Abstract
Study Design:
Meta-analysis of evidence level I to IV studies.
Objective:
To compare decompression alone versus decompression plus fusion in the treatment of grade I degenerative spondylolisthesis (DS).
Methods:
Following established guidelines, we systematically reviewed 3 electronic databases to assess studies evaluating patients with grade I DS. We stratified all patients into 2 cohorts; the first cohort underwent a decompression-type surgery, and the second cohort underwent decompression plus fusion. We noted clinical outcomes, complications, reoperations, and surgical details such as blood loss. Descriptive statistics and random-effects models were used to determine the specified outcome metrics with 95% confidence intervals (CIs).
Results:
In both cohorts, the pain (legs and lower back) significantly decreased and the physical component of the Short Form 36 showed better patient clinical outcomes. The decompression cohort had a 5.8% complication rate (95% CI = 1.7-2.1), and the decompression plus fusion cohort had an 8.3% complication rate (95% CI = 5.5-11.6). The reoperation rate was higher in the decompression-only cohort (8.5%; 95% CI = 2.9-17.0) compared with the decompression plus fusion cohort (4.9%; 95% CI = 2.5-7.9).
Conclusions:
There does not appear to be any advantage of one procedure over the other. Patients undergoing decompression alone tended to be older with a higher percentage of leg pain, whereas patients additionally undergoing fusion tended to be younger with more lower back pain. The decompression-only cohort had fewer complications but a higher revision rate.
Introduction
Degenerative spondylolisthesis (DS) is a pathological disease characterized by forward slippage of the vertebral body, which can result in symptoms of neurogenic claudication and back pain. 1 -3 It most often occurs in patients older than 50 years, with women being more commonly affected than men. 4 A large majority of symptomatic spondylolisthesis patients have Meyerding grade I spondylolisthesis, the lowest grade in a classification system based on the degree of slippage. 5
Nonoperative treatment consists of physical therapy, epidural injections, chiropractic care, anti-inflammatory agents, and opioid analgesic agents. 4 However, indications to proceed from conservative treatment to surgical management include progressive neurological deficit, persistent severe back pain and/or leg pain, and bladder or bowel symptoms even after a 3-month trial of nonoperative interventions. 2 Recent large randomized controlled trials, including the Spine Patient Outcomes Research Trial (SPORT) trial, have shown that surgical management improves pain and function in patients with symptomatic DS compared with nonoperative options. 4,6 Surgical interventions for DS include decompression alone and decompression with fusion with or without instrumentation. Interbody fusion has additionally been presented as a viable option. 7 A 2009 North American Spine Society evidence-based clinical guideline presented a grade B recommendation for fusion with instrumentation when assessing its effectiveness in the treatment of lumbar DS. 8
Based on these guidelines and most research studies pertaining to DS, grade I deformity is often not considered separately from the higher grades. The question remains whether patients with grade I DS can be treated adequately with simple decompression without fusion. This is important because despite the many advancements in fusion techniques, complications of surgical arthrodesis, especially in the elderly, are well documented. 9,10 Still, lumbar decompression has complications of its own, including infection, dural tears, and hematomas, expected to occur in approximately 11% of cases. 11
Therefore, we conducted a meta-analysis of available literature stratifying patients with grade I DS into the 2 most common categories of surgical treatment—decompression alone (the decompression cohort) versus decompression plus fusion (the fusion cohort)—to determine differences in clinical outcomes, complications, and reoperation rates for patients undergoing surgical intervention.
Materials and Methods
Institutional review board approval was not required for this study. This meta-analysis is registered with Prospero: registration number CRD42017057587. Following the preferred reporting items for systematic review and meta-analyses (PRISMA) guidelines, we systematically reviewed the Medline, Embase, and Ovid electronic databases to assess for studies evaluating operative management of patients with grade I spondylolisthesis published from January 1996 until December 2016. 12 The keywords used for searching the databases were “degenerative spondylolisthesis management,” which yielded 1625 results. Cross-referencing strategies were applied to capture relevant cited sources within the articles obtained.
Inclusion criteria were (1) grade I spondylolisthesis indicating that the cranial vertebrae had translated up to but not more than 25% with respect to the caudal vertebrae, 1,4 (2) a cohort of grade I DS of at least 10 patients, (3) a cohort of decompression surgical treatment or fusion surgical treatment, and (4) minimum 1 year of follow-up. Fusion could be any form of posterolateral fusion (instrumented or un-instrumented) or any form of interbody fusion, including minimally invasive modalities. Both neurosurgery and orthopedic surgery articles were included.
A study was excluded if it was in a language other than English, involved spondylolistheses other than degenerative type in the grade I cohort (ie, congenital, postsurgical, isthmic, traumatic), or was unable to analyze patients with grade I DS separately from patients with other grades of DS. Studies were not excluded based on level of evidence. Level of evidence was determined based on the updated level of evidence guidelines provided in the Journal of Bone and Joint Surgery. 13 Three studies assessed ligamentoplasty for the treatment of spondylolisthesis, and those studies were excluded.
We specifically noted clinical outcomes, complications, reoperations, and surgical details such as blood loss. Pain and function were assessed using the Visual Analog Scale (VAS) for lower back pain, VAS for leg pain, Oswestry Disability Index, Short Form 36 (SF-36), Japanese Orthopaedic Association back pain evaluation, Numeric Rating Scale (NRS) for back pain, NRS for leg pain, and Neurogenic Claudication Outcome Score. Descriptive statistics were used to describe both cohorts, and random-effects models were used to determine the rates of the specified outcome metrics with 95% confidence intervals (CIs).
Titles and abstracts of the identified studies were reviewed, and full-text versions of studies being considered for inclusion were collected. The full-text versions were carefully assessed by 2 of the authors, and 20 studies met our criteria. A flowchart of the search methodology is displayed in Figure 1. Within these studies, we stratified all patients into 2 cohorts: the first group had patients who underwent a decompression-type surgery, and the second consisted of patients who additionally underwent a fusion procedure. A few different means of decompression surgeries were featured among the studies, with laminectomy and laminotomy being the most commonly performed. 14 -24 One study assessed laminoplasty, 17 and 1 study assessed semi-circumferential decompression. 25 In 5 of the fusion studies, patients underwent un-instrumented posterolateral fusion, 15,17,21,22,26 and in the remaining fusion studies, some form of interbody fusion was used. 19,27 -33
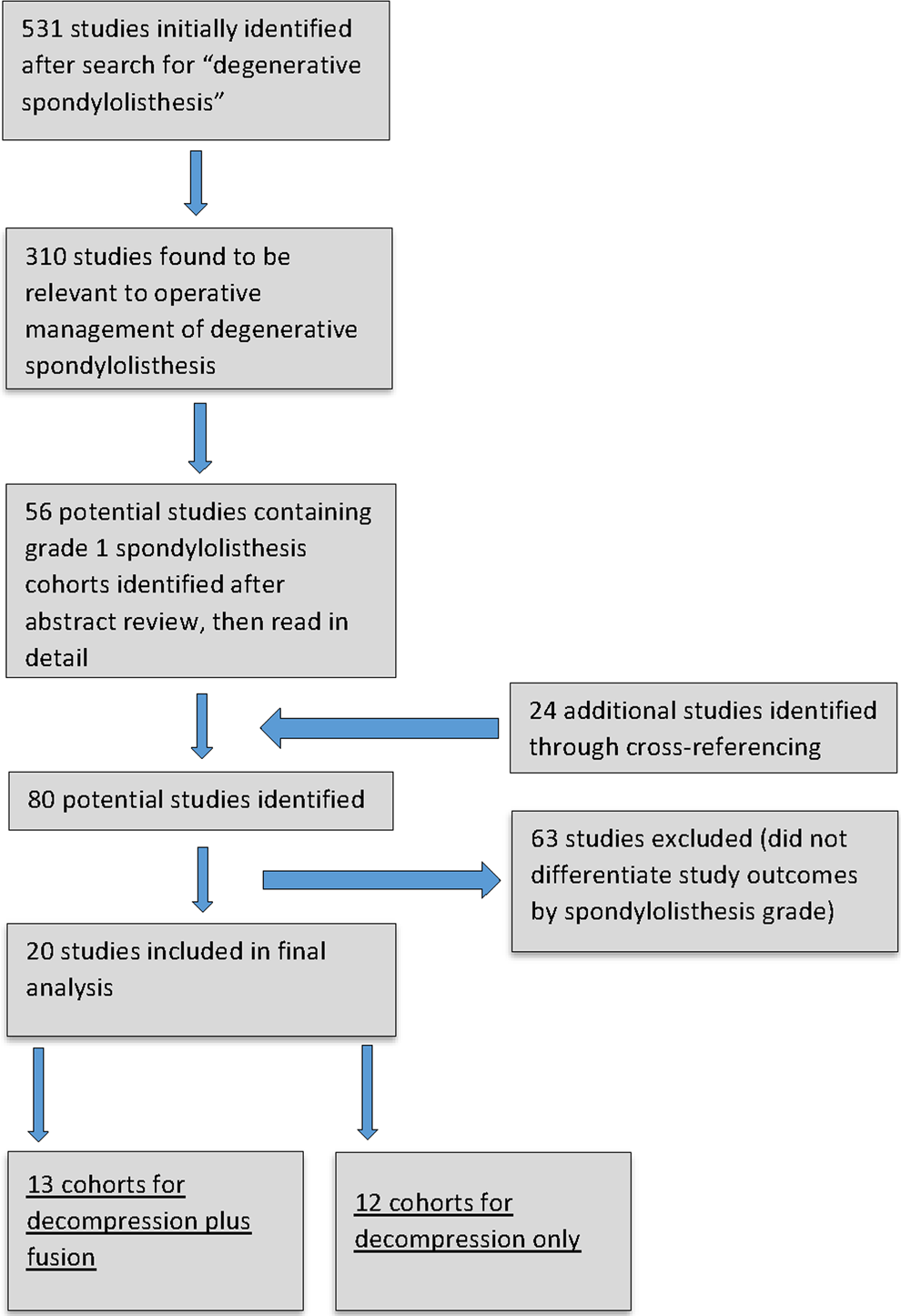
Flowchart of study selection.
We searched for specific end points within each study, and this information was transferred to an electronic spreadsheet (Microsoft Excel; Microsoft Office, Redmond, Washington). The following information comprised the Excel sheet: type of decompression and/or fusion performed, number of patients in the study, patient age, sex, body mass index (BMI), follow-up time, level of evidence, and outcomes. Operating room parameters, reoperation rates, complication rates, fusion rates, radiographic measures, and preoperative and postoperative pain and functioning scales were the outcomes being evaluated. Using random effects models, forest plots were obtained to compare the differences in outcome measures between the decompression cohort and the fusion cohort. This was accomplished with the aid of statistical software (MedCalc Software version 15.2; MedCalc, Ostend, Belgium). No outside funding was received for this work.
Results
A summary of the included studies is presented in Table 1. Twelve studies (n = 591 patients) were included in the decompression cohort, and 13 studies (n = 465 patients) were included in the fusion cohort. All included studies provided a description of the decompression performed. In the decompression cohort, the mean age was 66.4 years (range of means = 62-69 years), the mean BMI was 23.7 kg/m2 (range of means = 23-26 kg/m2), and 67% were women. The fusion cohort had a mean age of 60.6 years (range = 49-70), a mean BMI of 23.8 kg/m2 (range = 23-26 kg/m2), and 66% were women. The mean follow-up in the decompression cohort was 83.5 months, whereas the mean follow-up in the fusion cohort was 46.4 months.
Included Studies.
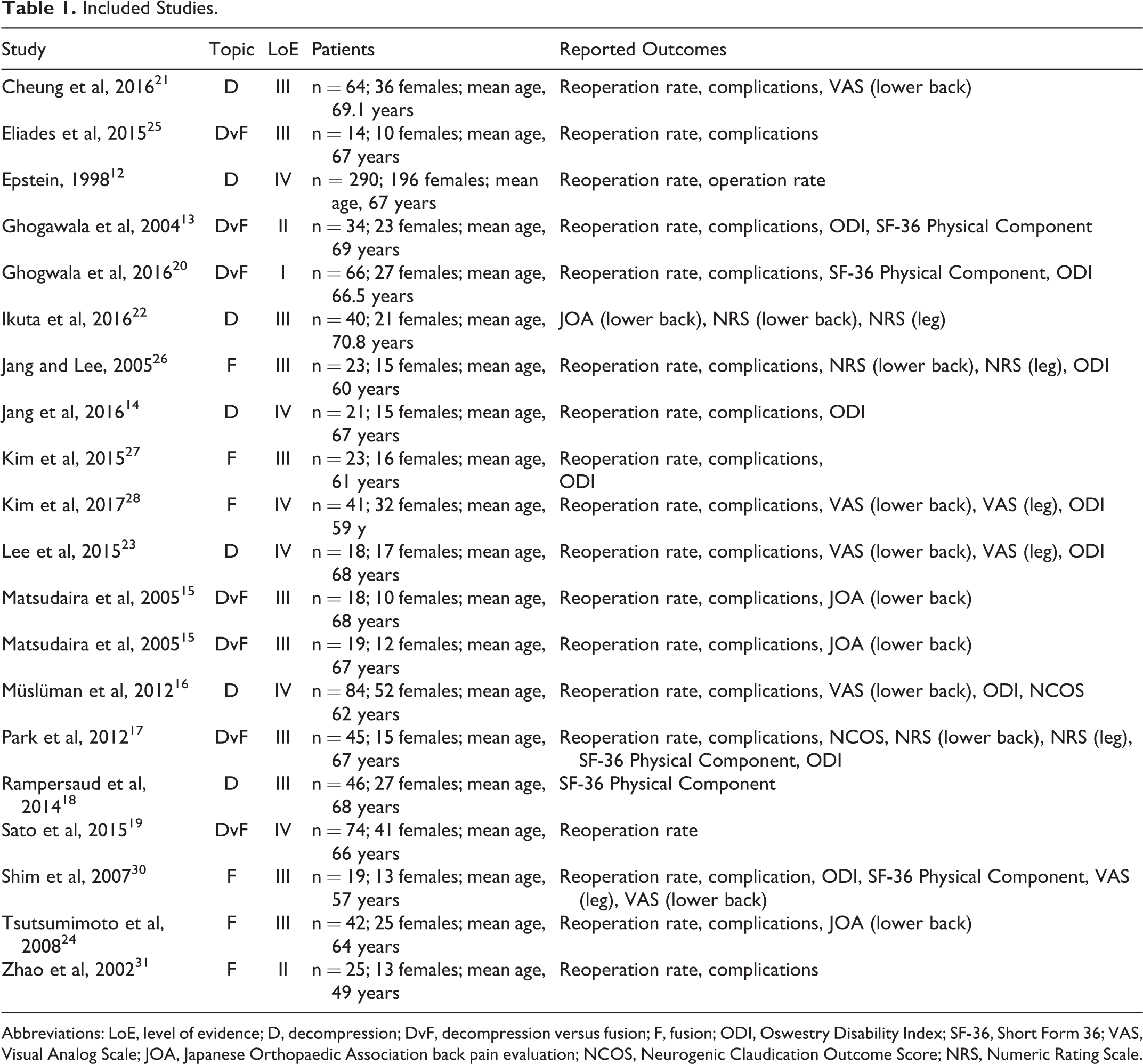
Abbreviations: LoE, level of evidence; D, decompression; DvF, decompression versus fusion; F, fusion; ODI, Oswestry Disability Index; SF-36, Short Form 36; VAS, Visual Analog Scale; JOA, Japanese Orthopaedic Association back pain evaluation; NCOS, Neurogenic Claudication Outcome Score; NRS, Numeric Rating Scale.
In both cohorts, pain (legs and lower back) significantly decreased and the physical component of the SF-36 showed improvements in patient reported outcomes. More specifically, the lower back pain in the decompression cohort improved from a mean of 5.3 to 3.5 points on the VAS, and the lower back pain the fusion cohort improved from 7.9 to 4.0 points on the VAS. However, the leg pain in the decompression cohort improved from 8.3 to 2.5 points on the VAS, and the leg pain in the fusion cohort improved from 7.5 to 3.8 points on the VAS. In both cohorts, the physical and mental components of the SF-36 improved: from 29.7 and 35.5 points, respectively, to 41.2 and 48.5 points, respectively, in the decompression cohort, and from 28.4 and 39.3 points, respectively, to 41.5 and 48.3 points, respectively, in the fusion cohort.
In terms of complications, the decompression cohort had a 5.8% complication rate (95% CI = 1.7-12.1), and the fusion cohort had an 8.3% complication rate (95% CI = 5.5-11.6). One wound infection occurred in the decompression cohort, and the remaining complications were either worsening of radicular symptoms or durotomy. Six wound infections complicated the fusion procedures. Worsening of radicular symptoms, durotomies, and subsidence of interbody cages comprised the remaining complications. Contrary to the complication rates, the reoperation rate was higher in the decompression cohort (8.5%; 95% CI = 2.8-17.0) compared with the fusion cohort (4.9%; 95% CI = 2.5-7.9).
Estimated blood loss (EBL) was significantly lower in the decompression cohort (220.7 mL) compared with the fusion cohort (512.1 mL), although only 1 decompression study and 4 fusion studies listed EBL. A summary of demographics, complication rates, and reoperation rates is presented in Table 2. Forest plots of the reoperation rates and complication rates are shown in Figures 2 through 5.
Demographics, Complication Rates, and Reoperation Rates by Cohort.
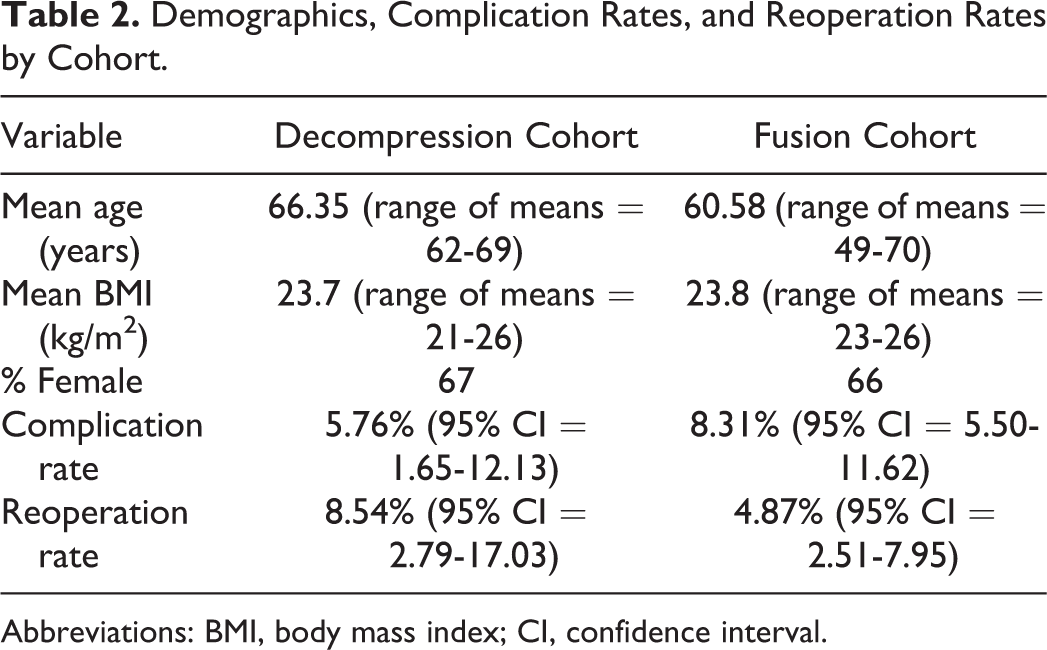
Abbreviations: BMI, body mass index; CI, confidence interval.
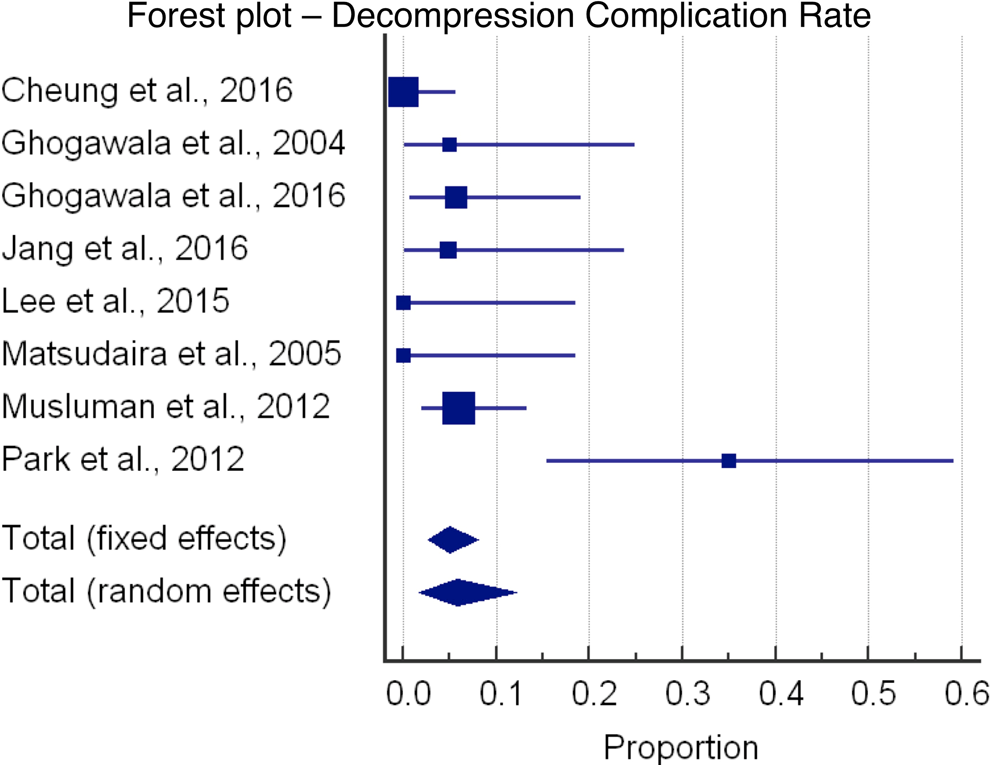
Forest plot showing decompression complication rate.
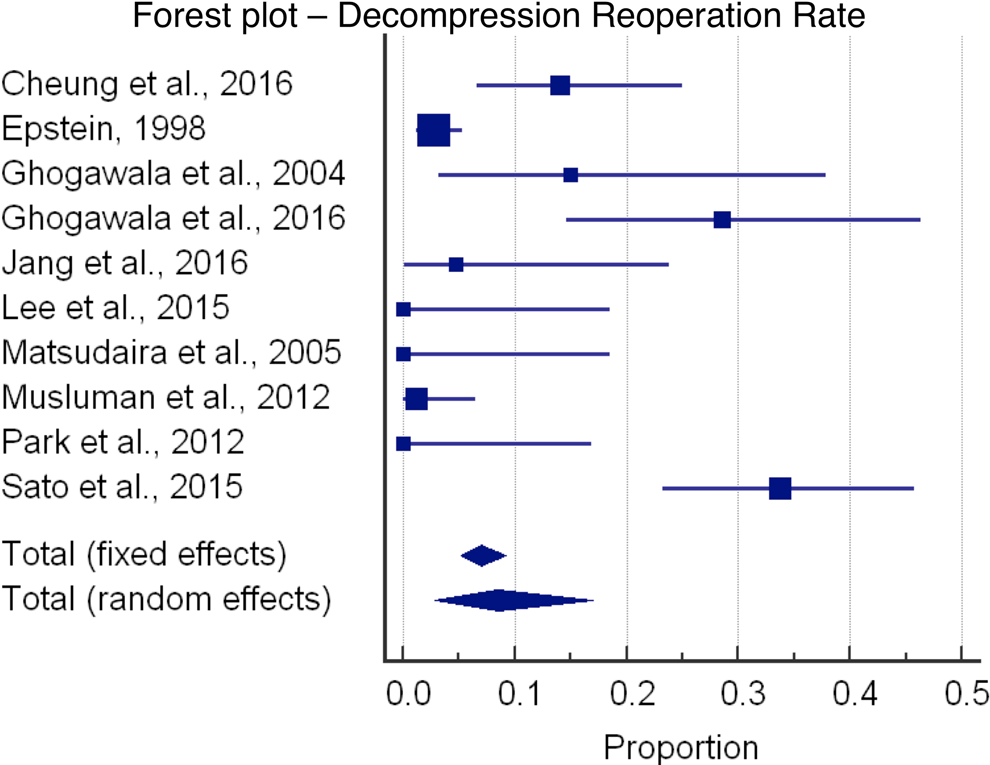
Forest plot showing decompression reoperation rate.
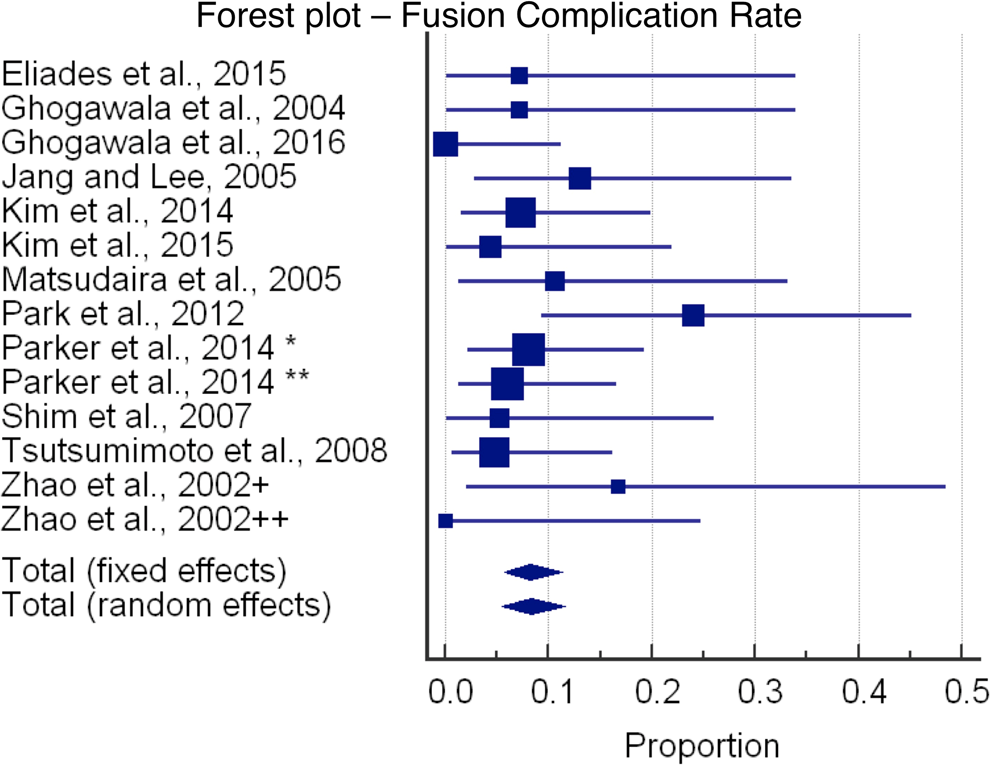
Forest plot showing fusion complication rate. *, transforaminal lumbar interbody fusion, open; **, transforaminal lumbar interbody fusion, minimally invasive surgery; +, posterior lumbar interbody fusion, 2 cages. ++, posterior lumbar interbody fusion, 1 cage.
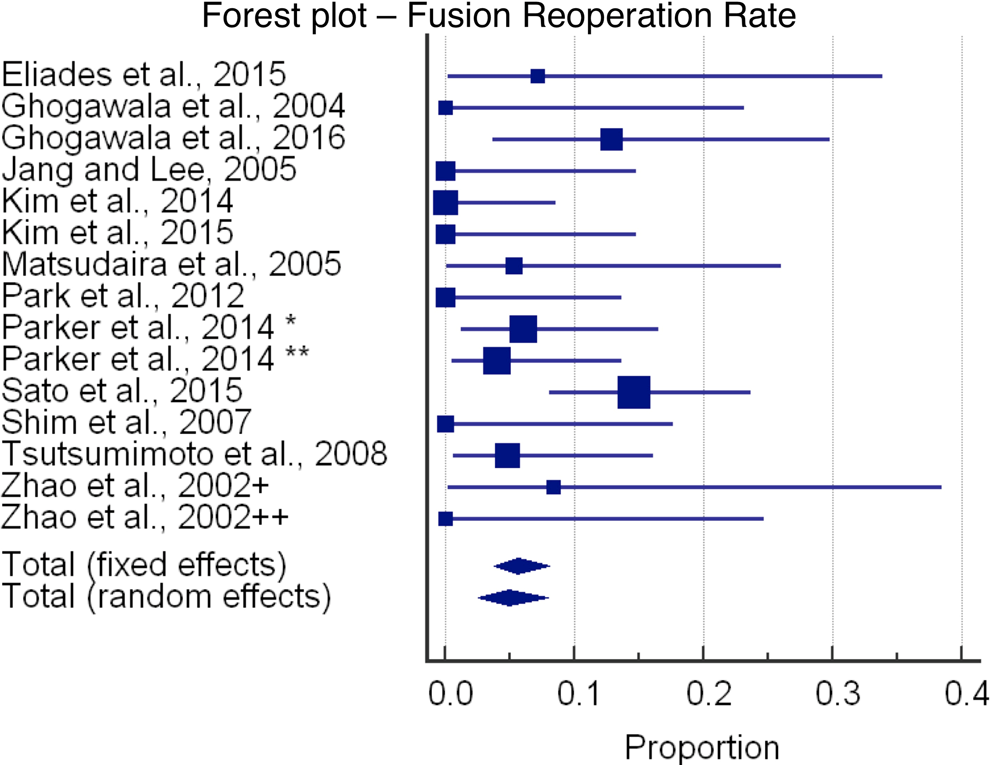
Forest plot showing fusion reoperation rate. *, transforaminal lumbar interbody fusion, open; **, transforaminal lumbar interbody fusion, minimally invasive surgery; +, posterior lumbar interbody fusion, 2 cages. ++, posterior lumbar interbody fusion, 1 cage.
Discussion
Grade I DS is the most common form of listhesis of the lumbar spine. The deformity is generally mild, and the degenerative process in general increases the degree of spinal stability. Some of these mechanisms of stabilization are in the formation of spurs, subchondral sclerosis, facet hypertrophy, and ligament ossification. 3 This helps explain why back pain can sometimes improve with conservative treatment and why progression of slippage occurs in only 30% of cases of DS. 34 Conservative treatment has been reported to be an effective first step for mild DS, and only 10% to 15% of patients ultimately require surgery. 35 However, the largest trial on DS, SPORT, demonstrated that surgical intervention achieved better results than nonoperative treatment for patients with symptomatic DS of all grades. Of the operative interventions, decompression combined with posterolateral fusion has been demonstrated in 1 trial to significantly improve outcomes compared with decompression alone for patients with symptomatic DS with spinal stenosis. 36 With this in mind, subsequent studies have focused on comparing outcomes after posterolateral fusion with or without instrumentation and have reported better outcomes with instrumentation. 2,37 -39
It has been reported that fusion has the advantage of preventing increased progression of listhesis and subsequent stenosis, 2 common causes of reoperation among patients with postoperative DS. 39 -41 According to a recent retrospective cohort study, patients with DS treated with decompression alone had increased postoperative instability, clinical failure, and revision rate compared with those treated with decompression plus fusion. 42 Our study also found that reoperation rates were higher in the decompression cohort. The most common reasons for reoperation in the decompression cohort in our study, in descending order, were recurrent stenosis, increased olisthy, and disc herniation. Despite the problems with decompression, a national database study 43 emphasized the dangers of complications after fusion in the elderly. Complications after fusion include permanent nerve root injury. Major medical complications, such as pneumonia, have been reported to occur more frequently compared with decompression alone. 43 Compared with patients aged 45 to 64, pateints who aged 65 to 84 were almost 70% more likely to experience complications and 5 times as likely to have complex disposition. 44 Our meta-analysis also found the complication rate to be higher in the fusion group. The SPORT data indicates that fusion is a safe procedure but certain subgroups (age younger than 67 years with neurogenic claudication symptoms) respond better to surgery. 45 Our study found that older patients tended to undergo decompression alone rather than decompression plus fusion. These patients tended to have greater degrees of leg pain. This may indicate some bias to treat older patients without fusion to avoid complications. Overall, SPORT data has shown that patients with predominant leg pain improve more significantly than do patients with predominant back pain. 46 Cost must also be considered, and fusion is clearly more expensive than decompression. 15
Our study had several limitations. We were limited by the available literature, and as with any meta-analysis, the limitations from each individual study apply to our study. We initially attempted to include only higher level studies, but many of those had to be excluded, thus diminishing the strength of our study. The studies were largely consistent regarding the levels of spine involved, with the majority including levels L4-5; however, some studies included other levels (L2-3, L3-4, L5-S1, or multiple levels). Analysis of complications was limited because a few of the studies reported reoperation statistics without reporting the reasons for reoperation as complications. The surgical techniques used and the decisions to use autograft or allograft were not uniform. Because of the lack of granular data, we had to broadly accept multiple types of decompression, such as minimally invasive and open decompressions, into the same cohort. A similar limitation applied to the fusion cohort because we had to integrate posterior instrumented fusion with other forms, such as interbody fusion. The lack of uniformity of surgical technique also renders it difficult to properly explain the reoperation risks and the most common causes for reoperation. Also, regarding reoperation rates, one study presented cumulative reoperation rates but enough information was provided in the data tables to extrapolate a value to allow for inclusion in our study. 22 Regarding our outcomes, an array of clinical scales were used in the literature for assessing patients with grade I DS. Different scales were used among the studies in our meta-analysis, but the same patient parameters, specifically pain and function, were covered in each of the scales. The statistical comparison of the patient parameters, SF-36, VAS (back), and VAS (leg) was limited because many of the studies lacked full reporting of the standard deviations. Most of the studies were retrospective and, as such, probably incorporate some degree of selection bias.
The results of this meta-analysis ultimately could not firmly support one operative treatment over the other, but this review sheds light on how decompression compares with fusion for the surgical treatment of grade I spondylolisthesis. Patients undergoing decompression alone tended to be older patients with predominant leg pain, whereas patients additionally undergoing fusion tended to be younger with predominant lower back pain. The decompression cohort had fewer complications but a higher revision rate. Based on the currently available data, there seems to be a stage set for prospective clinical studies to determine exactly which patients will do well with decompression alone and which will require fusion.
Footnotes
Acknowledgment
The authors thank Senior Editor and Writer Dori Kelly, MA, for invaluable assistance with the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Steven Ludwig: American Board of Orthopaedic Surgery, Inc, Board member; American Orthopaedic Association, Board member; AO Spine North America, Spine Fellowship Support, research support; ASIP, ISD, stock; Cervical Spine Research Society, Board member; DePuy, A Johnson & Johnson Company, IP royalties, paid consultant, paid speaker; Globus Medical, paid consultant, research support; Journal of Spinal Disorders and Techniques, Editorial Board; K2M Spine, research support; K2Medical, paid consultant; OMEGA, research support; Pacira, research support; SMISS, Board member; Synthes, paid consultant, paid speaker; Thieme, QMP, publishing royalties, financial or material support. Daniel Gelb: Advanced Spinal Intellectual Property, stock; DePuy-Synthes Spine, IP royalties, paid speaker; Globus Medical, IP royalties. Eugene Koh: Biomet, paid consultant; DePuy, A Johnson & Johnson Company, paid speaker. Kelley Banagan: Johnson & Johnson, employee; Orthofix, Inc, material support. The remaining authors have no conflicts of interest to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
