Abstract
Negative urgency (i.e., the tendency to act impulsively when experiencing negative affect) is robustly associated with psychopathology, but the mechanisms underlying negative urgency and its relation to mental health are not well understood. In addition to interfering with cognitive control, negative emotions may lead to impulsive behavior by enhancing reward processing of desired stimuli. In this study, we tested an emotion-enhanced reward-processing model of negative urgency in 153 women who spanned the spectrum of binge-eating severity. Participants completed two experimental tasks under both stressful- and relaxed-mood conditions while physiological, behavioral, and self-report indices of reward processing of palatable food were assessed. Contrary to hypotheses, reward processing of food was not heightened when stressed versus relaxed either in the full sample or in participants with greater negative urgency or binge-eating frequency/severity. Findings are discussed considering study limitations and previous mechanistic work on negative urgency.
Negative urgency, defined as the tendency to act impulsively when experiencing negative emotions, is a personality trait with critical importance for psychopathology. Of the five distinct personality pathways toward impulsive behavior specified in the (negative) Urgency, (lack of) Premeditation, (lack of) Perseverance, Sensation Seeking–Positive Urgency (UPPS-P) model (Whiteside & Lynam, 2001), negative urgency has emerged as most broadly related to maladaptive behavior (J. M. Berg et al., 2015). The unique clinical relevance of negative urgency lies in its integration of personality predispositions toward internalizing and externalizing disorders, with some evidence indicating that negative urgency maps onto the broad shared empirical variance among psychopathology symptoms labeled the “p” factor (Pearlstein et al., 2024). Negative urgency is thought to be distinct from related personality traits in that the rash action of individuals high on negative urgency is conditional on increases in negative affect (Cyders & Smith, 2008; Peterson & Smith, 2019). These rash actions are theorized to be negatively reinforced and maintained through decreases in negative affect. Given its transdiagnostic nature, treatments designed to target negative urgency are likely to have a greater clinical impact than treatments for individual disorders.
To develop treatments for negative urgency, it is critical to understand the mechanisms that underlie this personality tendency and its relation to psychopathology. Most theoretical accounts and initial mechanistic investigations of negative urgency have focused on how emotions interfere with “top down” cognitive-control processes, such as the ability to inhibit a prepotent (i.e., automatic) response (e.g., Bechara & Van Der Linden, 2005; Billieux et al., 2010; Chester et al., 2017; Chester, Lynam, Milich, Powell, et al., 2016; Gunn & Finn, 2015; Johnson et al., 2020; Wilbertz et al., 2014). There is evidence of a small but significant meta-analytic association between self-reported negative-urgency scores and prepotent response inhibition measured using behavioral tasks (Cyders & Coskunpinar, 2011), with effects stronger in clinical samples compared with community samples (Johnson et al., 2016). Regarding changes in behavior or neural activity in response to emotion, one study found that negative affect experienced after a social-exclusion manipulation predicted poorer performance on a measure of prepotent response inhibition (i.e., go/no-go task) in people with high but not low negative urgency (Chester et al., 2017). Another study reported that participants with elevated negative urgency recruited inhibitory brain regions more when no-go stimuli were overlaid with aversive images compared with pleasant and neutral images, suggesting the need to compensate for poor inhibitory control under negative mood (Chester, Lynam, Milich, Powell, et al., 2016). If impaired cognitive control underlies relationships between negative urgency and psychopathology symptoms, treatments aimed at improving cognitive control when distressed would be indicated. Cognitive-behavioral therapy (CBT), a leading treatment for many of the conditions associated with negative urgency, is thought to work by improving top-down cognitive control (Clark & Beck, 2010). However, a substantial portion of patients do not benefit from CBT, suggesting the need to consider other treatment options.
Another possibility is that negative affect leads to impulsive behavior through heightened “bottom-up” reward processing. Reward processing includes the motivation to approach a tempting stimulus (“wanting”) and responsiveness to the reward once obtained (“liking”; Berridge & Robinson, 2016). Preclinical data indicate that stress “sensitizes” mesocorticolimbic dopamine transmission and enhances the incentive salience of rewards and their associated cues (Berridge & Robinson, 2016). In contrast, hedonic pleasure obtained from a reward is less sensitive to changes in context, such as stress (K. S. Smith et al., 2011). In patients with alcohol, drug, and tobacco use disorders, imagining personally relevant stressful situations induces changes in reward processing and causes increased craving for and consumption of the addictive stimulus (relative to a neutral situation; e.g., Hyman et al., 2007; McKee et al., 2011; Sinha et al., 2009). In support of a reward-processing model of negative urgency, several neuroimaging and behavioral studies have linked negative urgency to increased reward-circuitry responding (e.g., Chester, Lynam, Milich, & DeWall, 2016; Cyders et al., 2014; Eiler et al., 2014; Weiland et al., 2014). For example, Chester, Lynam, Milich, and DeWall (2016) demonstrated that negative-urgency scores correlated with activation in the caudate nucleus, a brain region that links reward to external stimuli, in response to viewing alcohol images versus standard pleasant images. However, a critical limitation of many of these studies is that they have not tested whether negative urgency relates to increases in reward processing under negative-mood conditions and whether mood-induced changes in reward processing underlie relations between self-reported negative urgency and psychopathology. One exception is a study by VanderVeen and colleagues (2016) in alcohol-using adults. These researchers found that negative-urgency scores were related to greater mood reactivity, alcohol craving, and alcohol self-administration during a negative relative to a neutral-mood induction. Thus, negative affect may prompt a desire to approach a tempting stimulus, and enhanced approach motivation may lead to increases in impulsive behavior in individuals with elevated negative urgency. Regarding treatment, retraining of approach-motivation biases or stimulus-reward associations may be indicated for reward-processing alterations (e.g., Noël et al., 2013), pointing to the importance of considering alternative mechanistic models of negative urgency.
In the current study, we tested whether negative mood enhances reward processing of palatable food in individuals across the spectrum of binge-eating severity (i.e., from no binge eating to clinical eating disorders). Binge eating, defined as eating an unusually large amount of food in a short period of time accompanied by a sense of loss of control, is present in 10% to 15% of the population (Mitchison et al., 2017); approximately 5% of individuals meet the criteria of the fifth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5; American Psychiatric Association [APA], 2013) for a binge-type eating disorder (i.e., bulimia nervosa [BN] or binge eating disorder [BED]). Binge eating, BN, and BED are associated with substantial psychological distress, medical morbidity, and obesity risk (Kessler et al., 2013). Binge eating exhibits a robust, specific relationship with negative urgency. Negative urgency is the only impulsive personality trait to reliably relate to binge eating and eating disorders with medium to large effect sizes (for meta-analyses, see J. M. Berg et al., 2015; Fischer et al., 2008), and negative affect is a well-established antecedent of binge-eating episodes (Haedt-Matt & Keel, 2011). Examining a population whose impulsive behavior regularly occurs in response to negative affect allows for a particularly strong test of negative-affect-induced changes in reward-processing mechanisms. Finally, binge eating exhibits strong links to reward processing. The palatable foods commonly consumed during binge-eating episodes increase dopamine and opioid neurotransmission, similar to drugs of abuse (Berridge, 2009). Binge eating has been associated with neural, physiological, behavioral, and self-report indices of reward dysregulation (for reviews, see Harrison et al., 2010; Leenaerts et al., 2022; Wu et al., 2016). Of particular importance to the current study, initial data indicate that negative affect increases the reward value of palatable food in vulnerable individuals (women who report dieting or emotional eating) and in individuals with binge-type eating disorders (Arend et al., 2022; Schnepper et al., 2020, 2021; Wagner et al., 2012). However, no study has examined whether emotion-induced changes in reward processing explain the association between negative urgency and binge eating.
In the current study, emotion-enhanced reward processing of palatable food cues was assessed across multiple units of analysis (i.e., physiology, behavior, self-report) within the context of two paradigms (i.e., picture-viewing paradigm; Food-Effort Expenditure for Rewards Task [F-EEfRT]; Racine et al., 2019). During the picture-viewing paradigm, the postauricular reflex (PAR), a reflexive physiological index of positive emotion (Benning, 2018), and Self-Assessment Manikin (SAM; Bradley & Lang, 1994) ratings of valence and craving were obtained while participants viewed food and emotional images. Behavioral choices on the F-EEfRT were used to index willingness to work for food rewards, a component of approach motivation, and self-reports of “wanting” and “liking” for the resulting palatable food reward were obtained. Both paradigms were completed under negative- and neutral-mood conditions induced via personalized imagery scripts (Sinha & Tuit, 2012), a well-validated method used to examine emotion-induced craving. Study results have the potential to inform intervention approaches for conditions characterized by negative urgency by revealing whether targeting “bottom-up” reward processing is likely to result in improvements in emotion-based rash behaviors, such as binge eating.
Hypotheses were as follows:
Hypothesis 1: Self-reported negative-urgency scores will relate to emotion-enhanced wanting of palatable food, as indicated by PAR potentiation, SAM craving ratings, F-EEfRT choices, and food reward wanting ratings.
Hypothesis 2: Binge-eating frequency and severity will relate to emotion-enhanced wanting of palatable food using the same indices.
Hypothesis 3: Emotion-enhanced wanting of food will mediate the association between self-reported negative urgency and binge-eating frequency/severity.
Transparency and Openness
Preregistration
This study was not preregistered.
Data, materials, code, and online resources
Data and code for this study are available at https://osf.io/dc29m/files. Supplemental methods, tables, and figures are available in the Supplemental Material available online.
Reporting
We report how we determined our sample size, all data exclusions, all manipulations, and all measures in the study.
Ethical approval
The study protocol was approved by the McGill University Faculty of Medicine Institutional Review Board (A06-B26-17B) and the Douglas Institute of Mental Health Research Ethics Board (2019-204 IUSMD-18-20).
Method
Participants
Participants were women with (n = 105) and without (n = 48) binge eating recruited from Montreal, Canada, via flyers posted in the community, social media advertisements, and short presentations at local treatment centers. Eligible participants were individuals ages 18 to 60 who identified as a woman and were able to communicate in English or French. Women with binge eating were required to report at least one objective binge-eating (OBE) episode over the past 3 months. Exclusion criteria were (a) a history of a neurological disorder, head injury, or loss of consciousness for ≥ 10 min; (b) uncorrected vision or hearing difficulties; (c) allergies to or unwillingness to eat study foods; (d) allergies to adhesives or latex; (e) current use of psychiatric medications with the exception of selective serotonin reuptake inhibitors; and (f) current major depressive episode, mania, psychosis, or moderate to severe substance use disorder. Additional exclusion criteria for women without binge eating were any history of binge eating, an eating disorder, or another psychiatric disorder.
A sample size of 200 participants was originally specified for this study based on feasible recruitment targets over the 3-year grant and a sensitivity power analysis revealing that 180 women (accounting for 10% physiological-data loss) would provide 80% power to detect even small interaction effects (Cohen’s f = 0.10) between self-reported negative urgency and mood condition in mixed analyses of variance, assuming α = .05 and a correlation between repeated measures = .50. Data collection occurred between October 2018 and August 2022, with a break from March 2020 to March 2021 because of the COVID-19 pandemic. This break in data collection resulted in us falling slightly short of our recruitment target. One hundred fifty-three women completed all parts of the study, and a sensitivity power analysis with the same parameters as above revealed that this sample size provided 80% power to detect effect sizes only slightly larger than those prespecified (Cohen’s f = 0.11).
For descriptive statistics and clinical characteristics for individuals in the final sample, see Table 1 (one person did not complete questionnaires). Sixty-three participants (41.4%) identified as a visible minority; 11.2% identified as Chinese, 6.6% identified as South Asian, 5.9% identified as Black, 4.6% identified as Latin American, 3.9% identified as Arab, 2.6% identified as Southeast Asian, 2.6% identified as Korean, 2% identified as West Asian, 0.7% identified as Japanese, 4.6% identified as other, and 3.3% preferred not to indicate their ethnicity. Most participants reported their sexual orientation as heterosexual (75%), 18.4% identified as bisexual/pansexual, 2.6% identified as lesbian, 0.7% identified as asexual, and 3.3% preferred not to answer. Median combined parental income among students (n = 101) was $75,000, and median household income among nonstudents (n = 51) was $50,500. Most (96.73%) participants completed the study in English, and 3.27% participated in French.
Descriptive Statistics and t Tests for Demographic and Clinical Characteristics
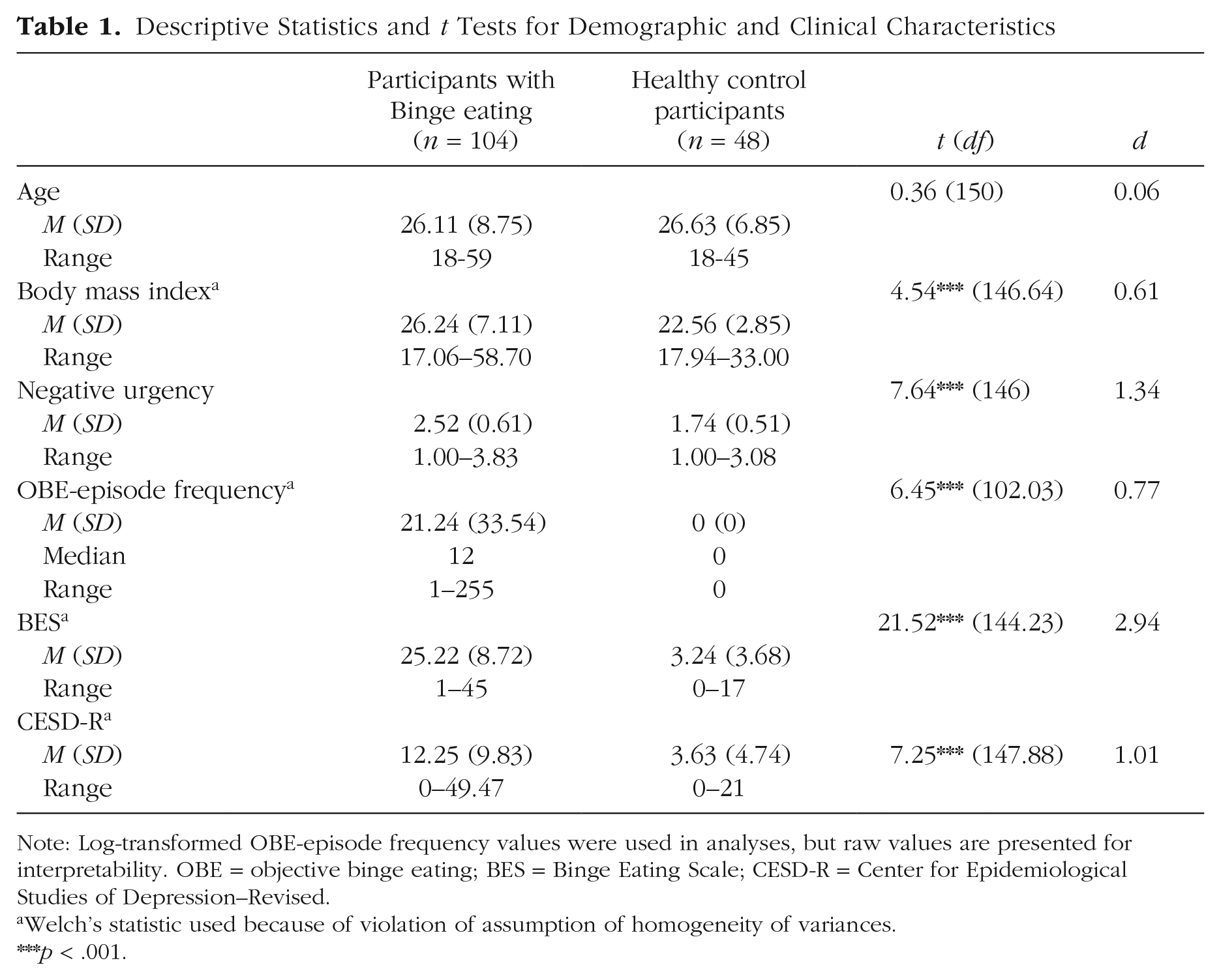
Note: Log-transformed OBE-episode frequency values were used in analyses, but raw values are presented for interpretability. OBE = objective binge eating; BES = Binge Eating Scale; CESD-R = Center for Epidemiological Studies of Depression–Revised.
Welch’s statistic used because of violation of assumption of homogeneity of variances.
p < .001.
Procedures
For a detailed flowchart of study procedures, see Figure 1.
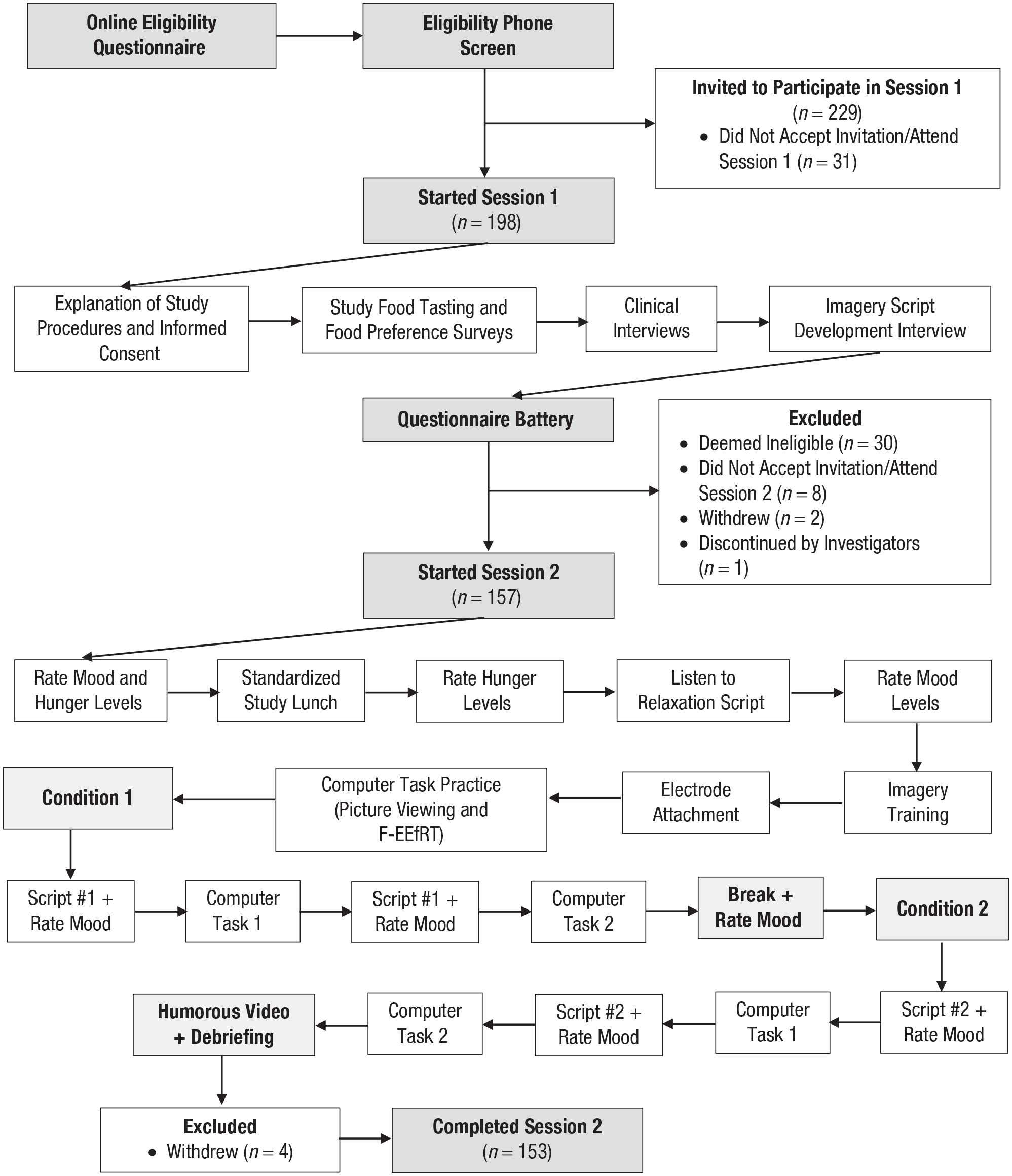
Flowchart of progression of study procedures.
Screening procedures
An online screening questionnaire was used to evaluate exclusion criteria and detect probable OBE episodes. Potentially eligible participants were then contacted via email to arrange a phone screen with a trained research assistant. Following verbal consent, questions from the Structured Clinical Interview for DSM-5 Disorders-Research Version (SCID-5-RV) were used to assess psychiatric inclusion (e.g., OBE episodes) and exclusion (e.g., current major depressive episode) criteria.
Session 1: clinical interviews
Session 1 procedures were completed with the study research coordinator, a graduate student, or a postdoctoral scholar either in person (before the COVID-19 pandemic; n = 93) or via Microsoft Teams (during the COVID-19 pandemic; n = 60). After providing informed consent and completing several food-selection tasks (described below), participants were interviewed using modules from the SCID-5-RV and a modified version of the Eating Disorder Examination to diagnose eating and other psychiatric disorders. Then, using the personalized imagery script methods outlined by Sinha and Tuit (2012), participants were asked to describe a stressful and relaxing event that they experienced in the past year. Stressful events were events that made participants “mad, sad, or upset” and they felt they “could not do much to change it” (Sinha & Tuit, 2012). Perceived stress was assessed on a 10-point scale from not at all stressful to the most stress they felt in the past year, and only events rated 8 or above were accepted. Relaxing events included situations that participants found to be calm and low energy (e.g., taking a bath, going for a walk). Five-minute stressful and relaxing scripts were written based on the events and were audio-recorded by a trained research assistant.
Session 2: experimental tasks
Session 2 took place in the laboratory (both before and during the COVID-19 pandemic) approximately 1 week following Session 1. Participants were asked not to eat, smoke, or consume caffeine for 3 hours before the session. They were provided with a standardized lunch (sandwich, yogurt, fruit cup, water) and were told to eat until they were comfortably full. To reduce individual variability in participants’ imagery abilities, which may affect the efficacy of the mood induction, participants then completed an imagery-training procedure (Sinha & Tuit, 2012).
Participants completed two experimental tasks (the picture-viewing paradigm and F-EEfRT) under two mood conditions (stressful and relaxed; within-subjects manipulation). Before completing each task, participants listened to one of their personalized scripts (either stressful or relaxing event) and rated their emotions before and after listening to the script. The order of the experimental tasks and the mood condition order were randomized across participants. Between the two mood conditions, participants were given a 10-min break that was followed by a 5-min relaxation script to return emotion ratings to baseline. Additional 5-min breaks were provided if ratings did not return to baseline following the initial break.
Measures
Because of the bilingual nature of the recruitment region, the study was available in either English or French. Validated French versions of measures were used if available (e.g., UPPS Impulsive Behavior Scale; Van der Linden et al., 2006). If unavailable, study measures were translated by bilingual members of the research team and back-translated by a bilingual professional translator blind to the original content.
Clinical interviews
Twenty-seven (17.5%) audio-recorded interviews were coded by an independent rater to calculate interrater reliability estimates.
Modified Eating Disorder Examination
The modified Eating Disorder Examination (EDE; Fairburn et al., 2014) is a semistructured interview that evaluates current (past 3 months) and lifetime presence of eating-disorder symptoms. A systematic review of the psychometric properties of the EDE supports its use (K. C. Berg et al., 2012). Our lab’s modifications to the original EDE include a more comprehensive assessment of loss of control eating and of the timeline of past eating pathology and the addition of a restriction module to evaluate compensatory and noncompensatory restrictive eating. The EDE was used to confirm eligibility, determine eating-disorder diagnosis, and quantify one of the primary variables: OBE episode frequency, defined as frequency of OBE episodes over the past 3 months. For a description of the eating-disorder diagnoses, see the Supplemental Methods and Table S1 in the Supplemental Material. Interrater reliability estimates for frequency of OBE episodes (κ = .80) and for eating-disorder diagnosis (κ = .76) indicated substantial agreement.
SCID-5-RV
The SCID-5-RV (First et al., 2015) is a semistructured clinical interview that evaluates diagnostic criteria for psychiatric disorders. The SCID is the most widely used tool to assess DSM disorders and has strong psychometric properties (First et al., 2015; Shabani et al., 2021). The SCID was used to confirm participant eligibility and characterize the sample on comorbid psychopathology. For a list of the diagnoses assessed and the percentage of the final sample meeting criteria for each, see Table S2 in the Supplemental Material. Interrater reliability for SCID diagnoses was generally substantial (κs = .65–1.0), except for moderate agreement for social anxiety disorder (κ = .51) and fair agreement for current alcohol use disorder (κ = .29).
Questionnaires
A questionnaire battery was completed online between Sessions 1 and 2.
UPPS-P
The Negative Urgency subscale of the UPPS-P (Lynam et al., 2006) includes 12 items rated from 1 (agree strongly) to 4 (disagree strongly). The Negative Urgency subscale has strong psychometric properties, including good internal consistency (α = .86; Whiteside & Lynam, 2001), test–retest reliability over a 1-month period (r = .86; Weafer et al., 2013), and convergent and discriminant validity (G. T. Smith et al., 2007). In the current sample, the internal consistency estimate for the Negative Urgency subscale was good (α = .87).
Binge Eating Scale
The Binge Eating Scale (BES; Gormally et al., 1982) is a 16-item measure that assesses behavioral, affective, and cognitive components of binge eating. Each item is composed of three or four statements, and participants are asked to select the one statement that best describes their eating behavior. BES scores range from 0 to 46, and cutoff scores can be used to categorize individuals as having no or minimal binge eating (score ≤ 17), mild to moderate binge eating (score = 18–26), and severe binge eating (score ≥ 27; Marcus et al., 1985). The BES has excellent psychometric properties, with strong internal consistency (α = .88) and 1-month test–retest reliability (r = .84; Duarte et al., 2015). BES scores were used as our measure of binge-eating severity. In the current sample, internal consistency was excellent (α = .96).
Center for Epidemiological Studies of Depression–Revised
Given the co-occurrence of binge eating and depressive symptoms and the fact that depression is often associated with decreased reward processing, we included depressive symptoms as a covariate. The Center for Epidemiological Studies of Depression–Revised (CESD-R) scale (Eaton et al., 2004) is a 20-item measure that assesses DSM-5 (APA, 2013) symptoms of major depression “over the past week or so” on a scale from 0 (not at all or less than one day) to 4 (nearly every day). Internal consistency estimates were excellent in past studies (α = .92; Van Dam & Earleywine, 2011) and good in the current sample (α = .88).
Experimental tasks
Both the picture-viewing paradigm and the F-EEfRT were presented using E-Prime 3.0 software (Schneider et al., 2016) and viewed on a 24-inch monitor.
Picture-viewing paradigm
The picture-viewing paradigm consisted of 60 images divided into five categories of 12 pictures each: aversive (six threat, six disgust/mutilation), pleasant (six erotic, six nurturant), neutral (six objects, six humans), high-calorie food (six standard, six personalized), and low-calorie food (six fruits, six vegetables). Affective images were drawn from the International Affective Picture System (Lang et al., 2008) and were chosen based on normative valence and arousal ratings in women. Food images were drawn from the Open Library of Affective Foods (OLAF; Miccoli et al., 2014) and were chosen based on normative ratings in female adults (Miccoli et al., 2016). From a list of 26 foods, participants chose six that they either typically consumed during a binge-eating episode or highly craved, and images of these six foods were chosen from the internet to include personalized stimuli in the picture-viewing paradigm. Because participants completed the picture-viewing paradigm twice, two image sets were created, with images within categories matched on normative ratings of valence and arousal, and participants were randomly assigned to the image set viewed in the stressful- versus relaxed-mood condition. Four quasi-random run orders were created for each image set so that two images from the same affective or food category were not presented consecutively.
Each image was presented for 6 s followed by a 3-s intertrial interval (ITI), during which participants saw a black screen. Immediately after each image, participants rated the image on the dimensions of valence, arousal, and craving using the SAM (Bradley & Lang, 1994). The SAM scales are 9-point pictorial rating scales originally developed to assess valence (1 = unhappy, 9 = happy) and arousal (1 = calm, 9 = excited). A SAM scale assessing craving was developed for use in addiction research and has been administered alongside OLAF food images (Miccoli et al., 2014; Racine et al., 2021).
F-EEfRT
The F-EEfRT Racine et al. (2019) is a multitrial, button-pressing task that was adapted from the original EEfRT (Treadway et al., 2009) to assess individuals’ willingness to work for food rewards. Participants choose between “easy” and “hard” trials and must make repeated manual button presses on the computer keyboard in a short period of time to have the chance of “winning” the trial. Instead of competing for monetary reward (easy task: $1.00; hard task: $1.24–$4.30; Treadway et al., 2009), participants completing the F-EEfRT compete for portions of a preferred food (easy task: one portion; hard task: two to five portions). As in the original EEfRT, the probability of receiving the reward if the trial is completed varies and is either 12%, 50%, or 88%. The F-EEfRT was previously validated in a sample of undergraduate women across the spectrum of binge eating (Racine et al., 2019).
In the current version of the task and consistent with some previous research (e.g., Gilman et al., 2015; Reddy et al., 2015), participants began the task by completing three calibration trials to tailor the tasks to individuals’ button-pressing ability: Easy trials required 33% of each participant’s average button-press rate over the three calibration trials in 4 s using the dominant-hand index finger. Hard trials required 90% of the average button-press rate over the three calibration trials in 12 s using the nondominant hand little finger. Before each trial, participants were informed of the probability of winning the trial (12%, 50%, or 88%) and of the reward magnitude (two, three, four, or five portions of food) associated with the hard task. Participants had 5 s to select an easy or hard task, after which the computer program made a forced choice. After each trial, participants received feedback on whether they completed the trial and the amount of food portions they won. Figure S1 in the Supplemental Material shows a single trial of the F-EEfRT.
Two randomly selected “win” trials determined the number of standardized portions of preferred food that the participant received as a reward. Two of the following four foods were chosen in Session 1 based on liking ratings after tasting (before the pandemic) or when imagining having recently eaten the food (during the pandemic): M&Ms, two-bite brownies, Lay’s regular potato chips, and Doritos chips. One food was used per mood condition, and the order of preference of food reward was counterbalanced. Participants rated their wanting of the upcoming food reward after learning the number of portions won and their liking of the food upon first bite using 100-point visual-analogue scales.
Psychophysiology
Psychophysiology indices
During the picture-viewing paradigm, two physiological reflexes previously shown to relate to reward reactivity were measured. The primary index, the PAR, is a vestigial ear muscle response whose activation is thought to reflect appetitive reactivity (Benning, 2018; Stussi et al., 2023). The PAR is heightened in response to pleasant relative to neutral stimuli (Benning et al., 2004), with particularly strong potentiation to food (Sandt et al., 2009). We have previously shown that undergraduate students with past-month binge eating have greater PAR potentiation to food images than students without binge eating (Racine et al., 2018). PAR has also been shown to be sensitive to appetitive conditioning (using pleasant odors and juice; Ebrahimi et al., 2019; Stussi et al., 2018) and to respond to both appetitive pictures used as rewards and to cues associated with an upcoming reward (Ebrahimi et al., 2019; Stussi et al., 2018).
The secondary index, the startle eyeblink reflex, is a widely used physiological index of threat reactivity. It is potentiated in response to aversive stimuli and (typically) inhibited in response to pleasant stimuli relative to neutral stimuli (Lang, 1995). Inhibition of the startle eyeblink reflex is sometimes interpreted as indicating a reward response (e.g., O’Hara et al., 2016; Reichel et al., 2014), although it may also represent ongoing attention to emotional foreground stimuli (Bradley et al., 2006). We have previously shown that startle-eyeblink-reflex magnitudes relate inversely to craving ratings for food (Suissa-Rocheleau et al., 2019).
Psychophysiology data collection and reduction
The PAR and startle eyeblink reflexes were measured using 4-mm sintered electrodes filled with Signagel conductive gel (Parker Laboratories Inc.) placed on either side of the tendon of insertion of the postauricular muscle of each ear and on the lower left orbicularis oculi muscle (startle eyeblink; Benning et al., 2004; Blumenthal et al., 2005). An 8-mm electrode placed on the forehead served as the ground. Electrical impedances were minimized by first cleaning and slightly abrading the skin with an alcohol wipe and NuPrep (Weaver and Company), and impedances were measured using a checktrode (UFI MKIII Model 1089).
Both reflexes can be elicited with auditory noise probes, which consisted of 50-ms, 95-dB bursts of white noise with near instantaneous rise and fall time presented via calibrated headphones (Etymotics ER3SE). During 83% of pictures, auditory probes occurred at 3-, 4-, or 5-s postpicture onset. During 9% of ITIs, a startle probe occurred to enhance unpredictability. Participants were exposed to four noise probes during a practice experiment for habituation.
Psychophysiology data were collected using a MindWare Bionex system (Mindware Technologies). A sampling rate of 1000 Hz and gain of 5000 were used during data acquisition. A 60-Hz notch filter was applied, data were rectified, and bandpass filters (8–500 Hz for PAR; 28–250 Hz for startle) were applied (Blumenthal et al., 2005; Racine et al., 2018, 2021). Because PAR is a microreflex, the waveforms from each image category (e.g., high-calorie food images) were averaged to create aggregate waveforms (Benning et al., 2004). PAR magnitude was scored as the peak activity 8 ms to 35 ms following onset of the startle probe minus the average activity during the 50 ms before the startle probe (Benning et al., 2004; Racine et al., 2018, 2021). PAR magnitudes from right and left ears were averaged when data from both ears were available. Startle-blink-reflex magnitude was scored on a per trial basis as the peak activity 30 ms to 120 ms poststartle probe minus average activity during the 50-ms prestartle probe baseline (Blumenthal et al., 2005; Racine et al., 2018, 2021).
Trained research assistants examined each PAR aggregate waveform and individual startle waveform to evaluate the presence of a response. A second rater independently processed the data when responses were unclear. Difficult files were reviewed by graduate students who oversaw data processing. Trials in which responses were indistinguishable from baseline because of noise or movement were omitted from analyses. Nonresponses were coded as 0 and included in analyses. Data from at least four (of 10) trials were required for an image category to be included in analyses. Participants who had mean PAR values of ≤ 2µV (n = 5) or mean startle values of ≤ 5µV (n = 10) were excluded from analyses.
Consistent with past studies (Benning & Ait Oumeziane, 2017; Racine et al., 2021), we examined the difference between PAR and startle-blink-response magnitudes during high-calorie food images compared with neutral images. PAR values were significantly skewed and kurtotic but were normalized with log transformation.
Data analysis
Mixed analyses of covariances (ANCOVAs) were used to test Hypotheses 1 and 2. Independent variables included (a) mood condition (0 = relaxed, 1 = stressed); (b) self-reported negative-urgency scores (Model 1), binge-eating frequency (log-transformed; Model 2), and binge-eating severity (Model 3); and (c) the interaction between mood condition and negative urgency, binge-eating frequency, or binge-eating severity. Dependent variables included (a) PAR-response magnitude during high-calorie food images minus neutral images, (b) SAM valence ratings to high-calorie food images, (c) SAM craving ratings to high-calorie food images, (d) proportion of high-cost/high-reward choices on the F-EEfRT, (e) wanting ratings for upcoming preferred food, and (f) liking ratings of preferred food on first bite. Covariates included mood-condition order (0 = relaxed first, 1 = stressed first), task order (0 = picture viewing first, 1 = F-EEfRT first), CESD-R scores, and (for F-EEfRT analyses) whether the highest rated palatable food was served first (0) or second (1). All continuous variables were standardized before analysis. An alpha value of .05 was used for omnibus effects, and a Sidak correction for multiple comparisons was used for post hoc tests.
To test Hypothesis 3, multilevel mediation analyses (2-1-2) using a multilevel structural-equation-modeling (MSEM) framework (Preacher et al., 2010) implemented in Mplus (Muthén & Muthén, 2017) were conducted. These analyses were carried out only if there was evidence that both negative urgency (independent variable) and either binge-eating frequency or severity (dependent variable) were related to a particular reward-processing index (mediator) in ANCOVAs. Covariates were the same as those included in ANCOVAs. Full-information maximum likelihood estimation with robust standard errors was used.
Results
Clinical characteristics
Participants with and without binge eating were compared on several demographic and clinical characteristics (see Table 1). There was no significant age difference between groups, but women with binge eating had a significantly higher body mass index than women without binge eating. As expected, women with binge eating had significantly greater scores on all clinical measures than control women. Median OBE-episode frequency over the past 3 months was 12, corresponding to the DSM-5 (APA, 2013) requirement of once a week for 3 months for a full-threshold BN or BED diagnosis. The average BES score was in the upper end of the mild to moderate range, and the average CESD-R score was below the cutoff for probable depression (i.e., 16).
Validation analyses
For details of validation analyses, see Supplemental Methods in the Supplemental Material.
Mood induction
Regarding the ability of the stressful and relaxation scripts to induce the intended moods, 100-point visual-analogue scale ratings of “stress” and “relaxation” were compared and are shown in Table S3 and Figure S2 in the Supplemental Material. Consistent with the intended effect of the mood induction, stress ratings significantly increased after listening to the stressful script and significantly decreased after listening to the relaxing script. Likewise, relaxation ratings significantly increased after the relaxing script and decreased after the stressful script.
Picture-viewing paradigm
To establish the validity of the picture-viewing paradigm, we compared image categories on SAM valence, arousal, and craving ratings and on PAR and startle-blink-response magnitudes (see Table S4 in the Supplemental Material). SAM valence ratings of emotional images followed the expected pattern (pleasant > neutral > aversive); high-calorie food images were rated comparably with pleasant images, and low-calorie food images fell between pleasant and neutral images. Aversive images received the greatest arousal ratings, followed by pleasant images and high-calorie food images, low-calorie food images, and neutral images. High-calorie food images elicited greater craving ratings than low-calorie food images, and both food-image categories received greater craving ratings than emotional images. There were no significant differences in PAR magnitude across image types. Thus, in contrast to prior studies (Benning & Ait Oumeziane, 2017; Benning et al., 2004; Racine et al., 2021), PAR magnitude was not potentiated during pleasant or high-calorie food images relative to neutral images. As expected, mean startle-blink magnitude was greatest to aversive images and lowest to pleasant images, but only pleasant images significantly differed from neutral images. Startle-blink magnitudes during food images were significantly lower than during aversive images but did not significantly differ from that during neutral images, and high-calorie food images also did not differ from that during pleasant images.
F-EEfRT
To establish the validity of the F-EEfRT, we examined the effects of trial-level parameters (probability of winning [12%, 50%, 88%] and reward magnitude of the hard task [two, three, four, or five food portions]) on choice behavior. Consistent with past research (Racine et al., 2019), there were significant main effects of probability, F(2, 188) = 134.54, p < .001, h2 p = .56, and reward magnitude, F(3, 242) = 38.68, p < .001, h2 p = .27, and a significant Probability × Reward Magnitude interaction, F(6, 564) = 6.83, p < .001, h2 p = .06, on the proportion of hard-task choices. For results, see Figure S3 in the Supplemental Material.
Hypothesis-testing analyses
For ANCOVA results, see Table 2. Contrary to hypotheses, the effect of mood condition on reward-processing indices and interactions between mood condition and negative urgency, binge-eating frequency, and binge-eating severity were almost uniformly nonsignificant. The one exception was a significant interaction between mood condition and negative-urgency scores on PAR magnitude during high-calorie food minus neutral images, F(1, 117) = 4.60, p = .034, h2 p = .038. Negative urgency was related (albeit nonsignificantly) to a greater positive difference in PAR magnitude during high-calorie-food images versus neutral images (i.e., PAR potentiation to food) in the relaxed condition, b = 0.09, SE = 0.05, p = .06, h2 p = .03, but not in the stressed condition, b = −0.04, SE = 0.04, p = .30, h2 p = .01.
Primary Analyses Examining Mood Condition, Negative Urgency/Binge Eating, and Their Interaction in Relation to Reward-Processing Indices
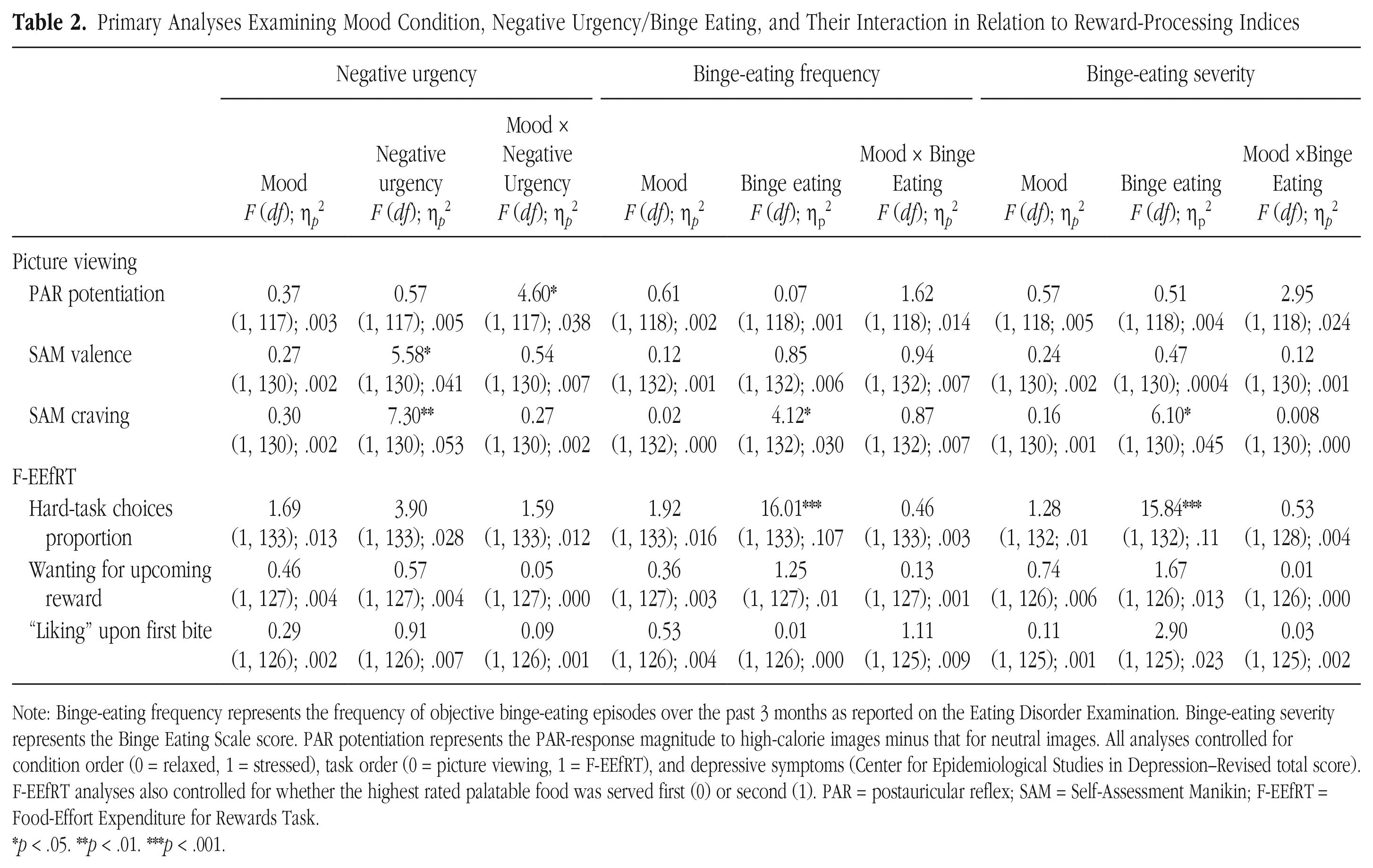
Note: Binge-eating frequency represents the frequency of objective binge-eating episodes over the past 3 months as reported on the Eating Disorder Examination. Binge-eating severity represents the Binge Eating Scale score. PAR potentiation represents the PAR-response magnitude to high-calorie images minus that for neutral images. All analyses controlled for condition order (0 = relaxed, 1 = stressed), task order (0 = picture viewing, 1 = F-EEfRT), and depressive symptoms (Center for Epidemiological Studies in Depression–Revised total score). F-EEfRT analyses also controlled for whether the highest rated palatable food was served first (0) or second (1). PAR = postauricular reflex; SAM = Self-Assessment Manikin; F-EEfRT = Food-Effort Expenditure for Rewards Task.
p < .05. **p < .01. ***p < .001.
As expected, several indices of reward processing were related to negative urgency and binge eating via main effects. Negative urgency but not binge eating was associated with greater pleasure ratings to high-calorie food images, whereas binge-eating frequency and severity but not negative urgency were associated with a greater proportion of high-cost/high-reward choices on the F-EEfRT. Negative urgency and both indices of binge eating were related to greater craving ratings of high-calorie food images.
MSEM results indicated that only negative urgency and not craving ratings of high-calorie foods was significantly independently associated with binge-eating frequency and severity. In both cases, the indirect effect of negative urgency on binge eating through craving was nonsignificant (estimates = 0.027–0.031; ps = .15–.17).
Exploratory post hoc analyses
Exploratory post hoc analyses were conducted to better understand possible reasons for null results. For details of these analyses, see Supplemental Results in the Supplemental Material. To summarize, we first considered whether the restrictive eating that would be more likely to be present in participants with BN-spectrum diagnoses than BED-spectrum diagnoses might affect results by examining broad diagnosis instead of continuous binge-eating symptoms as a moderator, but no main or interaction effects emerged (see Table S5 in the Supplemental Material). We also wondered whether the design decision to complete a within-subjects mood manipulation with both mood conditions experienced on the same day affected results (e.g., because of carryover effects or habituation to study procedures), but reanalyzing our data in a between-subjects fashion (i.e., by examining only data from the first mood condition participants underwent) did not reveal any additional significant results. We considered whether individual differences in stress responsiveness might be relevant by investigating stress ratings after the stressful mood induction (controlling for stress before induction) as a moderator of the impact of negative-urgency and binge-eating variables on reward-processing outcomes. The three significant interactions that emerged suggested that for individuals with elevated negative urgency and binge-eating frequency, reward processing was greater when stress ratings were low but not high, which is opposite to the hypothesis of emotion-enhanced reward processing. Finally, given the lack of difference in PAR across picture types, we examined startle-blink magnitude as an alternative physiological index of reward processing. Only one significant interaction effect emerged (i.e., between mood condition and OBE-episode frequency), and the pattern was again such that individuals with greater OBE-episode frequency experienced greater reward processing (indexed by an attenuated startle blink reflex to high-calorie food vs. neutral images) in the relaxed versus stressful mood condition.
Discussion
This study was designed to test whether negative mood enhances reward processing in individuals with elevated negative urgency and whether emotion-enhanced reward processing underlies the relationship between negative urgency and binge eating. We conducted a within-subjects mood manipulation (stressed vs. relaxed) using personalized imagery scripts, assessed several indices of reward processing measured across multiple levels of analysis, and examined dimensional assessments of binge eating in individuals recruited to represent the spectrum of binge-eating severity. Overall, results did not support a model in which negative urgency is characterized by enhanced reward processing of food under negative mood conditions given that interactions between mood condition and negative-urgency scores were largely nonsignificant. In the few instances in which significant interactions emerged, effects were opposite to hypotheses—the impact of individual differences on reward-processing outcomes instead were seen under relaxed or lower stress conditions. Findings have implications for understanding the construct of negative urgency and the role of reward processing in the maintenance of binge eating.
The construct of negative urgency
Negative urgency has been conceptualized as a conditional trait—the impulsive behavior of individuals high on negative urgency is thought to emerge only under negative-mood conditions. However, the conditional operation of negative urgency has been difficult to capture in both experimental studies like ours and ecological momentary assessment (EMA) studies that examine whether trait negative urgency strengthens the momentary relationship between negative affect and impulsive behaviors (e.g., Feil et al., 2020; Sperry et al., 2016). More broadly, the overlap between the self-reported personality trait of negative urgency with biological, behavioral, and within-subjects measures has been limited. In this study, negative urgency was related to greater self-reported valence and craving ratings of high-calorie foods overall but not to more positive appraisals of these foods specifically under negative-mood conditions.
Clearly, negative urgency is important for psychopathology (J. M. Berg et al., 2015; Pearlstein et al., 2024). Research has demonstrated that negative urgency is more than negative affectivity, lack of planning impulsivity, and their interaction (Peterson & Smith, 2019), and negative urgency relates to psychopathology beyond these variables (e.g., King et al., 2022; Peterson & Smith, 2019; Racine et al., 2013). Moreover, elevated negative urgency predicts poor treatment outcomes among individuals with various disorders (e.g., Manasse et al., 2016; Mestre-Bach et al., 2020; Peckham et al., 2019). Perhaps rather than continue to try to “map on” self-reported negative urgency to biobehavioral mechanisms or within-subjects processes, one can instead recognize negative urgency as an individual difference marker of psychopathology risk and maintenance, identifying individuals who may eventually develop psychopathology or who may benefit from particular treatments (e.g., dialectical behavior-therapy techniques). Mechanistic research can continue to focus on identifying the individual-level processes that link affect to poor behavioral control, which may differ across individuals or circumstances.
Reward processing and binge eating
Regarding the implications of our results for binge eating, we found that binge-eating frequency and severity were related to greater craving ratings of high-calorie food images and an increased willingness to expend effort for a preferred food reward. These findings replicate previous work examining food craving and effort expenditure in populations with binge eating (e.g., Bodell & Keel, 2015; Leehr et al., 2015; Racine et al., 2019; Reents & Pedersen, 2021). When combined with null results for valence and liking ratings, findings suggest that greater binge-eating frequency/severity may be more strongly associated with excessive wanting rather than liking of high-calorie foods (Berridge & Robinson, 2016) irrespective of emotional state. Our study is novel in its consideration of the interaction between negative affect and reward processing in relation to binge eating. Both have been conceptualized as independent mechanisms underlying binge-type eating disorders, but few studies have considered them together. Exceptions include two studies that examined differences in food-cue reactivity (defined as self-reported pleasantness and drive to eat and corrugator-muscle response) under negative- versus neutral-mood conditions in patients with BN and BED (Arend et al., 2022; Schnepper et al., 2021). There is also a recent EMA study that found that increases in craving mediated the association between negative affect and engagement in binge-eating episodes in patients with BN (Leenaerts et al., 2023). The absence of emotion-enhanced reward-processing effects in our study may be due to our inclusion of individuals with subthreshold forms of BN, BED, and binge eating, who may not exhibit the same conditioned effects of emotion on food reward as individuals with a more severe or longer duration of illness. Another important consideration for future research is the existence of mechanistic subtypes of patients who binge eat for some reasons but not others. For example, past research has identified both high dietary restraint and high dietary restraint/negative affect subtypes of BN and BED (e.g., Grilo et al., 2001; Stice & Agras, 1999). It may be that individuals who belong to the dietary-restraint-only subtype are less likely to binge eat in response to negative emotions or craving.
Design considerations
Given that we did not replicate previous studies that found that a negative or stressful mood enhanced reward processing of food in individuals with binge eating (e.g., Arend et al., 2022; Schnepper et al., 2021), we also must consider study-design elements as possible reasons for null results. The first was the choice to have individuals undergo both a stressful and a relaxed mood induction on the same day. This was done for feasibility reasons and was based on previous successful use of this procedure (e.g., Hyman et al., 2007; Sinha et al., 2000), although more recent studies using personalized imagery scripts conducted mood inductions on different days (e.g., McKee et al., 2011; Sinha et al., 2009). Although post hoc between-subjects analyses indicated no differences in outcomes between individuals who underwent the stressful- versus the relaxed-mood induction first, we may have seen a different pattern of effects with a within-subjects mood manipulation completed on different days. A second consideration is that the mood induction may not have persisted throughout the tasks, and thus behavior may have been affected toward the beginning but not toward the end of the tasks. Previous studies of mood manipulations in relation to negative urgency or binge eating have used an adjunctive musical mood-induction procedure (VanderVeen et al., 2016) or included phrases from a personalized negative script throughout the experimental task (Arend et al., 2022; Schnepper et al., 2020, 2021). Finally, it is possible that fatigue affected our results given that the study protocol lasted 5 hours and participants were required to complete the same tasks twice.
Another important design consideration for future studies examining the impact of acute stress on reward sensitivity is how to treat factors such as depression and chronic stress. Because blunted reward processing increases risk for depression and characterizes the depressed state (for a review, see Keren et al., 2018), we chose to exclude participants experiencing a current major depressive episode. Nonetheless, we had significant variability in current depressive symptoms and a large subset of the sample with past major depression or persistent depressive disorder. Note that when significant results were detected in the current study (mostly in the context of exploratory post hoc analyses), we found stronger associations between negative urgency and binge eating with reward-processing indices under relaxed or low-stress conditions. Thus, unlike findings from the addiction literature on which the current study’s design and hypotheses were based (Berridge & Robinson, 2016; Sinha & Tuit, 2012), these findings correspond better with the blunted reward processing observed in response to both laboratory and self-reported stressors, with stress-related blunting of reward processing thought to increase risk for depression (e.g., Burani et al., 2023; Ethridge et al., 2020; Lincoln et al., 2019; Porcelli et al., 2012; Vidal-Ribas et al., 2019). Considering how a history of depression and life stress interact with the experience of acute stress to affect reward processing is an important avenue for future research.
Several strengths and weaknesses of our study have already been mentioned. Additional strengths include the use of personalized food images in the picture-viewing paradigm and preferred food rewards in the F-EEfRT, which maximizes the likelihood that individuals will experience a reward response. In addition, many experimental controls were incorporated into the protocol (e.g., standardized lunch, imagery training) to minimize confounding factors. Weaknesses include the decision to recruit only women. This was in part due to feasibility but also because of nonhuman animal and human data supporting sex differences in stress and reward responsivity (e.g., Bale & Epperson, 2015; Becker & Chartoff, 2019; Ter Horst et al., 2009; Warthen et al., 2020) and differences in the types of stimuli women and men find rewarding (e.g., Spreckelmeyer et al., 2009). Nonetheless, it is critical to examine the mechanisms that underlie binge eating in men because men are vastly underrepresented in eating-disorders research and it is possible that our theoretical and clinical models do not apply equally to men and women. Furthermore, our decision to exclude participants with current depression meant that the eating-disorder and comorbid-psychopathology severity of our participants was lower and likely less representative of the population of people with binge eating (e.g., very few participants from our partner tertiary-care hospital for eating disorders were eligible). Another limitation is the fact that our picture-viewing paradigm included affective images, which may have contaminated our intended mood manipulation, and our study design did not allow for a direct examination of this possibility. However, because participants viewed the same number of each type of affective image in both mood conditions and our analyses examined differences in reward processing by mood condition, we do not believe this should have substantially affected results. Finally, we fell slightly short of target sample size, which decreased power for detecting significant effects.
Conclusions
In sum, this study did not support the hypothesis that emotion-enhanced reward processing of food is associated with self-reported negative urgency and its links with binge eating. Negative urgency may relate to emotion-enhanced reward processing in other samples of people with binge-type eating disorders (e.g., individuals with a more severe or longer duration of illness) or in other clinical populations (e.g., individuals with an alcohol or substance use disorder), or it may be that the focus should remain on other mechanisms (e.g., decreased cognitive control; Johnson et al., 2020). Nonetheless, negative urgency is a clinically significant individual-difference variable that should be incorporated into screening and intervention programs designed to prevent and treat mental-health conditions.
Supplemental Material
sj-docx-1-cpx-10.1177_21677026241267996 – Supplemental material for Testing a Reward-Processing Model of Negative Urgency in Women With and Without Binge Eating
Supplemental material, sj-docx-1-cpx-10.1177_21677026241267996 for Testing a Reward-Processing Model of Negative Urgency in Women With and Without Binge Eating by Sarah E. Racine, Vittoria Trolio, Alexia E. Miller, Adrienne Mehak, Ege Bicaker, Samantha Wilson and Stephen D. Benning in Clinical Psychological Science
Footnotes
Acknowledgements
We acknowledge and thank Jennifer Wildes for her assistance in conceptualizing the initial grant; Michael Treadway for providing us with a copy of the Effort Expenditure for Rewards Task (EEfRT) and allowing us to modify it; Léah Suissa-Rocheleau for programming the food EEfRT in E-Prime; Chloe White and Lisa Zhu for their roles in project administration; Sarah Schell, Amy Gregory, and Erin Macdonald for conducting clinical interviews for the project; and the many undergraduate research assistants who ran experimental sessions. A preprint of this article is available at ![]() .
.
Transparency
Action Editor: Kelsie T. Forbush
Editor: Jennifer L. Tackett
Author Contributions
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
