Abstract
Study Design
Narrative Review.
Objective
Contextualized by a narrative review of recent literature, we propose a wound complication prevention and management algorithm for spinal oncology patients. We highlight available strategies and motivate future research to identify optimal and individualized wound management for this population.
Methods
We conducted a search of recent studies (2010-2022) using relevant keywords to identify primary literature in support of current strategies for wound complication prevention and management following spine tumor surgery. When primary literature specific to spine tumor cases was not available, data were extrapolated from studies of other spine surgery populations. Results were compiled into a proposed clinical algorithm to guide practice considering available evidence.
Results
Based on available literature, we recommend individualized stratification of patients according to identifiable risk factors for wound complication and propose several interventions which might be employed preventatively, including intrawound antibiotic administration, negative pressure wound therapy, and primary flap closure of the surgical wound. Of these, the available evidence, weighing possible risks vs benefits, most strongly favors primary flap closure of surgical wounds, particularly for patients with multiple risk factors. A secondary algorithm to guide management of wound complications is also proposed.
Conclusions
Wound complications such as SSI and dehiscence remain a significant source of morbidity following spine tumor surgery. Triaging patients on an individualized basis according to risk factors for complication may aid in selecting appropriate prophylactic strategies to prevent these complications. Future research in this area is still needed to strengthen recommendations.
Introduction
Oncologic spine surgery comprises a particularly complex subset of spine surgery, often involving procedures that require significant bony resection, use of autografts/allografts for arthrodesis, insertion of spinal instrumentation, and significant disruption of paraspinal musculature. Individualized goals of surgery may include neural element decompression, spinal stabilization, and/or deformity correction. Tumor resection goals vary depending on the nature of the primary tumor histology, and whether the spinal tumor is primary or metastatic.1,2 While malignant primary spinal column tumors are more often be treated through approaches such as aggressive en bloc resection with curative intent, metastatic disease often presents with multifocal lesions and is more frequently treated through a combination of palliative (stabilizing) surgery and adjuvant chemoradiation therapy.2-5 While surgery for spinal metastases overall has decreased since the introduction and continued advancement of stereotactic radiosurgery and other adjuvant therapeutic strategies, symptomatic spinal metastases remain quite common in patients with metastatic cancer, with increasing prevalence across an aging population.1,2 Conversely, and owing mostly to therapeutic advances in systemic and radiation therapy, recent evidence suggests an expanding role for surgical management of spinal metastases for symptom palliation and improvement in quality of life, including for older patients, those with shorter life expectancies, and those with an indeterminate degree of spinal instability.1,6-8
Despite trends towards more minimally invasive surgical approaches to help reduce complications whenever possible,8-11 many patients undergoing surgery for spinal neoplasms still require a large surgical exposure to accomplish tumor resection and spinal stabilization.2-5,12 Overall complication rates following surgery approach 20%, depending on treatment setting.1,13 Furthermore, patients with spine tumors are frequently exposed to preoperative chemoradiation and steroids, which can cumulatively affect wound healing.14-16 Unfortunately, surgical site infection (SSI) and other postoperative wound complications commonly impact patient outcomes, inflate healthcare expenditures, and contribute to readmission rates across spine surgery indications, but especially so for spinal oncology cases.14,16-20 Preventing and managing such complications is thus a priority for patients who must undergo complex open procedures amidst multiple risk factors. In spinal metastasis patients particularly, postoperative delays in adjuvant radiation and/or systemic therapy due to additional time required for resolving SSI and other complications may impair local control of disease, shorten life, and reduce the time patients can spend with loved ones at home. Hence, mitigating against wound complications is paramount as a primary goal of surgical therapy to best prepare patients for the challenging disease course that often follows.
Given the unique needs of spinal oncology patients, we suggest that a standardized approach to preventing postoperative wound complications could help facilitate reduced overall complication rates as well as cost-per-patient. When prevention fails, a similarly standardized approach for diagnosing and treating complications may be a reasonable means for optimizing outcomes. Building upon other recent reviews of this topic,21-25 we propose an initial algorithm based on the available evidence for the prevention and management of postoperative wound complications in spinal oncology surgery, with the aim of encouraging consensus around a standardized approach to care.
Methods
To identify the most recent articles pertinent to this narrative review to establish initial evidence-based guidelines, we conducted a systematic search of PubMed including all articles published between 2010 and 2022 with “spine” or “spine surgery” in the search phrase or medical subject headings. We further sub-selected articles containing search terms referring to subsets of wound complications and/or available therapies, including the following terms, individually and in combination: “dehiscence,” “surgical site infection,” “plastic surgery,” “wound reconstruction,” “debridement,” “antibiotics,” or “negative pressure wound therapy.” The search was limited to English-language peer-reviewed articles with online full texts. Articles were categorized thematically by whether they focused on “prevention” or “management,” and level of evidence was evaluated for articles studying specific interventions using Grading of Recommendations Assessment, Development, and Evaluation (GRADE) criteria.26,27 Articles regarding a particular intervention or strategy were selected based on pertinence to spinal oncology cases, though spinal oncology-specific studies were frequently not available, requiring that results be extrapolated to this sub-population. This article involved no primary data collection from human subjects; all data were obtained from published literature. As such, this article did not require IRB authorization nor informed consent from human subjects.
Review
Among postoperative wound complications, SSI and dehiscence represent major sources of postoperative morbidity following oncologic spine surgery. Accordingly, it is imperative to identify methods to prevent these complications and effectively manage them when they occur. Here, we provide a narrative review of major operative and nonoperative approaches to wound complication prophylaxis and treatment, including emerging strategies which are gaining popularity at the time of this review. Though no algorithm for managing wound complications following complex spine surgery has been comprehensively validated, a working algorithm is presented based on available evidence on various management approaches that have yielded better outcomes in particular subpopulations of patients experiencing postoperative wound complications. Such an algorithm can be framed according to established subclassifications of SSI (Figure 1), while wound dehiscence is considered as a separate diagnostic entity.
28
Classification of postoperative wound infections and dehiscence by tissue layer.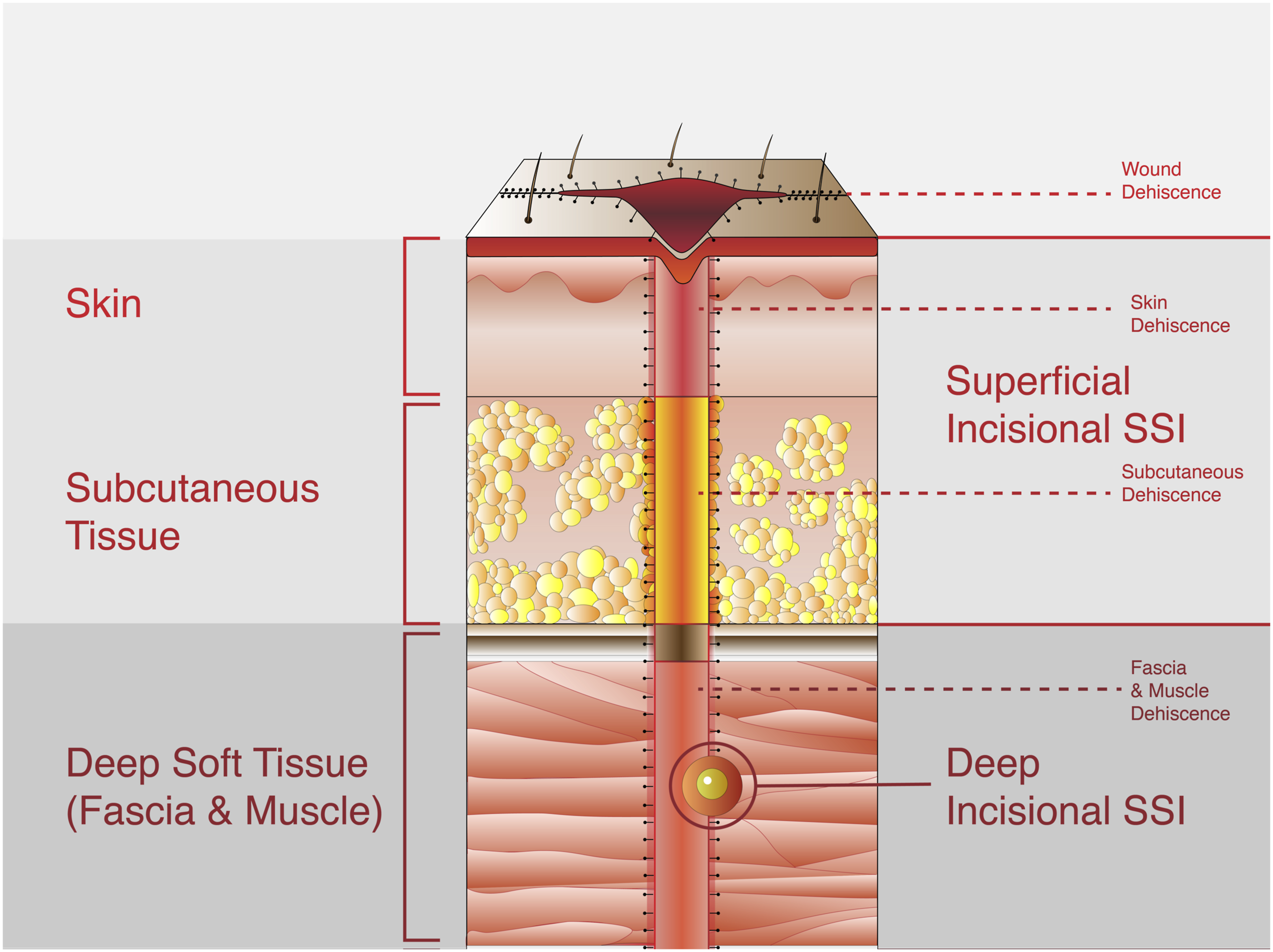
Prevention: Identifying Risk Factors
Patients may be considered at “high risk” for wound complications based upon anticipated procedure and tumor specific factors such as prolonged operative time, high blood loss, and necessity for multiple stages, or patient-level factors such as high comorbidity burden.29-31 Repeat surgery, prior SSI, age, immunocompromised status, chronic corticosteroid use, smoking, obesity, patient acuity, and prolonged procedural time have all been identified as risk factors for postoperative wound complications across spinal indications.32-47 Other patient comorbidity and frailty-related factors which are individually associated with SSI and/or dehiscence include malnutrition (as detected through preoperative hypoalbuminemia),48-51 coronary artery disease and diabetes mellitus.52,53 A composite risk score developed by Piper et al. in terms of these factors was found to be associated with SSI, reoperation, and mortality following spine surgery. 38 Similar risk stratification systems in terms of multiple risk factors have been developed and applied to spinal oncology patients specifically.14,16,54 Spinal oncology patients frequently undergo more invasive spinal approaches to minimize risk of tumor recurrence (depending on patient-specific resection strategy), neoadjuvant or adjuvant radiation and systemic therapy, and steroids in the context of epidural spinal cord compression.14,16,55,56 Differences in risk of postoperative wound infection between specific radiation and chemotherapeutic regimens have not been thoroughly investigated and may depend on the tumor histology and other risk factors in these patients.1,2,14,16 As a guiding principle, recognizing and reducing modifiable risk factors should be considered the first step to patient-specific medical optimization prior to elective and even urgent spine surgery. This can routinely consist of at least blood glucose level stabilization in patients with diabetes and nutritional intervention in those at risk of malnutrition based on preoperative laboratory assessment.48-50 There is some evidence that preoperative exercise regimens may also help to reduce morbidity following non-urgent spine surgery.57-59 While challenging when surgery is urgent, smoking cessation can also be explored as part of preoperative optimization. 35 Ultimately, when multiple non-modifiable risk factors are still present despite optimization, the treating team should consider applying some of the prophylactic options described subsequently.
Prevention: Perioperative and Intrawound Antibiotics
Preoperative IV administration of antibiotics is a widely used wound complication preventative measure across surgical practice. A prospective study conducted by Amelot et al found that preoperative administration of antibiotics in non-instrumented spine surgeries was effective in reducing postoperative wound infections when compared to cases without the use of preoperative antibiotics (1.7% vs 4.9%, HR .283, 95% CI .164-.488). 60 Though this study provided moderately strong evidence in a large prospective cohort, the group was overall a relatively low-risk cohort given inclusion of only non-instrumented cases.
Administration of intrawound antibiotic powder has been more extensively studied for SSI prophylaxis in spine surgery. In 1 retrospective review, Molinari et al. reported the efficacy of intraoperative administration of intrawound vancomycin powder for wound infection prophylaxis. 61 The study demonstrated a <1% rate of deep wound infection after the administration of 1 gram of powdered vancomycin below the deep fascia prior to closure. Lemans et al. further supported the effectiveness of intrawound antibiotic powder by reporting a significant reduction in both deep and superficial wound infections. 62 Additional studies showed reductions in SSI rates particularly when intrawound antibiotic administration was implemented as part of a larger SSI prevention regimen.15,45,63,64 For example, a case-control series by Mesfin et al demonstrated that betadine irrigation combined with intrawound vancomycin powder administration significantly reduced complication rates when compared to the sole use of vancomycin powder administration (5% vs 12.8%). 15 Specifically in spine tumor surgery, Okafor et al. report an infection rate of 4.9% using intrawound vancomycin powder in a cohort primarily consisting of metastatic tumor cases. 65 Two 2019 meta-analyses reported moderate evidence that intrawound vancomycin was associated with a lower overall SSI rate following spine surgery, though 1 reported a higher polymicrobial and gram-negative SSI rate specifically.66,67 Interestingly, these results have not been replicated in SSI associated with cranial surgery. 68
In terms of antimicrobial effect, Naresh-Babu et al reported that intrawound administration of vancomycin powder resulted in persistent drug concentrations in the wound that are well maintained even after 72 h, while serum levels of intravenously administered antibiotics are more transient. 69 These results suggest that intrawound administration of antibiotics may lead to more durable antimicrobial prophylaxis at the surgical site. However, antibiotic resistance should also be considered when selecting any regimen, as routine use of intraoperative antibiotics could lead to a shift in microbial profile for SSI. 70 Further, some studies have noted a possible association between intraoperative vancomycin powder and seroma formation, though the clinical significance of this unconfirmed finding remains to be seen.70-72 Optimal intrawound delivery method, such as antibiotic powder vs antibiotic-impregnated calcium sulfate beads, is also an area of active study. 73 Future prospective and randomized controlled studies are likely required to confirm optimal combinational regimens and unintended consequences of intrawound antibiotics, though the existing literature has shown some initially promising results. Use of intrawound antibiotics should be carefully considered alongside other prophylactic options.
Prevention: Subcutaneous Drains
Subcutaneous drain placement is a commonly employed intervention with an unknown effect on postoperative wound complications. The effect of drain usage patterns on SSI prophylaxis has become a popular area of study, with some hypothesizing that indwelling drains increase infection rates. Multiple retrospective studies have been unable to show any statistically significant benefit to prophylactic subcutaneous drain placement in spine fusion cases.74,75 A retrospective study conducted by Adogwa et al found that subfascial drain placement resulted in a 2-fold increase in duration of hospital stay and a 14-fold increase in rate of post-operative blood transfusion. 76 However, data do not clearly indicate increased SSI with drain use, and studies specifically in spinal oncology cases are lacking. Given high overall drain application rates in spinal oncology cases with large defects, prospective studies should examine benefits and drawbacks to subcutaneous drain placement and appropriate patterns of use.
Prevention: Negative Pressure Wound Therapy (NPWT) and Closed Suction Irrigation (CSIS)
Negative pressure wound therapy (NPWT) applies the theory of Dersch et al that positive pressure reduces skin perfusion while negative pressure promotes it. A fixed foam dressing is placed and stabilized to create an airtight seal, with a negative-pressure pump over the wound drawing excess fluid out and onto the dressing. 77 Closed-incisional NPWT reduces shear forces across the incision and provides a barrier with the environment that aids in offloading direct pressure to the incision in the immediate perioperative period. Additionally, NPWT reduces wound colonization by bacteria, augments neovascularization, and hastens healing. 78 2 retrospective studies of NPWT plus conventional wound closure following spinal fusion observed reduced rates of SSI, dehiscence, and reoperation.79,80 While these studies still have not been extended to spinal oncology patients specifically, several more recent studies have prospectively investigated the efficacy of NPWT as a preventative measure for spinal wound complications more broadly. Dyck et al compared a prospectively enrolled posterior lumbar interbody fusion cohort who received NPWT with a retrospective control cohort that did not receive NPWT but had a similar risk profile; the study found a significantly lower SSI rate (10% vs 21%) in the NPWT group. 81 Similarly, Sun et al reported that the use of NPWT resulted in reduced 10-day postoperative pain, drainage volumes, and C-reactive protein levels. 82 Mueller et al demonstrated that NPWT may reduce SSI rate in instrumented spine surgery compared to standard dressings. 83 The results of these recent studies together suggest that the use of NPWT may reduce SSI rates and promote healing, though the cost-benefit ratio of these expensive devices has not yet been elucidated.
Management: NPWT and Closed Suction Irrigation Systems (CSIS)
As treatment of diagnosed SSI, NPWT has been used in cases of deep SSI and dehiscence with the intention of promoting growth of granulation tissue and increasing perfusion to the wound bed. 84 In a study regarding treatment of deep SSI after lumbar spinal fusion, Wang et al. reported significantly lower implant infection recurrence rate and higher implant retention rate in NPWT vs standard debridement. 84 Ousey et al’s 2013 systematic review cited the benefits of combination NPWT and antibiotics for treatment of deep wound infections, as well as the success of NPWT in combination with debridement and antibiotics for difficult to heal spinal wounds. 77 Despite these apparent benefits, which require confirmation in larger prospective studies, NPWT in spine surgery is not without risks. According to manufacturer labelling, using NPWT in spinal surgery involving durotomy is not recommended due to concern for CSF leakage and fistula. Nevertheless, a small study by Ridwan et al showed that the use of NPWT did not carry significant risk of CSF-related complications and that previous comorbidities also did not increase the risk for CSF-related complications in NPWT. 85 Another study by Lee et al further suggested that the use of NPWT in spinal wound infections with dural exposure is safe and does not pose additional risks when compared with cases without dural exposure. 86 As with prevention data, treatment of SSI with NPWT in spinal oncology cases has not been specifically studied.
Closed suction irrigation systems (CSIS) are an alternative approach for wound management and prophylactic infection reduction and has been studied as both an adjunct and alternative to NPWT. CSIS supplies saline lavage to consistently promote a clean wound bed but does not provide negative pressure. Zeng et al found no significant differences in treatment outcomes between patients treated with NPWT and CSIS in cases of deep SSI but suggested that use of CSIS was more cost effective than NPWT. 87 In a case series on combination therapy of NPWT and CSIS, Chen et al reported that combining NPWT and CSIS promotes cleaner wound beds and shortened healing time and hospital stay. It also reduced treatment costs as the promotion of a cleaner wound bed reduced the required frequency of redressing. 88 Further research is warranted to investigate if there is any benefit to using a combined approach.
Prevention: Primary Wound Closure With Complex Plastic Technique
Primary spinal wound closure by plastic surgery has been proposed to reduce rates of wound dehiscence and SSI.89,90 Cook et al investigated the benefit of multilayered plastic surgery closure and advancement flaps in neuromuscular scoliosis treatment in comparison with standard wound closure, reporting decreased revision and SSI rates among patients undergoing plastics closure. 91 The use of multilayer wound closure reduces the potential for dead space creation while preserving spinal hardware. As plastic surgery-assisted wound closure may increase operative time significantly, this approach may be infeasible in lower-resource centers. However, well-selected patients potentially stand to benefit. Several recent retrospective cohort studies of wound outcomes following spinal tumor resection have suggested that flap closure by plastic surgery may similarly mitigate risk of wound complication in this cohort of patients, particularly those with several baseline risk factors.14,92,93
A large retrospective study by Houten et al confirmed these findings, reporting that prophylactic muscle flap closures for high-risk patients (eg, those with higher comorbidity, injury, and/or tumor burdens) have lower complication rates than low-risk patients receiving a traditional incision closure. 94 Weissler et al further demonstrated that simply including a plastic surgeon in primary wound closure significantly decreased SSI rates and wound-related hospital readmission rates, regardless of flap mobilization use. 95 Though all plastic surgery closure cases included in this study were closed by a single surgeon, the study also showed that when the plastic surgeon closed the incision, wound complication rates were primarily associated with patient ASA classification instead of the invasiveness of the procedure itself. 95 While postoperative SSI and dehiscence can also be managed with plastic surgery techniques such as flap closure, these recent data suggest that preemptive muscle flap closure in high-risk patients undergoing open spine surgery may decrease risk of wound breakdown in the first place.14,96-101 While these studies were limited by lack of prospective randomization, they do provide low-level evidence replicated across centers which supports plastics primary wound closure in high-risk oncology patients.102-104 Furthermore, there has long existed a rationale in specific subsets of cases for plastic surgery involvement, such as in large sacral tumor resection cases in which specialized techniques (eg, V-Y flap) may reduce complications.105,106
Despite retrospective evidence indicating the possible benefit of employing plastic surgery and/or flap mobilization for primary wound coverage, there is significant room for further research in this area. Unresolved are the optimal intensity and/or energy settings of diathermy during cutting and coagulation, strategies for minimizing wound complications through reduced retractor time or extent of retractor use over the duration of the case, and other methods of minimizing soft tissue disruption. Further, randomized studies of neurosurgery vs plastic surgery closure using similar techniques have not been performed, so the nature of the derived effect reported across retrospective comparative studies is poorly understood. Future research in all these areas could further improve our understanding of effective prophylaxis strategies which might be employed during the index surgery.
Management: Flap Reconstruction
When wound dehiscence occurs as the result of SSI, reconstructive flap procedures can be used if soft tissue coverage of the wound is unable to be achieved following treatment through debridement, antibiotics, irrigation, and negative pressure treatment. Customarily, well-vascularized musculocutaneous flaps have been used to pad hardware/reduce skin erosion over implants, eliminate dead space, improve vascularization to promote healing and generate the ability for a tension-free wound closure.97,107 Paraspinous and latissimus muscle flaps are considered the gold standard approach for soft tissue reconstruction of large spinal defects, with trapezius and gluteal flaps also commonly applied depending on lesion and defect location.96,97
Although less frequently utilized, perforator flaps (which are made of skin, subcutaneous tissue and fascia and rely on blood supply from a perforating artery and parallel vein) can preserve the peripheral nerve muscle innervation and decrease the chance of muscle atrophy. Importantly, this beneficial effect is derived by carefully maintaining the muscle’s blood supply during flap advancement, saving its functional integrity. de Weerd et al studied the use of perforator flaps in a small series of 9 patients with midline defects following deep SSI. 107 After debridement, the perforator flaps were able to maintain the stability of hardware with positive SSI resolution, indicating promise for increased use of such flaps in large, complex wound dehiscence cases as the result of deep SSI. 107 As discussed previously, earlier flap closure by surgeons specialized in tissue reconstruction may minimize the need for revision.
Management: Antibiotics Alone
Antibiotic therapy is often used to treat both superficial and deep SSIs following spine surgery (Figure 1). For superficial infections, there has been success with treatment consisting of local wound care and oral and/or intravenous antibiotic therapy. In a retrospective study, Pull ter Gunne et al reported that isolated superficial spinal SSIs were successfully treated with antibiotic treatment and local wound care in 72.9% of cases, without need for surgical debridement. 108 Of these, 56% of SSI limited to superficial fascia required only first generation oral cephalosporin (or clindamycin in cases of allergy) while the remaining 44% of superficial SSIs eventually required IV antibiotics. Several studies further discussed the optimal duration of antibiotic therapy for suspected postoperative spinal wound infection. Two studies proposed 12-week courses of IV antibiotics as safe and effective means to treat spinal implant infections,109,110 though this regimen has recently been challenged by studies showing that both 6-week and 8-week treatment regimens, when applied in the context of proper debridement, antibiotic, and implant retention (DAIR) protocols, are as effective as 12-week protocols.111-114 Importantly, close monitoring of patients with presumed superficial SSI treated with antibiotics is essential to confirm efficacy of treatment or transition to more invasive measures as needed.
Management: Debridement, Antibiotics & Instrumentation Retention (DAIR) Protocols
Compared with superficial wound infections, deep infections have almost universally been managed with surgical washout and debridement in combination with antibiotic therapy. Debridement serves to control infection and address potential compression of neurologic structures. Treatment is typically initiated with IV antibiotics for a given duration, followed by oral antibiotics which continue following washout. 115 Acute deep wound infections often require longer treatment duration than delayed infections, and decisions regarding whether to retain or remove instrumentation often arise in these cases. 116 On the other hand, delayed infections may require instrumentation removal due to biofilm formation, which may lead to re-infection. 117 However, while instrumentation removal can lead to better response to shorter treatment course after surgical debridement, some studies have reported cases of cleared infection without removal of hardware.110,113,114,117 Manet et al. reported that an appropriate DAIR protocol can effectively clear SSI with instrumentation in situ. 113 Another protocol by Yin et al showed a great efficacy in treating late-onset deep SSI using closed suction drains with DAIR, and reported no recurrence of infection without hardware removal. 114 Transitioning IV antibiotics to long-term oral suppressive antibiotics in debridement cases with retained hardware can offer a high SSI resolution rate in select cases. 117
Retention of hardware is optimal, when possible, since removal can be associated with spinal deformity and loss of surgical correction. 117 Fortunately, advances in hardware biocompatibility have largely reduced the need for hardware removal in delayed infections. For instance, titanium spinal implants have a lower infection rate than those made of stainless steel. 118 Other modern reconstruction materials, such as polyetheretherketone (PEEK), also appear to benefit from low infection risk.119-121 If hardware must be removed, replacing instrumentation with new hardware following debridement has been associated with reduced incidence of infection recurrence and attainment of stability. 116 Thus, surgeons should consider the immediate risk of destabilization after a hardware removal and, if necessary, plan to replace hardware immediately or through a subsequent procedure. Such consideration may be particularly important for delayed, deep postoperative spinal infections when osseous fusion may or may not already be underway.
Discussion
Proposed Algorithm
SSI Prevention Interventions and Level of Available Evidence.

Studies which were exclusively limited to spine tumor patients are marked with (*).
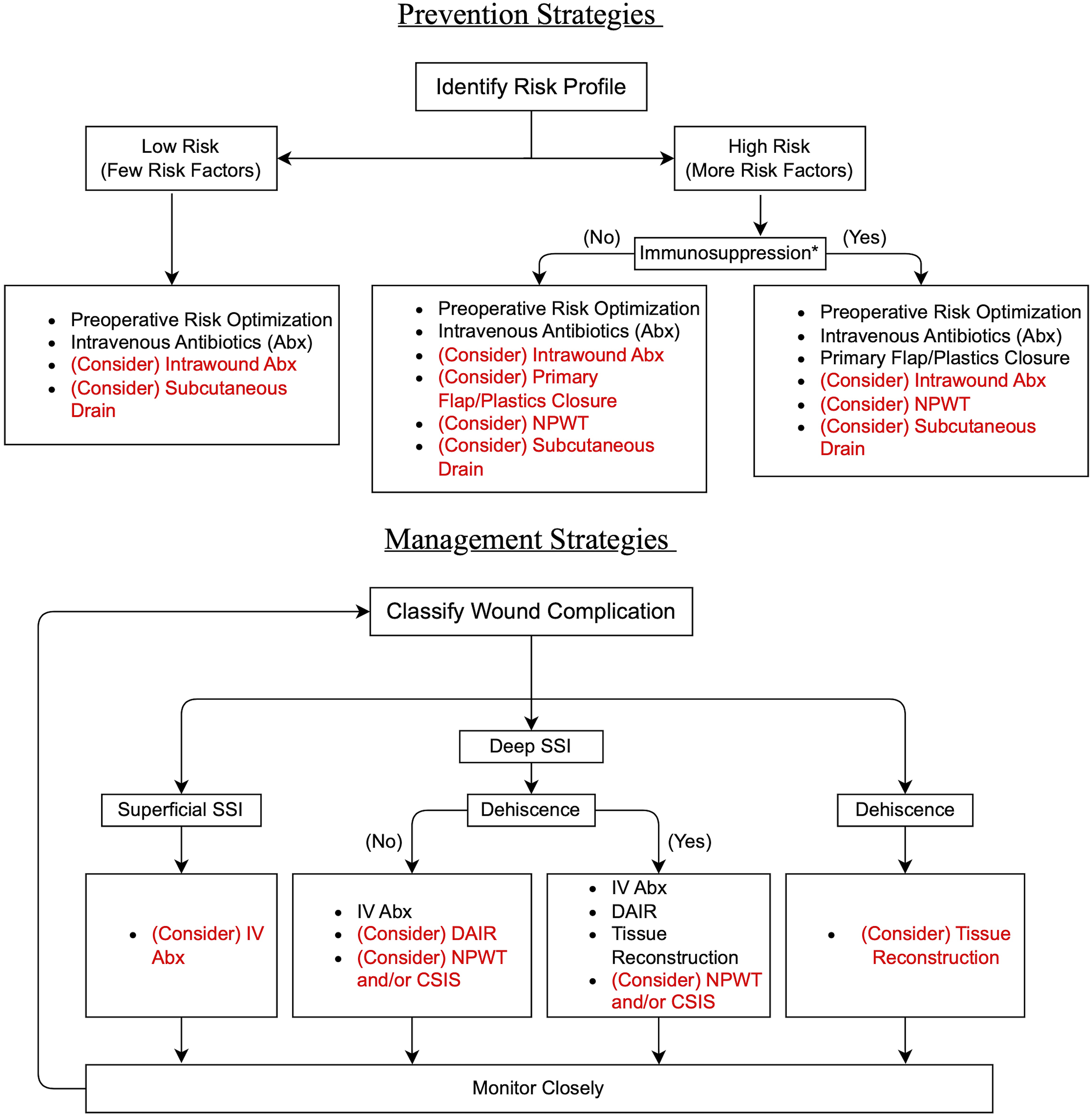
Proposed prevention and management algorithm flowcharts. *Immunosuppresion includes recent use of chemotherapeutic agents, radiation to the surgical site, chronic corticosteroids, or chronic metabolic, endocrine, or auto-immune disorder which may increase the risk for complicated wound healing.
Available Options for Preventing Spinal Wound Complications
In spinal oncology cases, risk factors for wound complications are often numerous and only partially modifiable prior to surgical resection. For primary prevention of wound complications, prophylactic options are accordingly recommended based on type and quantity of risk factors present preoperatively. In our proposed algorithm, management is organized by classification of wound complications, and interventions are recommended based on close monitoring of wound healing and repeated reassessment. In this way, treatment approaches recommended by the algorithm are modulated according to progressive wound healing or worsening. To prevent and reduce the rates of wound complications, it is increasingly important to acknowledge and classify preoperative risk factors.16,54,106 While we lack sufficient evidence to recommend a quantitative threshold of present risk factors that should prompt any particular prophylactic intervention, the proposed concept is that cumulative risk should merit the spine surgeon to consider applying a selection of the available preventative measures. Future research leveraging large prospective databases monitoring surgical outcomes should delineate a formal risk factor grading system specific to spinal oncology which can be used to stratify potential benefit of preventative options; until that point a conservative threshold (eg, 1-2 factors) is warranted. In its current form, our algorithm stratifies patients into “low risk” and “high risk” categories, and suggests further triage based upon the nature of specific risk factors which place patients at further risk of immunosuppression within the high-risk group.
Our literature review revealed limited potential harm across an array of available interventions, none of which can be recommended with a high degree of confidence based on available data (beyond standard pre-operative optimization and IV antibiotics). Intrawound antibiotics are recommended only as a consideration despite some of the strongest evidence of potential benefit, given the possible but unconfirmed association with seroma formation.70-72 Particularly in the presence of multiple risk factors, available evidence points to the potentially beneficial use intrawound antibiotics, prophylactic NPWT, and/or primary musculocutaneous flap closure to minimize the risk of wound breakdown or infection. Subcutaneous drain data were equivocal. Across these interventions, data specifically in spinal oncology cases were found to be limited; the field would greatly benefit from further prospective comparative trials examining wound complication reduction strategies among these patients specifically. Accordingly, we only make strong recommendation for flap closure techniques since there is an accumulation of spinal oncology-specific data for this option.14,92 This may need to be updated as further evidence emerges from large spine tumor registry datasets.
While the presence of many risk factors may elevate risk, the specific risk factors present are also be important. Spinal oncology patients with extensive prior radiation therapy and/or chemotherapy may particularly benefit from the use of prophylactic flap closure, given the propensity for poor tissue healing.14,16 For this reason, our algorithm separates out these patients from among all the patients in the high-risk group with a more specific recommendation in favor of primary musculocutaneous flap reconstruction of the tissue defect in the presence of preoperative radiation or other immunosuppressive risk factors and comorbidities. The existing literature has mostly highlighted the role of plastic surgery involvement in flap closure approaches, however future work could also investigate whether the same benefit is derived when spine surgery teams adopt similar techniques. Indeed, we do not yet understand which specific aspects of musculocutaneous flap closure, which has primarily been reported as being utilized by plastic surgeons, drive the observed risk mitigation.
Management of Postoperative Wound Complications
Overall, evidence to guide postoperative complication management recommendations is of even poorer quality, mostly owing to difficulty defining uniform endpoints which represent “successful” treatment and low overall frequency of cases in any single-institutional cohort. Within our algorithm, recommended methods of managing wound complications vary according to classification and severity. First, the presence of superficial vs deep SSI should be identified to guide treatment decisions (Figure 1). Superficial SSI may require only oral antibiotics and close monitoring to clear the infection. Deep SSI, however, often requires a more extensive debridement and intravenous antibiotic regimen to remove necrotic tissue, reduce bacterial colonization, and hasten healing. If deep SSI is not accompanied by dehiscence, comprehensive DAIR protocols have been established to effectively treat infections and reduce the risk for instrumentation removal. Based on the extent of infection and local resources, NPWT and/or CSIS may be considered as strategies to keep the wound bed clean and encourage the development of granulation tissue.93,109 Complications which involve cerebrospinal fluid leaks should, based on manufacturer packaging, avoid these strategies, though early evidence is reassuring that use even in these contexts may present relatively low risk.
More serious cases of deep SSI can be accompanied by progressive dehiscence due to tissue breakdown. In these case, DAIR protocols coupled with tissue reconstruction is likely the best approach to prevent additional tissue injury. In the event of sole dehiscence without signs of infection, consideration should be given to tissue reconstruction after analyzing the severity of the breakdown. Small areas of dehiscence free from signs of underlying infection can often be appropriately managed with dressings and close outpatient monitoring.
Limitations
While evidence directly pertinent to spinal oncology cases is centered throughout this review, relevant articles studying other open spine surgery cohorts are also considered when findings are pertinent to prevention and management strategies that might be reasonably applied in oncology cases, and when spinal oncology cohort data are scarce. Further, while multiple anterior approaches to the spinal column have been introduced to target specific subsets of spinal neoplasms (eg, transmandibular/transoral approaches to upper cervical chordoma), we focused on prevention and management of posterior spinal wound complications primarily. Considerations not addressed in this review may also apply when managing and preventing complications of wounds created during anterior approaches. Finally, we focus on the most common complications, primarily SSI and dehiscence. Lower frequency but potentially more devastating complications, such as cerebrospinal fluid leak and hardware erosion through the skin are not covered in much detail throughout this review, nor in the proposed algorithm. Similarly, seroma and hematoma within the wound bed are not addressed; though these may occur at comparable frequency as SSI, their presence may or may not be clinically significant, particularly in the absence of symptoms or other complications.
Conclusion
Wound complications such as SSI and dehiscence remain a significant source of morbidity following spine tumor surgery. Triaging patients according to baseline risk may aid in selecting appropriate prophylactic strategies to prevent these complications. A narrative review of the available literature enabled the development of a rudimentary algorithm for both prevention and management of wound complications following oncologic spine surgery. Such algorithms may be helpful for guiding and standardizing best practices. While this approach provides a useful framework, further research including evaluation of the algorithm in a subsequent group of patients is warranted to evaluate the efficacy of this algorithm, and to further optimize across future iterations as new evidence emerges.
Footnotes
Acknowledgments
The authors would additionally like to thank Kendall Rivera-Lane BA for contributing the original illustration for this article; Samathalee Obiorah MD, Albert Woo MD, and Luke Soliman BS for contributing to an earlier version of the drafted text; and Kyle Mueller MD for insights related to the use of negative pressure wound therapy in spinal surgery.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors disclose no financial conflicts of interest directly pertinent to this work. Please see supplemental ICMJE documents for full disclosures.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was organized and funded by AO Spine through the AO Spine Knowledge Forum Tumor, a focused group of international spine tumor experts. AO Spine is a clinical division of the AO Foundation, which is an independent medically guided not-for-profit organization.
Correction (June 2025):
This article has been updated to correct the affiliations for Alessandro Gasbarrini. A correction has been published.
