Abstract
Study Design:
Retrospective case control.
Objectives:
The purpose of this study is to compare clinical outcomes and rates of symptomatic caudal adjacent segment pathology (ASP) in posterior cervical fusions (PCF) constructs with end-instrumented vertebrae in the cervical spine (EIV-C) to PCF constructs that end in the proximal thoracic spine (EIV-T).
Methods:
Retrospective review of 1714 consecutive cervical spinal fusion cases was done. Two groups were identified: 36 cervical end-instrumented vertebra patients (age56 ± 10 yrs) and 53 thoracic EIV patients (age 57 ± 9 yrs). Symptomatic ASP was defined as revision surgery or nerve root injection (or recommended surgery or injection) at the adjacent levels.
Results:
EIV-C patients had a significantly higher rate of caudal-level symptomatic ASP requiring intervention compared with EIV-T patients (39% vs 15%, p = 0.01). The development of caudal-level ASP was highest at C7 (41%), followed by C6 (40%). The overall complication rate and surgical revision rates, however, were similar between the groups. Neck Disability Index outcomes at 2 years postop were significantly better in the EIV-T group (24.5 vs. 34.0, p = 0.05).
Conclusions:
Long PCF that cross the C-T junction have superior clinical outcomes and reduced rates of caudal breakdown, at the expense of longer fusions and higher EBL, with no increase in the rate of complications. Crossing the C-T junction affords protection of the caudal adjacent levels without adding significant operative time or morbidity.
Keywords
Introduction
Long posterior cervical fusions (PCF) are commonly performed for multilevel radiculopathy and/or myelopathy, especially in the revision surgical setting. Surgical goals are decompression, stabilization while minimizing number of segments requiring arthrodesis. Despite a stable radiographic appearance and excellent clinical outcomes following cervical decompression and stabilization, due to the long lever arm, the risk for the development of adjacent segment pathology (ASP) over time exists. Crossing the Cervico-thoracic junction theoretically lowers the risk for symptomatic caudal ASP due to the stabilizing effect of caudal adjacent levels by the ribs. However, to our knowledge, no data exists currently to support this practice. The purpose of this study is to compare clinical outcomes and rates of clinically symptomatic caudal ASP in long PCF constructs with the distal end-instrumented vertebrae in the cervical spine versus the proximal thoracic spine. We hypothesize that crossing the cervico-thoracic junction will reduce the rate of symptomatic ASP compared with constructs that end in the distal cervical spine.
Methods
This was a retrospective review of 1,714 consecutive cervical spinal fusion cases by a single surgeon at a single institution. After Institutional Review Board (IRB) approval, 2 independent surgeons uninvolved in the care of the patients analyzed all of the data. Inclusion criteria consisted of consecutive patients with a minimum 3-level PCF and at least 2 years follow-up that included at least the C5 vertebra. Two groups were identified: 36 cervical end-instrumented vertebra (EIV-C) patients (age 56 ± 10 yrs) who underwent a minimum 3-level PCF were compared with 53 thoracic EIV (EIV-T) patients (age 57 ± 9 yrs) who underwent a minimum 3-level PCF (average follow-up: 35 Months; range 18-82 Months). The EIV-C group consisted of patients with the distal fusion level at C5 (n = 4), C6 (n = 10), or C7 (n = 22) and EIV-T consisted of patients with the distal level at T1 (n = 22), T2 (n = 12), T3 (n = 11), or T4 (n = 8). The 2 groups were compared in terms of perioperative data, the incidence and radiographic parameters of clinically symptomatic caudal Adjacent Segment Pathology (ASP), and Neck Disability Index (NDI) score at 2 years follow up. Clinically symptomatic caudal ASP was defined as 1) revision surgery involving the caudal adjacent level or 2) interventional pain management (or recommended surgery or injection) for pathology at the caudal adjacent level, and analyzed for each end-instrumented vertebra.
Results
At baseline, there were no group differences with respect to age, gender, number of primary surgeries. However, a diagnosis of myeloradiculopathy was a more common indication in the EIV-C group (Table 1). Perioperative data reveals that EIV-T patients had significantly longer fusions and higher EBL, but there were no significant differences in operative times (Table 2). All patients in EIV-C but 2 and in EIV-T but 8 had anterior cervical fusion previously or at the same time with posterior fusion surgery. In EIV-C group, 5 patients received laminoplasty, and 6 patients received laminectomy. In EIV-T group, 2 patient received laminoplasty, and 14 patients received laminectomy. EIV-C patients had a significantly higher rate of clinically symptomatic caudal ASP requiring intervention compared with EIV-T patients (39% vs 15%, p = 0.01) (Table 3). Out of 14 patients in EIV-C group and 25 patients in EIV-T group whose entire x-rays were available and measurable, there was no statistically significance in terms of changes of cervical lordosis (Table 4). Neck Disability Index outcomes at 2 years postop were significantly better in the EIV-T group (24.5 vs 34.0, p = 0.05) (Figure 1). The development of caudal-level ASP was highest at C7 (41%), followed by C6 (40%), C5 (25%), T1 (18%), T3 (18%), T2 (17%), and T4 (0%) (Figure 2). The overall complication rate and surgical revision rates, however, were similar between the groups: EIV-C 8/36 (22.2%) Vs. EIV-T 10/53 (18.5%).
Baseline Demographics Between 2 Groups.
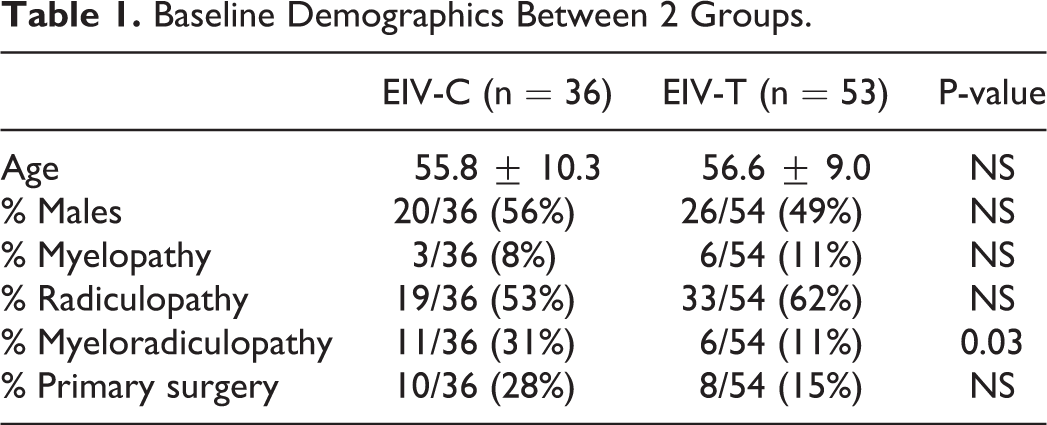
Perioperative Data of 2 Groups.
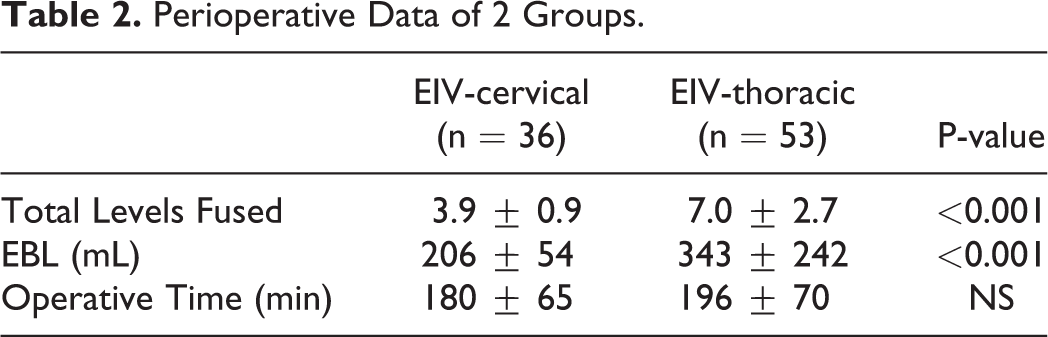
The incidence of Clinically Symptomatic Caudal ASP.
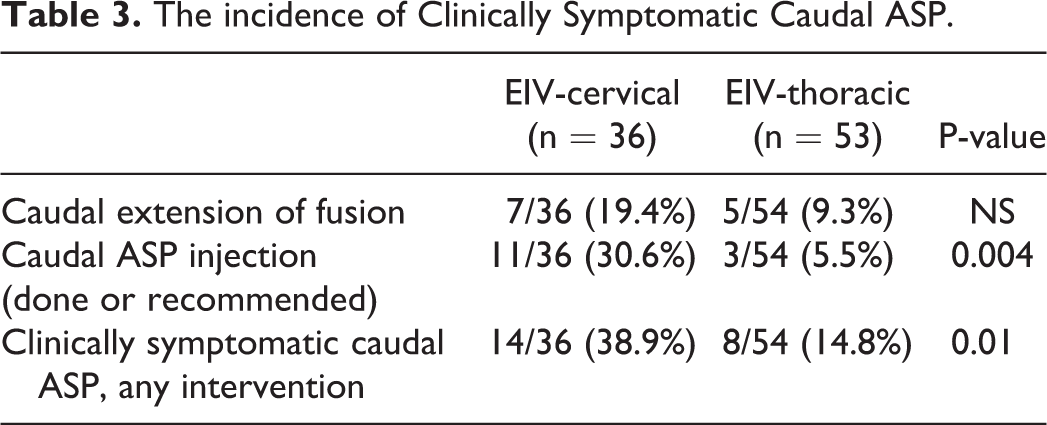
Radiographic Parameters of Clinically Symptomatic Caudal ASP.
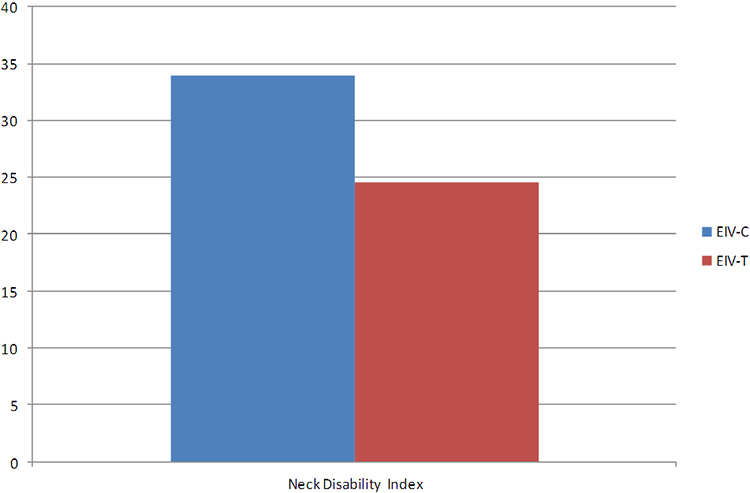
NDI outcomes at 2 years post-op were also significantly better in the EIV-T group.
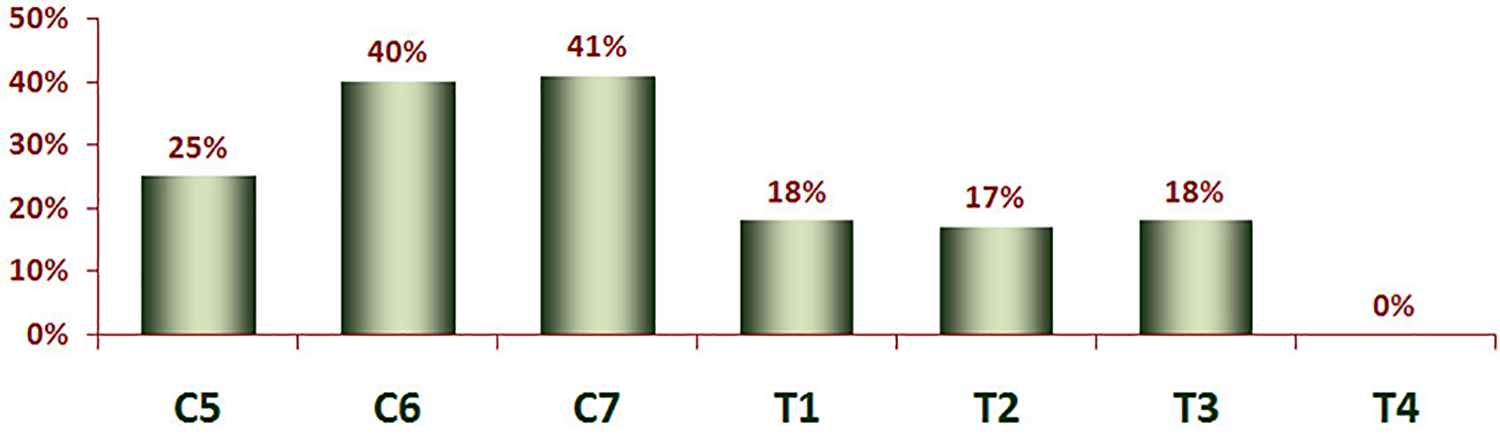
Clinically symptomatic caudal ASP per each end-instrumented vertebra.
Discussion
Long PCF are commonly indicated for multilevel degenerative, traumatic, neoplastic, and congenital cervical pathologies. The risk for development of ASP exists, despite the well-documented good clinical and radiographic outcomes, and may be attributable to the effect of a long lever arm. Crossing the Cervico-thoracic junction theoretically can reduce the risk of clinically symptomatic caudal ASP by ending the lowest instrumented vertebra located in the thoracic spine, which is stabilized by the rib cage. We compared PCF with the end-instrumented vertebrae at the cervical versus thoracic spine to assess whether crossing the C-T junction decreases the risk of clinically symptomatic caudal adjacent level pathology.
Our data suggests that patients who had the construct ending in the cervical spine (EIV-C) had a significantly higher rate of clinically symptomatic caudal ASP requiring intervention compared with patients who had the lowest end vertebra at the thoracic spine (EIV-T). At final follow up, the clinical outcomes were also significantly better in the EIV-T group.
The mechanism for development of adjacent segment pathology (ASP) after anterior cervical spinal fusion has long been debated, as both mechanical factors and progression of underlying patient disease have been purported as contributing factors. Hilibrand et al. reported 2.9% of annual incidence of symptomatic ASP after anterior cervical fusion, and 25.6% of newly developed symptomatic ASP within 10 years after anterior cervical fusion. 1 The risk factors for ASP after anterior cervical fusion include a reduced distance between the tip of the plate and adjacent level disc, preoperative disc degeneration, and multilevel fusion.2,3 Further, biomechanical studies have shown that multilevel fusion is another risk factor for ASP. Prasarn et al. showed that multilevel fusion increased ROM of the adjacent segment compared to single level constructs, possibly increasing the risk of ASP. 4 Hussain et al. also showed that increased stiffness due to multilevel cervical fusion not only can cause degeneration of the disc, but also can cause degeneration of the facet joint in their finite element study. 5
Posterior fusion tends to increase the incidence of ASP compared to anterior fusion. In a recent study, Lee et al. analyzed 1358 cervical spine surgery cases, and reported that posterior cervical fusions had 3 times greater risk for requiring revision surgery for ASP than anterior cervical fusion. 6 Therefore, efforts have been made to identify the factors that increase the risk of ASP, as well as to implement methods to reduce it in multilevel posterior spinal fusion. In the lumbar spine, one technique introduced to reduce ASP includes crossing the thoraco-lumbar junction in posterior lumbar fusion constructs for degenerative lumbar scoliosis, as the rib cage may afford additional stabilization. 7
Our results confirm the hypothesis and support the practice of crossing the C-T junction as one method to reduce the rate of clinically symptomatic caudal ASP in multilevel PCF, with similarly improved long-term clinical outcomes. Not fusing C5-6 and C6-7 has been previously identified as one of the risk factors for adjacent segment pathology after cervical fusion.8,9 Our study also confirmed these findings, although a key difference between these studies and the current one is the posterior-only approach evaluated in our cohort.
Our study had several methodological limitations. First, we did not correlate our result to patients’ radiographic outcomes except lordosis change. To that point, a similar study was performed recently at our institution that showed that sagittal malalignment after anterior cervical fusion increased the incidence of symptomatic ASP. 10 Therefore, a subsequent radiographic study may be necessary in order to confirm those findings in posterior cervical fusions. Second, our study was a retrospective review of a consecutive series from a single surgeon for a variety of surgical indications. Future studies may include a randomized prospective trial evaluating a more focused clinical cohort. By way of prospective study, we can also compare the timing of development of clinically symptomatic caudal ASP in-between 2 groups to better understand the pathogenesis of ASP. Third, although clinically symptomatic caudal ASP per the definition described in the current study was limited to those patients requiring revision surgery or injections at caudal adjacent levels, it should also be noted that a subset of patients may have also been symptomatic to a lesser degree and been recommended medications and physical therapy alone. Those subjects were not included in the current study. It is the typical practice of the senior surgeon to try non-invasive treatment first and then advance to injections next. Fourth, we did not include patients who presented with radiographic changes of ASP in the absence of clinical symptoms. Fifth, we did not analyze clinically symptomatic cranial ASP. In our cohort, extension of fusion cranially sometimes was performed even without symptom during the revision surgery to extend the fusion caudally in order to obtain the symmetric numbers of fusion above and below the apex of lordosis. Therefore, even though injection at cranial level might be true clinically symptomatic cranial ASP, extension of fusion cranially does not necessarily due to true clinically symtomatic cranial ASP. Finally, the numbers of fusion level between EIV-C and EIV-T were significantly different. However, despite the different numbers of fusion level, the incidences of clinically symptomatic caudal ASP were lower in EIV-T group despite the longer level arm. Therefore, we can deduce the crossing CT junction has a protective effect on clinically symptomatic caudal ASP.
As a direct result of this study, we now give serious consideration to crossing the cervico-thoracic junction when performing long instrumented posterior arthrodeses, instead of stopping at C7. It should be noted, however, that avoidance of ASP should not be the paramount goal of an operation. For example, in young patients who want to retain motion, and for whom additional surgery in the future does not pose undue risks, it may be advisable to just treat the symptomatic levels. Similarly, it may be advisable to keep the construct shorter for some, including the frail or very elderly, for whom longer constructs might pose more risks. The effects of an operation on ASP should therefore be just one of many factors that are considered when choosing surgical levels. Awareness of these effects can also allow the surgeon to more intelligently pick surgical levels and inform patients about the future ramifications of surgery.
To our knowledge, this is the first study to compare the incidence of clinically symptomatic caudal ASP between 2 groups: those treated with or without crossing the cervico-thoracic junction with long posterior cervical fusion constructs. In this single surgeon consecutive series, reviewed by 2 independent surgeons, we conclude that crossing the C-T junction results in a significantly reduced rate of clinically symptomatic caudal ASP, with improved 2-year clinical outcomes. Although the decision regarding whether to perform arthrodesis with an EIV in the thoracic versus cervical spine is a clinical decision made on a case by case basis, the results from the current study do support the clinical rationale for crossing the C-T junction as a means to reduce the rate of symptomatic caudal adjacent segment pathology, with no significant differences in complication rates or operative times.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
