Abstract
Study Design:
Retrospective cohort study.
Objective:
To determine if local administration of liposomal bupivacaine (LB) reduces postoperative pain scores and narcotic use in spinal deformity patients.
Methods:
Adult patients undergoing elective spinal fusion (7 or more levels) for scoliosis or kyphosis were selected for inclusion. Patients received either periincisional injections of combined liposomal and standard bupivacaine (n = 90, group L) or standard bupivacaine only (n = 69, group C). Perioperative pain scores (VAS [visual analogue scale]), opioid use, length of stay, functional outcome (ODI [Oswestry Disability Index]), and perioperative complications were recorded. No external funding was received for this study.
Results:
A total of 159 patients met inclusion criteria (mean age was 54.2 years of age). No significant baseline demographic differences were noted between the 2 groups. Group L experienced slight improvements in pain control on postoperative day (POD) 1 (P = .02). No difference in pain scores were otherwise noted. Group L transitioned off of intravenous (IV) narcotics faster with 52.6% less IV use by POD3 (P = .03). No differences in total narcotic consumption, perioperative complications, lengths of stay, and functional outcome scores were otherwise noted between the 2 cohorts.
Conclusions:
The use of LB in adult spinal deformity surgery does not appear to provide clinically important improvements in postoperative pain at the manufacturer’s recommended dosage. Furthermore, while patients receiving LB may transition more quickly off of IV narcotics, this does not appear to translate into an overall decrease in narcotic consumption, hasten return of bowel function, or decrease hospital lengths of stay. Future prospective randomized control trials are warranted. The use of varying dosages of LB may also help further clarify the true efficacy of LB in the setting of spinal deformity surgery.
Introduction
Adult spinal deformity (ASD) affects up to 60% of the older population. While only 6% of these patients are symptomatic, pain, which may be severe, is the most common associated symptom. 1,2 Consequently, ASD has been associated with poor health-related quality of life measures 1,3 and may even be more physiologically debilitating than other chronic disease states such as diabetes, congestive heart failure, and chronic obstructive pulmonary disease. 4 Fortunately, surgical deformity correction has been shown to substantially improve outcomes in these patients. 2,5 However, as these surgeries can be associated with significant morbidity, consequent postoperative pain may be quite severe.
Inadequate postoperative pain management has been shown to increase the risk of perioperative complications such as thromboembolic events and pulmonary complications in many surgical settings. 6 -8 Furthermore, poor pain control in the first 48 hours following spinal surgery has been correlated with only a 50% chance of achieving long-term satisfactory pain relief. 7 On the contrary, adequate pain management following spinal surgery has been associated with quicker mobilization, decreased narcotic consumption, and shorter hospital lengths of stay. 9,10 However, the most optimal pain regimen following spine surgery remains to be determined.
Surgical site infiltration with an amide anesthetic has long been utilized as an adjunctive modality of pain control in surgery. More recently, there has been a surge in interest in the utility of long-acting liposomal bupivacaine (LB) for improving postoperative pain control. In 2011, LB (Exparel, Pacira Pharmaceuticals, San Diego, CA) was approved by the US Food and Drug Administration (FDA) for use as a local anesthetic at doses up to 266 mg for surgical site infiltration. LB is composed of a phospholipid bilayer that encapsulates bupivacaine, which allows for slow release of the drug. This formulation results in extended delivery of the drug for up to 72 hours after initial administration. 11 To date, while findings are mixed, there is evidence to suggest that LB may improve pain control and decrease opioid requirements when utilized as part of a multi-modal pain control regimen. 12
In the setting of spine surgery, specifically, LB has been associated with improvements in postoperative pain control, decreased total narcotic utilization, and shorter hospital stays. However, the number of investigative studies is small and mainly limited to the setting of single-level surgery. 9,10,13,14 As such, the utility of this drug in the setting of adult deformity surgery has not been studied to date. Consequently, the primary objectives of this study were to determine whether LB (1) reduces postoperative narcotic utilization when compared to plain bupivacaine alone and (2) improves postoperative visual analogue scale scores (VAS) following adult deformity surgery. Secondary objectives were to assess the effect of LB on hospital length of stay and perioperative complication rates. We hypothesized that LB use would results in improvements in postoperative pain, ultimately decreasing narcotic utilization and hospital lengths of stay in patients undergoing ASD correction.
Materials and Methods
This was a retrospective cohort study with a historical comparative group conducted within a single surgeon’s clinical practice. All adult patients >18 years of age undergoing either elective primary or revision long instrumented fusion (≥7 levels) between 2012 and 2017 were included in the study. Exclusion criteria included tumor, infection, and trauma cases. All patients underwent spinal deformity correction at a single hospital.
Patient characteristics were obtained from the practice database. These included demographic information (age and sex), surgical diagnoses, surgical procedure, as well as baseline comorbidities and surgical history.
Outcome measures included postoperative VAS scores for back pain collected every 4 hours averaged over each postoperative day (POD), inpatient opioid use (converted to oral morphine-equivalents), and total hospital lengths of stay. Additionally, all perioperative complications were recorded. Finally, Oswestry Disability Index (ODI) scores were obtained at the initial preoperative visit and at the 6-week postoperative visit.
Intervention Versus Comparative Cohort
All patients underwent deformity correction under general anesthesia. Prior to incision, both groups received periincisional 30 mL of 0.5% bupivacaine with epinepherine (5 mg/mL). At the conclusion of the case, the Exparel cohort received 20 mL of LB (266 mg) diluted to 120 cc with NS (normal saline) into both the paraspinal musculature and subcutaneous tissue, bilaterally, along their entire lengths utilizing an 18-gauge spinal needle. The comparative cohort received 30 mL of 0.5% bupivacaine utilizing a similar distribution technique. All patients were then admitted to the hospital and began physical therapy for postoperative mobilization on day 1 following surgery.
The standard postoperative pain regimen included initial patient-controlled anesthesia with 0.2 mg dilaudid Q8 min with an additional Q1 hour nurse directed bolus of 0.4 mg as needed for breakthrough pain. Additional medications included 650 mg PO (per os) acetaminophen Q4 hour, PO oxycodone 5 mg 1 to 2 tablets Q4 hour, PO flexeril 5 mg Q8 hour, or PO valium 5 mg Q8 hour. Nonnarcotic naïve individuals additionally were prescribed oxycontin 10 mg PO BID.
Statistical Analysis
Unadjusted group comparisons were performed with the use of χ2 and student t tests. Shapiro-Wilks was used to test for normality. A χ2 test was used for categorical variables, and an independent Student’s t test was used to assess continuous variables. Generalized linear mixed models were used to compare the temporal trend of postoperative outcomes. A P < .05 was set as our measure of statistical significance.
Source of Funding
There was no external source of funding.
Results
Patient Characteristics and Surgical Factors
A total of 159 patients met inclusion criteria. There were 90 patients who received LB (group L) and 69 patients in the historical cohort who received bupivacaine alone (group C). Mean age was no different between the 2 cohorts. There was no difference in comorbidity profile and surgical diagnoses between the 2 cohorts. All descriptive characteristics of both cohorts are shown in Table 1.
Patient Baseline Characteristics and Surgical Factors.
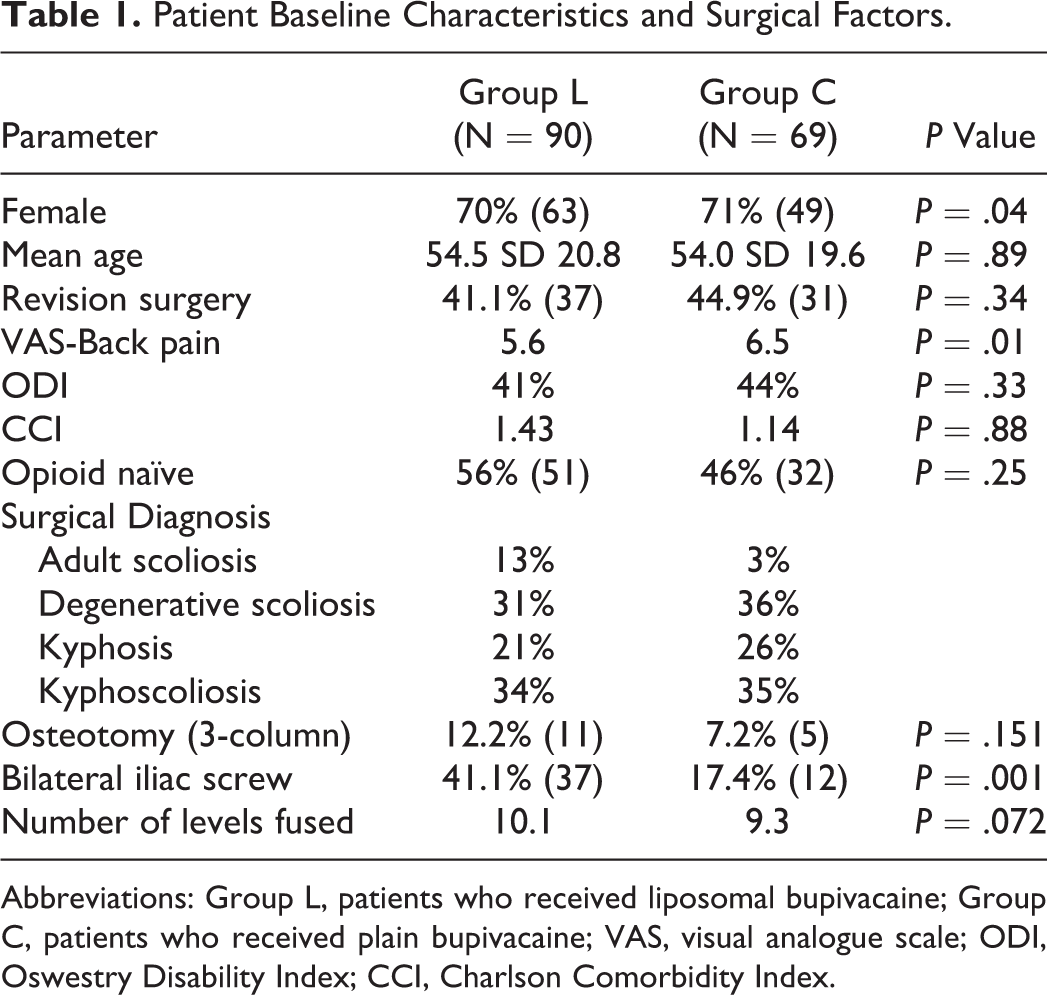
Abbreviations: Group L, patients who received liposomal bupivacaine; Group C, patients who received plain bupivacaine; VAS, visual analogue scale; ODI, Oswestry Disability Index; CCI, Charlson Comorbidity Index.
Postoperative Opioid Utilization
Group L received 259 mg of total morphine-equivalents versus 316 mg in Group C over the course of their hospitalization. Group L appeared to transition off intravenous (IV) narcotics significantly faster, with 52.6% less IV use by postoperative day 3 compared with the control arm (12.0 vs 25.4 mg, P = .03). Otherwise, no statistically significant differences were noted at any time point in opioid consumption between groups (IV, PO, and total consumption). Trends in narcotic utilization (IV, PO, and total consumption) are demonstrated in Figures 1 through 3. Finally, linear regression analyses revealed no difference between any of the trends in narcotic consumption.
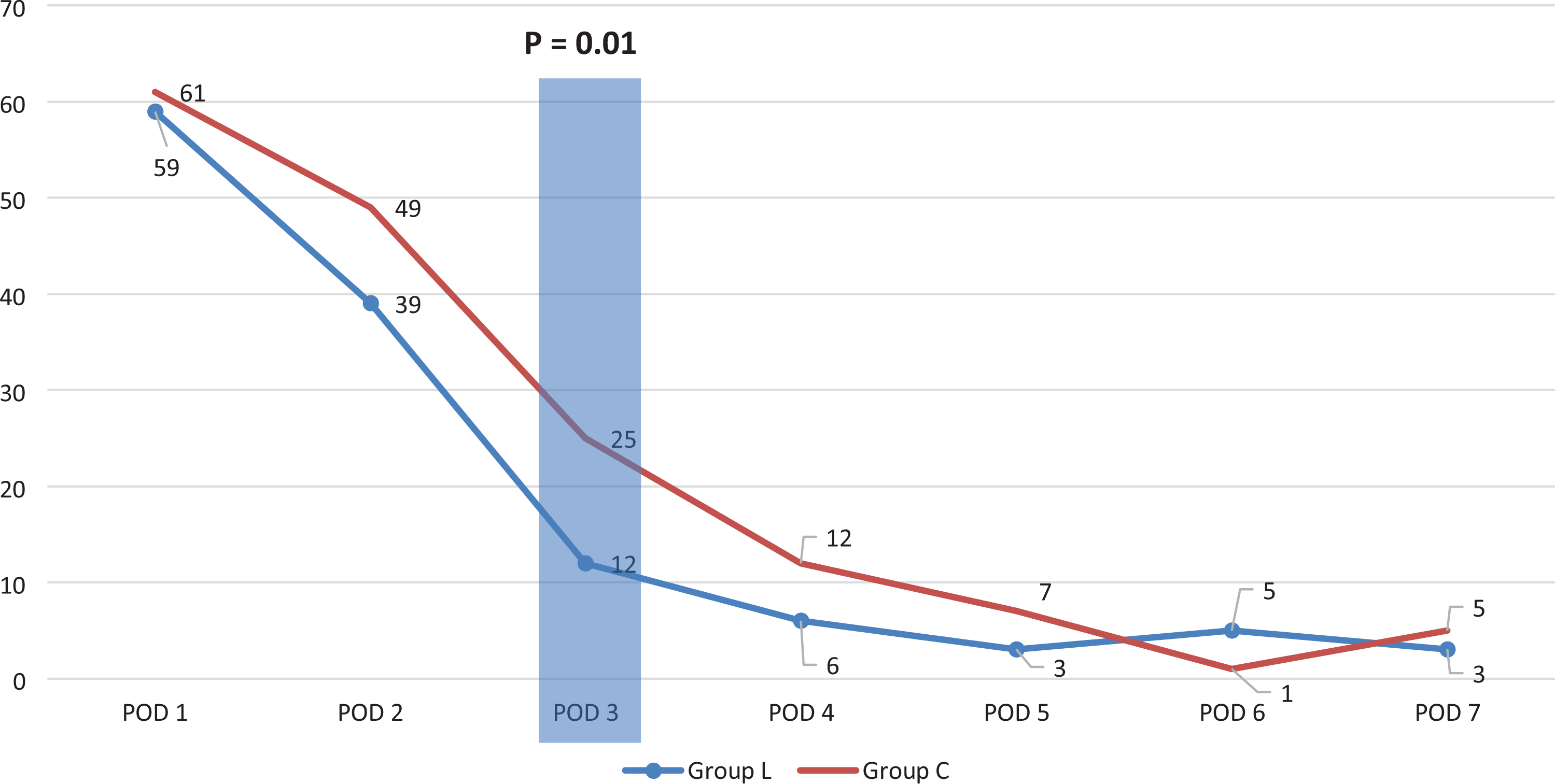
Inpatient intravenous (IV) opioid consumption (expressed in morphine-equivalents).
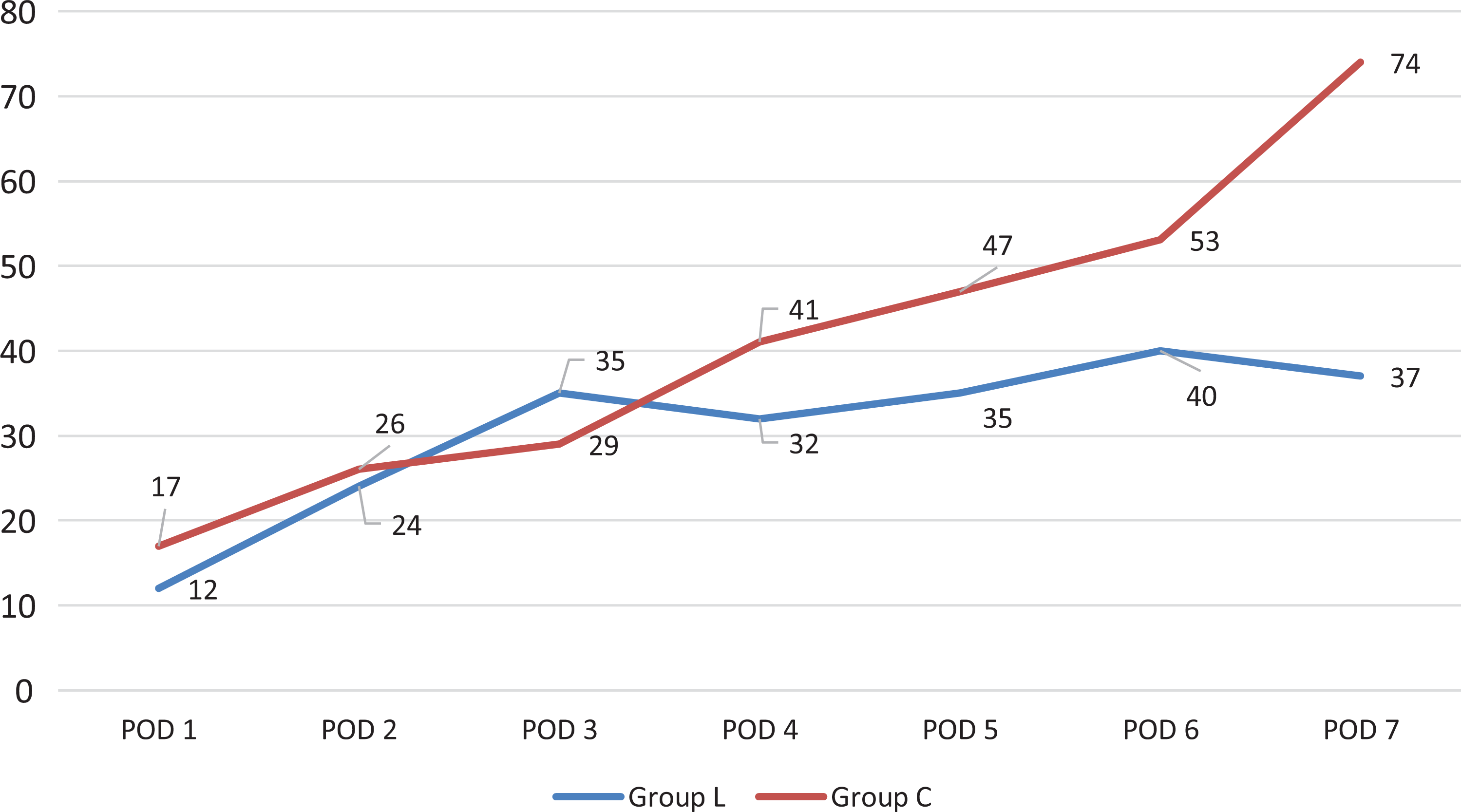
Inpatient (PO) opioid consumption (expressed in morphine-equivalents).
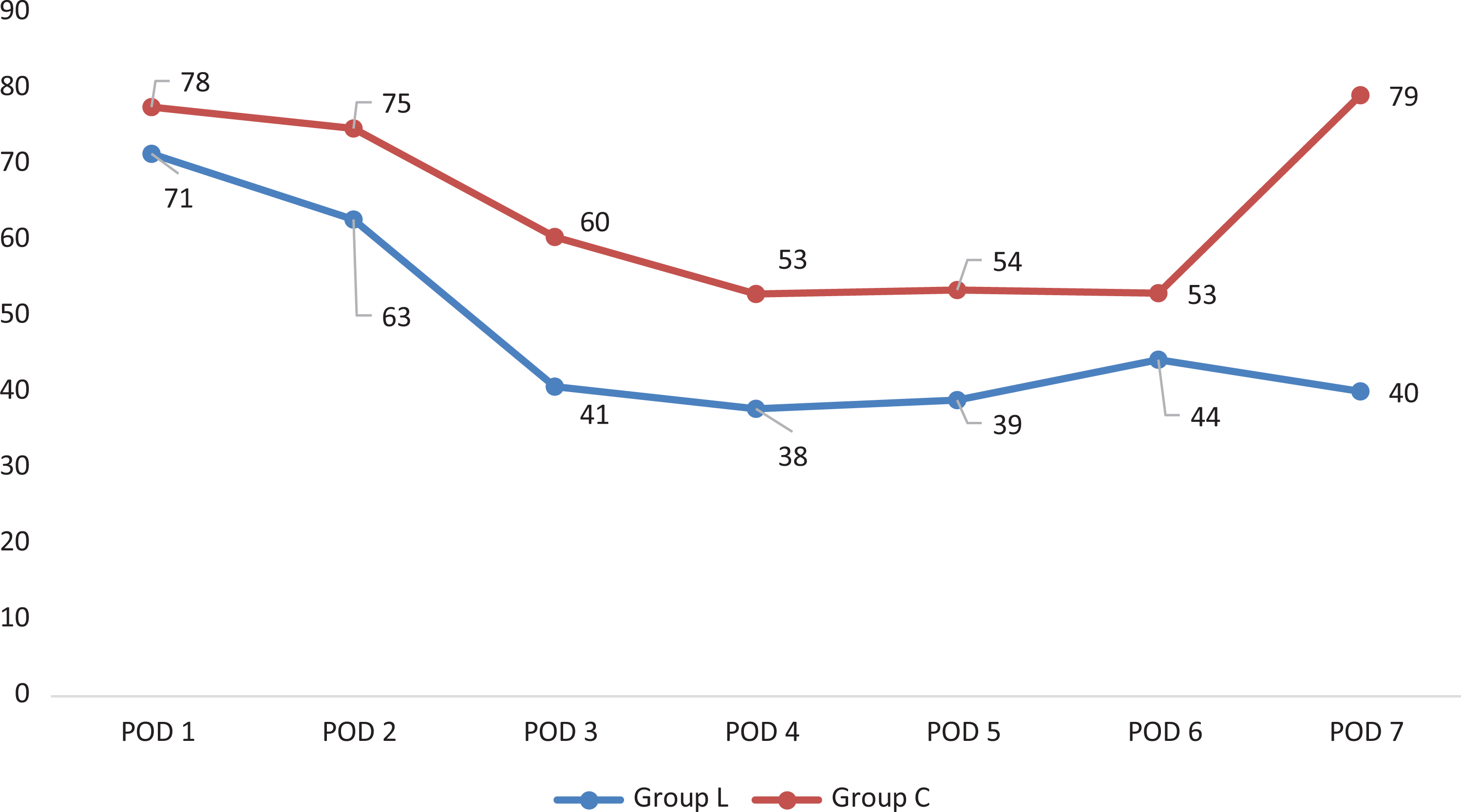
Inpatient total opioid consumption (expressed in morphine-equivalents).
Postoperative Pain Scores
On POD1 there was a mild difference in postoperative pain experienced between group L when compared with group C (4.8 compared to 5.7; P = .02). Otherwise, no further differences in pain scores were appreciated between the 2 cohorts following surgery (Figure 4). Linear regression analysis revealed no difference between the trends in pain scores between the 2 cohorts.
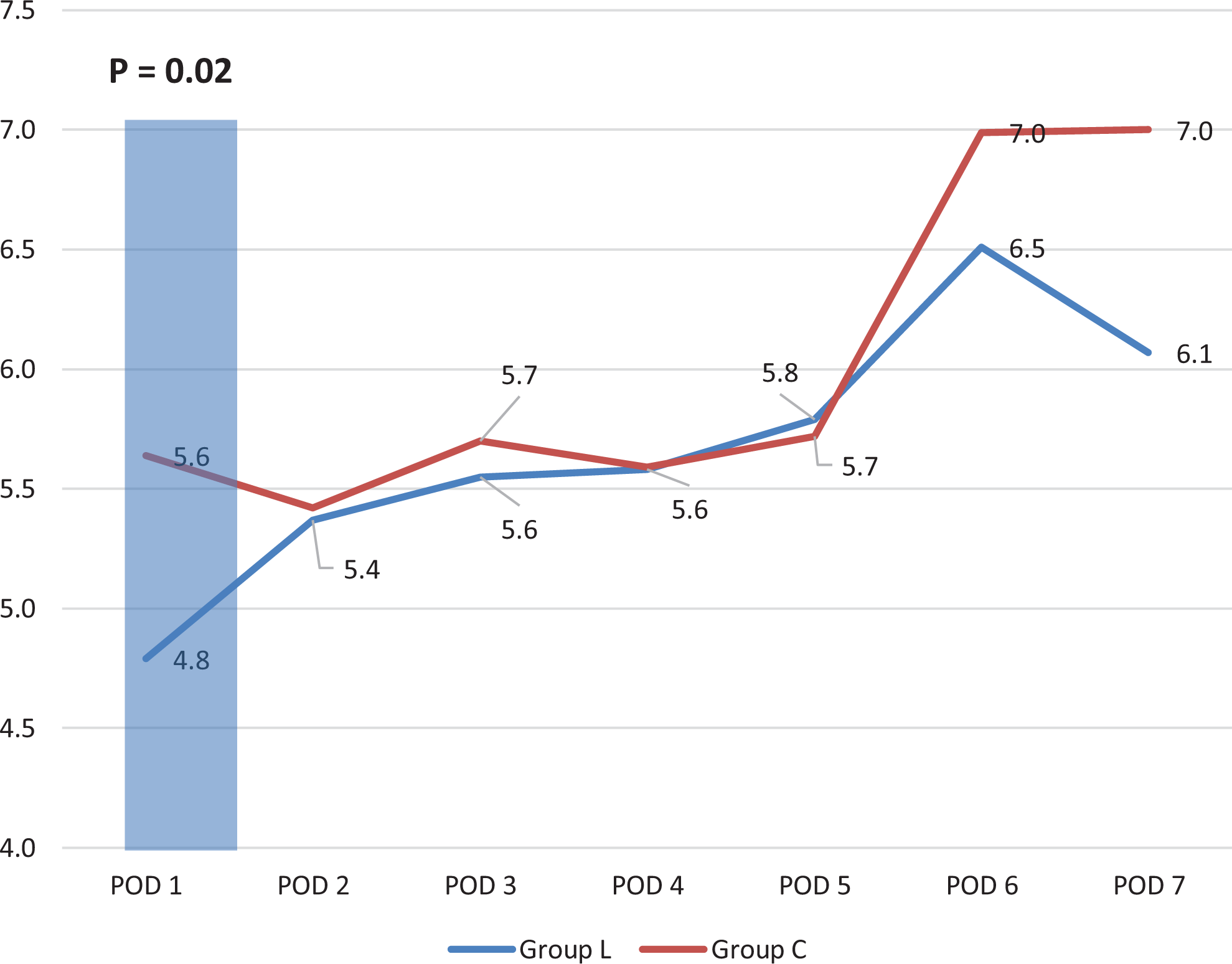
Inpatient pain scores.
Total Hospital Length of Stay and Perioperative Complications
No meaningful difference in lengths of stay were observed between the 2 cohorts (L: 4.7 vs C: 4.8). There were also no differences in postoperative complication rates overall (Table 2). Specifically, there was ileus (L: 7 [7.8%] vs C: 3 [4.3%]; P = .216) and superficial wound infection (L: 1 [1.1%] vs C: 0; P = .189).
Complications by Group.
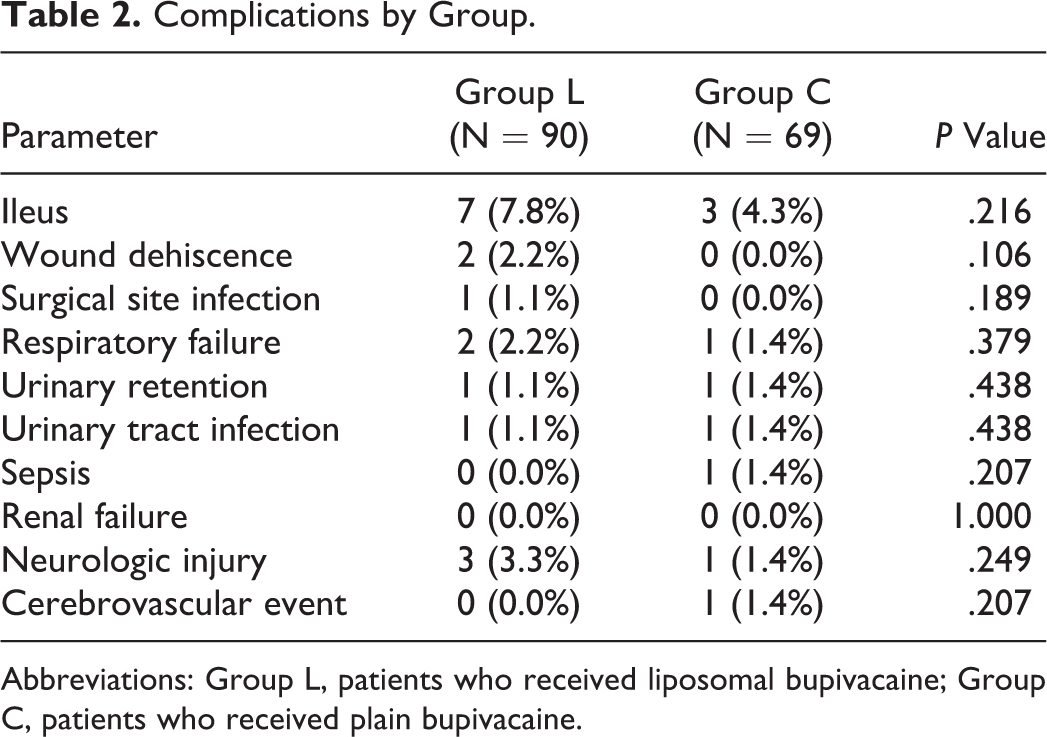
Abbreviations: Group L, patients who received liposomal bupivacaine; Group C, patients who received plain bupivacaine.
Patient-Reported Outcome Measures
No differences between preoperative and postoperative ODI scores were observed between the 2 cohorts.
Discussion
Traditionally, opioid medications have been the cornerstone of postoperative pain management in surgery. However, as narcotic overuse has become more prevalent, a substantial increase in the rate of opioid dependency, drug-related deaths, and surge in health care resource utilization 15 -18 has ensued. Consequently, new government regulations have emerged that place greater restrictions on the ability of physicians to prescribe narcotics with some laws even penalizing surgeons for excessive utilization. 19 Perhaps most concerning is the considerable side effect profile of opioid medications, which includes respiratory depression, gastrointestinal dysmotility, confusion, and even death. 20 Furthermore, excessive narcotic utilization in the postoperative period has been linked to paradoxically poorer long-term pain control. 21,22
Relatedly, a direct link between adequate pain management and improved perioperative outcomes has been established. Shorter hospitalizations, quicker postoperative mobilization, and a lower rate of perioperative complications are all cited benefits of satisfactory postoperative pain control. 9,10 Appropriate management of postoperative pain may even decrease rates of chronic pain syndromes. 14,23 Furthermore, since 2002, the Agency for Health Care Research and Quality developed the HCAHPS survey, a standardized national survey that allows for direct patient assessment of their perceived quality of hospital care with adequate pain management being one of the quality metrics observed. Inevitably, in light of these aforementioned issues, there has been a surge in interest in the development of the optimal multimodal postoperative pain regimen in all surgical settings. 24
In spine surgery, early evidence has demonstrated that multimodal pain regimens can result in improved postoperative pain control, decreased narcotic utilization, improved overall outcomes, and patient satisfaction following spinal surgery. 20,25 While the optimal pain management protocol remains to be determined, the use of adjunctive local infiltrative anesthetics has shown some utility. 20,25 -27 Most recently, as there has been literature to support the use of LB as part of a successful multifaceted pain control protocol in many surgical settings, 28- 31 investigations have been undertaken to determine whether or not this same benefit can be translated into the setting of spine surgery.
While limited in nature, these aforementioned studies have demonstrated that in the setting of single-level spinal surgery, LB may be of benefit. Kim et al found that in patients undergoing single-level transforaminal lumbar interbody fusion, the use of LB resulted in decreased postoperative pain and narcotic consumption in the first 24 hours following surgery. They suggested that this may have ultimately contributed to shorter hospital lengths of stay. 13 Similarly, in a recent retrospective case-control study by Tomov et al, adjunctive use of LB was associated with decreased IV opioid consumption in patients undergoing single-level transforaminal lumbar interbody fusion when compared with standard bupivacaine alone. 10 Finally, in patients undergoing open single-level microdiscectomies, LB was found to significantly decrease the amount of time that patients required IV narcotics postoperatively. 9 Brown et al performed a safety study in the setting of 1- to 2-level lumbar surgeries and found LB to be safe, but did not report on pain and opioid consumption. 32 Ultimately, as these studies were conducted in surgical settings associated with relatively low morbidity, the applicability of these findings to large spinal deformity correction may be limited.
Only 2 studies to date, that we are aware of, have evaluated the efficacy of LB in spinal deformity. Both retrospective studies investigated the use of LB as an adjunct to existing multimodal pain regimens in patients undergoing pediatric spinal deformity correction. 33,34 Cloyd et al found no difference in postoperative opioid use or postoperative pain scores with use of LB in their study. On the other hand, Chughtai et al found that the addition of periincisional LB at closure of surgery improved these aforementioned metrics. However, there are significant limitations to the latter study. First, they compared their results to a historical cohort of patients who had not received any apparent local anesthetic postoperatively. Furthermore, the results of this study should be interpreted with careful optimism as the authors reported a direct conflict of interest with the drug manufacturer. A recent review of all level-1 studies in orthopedic surgery investigating the efficacy of LB found that studies reporting a direct conflict of interest with the manufacturer were more likely to report more optimistic outcomes. In fact, of the 9/27 studies that reported superiority of outcomes with use of LB, 8 reported a direct conflict of interest with the manufacturer. 35
In our study, when compared to plain bupivacaine alone, while LB appeared to result in an improvement in pain scores on POD1, these differences were small, and likely clinically irrelevant. Otherwise, no further differences in pain scores were detected between the 2 groups. Interestingly, patients receiving LB did appear to transition more quickly off of IV narcotics with a 52% reduction in narcotic consumption observed in group L on POD3. However, this difference did not appear to result in any differences in overall narcotic consumption or hospital length of stay between the 2 cohorts.
There are several notable limitations in this study. First, the retrospective, nonrandomized study design and the consequent lack of appropriate blinding in the study limit the strength of our findings. Additionally, the use of a historical cohort potentially introduces selection bias. However, no statistically or clinically significant differences in patient characteristics were found when comparing the 2 groups. Also the relatively short study interval may have limited our ability to detect any meaningful changes in the clinical management of these patients. Interestingly, a higher proportion of patients in Group L underwent bilateral iliac screw fixation, which may have confounded results. Additionally, no cost analysis was performed. On average, the difference in cost between LB and plain bupivacaine is approximately $300. 36,37 While we would argue that in the setting of large deformity surgery this is a relatively trivial cost for the added benefit of decreasing overall narcotic consumption, we found no such difference in our study. Finally, the efficacy of the recommended dosage of LB, while perhaps useful in the setting of single-level surgery, may be significantly limited in large deformity surgeries given the dilutional effect of using the same dose in a much larger wound. Nonetheless, these limitations are accepted in exchange for the novelty of this study as this is the first study to evaluate the use of infiltrative LB as an adjunct modality of pain control in ASD surgery as far as we are aware.
Conclusion
The use of LB in ASD surgery does not appear to provide clinically important improvements in postoperative pain at the manufacturer recommended dosage. Furthermore, while patients receiving LB may transition more quickly off of IV narcotics, this does not appear to translate into an overall decrease in narcotic consumption, hasten return of bowel function, or decrease hospital lengths of stay. Future prospective randomized control trials are warranted. The use of varying dosages of LB may also help further clarify the true efficacy of LB in the setting of spinal deformity surgery.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
