Abstract
Study Design:
Secondary analysis of data from the multicenter, randomized, parallel-controlled Food and Drug Administration (FDA) investigational device exemption study.
Objective:
Studies on outcomes following anterior cervical discectomy and fusion (ACDF) in individuals with diabetes are scarce. We compared 24-month radiological and clinical outcomes in individuals with and without diabetes undergoing single-level ACDF with either i-FACTOR or local autologous bone.
Methods:
Between 2006 and 2013, 319 individuals with single-level degenerative disc disease (DDD) and no previous fusion at the index level underwent ACDF. The presence of diabetes determined the 2 cohorts. Data collected included radiological fusion evaluation, neurological outcomes, Neck Disability Index (NDI), Visual Analog Scale (VAS) scores, and the 36-Item Short Form Survey Version 2 (SF-36v2) Physical and Mental component summary scores.
Results:
There were 35 individuals with diabetes (11.1%; average body mass index [BMI] = 32.99 kg/m2; SD = 5.72) and 284 without (average BMI = 28.32 kg/m2; SD = 5.67). The number of nondiabetic smokers was significantly higher than diabetic smokers: 73 (25.70%) and 3 (8.57%), respectively. Preoperative scores of NDI, VAS arm pain, and SF-36v2 were similar between the diabetic and nondiabetic participants at baseline; however, VAS neck pain differed significantly between the cohorts at baseline (P = .0089). Maximum improvement for NDI, VAS neck and arm pain, and SF-36v2 PCS and MCS scores was seen at 6 months in both cohorts and remained stable until 24 months.
Conclusions:
ACDF is effective for cervical radiculopathy in patients with diabetes. Diabetes is not a contraindication for patients requiring single-level surgery for cervical DDD.
Keywords
Introduction
Diabetes mellitus (DM) is a chronic condition associated with abnormally high levels of glucose in the blood. DM may cause bone loss and/or osteoporosis. 1,2 Bone loss in the cervical spine may present as vertebral body height loss, spondylolisthesis, and neural compromise, leading to myelopathy, radiculopathy, neck pain, or a combination of these conditions. 3,4
Prior studies have analyzed the impact DM has on the outcomes of surgery on the lumbar spine and surgery for cervical spondylotic myelopathy (CSM) and degenerative cervical spine, with varying results. Whereas most studies found that DM adversely affects postoperative outcomes, 5 -9 one study found no impact of diabetes on outcomes of surgical decompression for CSM. 10
Anterior cervical discectomy and fusion (ACDF) is a common treatment for cervical radiculopathy or myelopathy that did not respond to nonoperative care. 11,12 Following discectomy, materials such as autograft, allograft, or demineralized bone matrix have been used to achieve fusion of the intervertebral disc space. Each of these materials has distinct advantages and disadvantages for attaining fusion. 13,14 i-FACTOR (CeraPedics, Inc, Westminster, CO) is a bone graft substitute, approved by the US Food and Drug Administration (FDA), and is indicated for single-level ACDF surgery. i-FACTOR Bone Graft is the only biologic bone graft made of a synthetic small peptide (P-15) bound to an anorganic bone mineral. This unique combination creates a surface-bound novel mechanism of action that enhances the body’s natural bone healing process, resulting in safe, predictable bone formation. Being surface bound, all cellular activity resulting from P-15 attachment is restricted to the implant surface, thus, preventing ectopic bone growth.
The aim of this analysis is to compare and report the 2-year radiological and clinical outcomes in those with and without diabetes undergoing single-level ACDF surgery. Participants were randomly assigned to receive i-FACTOR or local autograft irrespective of their diabetes status. Furthermore, we sought to evaluate if clinical and safety outcomes of patients with diabetes undergoing ACDF surgery differ between those receiving i-FACTOR and those receiving locally collected autograft.
Materials and Methods
Data was obtained from a prospective, multicenter, randomized, parallel-controlled FDA investigational device exemption trial of i-FACTOR versus autograft bone in individuals with cervical radiculopathy treated with single-level ACDF surgery (ClinicalTrials.gov NCT00310440). 15,16 A total of 319 patients were enrolled between June 2006 and May 2013 at 19 sites in the United States and 3 in Canada.
Inclusion criteria were as follows: age 18 to 70 years; failure to gain adequate relief from at least 6 weeks of nonoperative treatment; and radiographic evidence of single-level degenerative disc disease (DDD) of discogenic origin between the C3 and C7 vertebral levels (including at least 1 of the following: degenerated/dark disc on magnetic resonance imaging [MRI]; decreased disc height compared with adjacent levels on plain film radiographs, computed tomography (CT), or MRI; or disc herniation on CT or MRI). Additional criteria included the following: radicular symptoms by history and physical exam, preoperative Visual Analog Scale (VAS) pain level at neck or arm/shoulder >4, and Neck Disability Index (NDI) >30. Key exclusion criteria were as follows: multilevel symptomatic cervical DDD, previous cervical fusion and/or decompression at the index level, acute cervical injury or instability resulting from trauma (ie, subluxation >3 mm on flexion/extension film), and presence of systemic infection, active malignancy, myelopathy, rheumatoid disease of the cervical spine, or osteoporosis or osteomalacia.
A traditional anterior cervical approach was performed with intraoperative radiographic identification of the symptomatic surgical level. Surgeons performed an anterior cervical discectomy with achievement of neural decompression. Two treatment arms were evaluated and compared. In the first arm, following placement of i-FACTOR in a cortical allograft ring into the intervertebral disc space, an anterior cervical plate was placed spanning the disc space level; this plate was then fixated with a screw/plate construct. In the second arm, autologous bone from the adjacent vertebrae (obtained from the preparation of the endplates) was used to fill the central canal of the allograft, rather than placing i-FACTOR in the allograft.
Participants were followed up at 6 weeks and at 3, 6, 9, 12, 18, and 24 months postoperatively. Data collection included radiological evaluation of fusion, neurological outcomes, NDI (a patient-reported, 10-item questionnaire assessing neck pain disability 17 ), VAS neck and arm/shoulder pain scores, and the 36-Item Short Form Survey Version 2 (SF-36v2) Physical and Mental component summary (PCS and MCS) scores. Successful fusion was based on anteroposterior, lateral, flexion, and extension X-rays showing translational motion <3 mm, angular motion <58 degrees, and evidence of bridging trabecular bone between the involved motion segments. Qualitative evaluations of evidence of bridging bone were performed by 2 blinded radiologists from a central radiology laboratory; a third radiologist was involved in case of a tie. If there was lack of evidence of bridging bone on 12-month X-rays, CT was used to make the final determination of fusion status. The criteria for fusion on CT were trabecular bone formation patterns within the intervertebral disc space or bridging bone formation that crossed the interspace. The VAS is a 0 to 100 scale ranging from 0 (no pain at all) to 100 (worst pain possible). 18 The SF-36v2 is a health-related quality-of-life instrument resulting in a profile across 8 dimensions and 2 summary scores: the PCS score and the MCS 19 score.
Ethics
All sites obtained approval from an institutional review board or a research ethics board. All participants provided written informed consent to participate in the study.
Statistical Analysis
Participants were divided into 2 cohorts based on the prospectively recorded presence of diabetes prior to ACDF. Change in NDI functional outcomes, VAS arm and neck pain scores, and SF36-v2 PCS and MCS scores were compared between those with and without diabetes, using the mixed-model repeated-measures analysis. Both the unadjusted and adjusted models were run. The unadjusted model included the baseline score, follow-up visit, diabetes status, and the interaction between diabetes and follow-up visit. In the adjusted mixed model, the covariates included were baseline score, gender, age, and smoking status. Graft type, the interaction between graft type and visit, and the interaction between graft type and diabetes status were also included. Prior to analysis, missing values were imputed using the multiple imputation procedure. 20 All analyses were performed using SAS/STAT 9.4 for Windows.
Results
From June 2006 to May 2013, 319 individuals underwent ACDF. For more than 85% of the individuals, the levels involved C5-C6 or C6-C7. Of the 319 individuals, 35 (11%) had diabetes at the time of surgery and 284 (90%) did not. Average BMI was higher (32.99 kg/m2, SD = 5.72) in the diabetic cohort compared with the nondiabetic cohort (28.32 kg/m2, SD = 5.67; P < .0001) with a difference of 4.67. The majority of individuals in the diabetic cohort were female (21, 60%), and the nondiabetic cohort had a similar ratio (171, 60.2%). The mean age of the diabetic cohort was 53.3 years (SD = 8.79), compared with 45.9 years (SD = 9.40, P < .0001) in the nondiabetic cohort at the time of signed consent, with a difference of 7.4 years. There were 31 (88.57%) Caucasians in the diabetic cohort, compared with a similar ratio of 264 (92.26%) in the nondiabetic cohort. The most frequent cervical level affected was at C6-C7 (48.57%). The overall 1-year follow-up rate was 92%, and the 2-year follow-up rate was 84%.
The ratio of smokers in the nondiabetic cohort was significantly higher than in the diabetic cohort: 73 (25.70%) and 3 (8.57%), respectively; P = .0217. Preoperative scores of NDI, VAS arm pain, and SF-36v2 were similar between the diabetic and nondiabetic cohorts at baseline; however, the diabetic cohort presented with a significantly more severe VAS neck pain score at baseline (P = .0089; Table 1).
Baseline Data and Demographics.
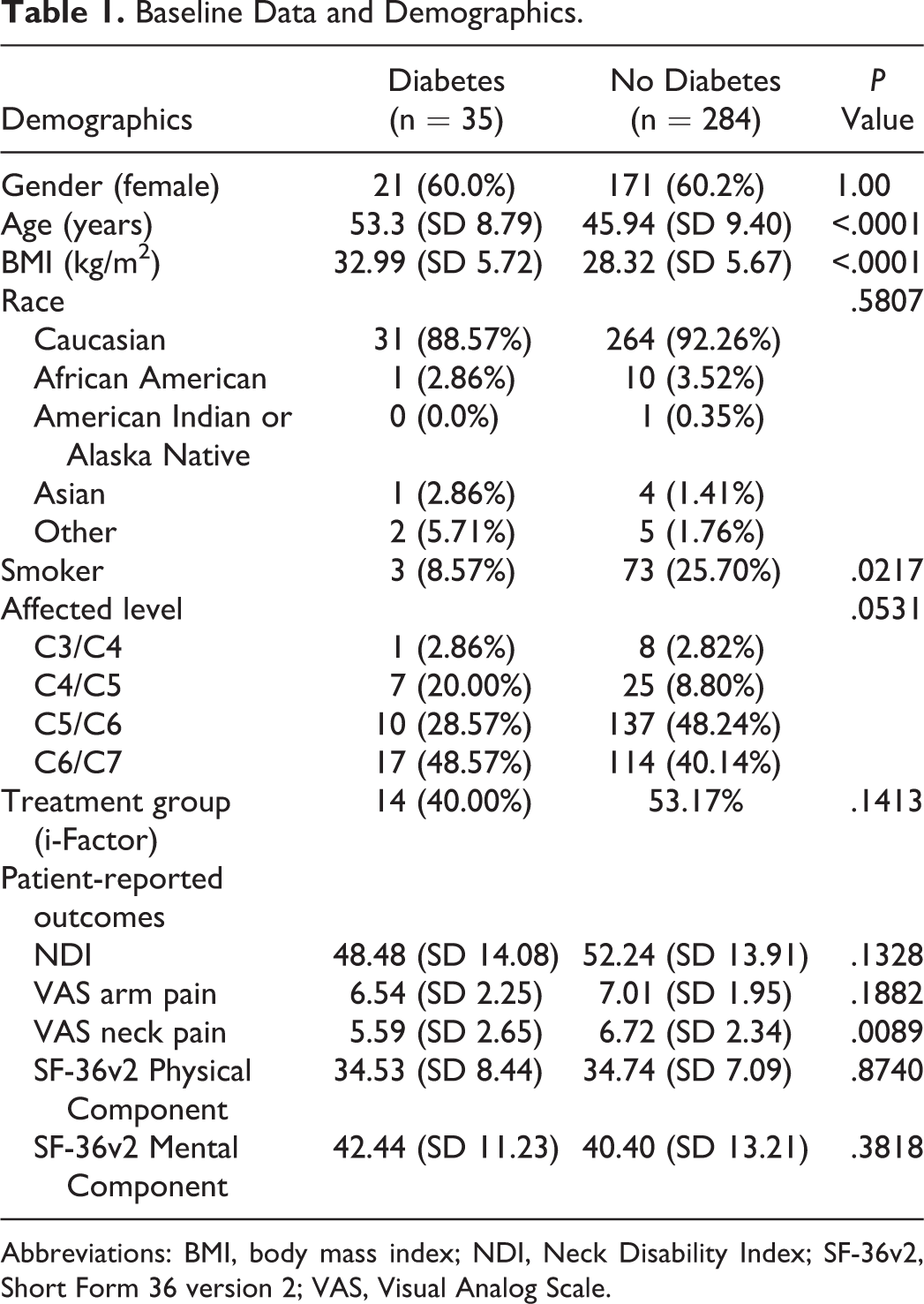
Abbreviations: BMI, body mass index; NDI, Neck Disability Index; SF-36v2, Short Form 36 version 2; VAS, Visual Analog Scale.
NDI improved significantly postoperatively in both the diabetic and nondiabetic cohorts. The improvement was seen at the 6-month follow-up; there was no further improvement after that time point. There were no differences in NDI change scores between diabetic and nondiabetic individuals at any time point (Table 2).
Postoperative Changes in Patient-Reported Outcomes.a
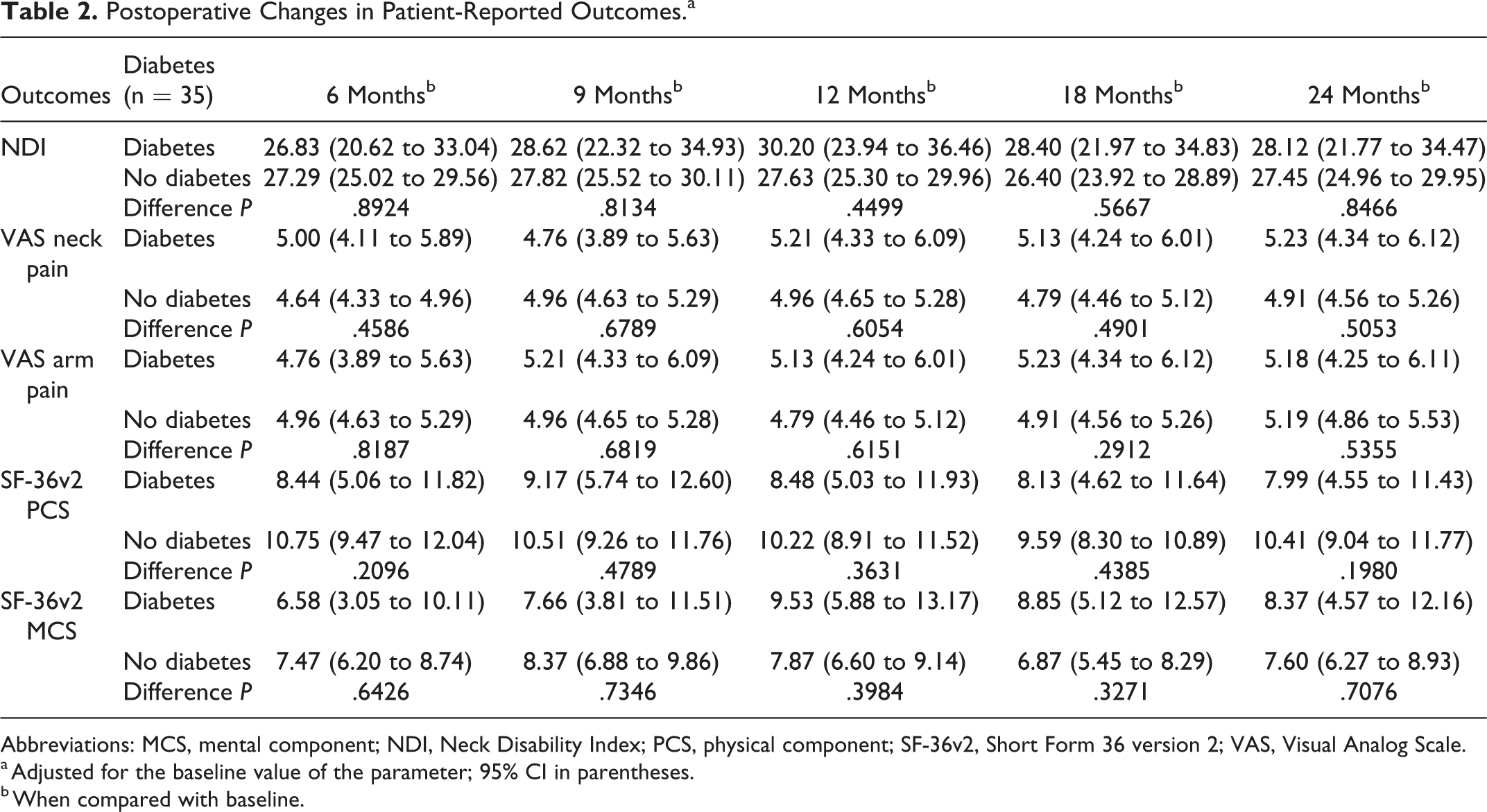
Abbreviations: MCS, mental component; NDI, Neck Disability Index; PCS, physical component; SF-36v2, Short Form 36 version 2; VAS, Visual Analog Scale.
a Adjusted for the baseline value of the parameter; 95% CI in parentheses.
b When compared with baseline.
VAS neck and arm pain scores improved postoperatively in both the diabetic and nondiabetic cohorts. The improvement was seen at the 6-month follow-up in both cohorts; additionally, at the 6-month follow-up, there were no significant differences between the 2 cohorts. After reaching improvement at 6 months, there were no further improvements in VAS neck and arm pain scores in either cohort (Table 2).
The SF-36v2 PCS and MCS scores improved postoperatively in both diabetic and nondiabetic cohorts. The maximum improvement was seen at the 6-month follow-up. There were no differences in SF-36v2 PCS and MCS scores between diabetic and nondiabetic individuals at any time point (Table 2). Following adjustment for covariates, there were no differences between any of the patient-reported outcome scores (Table 3).
Two-Year Patient-Reported Outcomes, Adjusted (Imputed).a
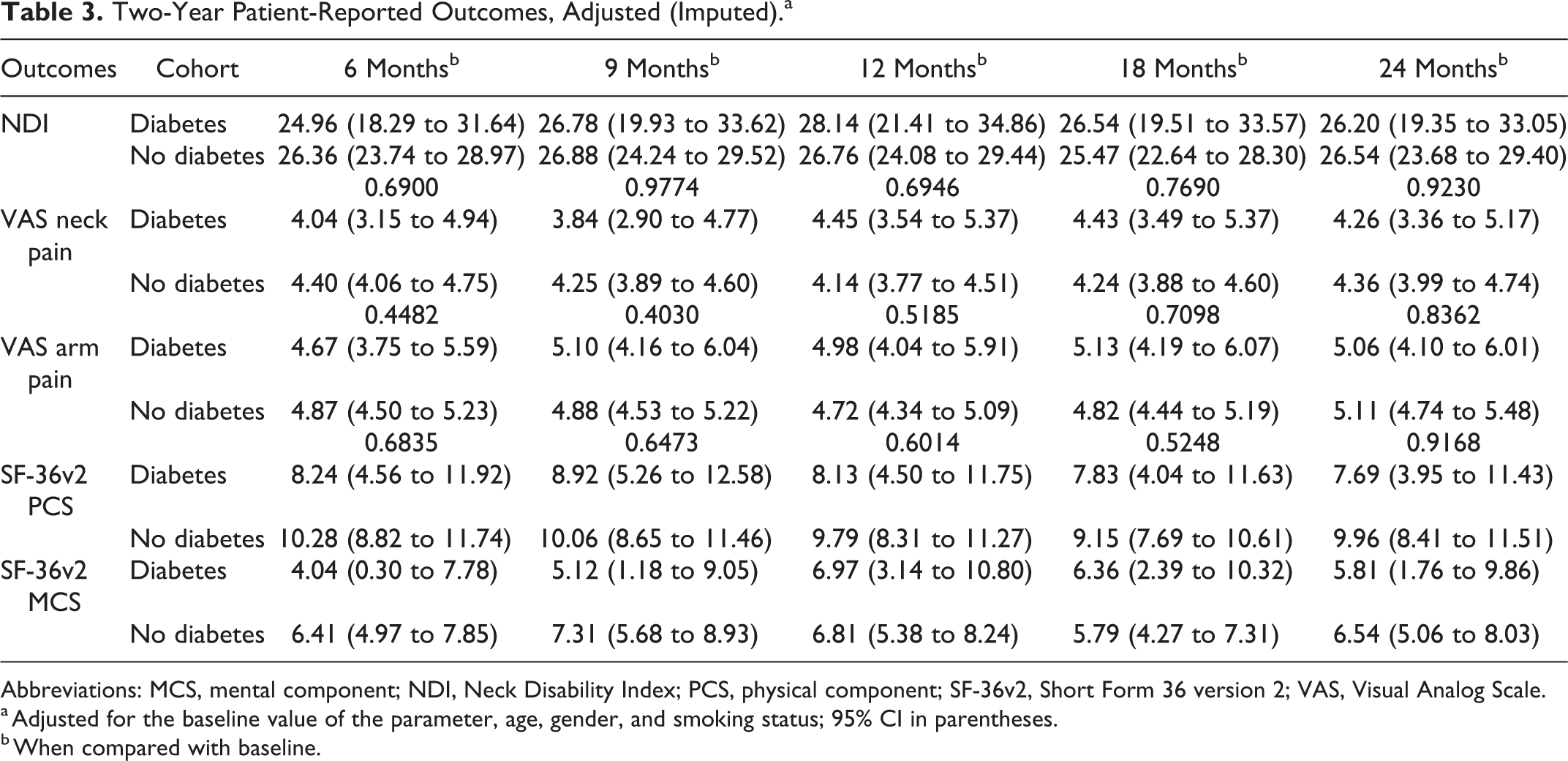
Abbreviations: MCS, mental component; NDI, Neck Disability Index; PCS, physical component; SF-36v2, Short Form 36 version 2; VAS, Visual Analog Scale.
a Adjusted for the baseline value of the parameter, age, gender, and smoking status; 95% CI in parentheses.
b When compared with baseline.
There was no significant interaction between i-FACTOR and autologous bone and diabetes status for any of the outcomes. At the 12-month follow-up, 32/34 (94.11%) individuals in the diabetic cohort and 223/257 (86.77%) in the nondiabetic cohort showed evidence of fusion (P = .2277). At the 2-year follow-up, 26/27 (96.29%) individuals in the diabetic cohort and 211/221 (95.47%) in the nondiabetic cohort showed evidence of fusion (Table 4).
Fusion Rates, per Cohort.
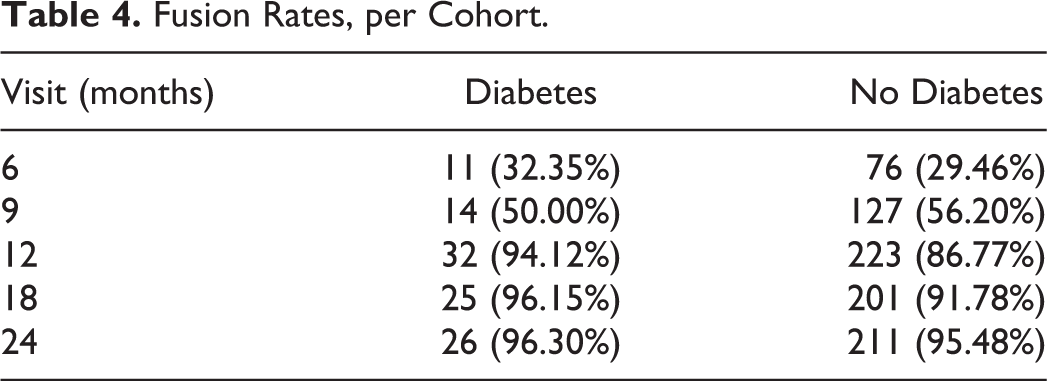
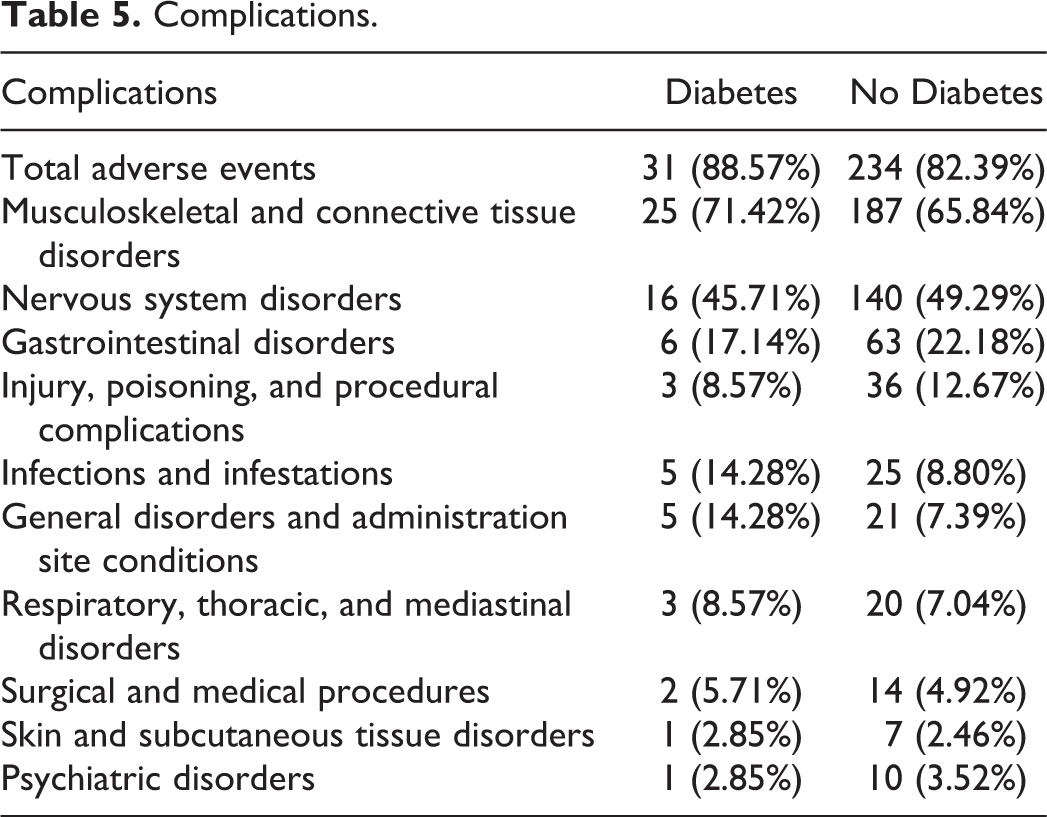
Table 5 shows the various adverse events seen in the cohorts. There were 31 (88.57%) adverse events in the diabetic cohort compared with 234 (82.39%) in the nondiabetic cohort. The most prevalent adverse events in both cohorts involved musculoskeletal and connective tissue disorders (71.42% in the diabetic cohort, 65.84% in the nondiabetic cohort), followed by nervous system disorders and gastrointestinal disorders. None of the adverse events in these comparisons reached statistical significance.
Complications.
Discussion
DM has been shown to be an independent risk factor for surgical complications, which include poor wound healing and perioperative ischemic events. 21 -23 When looking at the impact of diabetes on patients undergoing spine surgery, studies have noted decreased postoperative improvements, increased perioperative complications and mortality, worse patient-reported outcomes, and longer hospital lengths of stay after surgery. 5,24,25 However, some studies have shown that diabetes has minimal impact on patient outcomes. 26,27
Because of the disparate findings in the literature, we assessed the impact of diabetes on fusion rates, outcomes, and complications in those undergoing single-level ACDF surgery with either i-FACTOR or local autologous bone. We did not find evidence that individuals with diabetes have poorer outcomes following single-level ACDF for cervical radiculopathy compared with those without diabetes. We found no evidence that i-FACTOR has different clinical and safety outcomes compared with local autologous bone in patients with diabetes undergoing ACDF surgery. Both cohorts improved in NDI, VAS arm and neck pain scores, and SF-36v2 PCS and MCS scores from baseline to the 2-year follow-up. There was no indication that radiological fusion rates were less common in the diabetic cohort. As expected, the patients with diabetes had a higher BMI than patients without diabetes.
Several studies have shown that DM can negatively affect surgical results. Worley et al 8 found that diabetes was an independent risk factor for increased hospital length of stay and increased postoperative complications in patients undergoing surgery for CSM. Machino et al 28 found that patients with long-standing, poorly controlled diabetes undergoing laminoplasty for myelopathy had worse preoperative and postoperative modified Japanese Orthopaedic Association scores and slower and reduced neurological improvements. 28 Other studies have found that neurological outcomes in patients undergoing laminoplasty were negatively correlated with hemoglobin A1C levels. 24 Guzman et al 9 found that poor glycemic control increased the likelihood of mortality and perioperative complications in patients undergoing surgery for degenerative cervical spine disease. Armaghani et al 29 found that diabetes was related to worse patient outcomes at 2 years after surgery in a cohort of patients undergoing elective spine surgery.
Other studies have shown that diabetes does not have an impact on outcomes following spine surgery. Cho et al 30 found that diabetic patients undergoing surgery for adult deformity had similar Oswestry Disability Index scores, Scoliosis Research Society scores, and perioperative complications. Arnold et al 10 found that diabetes did not affect outcomes or complications in patients undergoing multilevel fusion for CSM.
There are limitations to this study. This secondary analysis looked at only single-level fusions, which historically have very high fusion rates. It is possible that results might be different in patients undergoing multilevel procedures. Also, only anterior cervical procedures were included in this analysis, whereas posterior cervical fusions may have a different set of complications. The younger age in patients with radiculopathy and not myelopathy may also have played a role. Other limitations were that the surgeons were not blinded to the assigned treatment arm, fusion was determined by X-ray, and a CT was obtained only if it was believed that there was a lack of bridging bone at the 12-month plain film X-ray analysis. In addition, the definition of diabetes was binary; the severity of glycemic regulation was not assessed.
Strengths of the study were its well-defined inclusion and exclusion criteria and the quality of data. Additional strengths were the large number of enrolled participants, the prospective identification of diabetes, and the long follow-up of 2 years.
Conclusion
After comparing the 2-year radiological and clinical outcomes in those with and without diabetes who underwent single-level ACDF surgery, we found that ACDF is a safe and effective surgery for the treatment of cervical radiculopathy in patients with diabetes. In this study utilizing i-FACTOR or local autologous bone, diabetes did not appear to be a contraindication for patients requiring single-level surgery for cervical DDD. Furthermore, the clinical and safety outcomes of diabetic patients undergoing single-level ACDF are similar between those receiving i-FACTOR and those receiving local autologous bone.
Footnotes
Acknowledgments
The authors thank Tamara Kopjar and Karen Anderson who provided medical writing services on behalf of Nor Consult, LLC.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Cerapedics, Inc, provided research funding to investigator sites to conduct the Food and Drug Administration (FDA) Investigational Device Exemption trial, including the research departments of the authors of this manuscript. No funding was received for other purposes. Dr Kopjar and his company were contracted by Cerapedics, Inc, to design and manage the clinical study. The remaining authors report no other conflicts of interest, including consultancy agreements, royalties, gifts received, or intellectual property with regard to the products (i-Factor Bone Graft) or company (Cerapedics, Inc.) involved in this scientific investigation.
