Abstract
Study Design:
Retrospective diagnostic analysis.
Objectives:
To establish a new predictor of surgical outcome after surgery for intradural extramedullary spinal tumor (IDEMT) in the thoracic spine, we introduced shape factor (SF), a mathematical description of the morphology of the spinal cord. SF was calculated by dividing object area by the square of perimeter.
Materials and Methods:
Forty-three consecutive patients with IDEMT, detected by magnetic resonance imaging at the thoracic level with myelopathic signs, were included. Preoperative transverse cross-sectional area (CSA) and perimeter of the spinal cord (perimeter) at the level of maximal compression were measured. SF was calculated as 4π × CSA/(perimeter)2. The association between clinicoradiological factors and surgical outcome of IDEMT was statistically analyzed.
Results:
Mean CSA, perimeter, and SF were 27.8 ± 15.8 mm2, 28.8 ± 6.1 mm, and 0.385 ± 0.14, respectively. A histogram distribution revealed that perimeter and SF, but not CSA, fit the normal distribution. The patients were subdivided into 2 groups according to postoperative modified Japanese Orthopedic Association Score (mJOA). [group F (favorable): n = 32, mJOA ≥ 9; group UF (unfavorable): n = 11, mJOA < 9). Group UF had significantly lower mean CSA and SF. In univariate analysis of possible predictive factors for IDEMT surgery, greater age, lower preoperative mJOA, and lower SF were significantly associated with unfavorable outcome. In multivariate analysis, lower SF was the only significant predictor of postoperative outcome (odds ratio = 2.66, 95% CI 1.10–6.39, p
Conclusion:
Measurements of CSA and perimeter, followed by calculation of SF, may provide valuable quantitative information for the outcome of surgery for IDEMT.
Keywords
Introduction
The success of surgical management of spinal intradural extramedullary tumors (IDEMTs) is impacted by several factors. For example, preoperative neurological deficits, the location of the tumor, and the presence of hyperintensity area in the spinal cord in T2-weighted MRI at the level of maximal compression of the spinal cord may predict satisfactory or acceptable functional recovery after surgery.1,2 However, few studies have sought to determine characteristic imaging features that affect the clinical results of IDEMT treatment. 3
Quantitative measurement of the spinal cord provides the basis for clinical assessment such as the relationship between the morphological features of the spinal cord and the severity of disease, including IDEMT. On the other hand, sagittal/transverse diameter and transverse cross-sectional area (CSA) of each spinal cord segment vary widely. 4 Therefore, the morphometric absolute measurement of the spinal cord is not directly associated with clinical symptoms, and is therefore not appropriate for evaluating the surgical outcome of IDEMT.
Recently, effective mathematical descriptions of the morphology of the material have been introduced in various biomedical fields, including orthopedic surgery. 5 One of the earliest and most widely applied descriptions is shape factor (SF), also known as compactness. SF is measured by dividing object area by the square of perimeter. 6 SF is an intrinsic feature of the shape of an object and may relate to its function. In addition, SF is dimensionless and considered to independent of the morphometric absolute measurement of the shape, leading us to hypothesize that SF of the spinal cord is associated with neurological function.
In this study, we carried out the quantitative measurements of CSA and perimeter of the spinal cord in cases of thoracic IDEMT at the level of maximal compression. We also calculated SF and investigated the association between SF and the surgical outcome of IDEMT.
Materials and Methods
Patients
Subjects were 43 consecutive patients with IDEMT detected by MRI at the thoracic level with myelopathic signs. Patients with significant additional diseases or previous surgery on the thoracic spine were excluded. Functional status was assessed using the modified score of the Japanese Orthopedic Association (mJOA score)
7
; the maximum mJOA score in healthy subjects is 11. Scoring of patients’ functional status was performed before and after surgery with median and mean observation periods of 44 and 45.3 months, respectively (range, 12–88 months). Surgery was performed using a standard dorsal approach with laminectomy in all patients. Gross total resection was achieved in all cases. There were no severe postoperative complications such as hematoma, surgical site infection, or sustained leakage of cerebrospinal fluid. The study was approved by the institutional review board at the Kyushu University Hospital (26-112) and
Imaging
Radiologic imaging was conducted in all patients within 1 month prior to surgery. All subjects underwent MRI examination of the thoracic spine in the supine position with 5-mm slice thickness at our institution using a 1.5-T MR system (Achieva; Philips Medical Systems, Best, The Netherlands). Axial planes in T2-weighted images were acquired. The preoperative CSA and perimeter of the thoracic spinal cord (perimeter) at the level of maximal compression were manually measured by SYNAPSE5
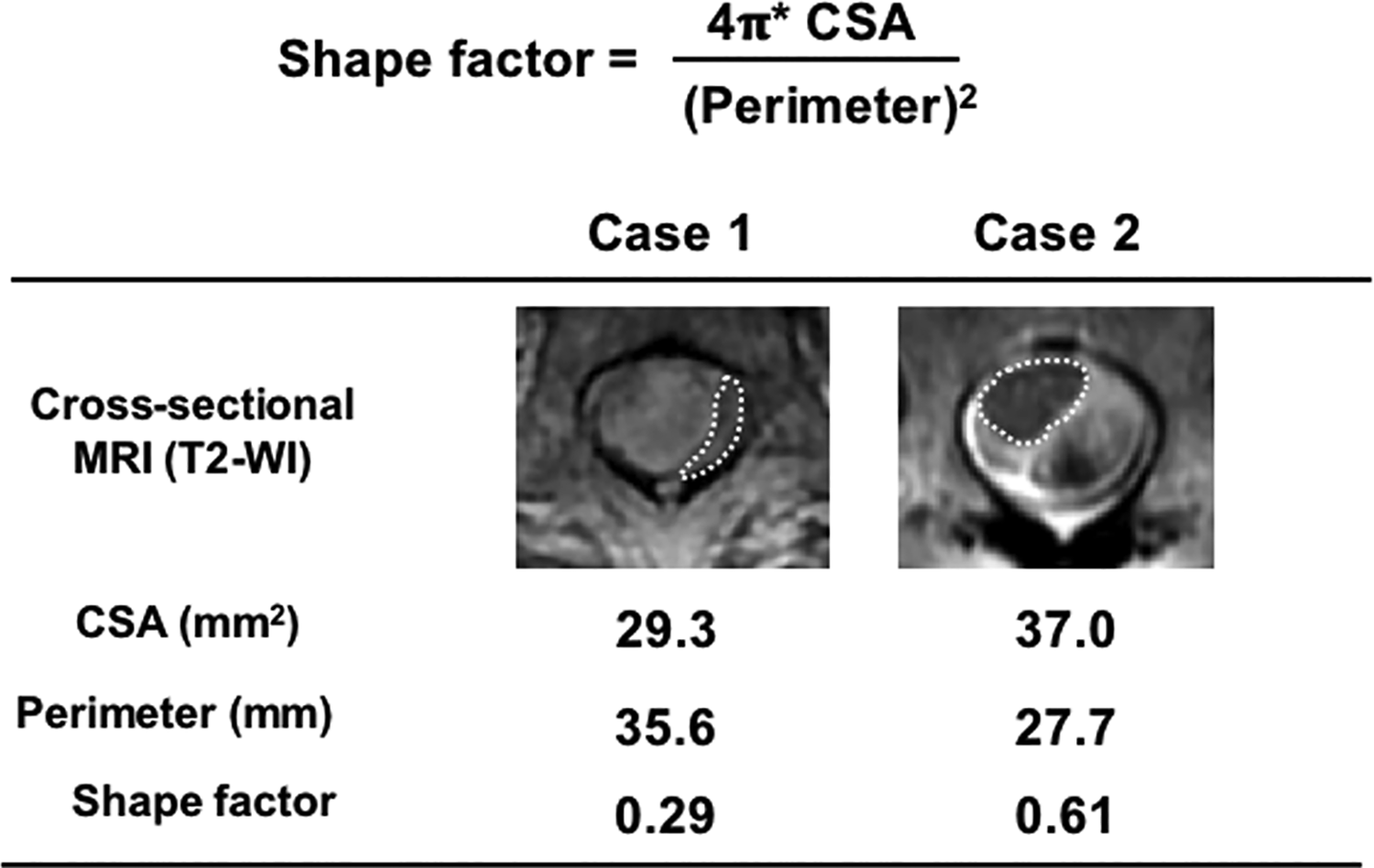
Calculation of shape factor: 2 representative cases. Spinal cord was manually demarcated (dotted line), and cross-sectional transverse area (CSA) and perimeter of the spinal cord were measured with the SYNAPSE5 software (FUJIFILM). SF was calculated as 4π × CSA/(perimeter)2. Case 1: meningioma at T2; Case 2: hemangioma at T8.
To test the reliability and reproducibility of evaluation of the measurements, 10 randomly selected cases, more than 20% of full sample size, were independently reviewed by 2 authors who specialized in the diagnosis and treatment of spinal disease and who were blinded to the clinical data. In addition, one author reviewed the data twice more over a 1-month interval. Intra- and inter-observer agreements were assessed using intraclass correlation coefficients (ICC). 8 We interpreted ICC values less than 0.5, between 0.5 and 0.75, between 0.75 and 0.9, and greater than 0.90 as indicative of poor, moderate, good, and excellent reliability, respectively. 9
Definition of Form Factor (SF)
SF serves as an indicator of the circle-like qualities of a shape; a value of 1 indicates a perfect circle. SF was defined as the ratio of the area of a shape to the square of its perimeter. 10 The formula for calculating SF was
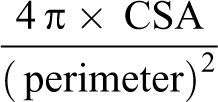
Representative calculations of SF are shown in Figure 1.
Statistical Analysis
Continuous variables were expressed as mean ± standard deviation or median and range. Shapiro–Wilk test was used to evaluate goodness of fit to the normal distribution. Categorical variables were expressed as counts and proportions. The correlation of 2 variables was calculated using the Spearman rank order correlation test. Unpaired Student’s t test or Mann–Whitney U test was used to detect significant differences between clinical subgroups. Receiver operating characteristic (ROC) curves and the area under the curve (AUC) was used to evaluate the accuracy of the SF as a predictive factor for differentiating favorable and unfavorable surgical outcomes. Univariate analyses using Student’s t test and the Chi-squared test or Fisher’s exact test and multiple logistic analysis were performed to assess the relationships between surgical outcome and the following clinicoradiological factors: preoperative mJOA score, coexistence of T2-high lesion in the spinal cord, location of the tumors, and SF. A value of p < 0.05 was considered to be significant. These statistical analyses were performed using JMP statistical analysis software (version 14; SAS Institute, Cary, NC, USA).
Results
Of the 43 patients, 23 had neurinoma, 15 had meningioma, and 5 had other histology. There were 20 males and 23 females. Mean and median ages were 58.3 ± 19.7 and 60 years, respectively (range, 2–92). Preoperative mean and median mJOA scores were 6.6 ± 2.4 and 7, respectively, increasing postoperatively to 9.4 ± 1.9 and mJOA score decreased in 2 patients. Patients with postoperative mJOA scores of ≥ 9 were classified as having favorable outcomes (group F, 32 patients), and those with a score < 9 points were classified as having unfavorable outcomes (group UF, 11 patients).
Mean CSA was 27.8 ± 15.8 mm2 (median 25.4, range 5–79.5) and mean perimeter was 28.8 ± 6.1 mm (median 27.4, range 15.6–42). Mean SF was 0.385 ± 0.14 (median 0.379, range 0.09–0.66). ICC (1.1) for CSA, perimeter, and SF was 0.90, 0.87, and 0.89, respectively. ICC (2.1) for CSA, perimeter, and SF was 0.96, 0.85, and 0.83, respectively. Histograms of CSA, perimeter, and SF are shown in Figure 2. A test of goodness of fit to the normal distribution revealed that perimeter (p = 0.22) and SF (p = 0.38), but not CSA (p = 0.0018), fit the normal distribution (Figure 2). CSA and SF were slightly correlated (r = 0.32), whereas perimeter and SF were not correlated (r = 0.003) (Figure 3). Hyperintensities in T2-weight images (T2-high) at the level of the maximal compression of spinal cord were observed in 10 cases (23.3%).
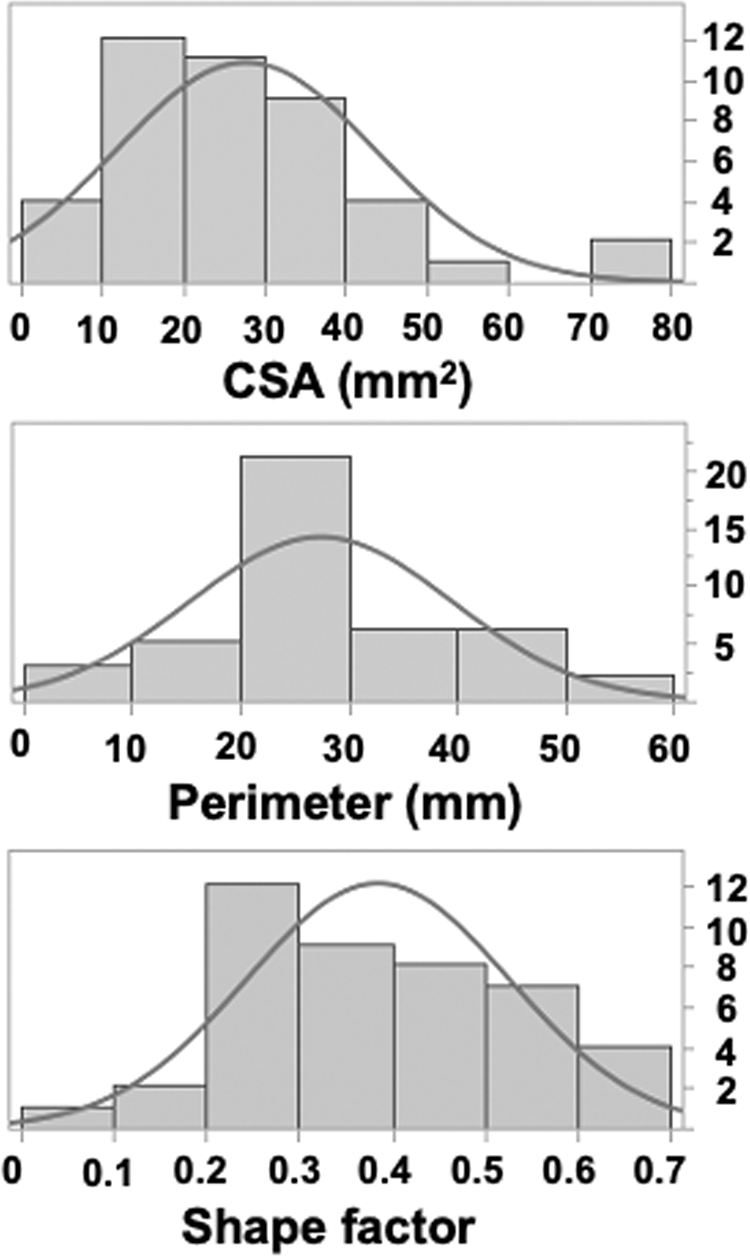
Histogram distribution of CSA, perimeter and SF. Histograms of CSA, perimeter, and SF are shown. Test for the goodness of fit to the normal distribution (overlay curve) revealed that perimeter (p = 0.22) and SF (p = 0.38), but not CSA (p = 0.0018), fit the normal distribution.
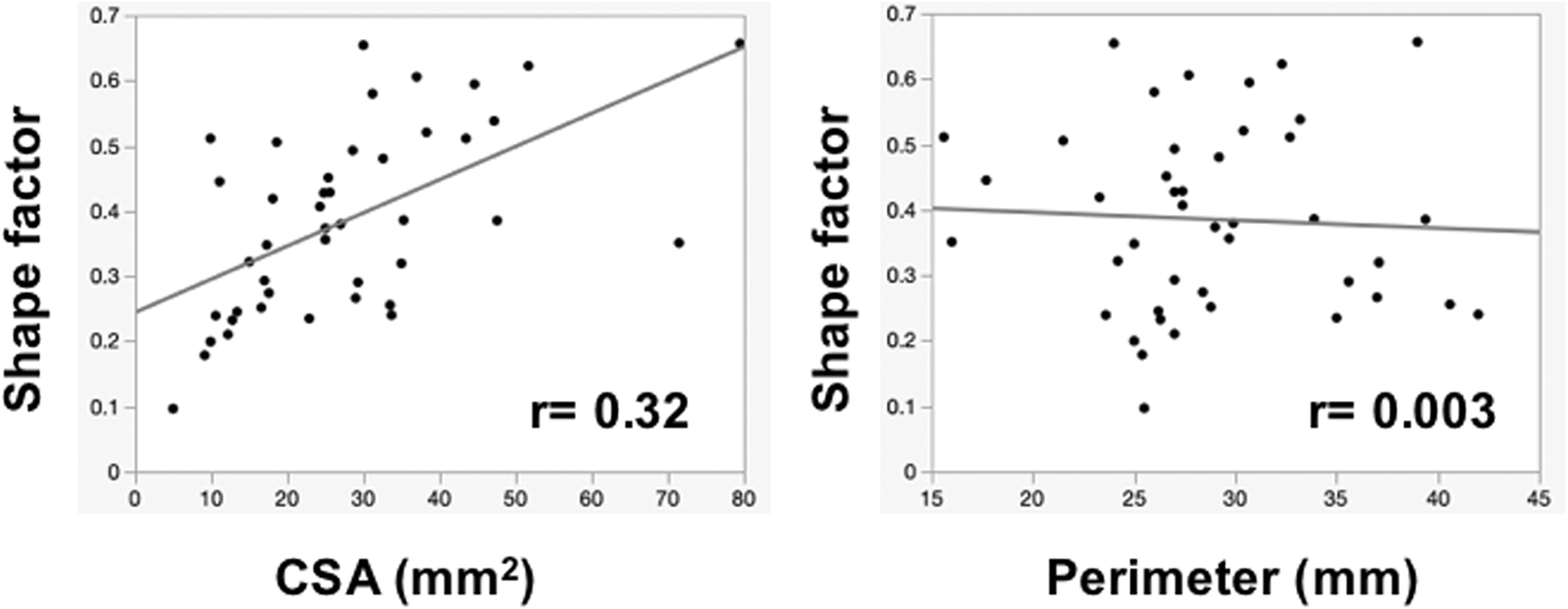
Correlation of CSA and perimeter with SF. CSA and SF were slightly correlated (r = 0.32), whereas perimeter and SF were not correlated (r = 0.003)
Regarding the association of CSA, perimeter, and SF with surgical outcome, group UF had a significantly lower mean CSA (22.7 ± 4.7 mm2) and SF (0.27 ± 0.04) than group F (CSA: 29.5 ± 2.8 mm2; SF: 0.42 ± 0.02) (p = 0.037 and 0.0012, respectively). However, perimeter did not significantly differ between group UF and group F (27.0 ± 1.8 mm vs. 29.4 ± 1.1 mm) (Figure 4).
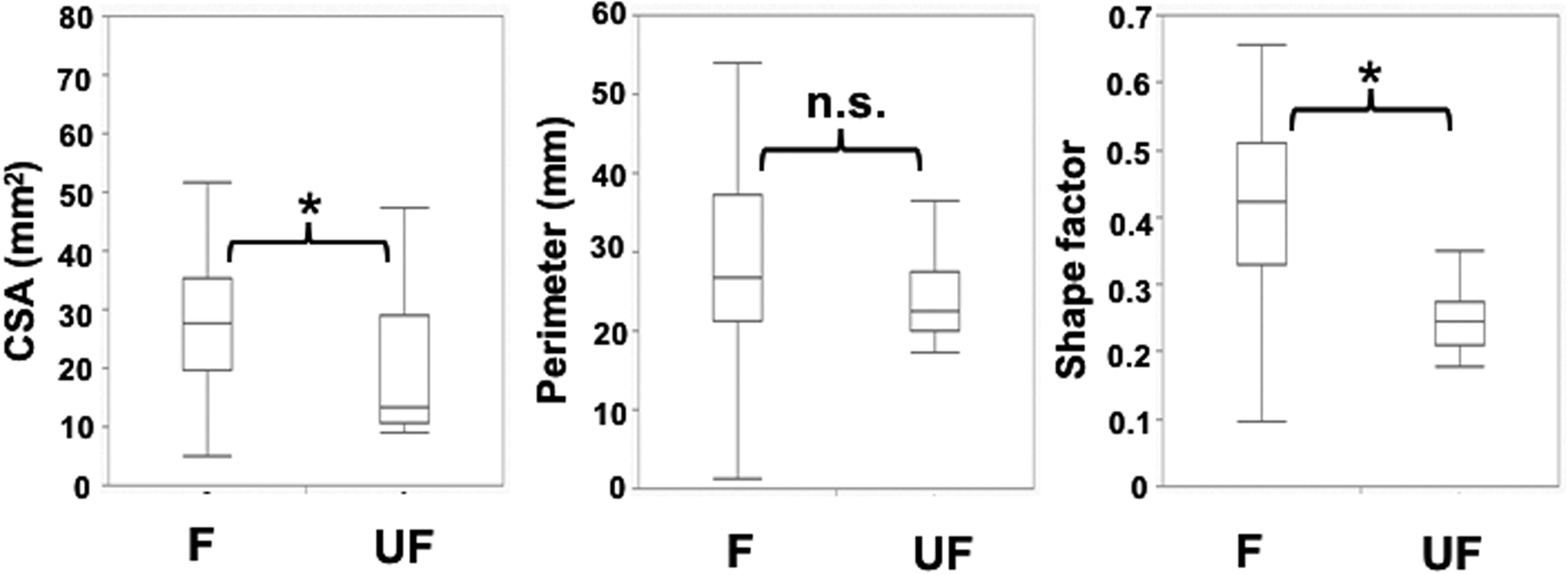
Association of CSA, perimeter, and SF with neurological recovery after surgery for IDEMT. Group UF had a significantly lower mean CSA (22.7 ± 4.7 mm2) and SF (0.27 ± 0.04 mm) than group F (CSA: 29.5 ± 2.8 mm2; SF: 0.42 ± 0.02 mm) (p = 0.037 and 0.0012, respectively). However, the perimeter did not significantly differ between group UF and group F. UF: unfavorable; F: favorable, *: p < 0.05; n.s: not significant.
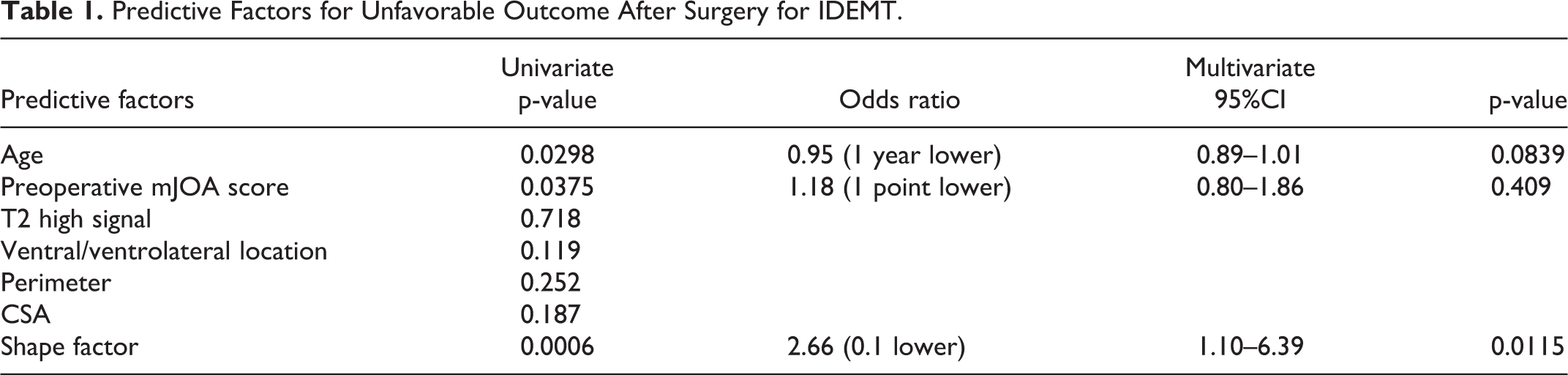
Several factors, including patient’s age, preoperative neurological impairment, location of the tumor, and T2-high at the level of maximal compression of the spinal cord, have been reported as predictors of neurological recovery after surgery for IDEMT (1-3). Hence, we next compared the predictive significance of various factors. In univariate analysis, higher patient’s age, lower preoperative mJOA score, and lower SF were significantly associated with unfavorable outcome (Table 1). In multivariate analysis, SF was a significant and independent prognostic predictor of postoperative favorable outcome (odds ratio = 2.66, 95% CI 1.10–6.39, p
Predictive Factors for Unfavorable Outcome After Surgery for IDEMT.
Next, to evaluate the predictive value of SF, we carried out ROC curve analysis to predict postoperative favorable outcome. AUC for SF was 0.844, and the optimum cut-off value of SF for detecting postoperative favorable outcome was 0.29 (sensitivity 87.5%, specificity 69.3%) (Figure 5).
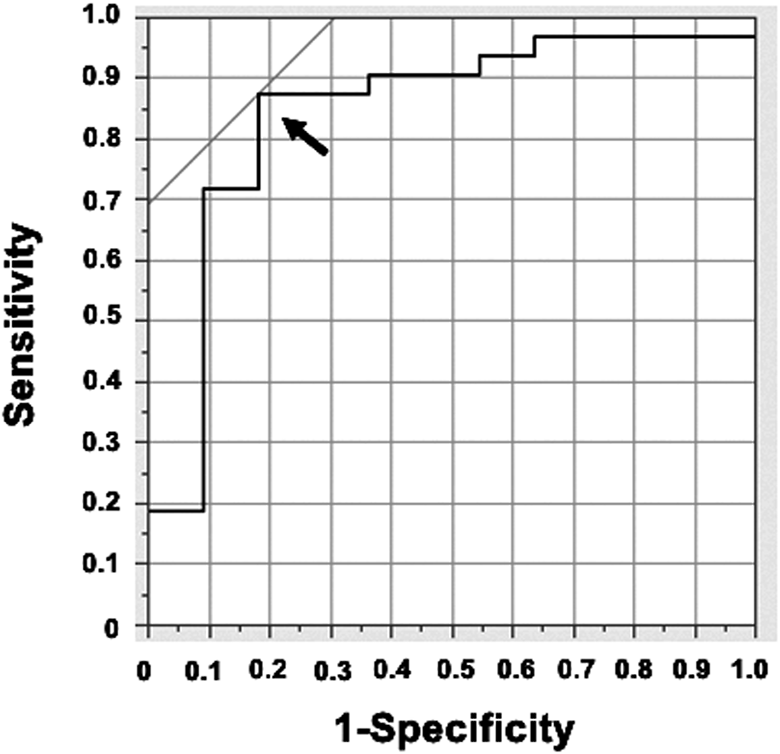
ROC curve analysis of shape factor for predicting postoperative favorable outcome. The optimum cut-off point (arrow) based on ROC curve analysis for SF was 0.29.
Discussion
Several reports have shown that preoperative neurological impairment is a predictive factor for postoperative favorable recovery in case of spinal meningioma, a predominant histology of IDEMT. 11 However, the radiological factors that predict the surgical outcome of IDEMT have not been completely elucidated. Ventral location of the tumors increases the surgical morbidity of spinal meningioma. 12 The relationship between the postoperative functional outcome and the presence of a hyperintense signal in T2-weighted imaging within the spinal cord has also been investigated in the surgical outcome of IDEMT.2,13 However, the associations of tumor size and CSA with functional outcome remain unclear. 1
The morphology of the spinal cord, including CSA, could be of prognostic significance in cervical myelopathy. 14 However, the absolute size of the spinal cord varies widely among patients.4,15 Furthermore, there is an intra-individual variation in the cross-sectional area of the spinal cord, e.g. the area increases caudally, reaching a peak at C5, and then decreases with a minimum at segment T7–T8. 4 These inter- and intra-individual variations of the morphology of the spinal cord may cause difficulties when interpreting morphometric analyses using absolute values. 15 Therefore, we anticipated that CSA would not be a good indicator of the clinical results of IDEMT surgery.
In this study, we proposed that SF could serve as a promising predictive factor for the surgical outcome of IDEMT. SF has several distinct features relative to morphometric absolute measurements such as CSA and perimeter of the spinal cord. First, SF is dimensionless and invariant to the scale of the parameters. Next, SF, but not CSA, fits the normal distribution, suggesting that SF is a more standardized value than CSA, and is therefore suitable for predicting the postoperative neurological recovery of IDEMT. In addition, SF is a relatively robust description that can be derived from simple and reproducible measurement of CSA and perimeter. Based on these advantages, we believe that SF reflects quantitative information that links the morphological features of the spinal cord with its neurological function. Consistent with this, group UF had significantly lower SF than group F, and SF was an independent and significant predictor of postoperative outcome in multivariate analysis.
The ventral/ventrolateral location of tumors is a prognostic factor for the outcome after surgical intervention for IDEMT. Spinal meningiomas are more prevalent in the thoracic region and are often located ventral and ventrolateral to the spinal cord. 16 Increased surgical morbidity in ventrally/ventrolaterally located meningioma has been reported. 11 However, we observed no significant correlation between postoperative favorable outcome and ventral/ventrolateral location of the tumors in patients with IDEMT. These results indicated that the ventral/ventrolateral location of the tumors is not a dominant factor for the development of thoracic myelopathy.
Patient age and severity of preoperative neurological impairment are predictive factors in surgical morbidity and complete recovery in IDEMT.17,18 Consistent with this, patient age and preoperative neurological impairment assessed by mJOA score are significantly associated with insufficient postoperative recovery in univariate analysis. Thus, the combination of these factors with SF might increase the value of SF for predicting the surgical outcome of IDEMT.
This study had several limitations. First, it was a retrospective study, and the number of patients was relatively small. Consequently, we could not include a sufficient number of possible predictive factors, such as histology of the tumor, in the multivariate analysis. In addition, it was difficult to determine the reason for unfavorable postoperative outcome because both the surgical insult itself and the preoperative tumor-induced spinal cord compression could affect surgical outcome. Finally, we measured CSA and perimeter manually. Even though intra- and inter-observer variabilities were very low, automated measurement of those parameters 19 would be more objective, and could increase the reliability of those measurements.
In conclusion, we showed that among the several previously reported predictive factors for surgical treatment of IDEMT, SF was most helpful in predicting surgical outcome. Therefore, measurements of CSA and perimeter, followed by calculation of SF, may provide valuable quantitative information regarding the prospective outcome of surgery for IDEMT.
Footnotes
Authors’ Note
The study was approved by the institutional review board at the Kyushu University Hospital (26-112) opt-out method was applied to obtain the consent of the patients.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (#18K09067).
