Abstract
Study Design:
Multicenter prospective study.
Objectives:
To validate the Early-Onset Scoliosis 24 Questionnaire (EOSQ-24) questionnaire for the Brazilian Portuguese language, a widely used tool for assessing the impact of different treatments and interventions in EOS patients.
Methods:
The EOSQ-24 questionnaire was cross-culturally adapted following guidelines already published. After language adjustments by a group of experts, the final version of the Brazilian Portuguese EOSQ-24 was applied to a group of 76 patients (35 male and 41 female). Internal consistency was evaluated using the Cronbach α coefficient and item-total correlations. Continuous variables were recorded as median values and interquartile ranges and categorical variables as percentages.
Results:
In the study group, 76 patients were evaluated. The total EOSQ-24 Cronbach α coefficient was 0.883, indicating excellent reliability. The internal consistency of EOSQ-24 was assessed in 3 domains: patient quality of Life, parental burden, and satisfaction (Cronbach α: 0.816-0.934). The range across all subdomains was 0.473 to 0.934. Floor effects for the 24 items were between 1.3% and 43.4% and ceiling effects, between 3.9% and 42.1%.
Conclusions:
The Brazilian Portuguese adaptation of the EOSQ-24 shows excellent reliability and can be a valid tool for psychometric assessment of children with EOS.
Keywords
Introduction
The concept of scoliosis is understood as a complex 3-dimensional deformity, accompanied by a deviation of the spine in the coronal plane, reduction of kyphosis in the sagittal plane, and rotation in the axial plane. 1,2 Recently, early-onset scoliosis (EOS), occurring before 10 years of age, was segregated from general scoliosis. In 2012, because of the rapidly expanding volume of medical knowledge about EOS and the impact on survival and treatment, the Scoliosis Research Society (SRS), the most prominent scoliosis research body, published an update on the subject. 3,4
EOS involves a group of deformities of diverse etiologies and natural history, as well as neuropsychomotor impairment and comorbidities, observed through different conformations of the spine curvature. 5 The most common causes of EOS are idiopathic, congenital, neuromuscular, or syndromic. 6
The deformity may inhibit the development and function of the heart and lungs, representing 2 of the most severe consequences of EOS. 7 -9 According to the etiology and evolution of the disease, motor function impairment of the patient is observed, with a consequent decrease in recreational activities and routine activities, such as brushing and bathing, and a compromise of cardiopulmonary, intestinal, and bladder function, together with intense pain. 10
Currently, the specific instrument for assessing the quality of life of patients with EOS is called the Early-Onset Scoliosis 24 Questionnaire (EOSQ-24), developed in the United States in 2011, and applied to the caregivers of children. This questionnaire consists of 24 items, with 11 designed domains to evaluate children’s health-related quality of life and the overload to children’s caregivers. This tool was initially applied in English and showed high internal consistency and reliability 11 as well as validation and application to other languages, such as Chinese, 12 Spanish, 13 Turkish, 14 Norwegian, 15 German, 16 Arabic, 17 and Dutch. 18
In Brazil, there is no available transculturally adapted version validated by international norms to our language. Thus, we cannot objectively evaluate the outcome of any intervention in children with EOS in Brazil, nor can we compare the results of the treatment with those in the international literature. In addition, the functional outcomes focused on caregivers and patients, and performed via questionnaires, are the most accepted form not only for validation of protocols of quality of care, but also for the definition of financing policies from funding sources. Therefore, the purpose of this work was to validate a cross-culturally adapted version of the EOSQ-24 for the Portuguese language spoken in Brazil and to determine the reliability of this questionnaire according to internationally accepted recommendations.
Materials and Methods
The research was approved by the research ethics committee of the Faculty of Medical Sciences of Santa Casa de São Paulo (Approval Number 56858516.0.0000.5479).
Transcultural Adaptation of the Questionnaire
The authors contacted the original EOSQ developers, receiving authorization and support for a Portuguese translation. The transcultural adaptation of the questionnaire in this study was conducted according to guidelines already published in 2000 19 and followed the steps described below.
First, the original questionnaire was translated from English into Portuguese by 2 Brazilian individuals, nonauthors of the work, of whom one had no medical knowledge, named Translator 1 and Translator 2. The native language of both translators was Portuguese, and both were fluent in English. These 2 translations were later collected and revised by a third translator, fluent in both languages as well. A consensus was carried out with both translators and the authors of the study, followed by discussions and finalized with a pretest Portuguese version of EOSQ-24.
Starting from the Portuguese EOSQ-24 pretest, 2 bilingual translators, whose mother language was English and who were fluent in Portuguese, who did not have medical training, and who were unaware of the original version of the questionnaire or of the subject treated translated the pretest version back to English. The expert committee, comprising the translators, 2 orthopedic spine surgeons, and 3 general practitioners, reviewed the translations. After this phase, they performed the comparison of semantic equivalence of both languages and the conceptual meaning of the retrotranslations with the original EOSQ-24, correcting any ambiguous expressions. Thus, the prefinal version of the EOSQ-24 questionnaire was translated into Portuguese.
The prefinal version was then applied to 20 Brazilian patients through their caregivers at the outpatient clinic of the Department of Orthopaedics and Traumatology of the Santa Casa de Misericórdia Hospital São Paulo, Hospital das Clínicas and Associação de Assistência à Criança Deficiente, in São Paulo, Brazil, by 2 research assistants. The assistants interviewed those responsible for the children, requesting them to answer the questionnaire according to their understanding and indicating the best answer for each question. Finally, the interviewees indicated any doubts about ambiguous expressions in the questions. The expert committee met again and modified the questions pointed out with ambiguous expressions in the pretest, developing the final version of the EOSQ-24 questionnaire in Portuguese, which was then applied to 76 individuals from the 3 centers involved in the study. The study was approved by the Ethical Committee of the 3 Centers, and informed written consent was obtained from each child’s parents.
Early-Onset Scoliosis 24 Questionnaire
The questionnaire consists of 24 items in 11 subdomains: general health, pain, respiratory function, mobility, body function, daily life, fatigue, emotional development, parental burden, financial impact, and satisfaction. 20 The 3 domains were quality of life, parental burden, and patients’ satisfaction. 11
The assessment of parents and/or caregivers considered each item relevant to their child’s health condition (relevance) and whether the items were clear enough to understand (clarity). For this purpose, the 5-point Likert scale was used (1, I strongly disagree with, to 5, I totally agree). 21 The Portuguese version of the EOSQ-24 questionnaire was similar to the original EOSQ-24 scoring system, with lower scores meaning greater disability. Domain scores were calculated as follows: (Value of the chosen item − 1)/(4 × 100) for domains of 1 item and (Algebraic average of items − 1)/(4 × 100) for domains composed of more than 1 item, which ranged from 0 to 100. The EOSQ-24 total score was the average score of 11 subdomains calculated as follows: (Items point average for subdomains − 1)/(4 × 100), rescaling the scoring metric from 1 to 5 to 0 to 100. The clarity and comprehension of EOSQ-24 translated into Portuguese was assessed using the 5-point Likert scale used for parents and/or caregivers. 11,22,23
Internal Consistency
Internal consistency was assessed using the Cronbach α coefficient. This test indicates the homogeneity of the distinguishing factors between items within a questionnaire or subdomains of the questionnaire. Low Cronbach α indicates a lower correlation between items designed to measure the same construct, whereas a high Cronbach α indicates redundancy between 1 or more items. The recommended value for Cronbach α is between 0.70 and 0.95. 23
An item total correlation higher than 0.3 indicated a good correlation with the domain and good discriminative validity; correlations between 0.2 and 0.8 indicated good internal consistency of the item. 23,24
Ceiling and Floor Effects
Ceiling and floor effects represent limitations of an instrument’s ability to assess the full spectrum of a condition’s severity within the items it contains. They refer to content validity and indicate that extreme items are missing on the scale. Descriptive statistics (means, SDs, and quartiles) were calculated to determine the distribution for domains. These effects were analyzed by calculating the frequency of participants giving the minimum and maximum scores possible. 4,25,26 Within the orthopedic community, ceiling and floor effects are defined as 15% (or more) of individuals in the sample reaching the best or the worst score levels, respectively. 27 -30
Statistical Methods
Continuous variables were recorded as means, SDs, medians, and interquartile ranges (Shapiro-Wilk test). Categorical variables were recorded by the absolute number of occurrences and the percentages involved. For internal consistency analysis, the Cronbach α reliability test was used in each group of questions, characterizing a domain of the questionnaire, in addition to the overall internal consistency index involving the whole questionnaire. For analysis of the ceiling and floor effects, 15% of the patients who obtained the lowest or the highest possible scores were considered. Data analyses were performed using SPSS 23.0 for MAC program (IBM SPSS Inc, Chicago, IL). A value of P <.05 was used for statistical significance.
Results
The patients’ parents and/or primary caregivers (41 women, 35 men; 76 total) with EOS were asked to complete the EOSQ-24. The average age was 11.88 years, with a SD of 4.15, and the distribution was normal, ranging from 3 to 24 years (P = .107; Table 1; Figure 1). The response rate was 100%, and all questionnaires were completed; however, 21 of the 1840 (1.15%) responses were inappropriately marked and not tabulated. The internal consistency represented by Cronbach α value and the ceiling and floor effects for each domain, subdomain, and item of the EOSQ-24 Brazilian version as well as the means, SDs, and median values are presented in Table 2.
Demographic and Clinical Characteristics of Patients With EOS.
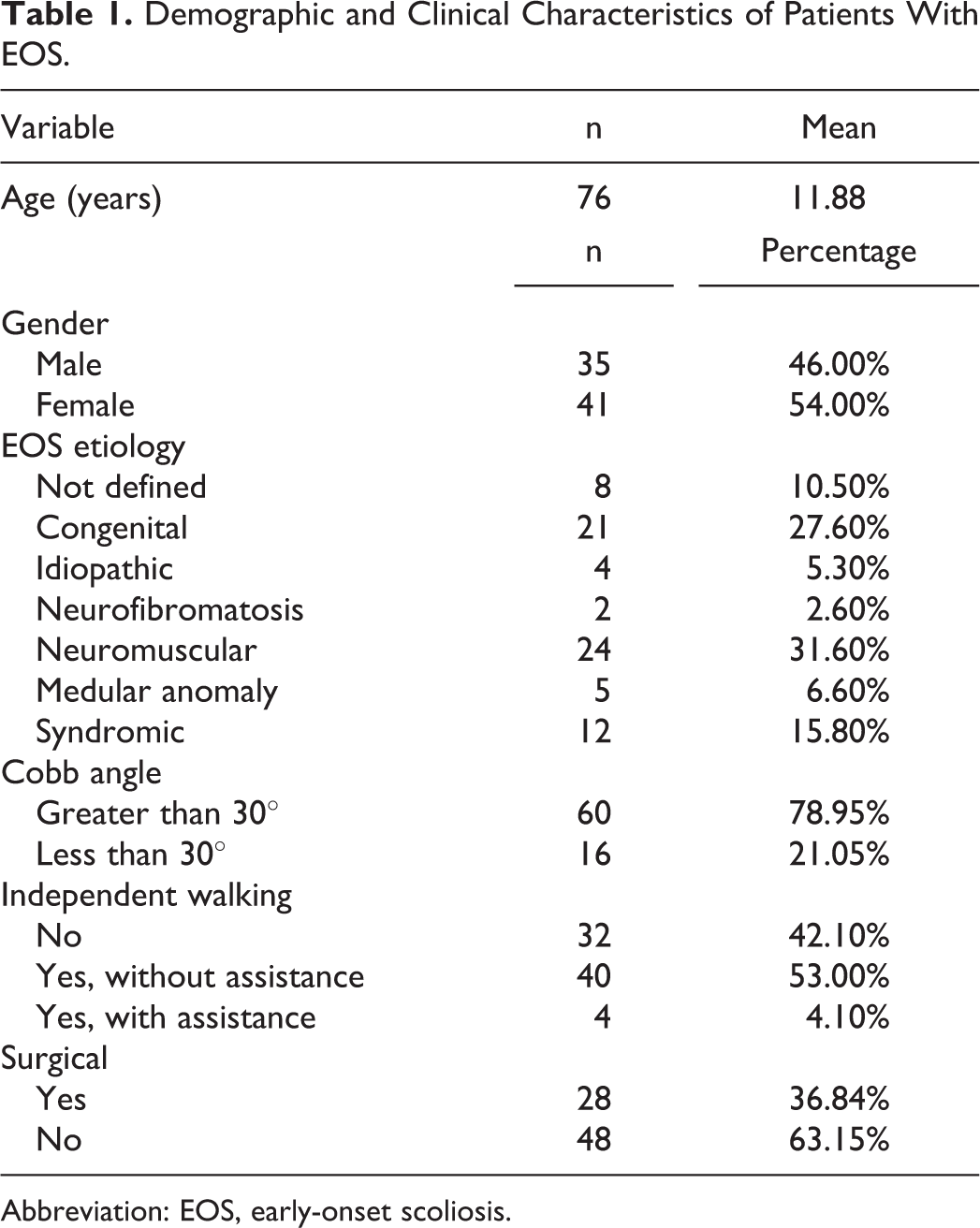
Abbreviation: EOS, early-onset scoliosis.
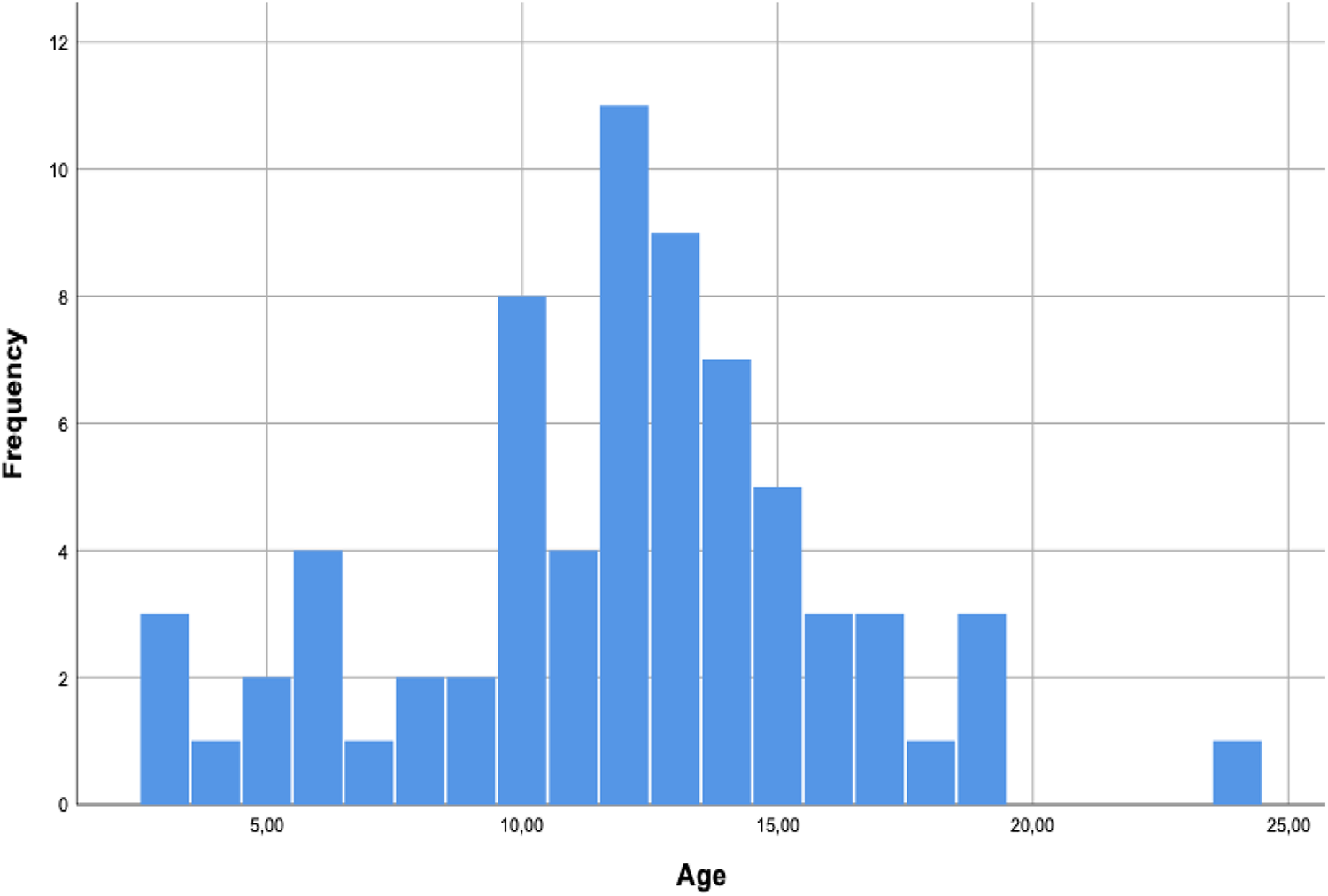
Age-normogram related for EOS patients.
Descriptive Statistics of EOSQ-24 Items, Subdomains, and Domain Scores.
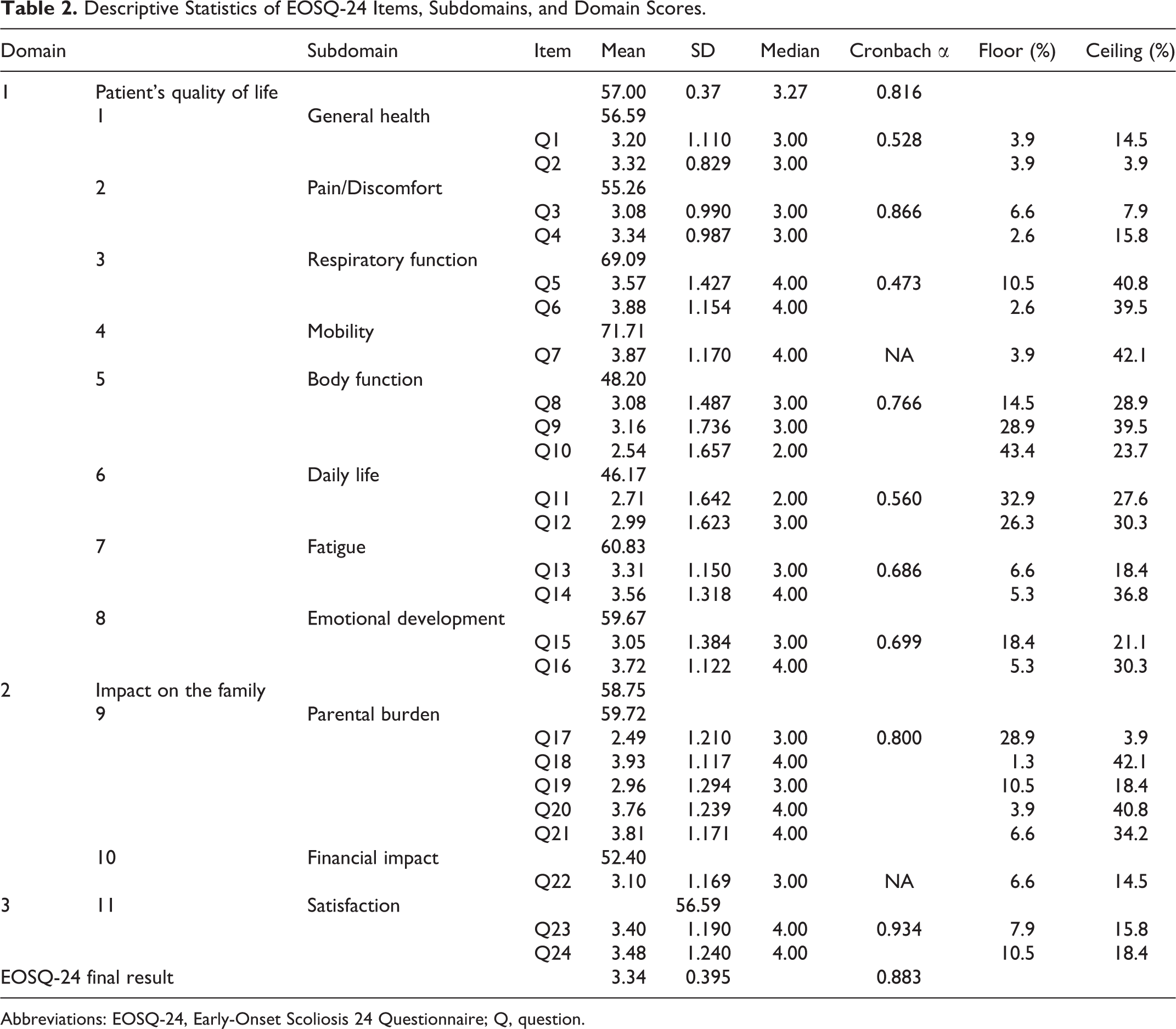
Abbreviations: EOSQ-24, Early-Onset Scoliosis 24 Questionnaire; Q, question.
Internal Consistency
Mean, SDs, ceiling and floor effects of EOSQ24 item scores, and subdomain, domain, and total scores can be seen in Table 2. The total Cronbach α coefficient for EOSQ-24 score was 0.883, indicating excellent reliability. The internal consistency of the 3 domains of the EOSQ-24—patient’s quality of life, family burden, and satisfaction—were excellent (Cronbach α: 0.816-0.934). The range across all subdomains was 0.473 to 0.934.
Ceiling and Floor Effects
For the study population, the mean score of individual EOSQ-24 items was between 2.49 (Q17) and 3.93 (Q18), both from the parental impact domain; the mean scores of the domains ranged from 46.17 (daily life) to 71.71 (transfer; Table 2).
The response pattern showed that 12 items (50%) were not skewed because items Q1, Q2, Q3, Q4, Q8, Q12, Q13, Q15, Q17, Q19, and Q22 showed a median value of 3 (Table 2). The other items were skewed because items Q5, Q6, Q7, Q14, Q16, Q18, Q20, Q21, Q23, and Q24 showed responses redirected to pulmonary function, transference, fatigue, emotion, parental impact, and satisfaction.
The items Q10 and Q11, physical function and daily life showed a median value of 2, resulting in a ceiling effect of 1.3% to 43.4% and a floor effect ranging from 3.9% to 42.1% of patients (Table 2).
Discussion
Cross-cultural translation and adaptation of the original EOSQ-24 are presented here, following international guidelines, together with an evaluation of its reliability and discriminative capacity. The Brazilian Portuguese version of EOSQ-24 presented satisfactory results and can be applied in our country. The internal consistency of the EOSQ-24 as measured by the Cronbach α coefficient was good for the 24 items and the 11 domains. To our knowledge, there is no Brazilian Portuguese version available on the Cronbach α coefficient for comparison. 11,14 The internal consistency of the present questionnaire was excellent (0.883), compared with the recently validated Chinese (0.896), Spanish (0.897), Turkish (0.909), and Norwegian (0.950) questionnaires. 12 -15
Recently, the translation and cross-cultural validation of the EOSQ-24 questionnaire was published in German, 16 Arabic, 17 and Dutch 18 and showed good validity and reliability. The Dutch 18 study did not have a follow-up, necessary to validate EOSQ-24 for use in a clinical setting. The study discriminated patients with different curve severities, diagnoses, and outpatient status. The German 16 version of the questionnaire was applied to parents or guardians of 67 patients, and about 55 patients were surgically treated with growth-friendly implants, different from our nonsurgical sample (63.15%). Finally, the Arabic 17 version also used the concept of EOS before the age of 10 and was applied to caregivers of 58 patients who were surgically treated. The results are similar to ours, with few losses and low floor and ceiling effects.
Items correlation presented good discriminatory validity for all domains. However, the respiratory function subdomains, daily life and emotional development, showed low reliability. One possibility is the diagnosis heterogeneity for our population, similar to that in the Turkish study. 14 It is worth mentioning that 31.6% of our patients have neuromuscular or syndromic scoliosis involving different parental overload compared with patients with congenital or idiopathic scoliosis. The heterogeneity can be explained by differences in the nonambulatory condition of 9 children and the severity of each deformity (Cobb angles). 31 Note that only 18 out of 76 patients had measurements of the Cobb angle smaller than 30°. Moreover, the score distribution of the parents’ responses involved patients with different ages, etiologies, and treatment modalities.
EOS is a condition that limits the daily activities of children, leading to a significant impact on the quality of life of caregivers/parents. Early morbidity and mortality justify the search for interventions to alter the natural history of this potentially lethal deformity. 32 A short decompensated trunk, in addition to aesthetic questions, leads to respiratory insufficiency syndrome and dysfunctions that inevitably affects the lives of these children. 7 Pulmonary parenchyma develops up to 10 years of age, and arthrodesis before this age is associated with adverse functional outcomes of lung function. 3 Interventions should reduce or stop the deformity growth and improve body function, together with psychosocial issues related to aesthetic-functional improvements in these children. 20 The options include serial plasters and instrumentation systems to allow the spine growth with periodic distensions. However, frequently described adverse effects, such as implant releases, subcutaneous adipose tissue lesions, infections, and impossibility to control the curve progression are reported. 3,6,7,32
Currently, there is no consensus about the ideal intervention or the best treatment of children with EOS. Numerous procedures involving sedation and anesthetics can affect the growing brain and family psychological dynamics. 20 Outcomes are always multidimensional and include what matters to children and their caregivers and not just to the surgeon. In fact, the procedures must cover the whole cycle of care of those responsible. Quality intervention includes outcomes that affect the child’s life, not just the radiological aspects. This criterion is recommended in decision-making for evidence-based medicine and funding from paying sources. 4,33
In 2012, there was a narrative review of the literature regarding the translation and validation of questionnaires developed by SRS, such as SRS-22, SRS-24, and SRS-30. Eight publications described the translation and validation of SRS-22 and SRS-24 questionnaires in Spanish, Japanese, Turkish, Chinese, Italian, and German. 34 Finally, a Brazilian version of the SRS-30 questionnaire was presented after testing 20 patients and recommended that, in Brazil, questionnaire completion should be assisted by a professional, preferably in the health area, because some patients could not understand the questions. 34
The treatment of EOS aims to improve not only the natural history but also the health-related quality of life of children, in addition to minimizing the overload of caregivers. Unfortunately, measurements of quality of life are difficult because of the heterogeneity of the population, and adult health status measures are not the same for children. The EOSQ-24 was designed to solve this issue through a psychometric approach to this population. 11,20 Thus, based on this transcultural study and the adaptation of the original English version of the EOSQ-24 to the Brazilian Portuguese version, with excellent reliability and discriminative capacity, it is possible from now on to conduct clinical research studies in this population
This study has limitations. First, even though the EOSQ-24 version for Brazilian Portuguese has internal consistency and discriminative validity, additional testing should be conducted in specific disease groups—in this case, using the adapted questionnaire to evaluate psychometric properties with convergent validity, temporal reliability, and responsiveness. Second, despite having been carried out in 3 reference centers in São Paulo, our country has continental dimensions, and the Brazilian population is culturally heterogeneous to justify a multicentered approach.
Conclusions
The version of the EOSQ-24 questionnaire for the Brazilian Portuguese language is presented here and validated cross-culturally (Supplemental Material). The EOSQ-24 questionnaire showed good internal consistency for application to caregivers of patients with EOS (Cronbach α of 0.883). Therefore, the EOSQ-24 can be used for clinical and research projects.
Supplemental Material
Supplemental Material, Manuscript_finalR1 - Validation of the Brazilian Portuguese Version of the 24-Item Early-Onset Scoliosis Questionnaire
Supplemental Material, Manuscript_finalR1 for Validation of the Brazilian Portuguese Version of the 24-Item Early-Onset Scoliosis Questionnaire by Rodrigo G. M. De Mendonça, Lucas M. Bergamascki, Karla C. M. da Silva, Olavo B. Letaif, Raphael Marcon, Alexandre F. Cristante, Hiroko Matsumoto, Michel G. Vitale and Robert Meves in Global Spine Journal
Supplemental Material
Supplemental Material, Portuguese_EOSq-24v - Validation of the Brazilian Portuguese Version of the 24-Item Early-Onset Scoliosis Questionnaire
Supplemental Material, Portuguese_EOSq-24v for Validation of the Brazilian Portuguese Version of the 24-Item Early-Onset Scoliosis Questionnaire by Rodrigo G. M. De Mendonça, Lucas M. Bergamascki, Karla C. M. da Silva, Olavo B. Letaif, Raphael Marcon, Alexandre F. Cristante, Hiroko Matsumoto, Michel G. Vitale and Robert Meves in Global Spine Journal
Footnotes
Authors’ Note
Authors contributions are as follows: RGMDM, R Meves, R Marcon, AFC: data acquisition. RGMDM, LMB, KCMdS, OBL: analysis and/or interpretation of data. RGMDM, LMB, KCMdS, R Meves: drafting the manuscript. OBL, AFC, R Marcon, HM, MGV: revising the manuscript critically for important intellectual content. RGMDM, R Meves, R Marcon, AFC, LMB, KCMdS, OBL, HM, MGV: approval of the version of the manuscript to be published.
Acknowledgments
The authors would like to thank Dan Kiyomoto for his invaluable assistance with the statistical analysis of the data. The authors also thank Tânia Fernanda C. da Silva, the Research Coordinator at FMUSP. We acknowledge the enterprise Escrever Ciência for scientific editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
The supplemental material is available in the online version of the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
