with a lower infection risk, likely due to less muscle destruction, blood loss and anatomic dead space formation, as hypothesized by Kasliwal et al. 1 Titanium as instrumentation material has demonstrated promising results and is discussed later as part of the prophylaxis section.23-25 Ho et al identified a more distal level of posterior fusion with instrumentation (thoracic as compared to lumbar) as risk factor for infection. However, the infection group only included 30 patients and a multivariate logistic regression analysis could not show significance. 26 Schimmel et al identified a higher number of spinal levels as a risk factor for infection in a patient group of 1.568 patients treated with spinal fusion, developing a total of 36 infections. 27
Symptoms and diagnosis
To this point there are no clinical studies present that describe the sensitivity, specificity, positive or negative predicative value of specific symptoms in the context of PSII. In addition to classical symptoms of a local infection (redness, pain, swelling, loss of function, warmth), a progression of symptoms following an operation is characteristically, differentiating PSII from physiological / normal postoperative pain. 28 Wound drainage (seroma, purulence) is the most common local, fever the most common systemic presentation. 29 High-grade microbes (Staphylococcus aureus, Streptococcus spp.) are primarily responsible for acute postoperative cases (high fever, sepsis, postoperative wound healing), low-virulent microbes like Coagulase-negative staphylococci or Cutibacterium are causing chronic PSII, resembled by implant loosening, gradual development of fistulas and neurological deficiencies. 6
The initial diagnosis of PSII should include a detailed anamnesis, including prior spinal operations and a full body clinical examination. 6 In suspected PSII, CRP can be determined next. A CRP < 5mg/l should never be considered an absolute exclusion criterion for a PSII, especially when considering patients with chronic and low-grade infections.30,31 Procalcitonin, ESR and a leukocytosis might be alternative diagnostical tools, although their predictive value specifically in the context of PSII has not systematically determined yet. 32 Blood cultures should be taken in any patients showing signs of systemic infections or sepsis. 33 An MRI with Gadolinium enhancement is the gold standard for imaging in suspected PSII, and might show epidural fluid collections, bone destructions and marrow inhomogeneity. 28 However, CT and plane X-Ray can be used as a faster and cheaper way to rule out or show additional pathologies like fractures or implant loosening, and might show lytic lesions, loosening or pseudarthrosis as indirect signs of an infection. 34 Positron emission tomography might be an alternative in undetermined cases following the prior mentioned diagnostical tools. 35 In undetermined cases, CT guided biopsy might offer a possible solution. 36 Alternative or additional diagnosis, including aseptic implant loosening, spinal tissue infection without implant involvement and skin infections like erysipelas should be excluded at any stage. Identification of possible infection areas is essential and should include hematogenous foci (teeth, heart, lung, urinary tract, foreign devices), local foci (skin, surrounding tissue, bone) or trigger (operations, dental procedures, immunosuppressive medications). Following preoperative diagnosis, intraoperative diagnosis should be performed similar to PJI, and thus should include sonication of removed implants, microbial detection out of the intraoperative samples, and histopathology. 37 Akbar et al estimated the probability of microbe detection to be 50% using 2 to 3 intraoperative tissue, and up to 70% using 5 samples, based on results from PJI. 38 While present studies indicate high rates of microbe detection using spinal implant sonication, 39 histopathology demonstrated poor results in identifying infection. A study by. Bürger et al calculated a sensitivity of 51% for the detection of PSII using histopathology in a study cohort of 47 patients, and concluded that histopathology is insufficient, especially when detecting low-grade microbes such as Cutibacterium. 40
Treatment and prophylaxis
The treatment of PSII remains controversial. In 2014 Lall et al systematically analyzed all PubMed studies comparing/discussing the principle of spinal implant retention versus removal. He could not identify a single level of evidence I or II study. 41 In general, a common rather than an isolated surgical and antimicrobial approach is considered a preferred strategy, combined with an interdisciplinary team of microbiologist, orthopedic-/neurosurgeons, and pathologist in a specialized and centralized department.42,43 While unstable implants and patients presenting with (impending) neurological damage should undergo imminent implant removal, patients unable to undergo surgery (cardiovascular, anesthesiologic and surgical contraindications, limited life expectancy, personal refusal) can be covered by long term suppressive antibiotic therapy, 6 although long term results of suppressive antibiotic strategies are still undetermined. Cases involving completed biofilm formation on spinal implants (chronic infections) 44 and specific microbes that cannot be eradicated by antimicrobial therapy alone (difficult to treat (DTT) microbes) 6 should undergo implant exchange due to high failure rates.45,46 DTT microbes are considered microbes resistant to biofilm active antimicrobial therapy, and include Rifampin resistant gram-positive bacteria, fluroquinolone resistant gram-negative bacteria, and fungi/candida. 6 The spinal stability is the critical factor limiting complete and multilevel implant removal, especially considering instable fractures and the following patient mobilization. Thus, exchange for stabilization rather than removal is necessary in instable cases. Debridement and implant retention with antibiotics (DAIR) is a possible treatment strategy for acute infections.47-49 Thereby, a second look surgery might improve the therapeutic outcome in DAIR. 50 In this context, the role of DAIR in chronic cases unable to undergo implant removal / exchange due to instability or perioperative factors remains unknown.
The exact length of administered antibiotics (AB) remains controversial. Trampuz et al have suggested AB administration for a total of 12 weeks in cases of implant retention and one-stage exchange. According to the authors, a possible empiric intravenous (i.v.) therapy is Vancomycin (2 x 1 g) together with Ampicillin/Sulbactam (3 x 3 g), or together with Piperacillin/Tazobactam (3 x 4.5 g) in cases of multiple previous revisions. 6 In contrast, Bullmann et al have suggested only 6 weeks of AB following the surgery. 51 All authors agree that, as soon as a specific microbe or a combination is detected, a targeted eradication therapy must be started immediately. The exact combination depends on the microbe’s susceptibility, patient weight, renal and liver function. In a 10-year cohort study, Köder et al (2020) were able to show that PSII treated with biofilm active AB demonstrated a significantly higher infection free survival rate as opposed to non-biofilm active AB. 52
Despite general trends toward specific treatment paradigms, alternative treatment strategies are still present and should be considered dependent on the individual case. For example, a study by Yin et al (2018) was able to show that aggressive debridement combined with vacuum closed-suction drain and AB was able to resolve late onset PSII with implant retention in 41 of 42 cases. 53 A further study by Bosch-Nicolau et al was able to show that 8 weeks of AB was not inferior to 12 weeks in acute PSII treated with DAIR. 54
Given the complexity of PSII treatment, an adequate prophylaxis avoiding an infection in the first place is necessary. Prospective RCTs by Meneske and Bible et al covering spinal implants before and during surgery,55,56 as well as metanalysis by Stepanow and Khan et al using Vancomycin powder,57,58 demonstrated significantly lower infection rates in spinal implant surgeries. However, the risk for development of resistant bacteria is a possible downside in the long run, and thus the indication for prophylactic local AB administration should be critically reviewed. 59 Further strategies reducing the rate of PSII include AB prophylaxis, 60 an anterior surgical approach, 61 regular glove changing,62,63 minimal invasive surgery, 64 and AB-impregnated fibrin sealant. 65
Antibiotic prophylaxis is a critical factor when avoiding PSII in the first place and should be based on the patient’s weight, liver and renal function, possible allergies, incision to closure time, blood loss, and previous surgeries. Table 3 is showing an overview over all included clinical original studies dependent on antibiotic prophylaxis and implant type (exclusion of narrative reviews; systematic reviews and prospective RCT are listed separately in Table 4 and 5).66-69 All authors cited in this systematic review, except Tofuku et al 65 (proposing a synthetic penicillin), used a first- (cefazolin, cephalothin) or second-generation (cefuroxime, cefamandole, cefonicid) cephalosporin as first line preoperative antibiotic prophylaxis.10,15,22,26,27,29,39,49,50,54,61 Some authors additionally used a second combined preoperative antibiotic prophylaxis such as an aminoglycoside in selected cases.22,70,71 In cases of known allergy to penicillin or cephalosporin, vancomycin was used/suggested by all authors,10,15,29,50,61,72 except Bosch-Nicolau et al that suggested a combination of clindamycin and gentamycin in known beta-lactam allergy instead. 54 While this cephalosporin and aminoglycoside-based prophylaxis is able to reduce the rate of low-virulent organisms such as S. aureus, resistant nosocomial microbes as Pseudomonas aeruginosa and MRSA are resistant to this first-line prophylaxis. 71
Overview Over All Included Clinical Original Studies (Prospective RCT Are Listed Separately in Table 4).
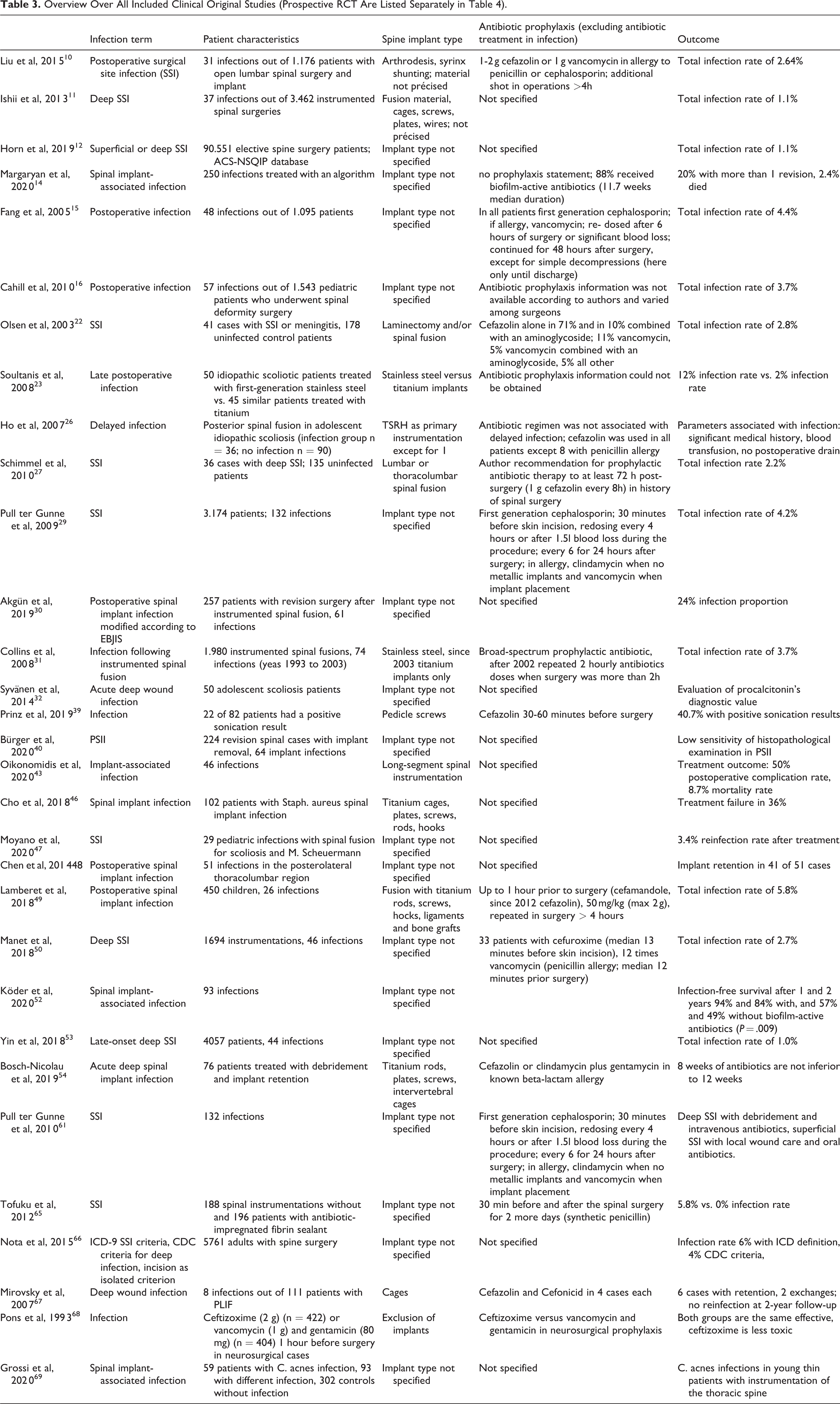
Overview Over All Identified Prospective RCT Concerning Spinal Implant Infections.
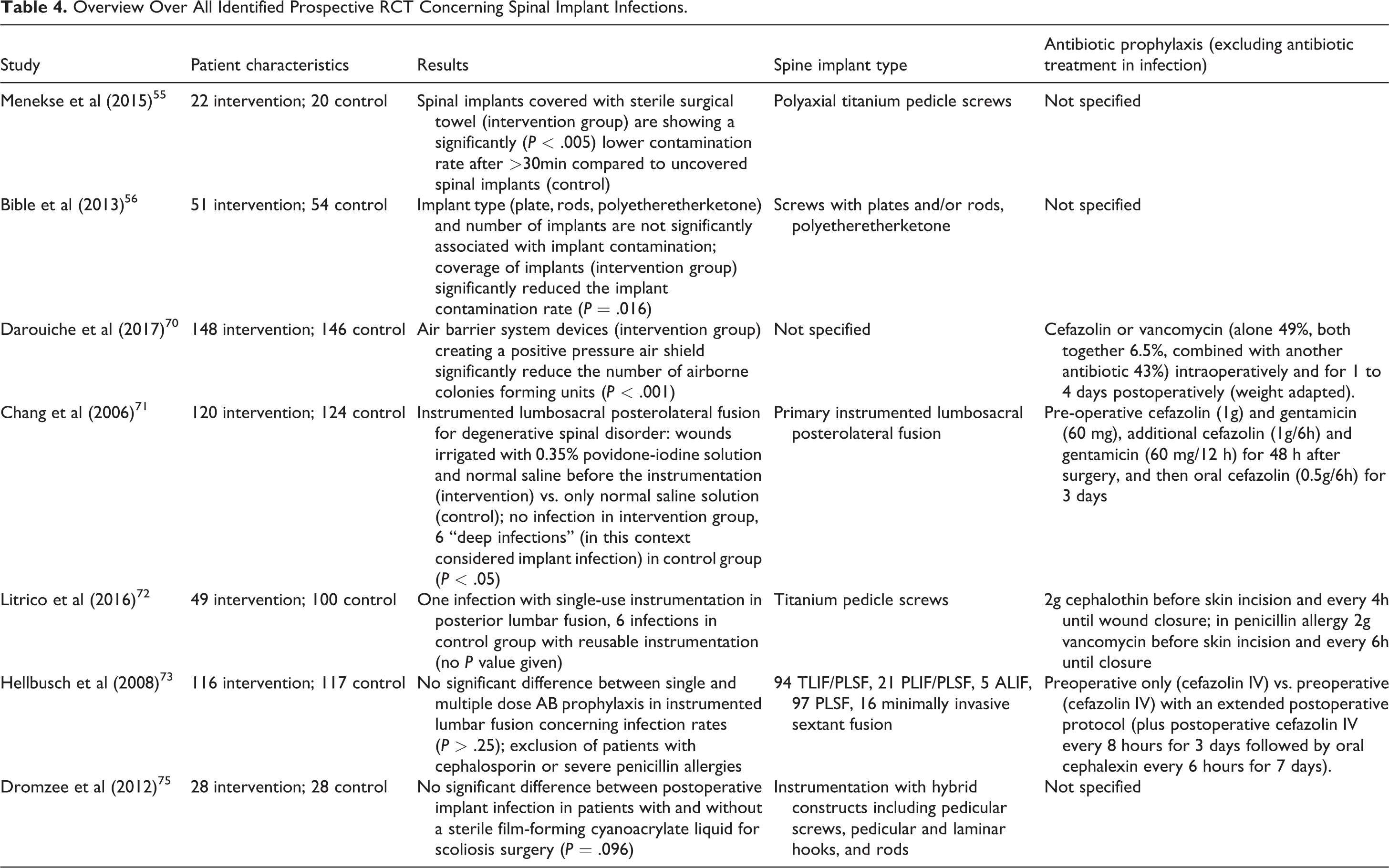
Overview Over All Identified Meta-Analyzes Concerning Spinal Implant Infections.

While the selection of first line prophylaxis and first line alternative in allergies is relatively consistent among authors, both dosage and time interval of prophylaxis presents itself inconsistently. Some authors suggest a constant amount of prophylaxis (usually 1-2 g cefazolin), 10 while some propose a body weight adapted dosage (e.g., 50 mg/kg). 49 All authors agree that cephalosporin prophylaxis should be started at a maximum of 1 hour prior to incision.10,29,39,49,50,61,65 A repeated prophylaxis was performed by Collins et al when surgery was more than 2 hours, 31 by Liu, Lamberet and Litrico et al in operations longer than 4 hours,10,49,72 by Fang et al after 6 hours of surgery or significant blood loss, 13 and by Pull ter Gunne et al after 4 hours or 1.5-liter blood loss.29,61
In contrast, some authors suggest a prolonged antibiotic prophylaxis not only in selected cases but rather as a postoperative standard in all cases, independently of surgery time or blood loss. Pull ter Gunne et al continued the antibiosis for 1 day after surgery,29,61 Fang and Tofuku et al for 2 days after surgery,15,65 Darouiche et al for 1 to 4 days, 70 and Chang et al for up to 5 days. 71 Prospective RCT and systematic analysis covering antibiotic prophylaxis are limited. A prospective RCT by Hellbusch et al could not identify a significant difference between single (preoperative i.v. cefazolin only; n = 117) and multiple doses of antibiotic prophylaxis (preoperative i.v. cefazolin plus postoperative cefazolin i.v. every 8 hours for 3 days followed by oral cephalexin every 6 hours for 7 days; n = 116) in instrumented lumbar fusion concerning infection rates (P > .25). 73 Similar to that, a systematic review by Phillips et al (2020) 74 could not identify a statistically significant difference concerning PSII development between extended postoperative and preoperative antimicrobial prophylaxis in instrumented surgery (P = .93; 888 patients).
Titanium as instrumentation material has demonstrated promising results. A clinical study by Soultanis et al compared 50 scoliosis patients treated with posterior stainless-steel instrumentation, and 45 patients treated with titanium implants. In the course of the follow-up, 6 patients in the stainless-steel, and 1 patient in the comparison group demonstrated signs of late infection. 23 The superiority of titanium has additionally been shown in several in vitro studies.24,25 According to Arens and Meneske et al titanium is the spinal implant of choice due to MRI compatibility, higher biocompatibility, and less risk of infections compared to stainless steel.25,55 For that reason, titanium has become the standard spinal implant in many hospitals. For example, stainless steel was replaced by titanium implants only since 2003 in the hospital reported of by Collins et al. 31 In contrast, Polyethyletherketone (PEEK) is showing a higher infection rate compared to titanium, likely due to its surface allowing for better biofilm formation. 1 However, as demonstrated by Table 4 and Table 5 studies directly comparing titanium, stainless steel and PEEK are limited to the cited study of Soultanis et al and in vitro studies. As many studies do not report a specific material type at all, and the studies are highly heterogenous concerning patient characteristics and intervention, a further comparison of different material among studies is not possible.
Additional Analysis
Existing meta-analysis and prospective RCT
A total of 82 results of the 572 overall identified articles were sub analyzed for prospective RCT and meta-analyzes. As demonstrated by Table 4, only 4 RCT were able to show statistically significant results: air barrier system reduces the number of airborne colonies forming units during spinal implant surgery, covering with surgical towels reduces the rate of spinal implant contamination, and 0.35% povidone-iodine solution reduces the rate of infection following instrumentation.55,56,70,71 The remaining RCT dealing with PSII could not show significant differences between single-use and reusable instrumentation, postoperative infection in patients with and without a sterile film-forming cyanoacrylate liquid, and between single and multiple dose AB prophylaxis.72,73,75
Like with RCT, only 4 meta-analyzes were able to demonstrate statistically significant results (Table 5): Vancomycin powder reduces the rate of spinal implant infection, sacral 2 alar iliac screw fixation showed a lower risk of infection compared to iliac screw fixation, and pedicle screws demonstrated a significantly higher rate of reoperations for reinfection than hybrid instrumentation.57,58,76,77 In contrast significantly reduced risk for infection was not identified with extended postoperative and preoperative antimicrobial prophylaxis, or different screw fixation techniques.74,78,79,80-84
After analyzing the context of the study, the term “deep spinal infection”71,77 and the unspecified term “infection (rate)”57,58,72,76,78 were considered equivalent to the term PSII in the included RCT and meta-analyzes, as opposed to solely wound healing delay and superficial infection. In addition, although not directly reporting of implant infection, studies dealing with “contamination rates” of spinal implants were considered eligible for inclusion, given the limited absolute number of studies.55,56,70
Potential Bias of RCTs (RoB 2.0).
Risk of Bias in Non-RCT (ROBINS-I).
Synthesis of Result
As no metanalysis was performed no confidence intervals and measures of consistency were calculated.
Discussion
Limitations
Up to this point, level of evidence I and II studies in the field of PSII, especially prospective controlled and double blinded studies, as well as meta-analyzes are limited. To the authors best knowledge, not a single prospective double blinded level of evidence I study addressing different treatment strategies for PSII with more than 100 patients is existing at this point. In addition to a limited level of evidence, the absolute numbers are a limiting factor compared to other fields like arthroplasty in which data sets with a few 1.000 patients are present 85 (reference 79 was not included in the systemic PRISMA search). This problem is aggravated by non-standardized definitions of spinal implant infections. Different articles are using different terms like “spinal site infection,” “superficial / deep infection” or “wound infection.” In 2015 Nota et al already noted that different definitions of the term “surgical site infection” were present, and that the different terms lead to varied frequencies of infection rates after spinal surgery within the same population. 66 The term “postoperative spinal implant infection” as used in this article still needs to be defined on an international level. Similar to arthroplasty were several internationally accepted definitions and even a consensus process is present 86 (reference 86 was not included in the systemic PRISMA search), an international consensus for the term “spinal implant infection” is necessary. The initially mentioned definition of Trampuz et al might be a first step toward this direction. 6 However, the definition has neither been evaluated in clinical studies, nor been accepted as an international standard, like its EBJIS counterpart. In addition, it remains unknown, if results and definitions initially developed in the field of PJI are directly transferable toward the field of PSII after all. For example, instability within knee and hip joint are of less direct importance compared to the spine where imminent neurological damage can result as a consequence. Thus, implant exchange or even a complete removal like a girdlestone resection arthroplasty might be an option in hip PJI, but are not directly transferable toward the field of PSII. An additional problem includes the heterogeneity even within the group of spinal implant infections, with some infections referring to pedicle screws, others exclusively to cages 67 and further studies to plates. In this context, it remains unknown if cement is considered a spinal implant. Besides, special patient groups, for example scoliosis surgery patients, known to demonstrate comparable high rates of late-onset infections, 87 are oftentimes analyzed together with elderly patient groups treated for fractures caused by osteoporosis or trauma.
A final problem, which however also might be a general problem of research in orthopedics, includes the redundancy of cited literature. For example, nearly all articles describing or discussing the field of PSII, including this article, cite the 2-20% infection rate stated by Kasliwal et al without even mentioning different results. 1
Summary of Evidence
As criticized by prior authors, it seems necessary to define the term “spinal implant associated infection / spinal implant infection / spinal instrumentation infection / postoperative spinal implant infection.” While Trampuz et al have already proposed concrete definition criteria, this article suggests an additional more abstract definition of the term. A possible definition can be adapted on the basis of the term “(postoperative) surgical site infection” and has in return to be differentiated by the term in the way that not any spine operation is necessarily using foreign material (e.g., microsurgical nucleotomia following a mediolateral disc prolapse). Based on review results1-3 we therefore suggest the following general definition. A PSII is: (1) an infection of any foreign spinal implant, (2) excluding autologous/allogenous bone material and cement, (3) independently of the original cause of surgical integration, onset of symptoms and route of infection, (4) with the possibility of peri implant biofilm formation. A possible downside of the presented term PSII includes a strong focus on a recent operation, and thus the term “spinal peri-implant infection” might be another option to put an additional focus on hematogenous infections without direct prior surgery. In addition, the role of cement and autologous/allogenous bone material as part of the definition was discussed controversially in our working group. Ultimately the authors decided to exclude bone material and cement from the definition, because (1) the PSII definition was primarily based on existing definitions of PJI, (2) biofilm formation on prosthesis surface differs from the 1 on cement, and (3) isolated infections of integrated bone material and (antibiotic augmented) cement is rare compared to instrumentation. 37
Based on the classification of PSII into acute or chronic infections, the operability, the stability of the implant, the types of involved microbes, and the paradigms mentioned in the results part, an additional simplified algorithm-based treatment model was developed for this article (Figure 2). In general, acute PSII with implant stability should be treated with implant retention and debridement, chronic cases without the involvement of difficult to treat microbes (DTT) with a one-stage exchange, given completed biofilm formation on implants, and subsequently reduced options to eradicate bacteria with isolated debridement and AB. AB suppression therapy is an option for inoperable cases and cases involving DTT microbes, until an implant exchange is possible.
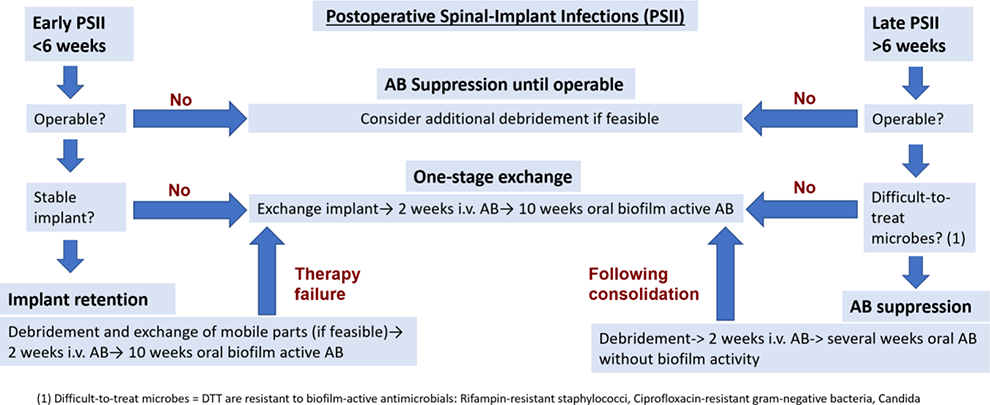
A simplified therapy algorithm for PSII.
Future Approaches
Future training, research and patient care in spinal implant infections should include a combined approach by neurosurgeons, microbiologists, infectiologists, and orthopedic surgeons. Such an approach might not only offer better clinical results, but would also increase the number of patients for studies. Additionally, implications from other fields like arthroplasty and neurosurgery should be critically reviewed and transferred in an adapted way to the field of PSII. For example, RCT should compare existing knowledge about AB prophylaxis in neurosurgery with current concepts in PSII. 68 An example of such a knowledge transfer is the usage of sonication, initially developed in the field of arthroplasty, which has been successfully established in spinal surgery in recent years. 37 Finally, personalized medicine might play a stronger role in the future, as specific risk factors now can be contributed toward specific patient groups, like for example a stronger association of Cutibacterium acnes in young, thin patients with instrumentation of the thoracic spine. 69
Conclusion
International consensus on a standardized definition of the term “spinal implant infection” is necessary. Existing concepts should be critically reviewed, given their limited level of evidence and small patient numbers. A combined approach by neurosurgeons, orthopedic surgeons and infectiologists is essential when dealing with complex patient cases. Both a possible definition and standardized treatment algorithm has been proposed by this article.
Footnotes
Authors’ Note
(I) Conception and design: DK, MP
(II) Administrative support: MP
(III) Provision of study materials or patients: review article / n.a.
(IV) Collection and assembly of data: All authors
(V) Data analysis and interpretation: All authors
(VI) Manuscript writing: All authors
(VII) Final approval of manuscript: All authors.
ICMJE Recommendations for the Conduct, Reporting, Editing, and Publication of Scholarly Work in Medical Journals were considered.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

