Abstract
Although substance use disorders are widely known to be influenced by myriad etiologic factors, recent research promotes the notion that liability toward addiction broadly construed can be described by a single, unitary dimension that we term “general addiction liability.” Here, we revisit the concept of general addiction liability by placing it at greater theoretical and empirical risk. To do so, we used data from two epidemiologic samples (N range = 262–8,552) and employed varied quantitative methods to examine the associations between alcohol, cannabis, tobacco, and opioid use disorders. We did not find strong evidence for general addiction liability. Nevertheless, consequence-based features (e.g., social/interpersonal harm, hazardous use) tended to form cross-substance connections. We contextualize our findings in the broader literature on addiction liability and offer several explanations for why we and others arrive at competing conclusions with respect to the robustness and nature of general addiction liability.
What causes substance addiction? This critical question has perplexed clinical scientists and nonscientists alike for hundreds of years. As it turns out, the answer to that question is “It’s complicated.” Abundant research has suggested that substance use disorders (SUDs) are moderately heritable (e.g., Deak et al., 2019; Verhulst et al., 2015), but efforts to identify their specific susceptibility genes and neurobiological mechanisms have left researchers largely empty-handed (Edenberg & McClintick, 2018; Hart & Kranzler, 2015; Salvatore et al., 2019). Resulting from this disappointment are intricate models composed of dozens of boxes and arrows (Sher, 1991) that gesture to a broad array of etiologic influences responsible for SUD, ranging from the biological to the psychological to the environmental. Indeed, over time, etiologic models have increasingly acknowledged that myriad influences play a role in the development of addiction (Bickel et al., 2019; Boness et al., 2021; Chassin et al., 2013).
And yet, recent research has promoted a competing possibility: that liability toward substance addiction broadly construed can be described in terms of a unitary dimension termed the “addiction risk factor” (Hatoum et al., 2022), or what we term “general addiction liability.” In this article, we revisit the notion of general addiction liability by adopting varied quantitative methods to examine its robustness and to probe particular aspects of SUDs that appear to contribute to increased comorbidity among them.
General Addiction Liability
Two sets of observations have led to the notion of a general addiction liability. First, others have argued that polysubstance use is the rule rather than the exception among people who use substances (e.g., Merikangas & McClair, 2012). In particular, people who use common drugs—alcohol, cannabis, nicotine, and opioids—have increased odds of using two or more drugs (Barrett et al., 2006; Everett et al., 1998; cf. Patton et al., 2007). In addition, use of more commonly used drugs (e.g., alcohol, cannabis) is prospectively associated with later use of other substances (e.g., cocaine, barbiturates, MDMA; Fergusson et al., 2008), and simultaneous use (or co-use) of alcohol with tobacco and other drugs (e.g., cocaine) tends to result in greater use of both (Barrett et al., 2006).
Second, SUD diagnoses are ostensibly moderately to highly comorbid. Although the fifth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5; American Psychiatric Association, 2013) frames its disorders as discrete (or nonoverlapping), considerable evidence suggests that a diagnosis of one SUD substantially increases risk for another. A large body of evidence raises the possibility that comorbidity among SUDs is due to shared phenotypic and genetic risk such that a common SUD dimension explains a substantial proportion of the phenotypic and heritable variation in SUDs (Agrawal et al., 2004; Hatoum et al., 2022; Kendler et al., 2007; Kendler, Jacobson, et al., 2003). Others have expanded this common SUD dimension to include conditions of behavioral and emotional undercontrol, or externalizing, such as conduct problems and antisocial personality disorder. Again, these researchers found that a common, heritable dimension explains much of the phenotypic variation in SUDs (Kendler, Prescott, et al., 2003; Krueger et al., 2002; Slutske et al., 1998). Still others have further extended these models by arguing that liability toward SUD can be modeled indirectly—by way of the same indices of behavioral and emotional undercontrol and not with explicit coverage of SUD—and that such a dimension is prospectively associated with substance use and SUD (Hicks et al., 2012; Vanyukov et al., 2009).
Most recently, Hatoum and colleagues (2022) used genomic structural equation modeling with summary statistics from genome-wide association studies (GWASs) to extract a common genetic factor for problematic alcohol use, problematic tobacco use, cannabis use disorder (CUD), and opioid use disorder (OUD; for a conceptual depiction, see Fig. 2). Each GWAS was conducted on diagnoses or summary scores of SUD-relevant phenotypes from cross-sectional data. Hatoum and colleagues concluded that shared genetic risk for these SUDs was unidimensional, with the strongest representation from OUD and CUD and weaker representation for problematic alcohol and tobacco use. The general-addiction-liability dimension explained 97% of the genetic variance in OUD, 67% in CUD, 29% in problematic alcohol use, and 11% in problematic tobacco use (estimates pulled from Hatoum et al.’s Supplemental Figure 3).
Arguments Against General Addiction Liability
The notion that a general-addiction-liability dimension causes variation in all forms of SUD is exciting; that scores on a single dimension could quantify risk for all SUDs across the life span is parsimonious and would imply that many, if not most, etiologic factors implicated in individual SUDs are shared across them. Still, there are important reasons why a general addiction liability may be untenable based on theory and data. We focus on three arguments against general addiction liability here.
Reason 1: The origins of general addiction liability are closely tied to factor analysis
Confirmatory factor analysis (CFA) is far and away the modal quantitative method used to proxy general addiction liability. For instance, Hatoum and colleagues’ (2022) general-addiction-liability dimension contained four broad indicators of SUD (one each for alcohol, cannabis, tobacco, and opioids), and on the basis of strong model-fit statistics, they concluded that common addiction liability conformed to a unidimensional structure. Nevertheless, model-fit statistics alone should not be used to conclude that a construct is unidimensional in nature (Bonifay et al., 2017).
In fact, Watts and colleagues (2021) showed that estimates of factor unidimensionality depend on how many indicators are used in the model and how well the intended construct(s) are assessed. Within the context of alcohol use disorder (AUD), they demonstrated that a single AUD factor fit the data well when one indicator per criterion (i.e., 11 items) was modeled, but fit quickly depreciated as multiple items per criterion were included in the model. Furthermore, multidimensional accounts of AUD were increasingly supported with more thorough measurement. All told, relying exclusively on model fit and factor analysis to inform theoretical models is limiting at best, and it can preclude detection of structures that are far more compatible with theory.
Reason 2: SUDs are phenotypically and etiologically heterogeneous
SUD diagnoses are widely regarded as unidimensional, once again because of empirical observations that single-factor solutions containing all SUD criteria (e.g., AUD) within a given substance (e.g., alcohol) fit the data well (Hasin et al., 2013). Researchers either implicitly or explicitly take these observations to mean that SUD criteria share a common cause or set of causes, further justifying the study of SUDs at the level of diagnosis and not criteria or symptoms.
Nevertheless, logically following from the findings of Watts and colleagues (2021) is that SUDs might be better conceptualized as multidimensional. In particular, these investigators found that neuroadaptations resulting from heavy and chronic substance use (i.e., tolerance, withdrawal) pulled away from each other and away from the consequence-based criteria (e.g., hazardous use, role interference; Martin et al., 2014). Their final model of AUD contained three dimensions: tolerance, withdrawal, and loss of control (see also Muthén et al., 1993). Moreover, Kendler and colleagues (2012) found that genetic variance in alcohol dependence could be partitioned into these same three dimensions. Not only are these multidimensional accounts of AUD consistent with observations for other substances, in which developing tolerance to tobacco is dissociable from withdrawal (e.g., Perkins, 2002), they are far more compatible with etiologically informed models of addiction based on preclinical animal models (e.g., Berridge & Robinson, 2016; Koob & Le Moal, 2001).
Additional, albeit more indirect, support for multidimensionality within SUDs pertains to the fact that AUD criteria have different comorbidity patterns. Withdrawal has been most closely tied to internalizing (Boschloo et al., 2015; Koob & Le Moal, 2001; Watts, Watson, et al., 2023), recurrent use in hazardous situations to higher degrees of externalizing and lower degrees of internalizing (Martin et al., 2011; McDowell et al., 2019; Watts, Watson, et al., 2023), continued use despite social/interpersonal harm or physical/psychological harm and role interference to higher degrees of externalizing and internalizing (Hoffman et al., 2019; McDowell et al., 2019; Quinn & Harden, 2013; Watts, Watson, et al., 2023), and tolerance to no other forms of psychopathology (Kendler et al., 2012; McDowell et al., 2019; Watts, Watson, et al., 2023). If widely replicated, these findings raise substantial concerns regarding unitary conceptualizations of SUDs (Hasin et al., 2013) and general addiction liability (Hatoum et al., 2022).
Reason 3: Modern models of addiction describe its development as dynamic
Modern models of addiction describe addiction as a staged, developmental process whereby certain symptoms elicit others and so on. According to Koob and Le Moal’s (2001) allostatic theory of addiction, people develop a tolerance to alcohol as they consume an excess amount of it, requiring more and more to achieve the same effect. Over time, tolerance is subserved by opponent processes that intensify over repeated drug exposures, and it is thought to alter one’s hedonic set point, resulting in withdrawal and negative affect that is either caused by allostasis (i.e., a shifting of one’s priorities to alcohol and away from other reinforcers and anhedonia toward non-drug-related reinforcers) or is a direct manifestation of withdrawal (i.e., sympathetic-nervous-system arousal, irritability). As one withdraws from a drug, one may experience a preoccupation with it (otherwise known as craving), and the resulting negative affect is thought to result in a lapse in control over abstention, reverting to consumption. Other models of addiction implicate additional mechanisms, such as habit and incentive sensitization (e.g., Berridge & Robinson, 2016). Taken together, modern models of addiction describe addiction as the causal interactions among multiple etiologically distinct phenomena. That is, criteria within a given SUD are not thought to share a set of etiologic influences or arise from a unitary latent entity.
Present Study
Factor analysis’s hegemonic influence on SUD classification (and modern quantitative classification efforts more generally; Haeffel et al., 2022) has encouraged, even guaranteed, premature conclusions regarding the nature of addiction and how it is structured across and within people. Indeed, most factor analyses of disorders have relied on data from large samples that were not designed to explore the structure of diagnosis. To adequately test multidimensional structures, it is essential to model input variables that permit statistical identification of distinct factors (Watts et al., 2021).
Compounding the problem, exclusive focus on model-fit statistics—and not theory—has led to a strong precedent for treating individual SUDs as unidimensional (Hasin et al., 2013), which, in turn, has led to the use of broad diagnoses to further model and argue that liability toward addiction broadly construed is unidimensional (Hatoum et al., 2022). Nevertheless, SUDs are increasingly described as heterogeneous and composed of features with distinct etiologies (Boness et al., 2021; Kendler et al., 2012; Muthén et al., 1993; Watts et al., 2021; Watts, Latzman, et al., 2023; Watts, Watson, et al., 2023). If individual SUDs have distinct etiologies and symptoms within SUDs cause or elicit one another, how do SUDs share a unitary etiology? This question implores the field to search beneath the level of diagnosis if it is interested in detecting etiologic factors relevant to addiction (Strain, 2021; Watts, Latzman, et al., 2023).
In this study, we revisit general addiction liability, this time modeling it with three key differences from Hatoum and colleagues’ (2022) approach. First, we modeled diagnostic criteria as opposed to disorders. Second, we evaluated the influence of factor analysis on the general-addiction-liability literature by approaching our study with the spirit of dual phenomenology. “Dual phenomenology” refers to the requirement that an incoming nuclear attack on the United States must be confirmed by two sources, radar and satellite, before engaging in retaliatory action to prevent a disastrous false alarm. Although quibbles over factor analysis have yet to cause a nuclear missile strike, at least to our knowledge, we extend the logic of dual phenomenology to psychiatric classification: If general addiction liability is a robust phenomenon, evidence for it ought to arise regardless of granularity of input (e.g., criteria, disorders) and across diverse methods, including but not limited to factor analysis. Here, we relied on three families of methods with different assumptions and unique abilities to inform the nature of general addiction liability—factor analysis, network analysis, and two-mode block modeling.
Third, we analyzed only people who report substance use as opposed to the entire sample of participants. In modeling the interrelations among SUD symptoms and cross-SUD associations, it is conceptually confusing to include in the analysis people who do not consume the substance(s) of interest (Buckland, 2008). People cannot develop tolerance to or withdrawal from a particular substance if they abstain from using it, nor can they experience downstream consequences of substance use without using the substance. Much as celibate individuals cannot get pregnant (Waldron et al., 2007), abstainers cannot develop addiction. Although SUD diagnoses do not explicitly incorporate a Criterion A (i.e., a required feature, such as experience of trauma in posttraumatic stress disorder), implicit in SUD diagnosis is that some minimum level of substance consumption is required to achieve a diagnosis. Indeed, most epidemiologic studies have adopted this assumption by gating out (or “skipping out”) of the SUD module if substance use was not endorsed (e.g., Grant et al., 2003) or was below some minimum threshold (e.g., Bucholz et al., 1994).
Transparency and Openness
This study was not preregistered and involved completely exploratory secondary analysis of existing data rather than new data collection. The National Epidemiologic Survey on Alcohol and Related Conditions (NESARC) data are available from the National Institute on Alcohol Abuse and Alcoholism on request (https://www.niaaa.nih.gov/research/nesarc-iii/nesarc-iii-data-access). Upon acquisition of the data, our code facilitates completely reproducible findings. To further facilitate reproducibility, we have included correlation matrices of all indicators included in our analysis. Our OSF project page contains all relevant documentation (https://osf.io/xdsnp/). Institutional review board approval (carried out in accordance with provisions of the World Medical Association Declaration of Helsinki) was obtained before conducting this study.
Method
Data
We used data from two large, nationally representative samples collected through the NESARC. NESARC surveyed persons 18 years or older living in the United States at the time of data collection. NESARC Wave 1 (N = 43,093) contains data collected in 2001–2002 (Grant et al., 2003; 57% female; 57% White, 19% Black, 2% American Indian/Native American, 3% Asian, 19% Hispanic or Latino). NESARC-III (N = 28,724) contains an independent sample of people in the United States collected in 2012–2013 (Grant et al., 2016; 56% female; 53% White, 21% Black, 1% American Indian/Native American, 5% Asian, 19% Hispanic or Latino). Because both samples are nationally representative, their median household income and education estimates mirror the U.S. population at the time of data collection.
Participants
Across both data sets, we used subsets of participants who reported using two or more substances in the past year. Rates of substance use for drugs other than alcohol, cannabis, tobacco, and opioids (e.g., tranquilizers, heroin) were too low to analyze here, so we focus on alcohol, cannabis, tobacco, and opioids. This analytic decision also conforms with the four phentoypes analyzed by Hatoum and colleagues (2022).
In NESARC Wave 1, rates of alcohol, tobacco, cannabis, and opioid use in the past year were 62.6%, 25.8%, 3.7%, and 1.6%, respectively. In NESARC-III, rates of alcohol, tobacco, cannabis, and opioid use in the past year were 70.5%, 28.0%, 10.3%, 4.7%, respectively. In contrast with frequently observed statements that polysubstance use is common, rates of polysubstance use in the epidemiologic data used here were relatively low (see Table S1 in the Supplemental Material available online). In NESARC Wave 1, 18.7% of participants (n = 8,058) used two substances in the past year, 2.4% used three (n = 1,034), and 0.5% used four (n = 215). In NESARC-III, 21.4% of participants (n = 6,146) used two substances in the past year, 5.9% used three (n = 1,694), and 1.3% used four (n = 373).
Thus, we restricted all analyses to the subsets of participants who used each pairing of substances in the past year: alcohol–cannabis (NESARC Wave 1: n = 1,510; NESARC-III: n = 2,745), alcohol–tobacco (NESARC Wave 1: n = 8,552; NESARC-III: n = 6,611), alcohol–opioids (NESARC Wave 1: n = 586; NESARC-III: n = 1,098), cannabis-tobacco (NESARC Wave 1: n = 520; NESARC-III: n = 1,808), cannabis–opioids (NESARC Wave 1: n = 262; NESARC-III: n = 1,059), tobacco-opioids (NESARC Wave 1: n = 410; NESARC-III: n = 718). Modeling 44 diagnostic criteria (4 SUDs × 11 criteria) among participants who engaged in all four types of past-year substance use (NESARC Wave 1: n = 215; NESARC-III: n = 373) was considered intractable. Still, if general addiction liability influences all SUDs, evidence for it ought to emerge among individuals who use two or more substances and among pairs of substances.
Diagnostic assessment
NESARC investigators used the Alcohol Use Disorder and Associated Disabilities Interview Schedule–Fourth Edition (AUDADIS-IV; Grant et al., 2003) in NESARC Wave 1 and the fifth edition (AUDADIS-5; Grant et al., 2015) in NESARC-III. Both forms of the AUDADIS are structured interviews that assess SUDs and other psychological disorders. They differ in terms of whether they assessed disorders and diagnostic criteria from the fourth (DSM-IV; American Psychiatric Association, 1994) or fifth edition (DSM-5; American Psychiatric Association, 2013) of the DSM, respectively. Thus, NESARC Wave 1 did not assess craving because it was added to SUD in DSM-5 (American Psychiatric Association, 2013).1
In NESARC Wave 1, we modeled all past-year DSM-IV (American Psychiatric Association, 1994) SUD criteria for alcohol, cannabis, and opioids with the exception of legal problems. We excluded legal problems given that it was removed from DSM-5 (American Psychiatric Association, 2013) based on evidence that it functions differently across gender, race, and ethnicity (Saha et al., 2006). NESARC Wave 1 contains a more limited assessment of tobacco-related SUD criteria, that is, DSM-IV (American Psychiatric Association, 1994) nicotine dependence criteria only, because DSM-IV (American Psychiatric Association, 1994) abuse criteria were not thought to apply to nicotine (Hughes et al., 2006, 2011). These criteria include tolerance, attempting to cut down/quit, using for large amounts/longer than intended, giving up activities to use, continued use despite physical/psychological harm, and withdrawal. In NESARC-III, we used the same SUD indicators as in NESARC Wave 1 but also included craving for alcohol, cannabis, and opioids.
Because there are multiple items assessing each criterion in the AUDADIS, we counted a criterion as present if one symptom assessing each criterion was endorsed with one exception. We counted alcohol withdrawal as present if participants endorsed three withdrawal symptoms or two symptoms and one relief/avoidance from withdrawal item to prevent withdrawal from functioning as a hangover index (Boness et al., 2016). For other substances, we followed DSM thresholds for the number of required symptoms to meet diagnostic criteria for withdrawal. Finally, withdrawal syndromes differ across substances, so we constructed withdrawal criteria by adhering to each substance’s specific criteria as outlined in the DSM (see Table S2 in the Supplemental Material). For instance, muscle aches during periods of abstinence indicate opioid withdrawal but not alcohol, cannabis, or tobacco withdrawal, so we included it only in our construction of opioid withdrawal.
Analytic Strategy
Approach
Figure 1 depicts our stepwise analytic approach beginning with exploratory factor analysis (EFA) and ending with two-mode block modeling along with what one should expect to see from results if general addiction liability is a robust phenomenon.
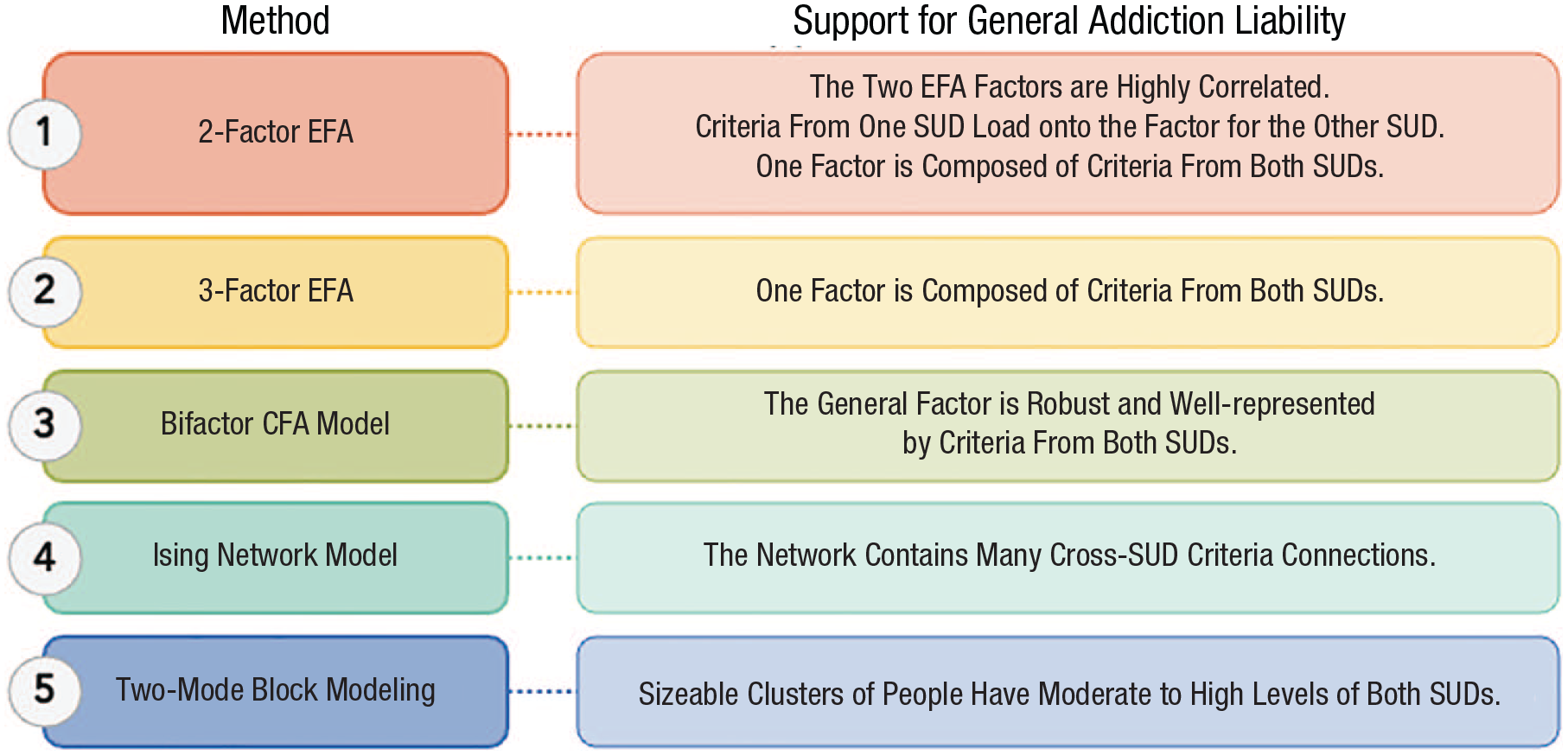
Analytic plan and what evidence would support general addiction liability. CFA = confirmatory factor analysis; EFA = exploratory factor analysis; SUD = substance use disorder.
Step 1: two-factor EFA
We began with EFA and extracted two-factor solutions to examine the degree of covariation among what we predicted would be one factor for each SUD (see Table S3 in the Supplemental Material). Because we analyzed the subsets of participants that endorsed past-year use of pairs of substances (i.e., alcohol–cannabis, cannabis–opioids), we extracted two-factor EFA solutions for each subset of participants, resulting in six two-factor EFAs per sample. To do so, we extracted EFAs on the tetrachoric correlation matrices of SUD criteria using an oblimin factor rotation and maximum likelihood factor extraction. All subsequent analytic steps followed suit.
In the two-factor EFA solutions, we interpreted support for general addiction liability because (a) the two factors are highly correlated, (b) criteria from one SUD load onto the factor for the other SUD, and/or (c) that one factor is composed of criteria from both SUDs.
Step 2: three-factor EFA
We used EFA to extract three factors (see Table S4 in the Supplemental Material). We did not anticipate that cross-SUD factors would form in Step 1 because we viewed it as much more likely that separate SUD factors would. That said, extracting three factors provides the opportunity to observe whether a cross-SUD factor would emerge when we forced an additional factor out of the data. If a cross-SUD factor formed, we interpreted that as evidence of general addiction liability. We followed the same analytic procedure as Step 1.
Step 3: bifactor CFA
We used CFA to extract a bifactor model with a general factor (i.e., general addiction liability) onto which all SUD criteria across the two SUDs were allowed to load and two specific factors, one for each SUD (e.g., AUD, CUD; see Table S5 in the Supplemental Material). If the general factor in the bifactor model was robust and well represented by criteria from both SUDs (i.e., it explains a substantial portion of variance in both sets of SUD criteria, with loadings > .40), we interpreted that finding as supportive of general addiction liability.
For bifactor CFA models, we used the lavaan R package (Rosseel, 2012) with the WLSMV estimator, which is most appropriate with categorical indicators, and we freely estimated all factor loadings. To identify the model, we fixed the factor means to 0 and their variances to 1. We further constrained correlations among all factors in the bifactor model to 0, as with standard applications.
Step 4: network analysis
In contrast with factor analysis, which can model broad SUD-like dimensions, network analysis affords examinations of criteria-by-criteria connections. In the context of general addiction liability, it might be that a subset of cross-SUD criteria forms dense connections, suggesting that general addiction liability is driven by a narrower set of SUD criteria. Such a possibility is somewhat testable with three-factor EFA models, but EFA does not facilitate examination of criteria-by-criteria connections. If networks contained many cross-SUD criteria connections, we interpreted that finding as evidence of general addiction liability.
Psychometric-network models comprise nodes (diagnostic criteria) and edges (estimated relations among diagnostic criteria). Using the mgm R package (Haslbeck & Waldorp, 2018), we estimated regularized Ising models that produce unique, conditional dependence associations that are similar to partial correlations. That is, the edge between two nodes reflects the association between the nodes after covarying all other nodes in the network. Because regularized Ising models estimate many parameters, they often rely on regularization with the least absolute shrinkage and selection operator (LASSO; Tibshirani, 1996) to avoid false positives. The LASSO shrinks all edge weights toward zero and sets small weights to exactly zero. We set the tuning parameter to zero for our main analyses and conducted sensitivity analyses across six other levels: 2.5, 5, 10, 15, 20, 25. 2 We considered nonzero edges that replicated across samples (i.e., NESARC Wave 1, NESARC-III; see Tables S7–S12 in the Supplemental Material).
Step 5: two-mode block modeling
Finally, although comorbidity is inherently a within-subjects problem, researchers routinely apply between-subjects or individual-differences models to inform comorbidity (e.g., factor models). We sought to examine whether evidence of general addiction liability arises within people, meaning that there are robust clusters of people who meet criteria for multiple SUDs in the population. To that end, we used two-mode block modeling, which simultaneously extracts K clusters of participants and L clusters of variables such that the submatrices formed by clusters of participants and criteria are either complete (all ones) or null (all zeros) to the extent possible (Brusco et al., 2021; Doreian et al., 2005). Thus, “two-mode” refers to the fact that psychopathology data can be partitioned into clusters of (a) criteria and (b) people. In the main text, we largely focus on the latter results, in part because the former largely mirrored the findings of our EFAs (for a brief summary of these findings, see Table 1; for all results, see Tables S13–S15 in the Supplemental Material).
Summary of Evidence for General Addiction Liability Across Methods
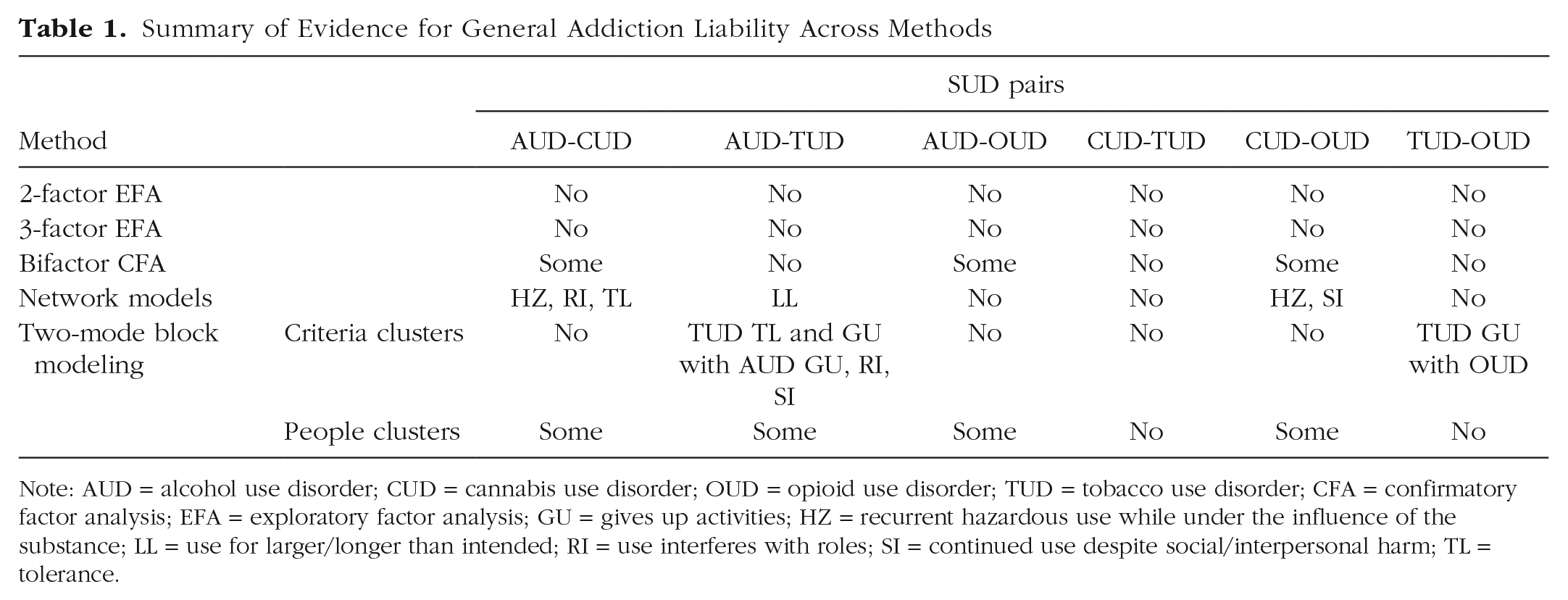
Note: AUD = alcohol use disorder; CUD = cannabis use disorder; OUD = opioid use disorder; TUD = tobacco use disorder; CFA = confirmatory factor analysis; EFA = exploratory factor analysis; GU = gives up activities; HZ = recurrent hazardous use while under the influence of the substance; LL = use for larger/longer than intended; RI = use interferes with roles; SI = continued use despite social/interpersonal harm; TL = tolerance.
Results
Replicating the typical examination of general addiction liability
To mimic the approach used by Hatoum and colleagues (2022), we modeled a general-addiction-liability factor in the entire samples of participants (past-year abstainers included) in NESARC Wave 1 (N = 43,093) and NESARC-III (N = 28,724). As with Hatoum and colleagues (2022), we modeled four SUD indicators (AUD, CUD, tobacco use disorder [TUD], and OUD diagnoses) in a one-factor CFA model (for a depiction of the model, see Fig. 2). Across both samples, these one-factor models fit the data well, and all disorders loaded substantially onto the general-addiction-liability factors that explained a substantial amount of the phenotypic variance in SUDs (NESARC Wave 1: 81%, NESARC-III: 76%). The strength of the loadings sometimes varied across disorders and samples. Both general-addiction-liability factors correlated perfectly (r = 1.0) with a count variable of the number of past-year SUD diagnoses.
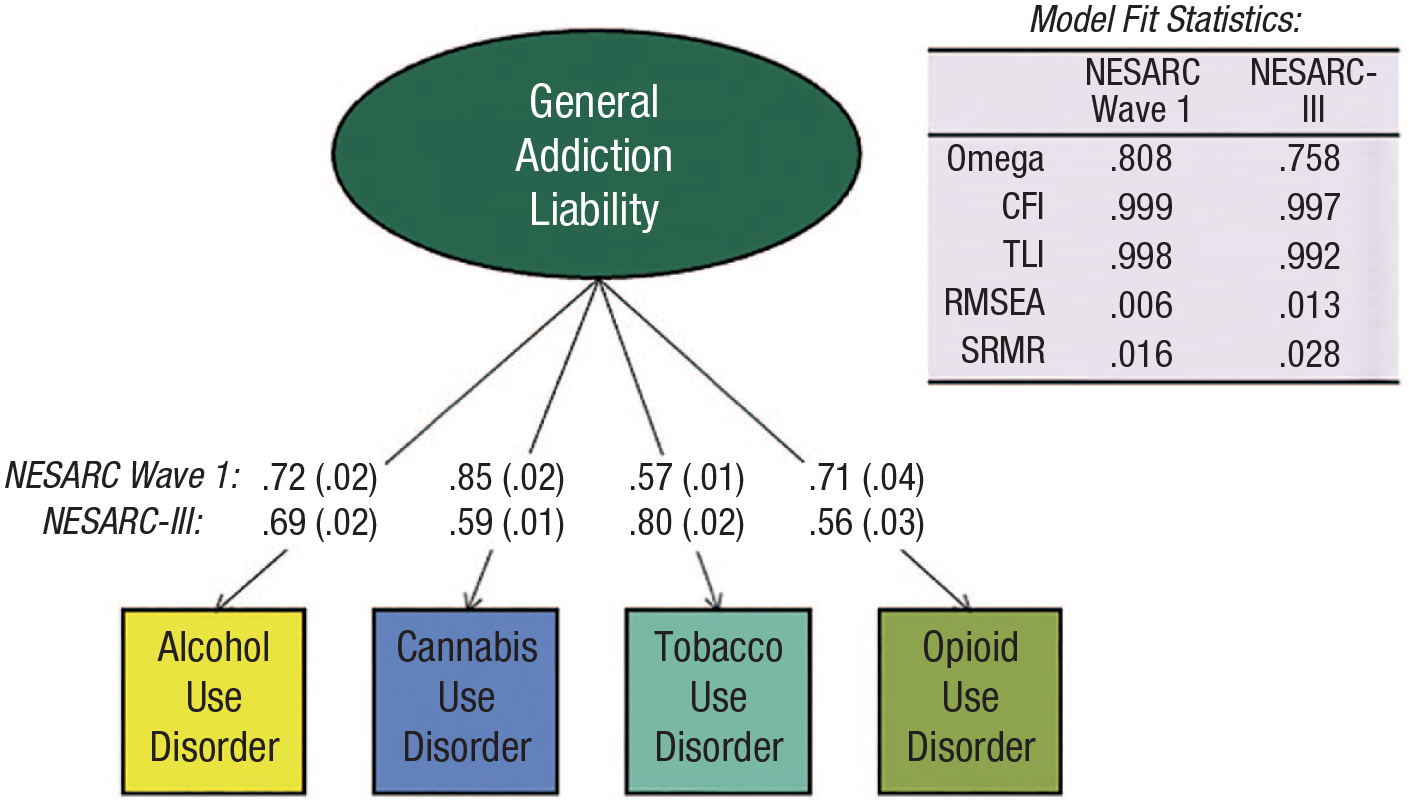
A replication of Hatoum and colleagues’ (2022) examination of general addiction liability. We present standardized factor loadings with their standard errors in parentheses.
On the basis of the strong fit of these models, many researchers might stop there and conclude that some general-addiction-liability dimension exists in nature (Hatoum et al., 2022). Nevertheless, as we mentioned earlier, strong model fit should never be taken as strong evidence of an underlying, causal latent variable (Bonifay et al., 2017; van Bork et al., 2017), particularly in the context of crude measurement (i.e., modeling four diagnoses; Watts et al., 2021). Instead, well-fitting latent variables should be further vetted with riskier tests. The remainder of our analyses involves riskier tests, relying on diagnostic criteria as opposed to disorders, varied quantitative methods, and subsets of polysubstance users.
A reanalysis of general addiction liability
Table 1 contains a general summary of our analyses.
Step 1: two-factor EFA solutions
We extracted two-factor EFAs to examine whether SUD criteria across two different SUDs (e.g., AUD-CUD) formed a factor or whether SUD criteria from one SUD loaded onto a factor reflecting another SUD. Two-factor EFAs revealed distinct factors for each SUD rather than factors that reflect a mix of cross-substance SUD liability (see Table S3 in the Supplemental Material). Correlations between EFA factors were low to moderate (rs ranged from .20 [CUD-OUD, NESARC-III] to .44 [CUD-TUD, NESARC-III]), and there was no evidence of criteria from one SUD (e.g., withdrawal from AUD) loading substantially (standardized λ > .25) onto a factor reflecting another SUD (e.g., CUD) that replicated across the two samples. All told, only 4% to 19% of the variance in one SUD was shared with the other.
Step 2: three-factor EFA solutions
We extracted three-factor EFAs to allow for the possibility that each set of SUD criteria would form its own factor but that a third factor might reflect “general” SUD liability with representation from criteria across both SUDs. In general, these three-factor solutions were overextracted, and the third factors were weak, such that they were not strongly indicated by many items (see Table S4 in the Supplemental Material). The third factors never contained positive loadings from criteria for both SUDs that replicated across samples.
Step 3: bifactor CFA models
We extracted bifactor models to force general factors across pairs of SUDs and examine their robustness, which varied depending on the model and sample (Fig. 3; see Table S5 in the Supplemental Material). General factors for AUD-TUD, CUD-TUD, and TUD-OUD were weak. AUD-TUD general factors were generally underrepresented by TUD (NESARC Wave 1: Mdn λ = .24; NESARC-III: Mdn λ = .33). The CUD-TUD general factor was underrepresented by TUD in NESARC Wave 1 (median λ = .27), and it was underrepresented by CUD in NESARC-III (median λ = .40). Finally, the TUD-OUD general factors tended to be best represented by TUD and were not well represented by OUD, but TUD indicators did not consistently load onto the general factors, either (NESARC Wave 1, TUD: Mdn λ = .42; NESARC-III, TUD: Mdn λ = .63; NESARC Wave 1, OUD: Mdn λ = .20; NESARC-III, OUD: Mdn λ = .29).
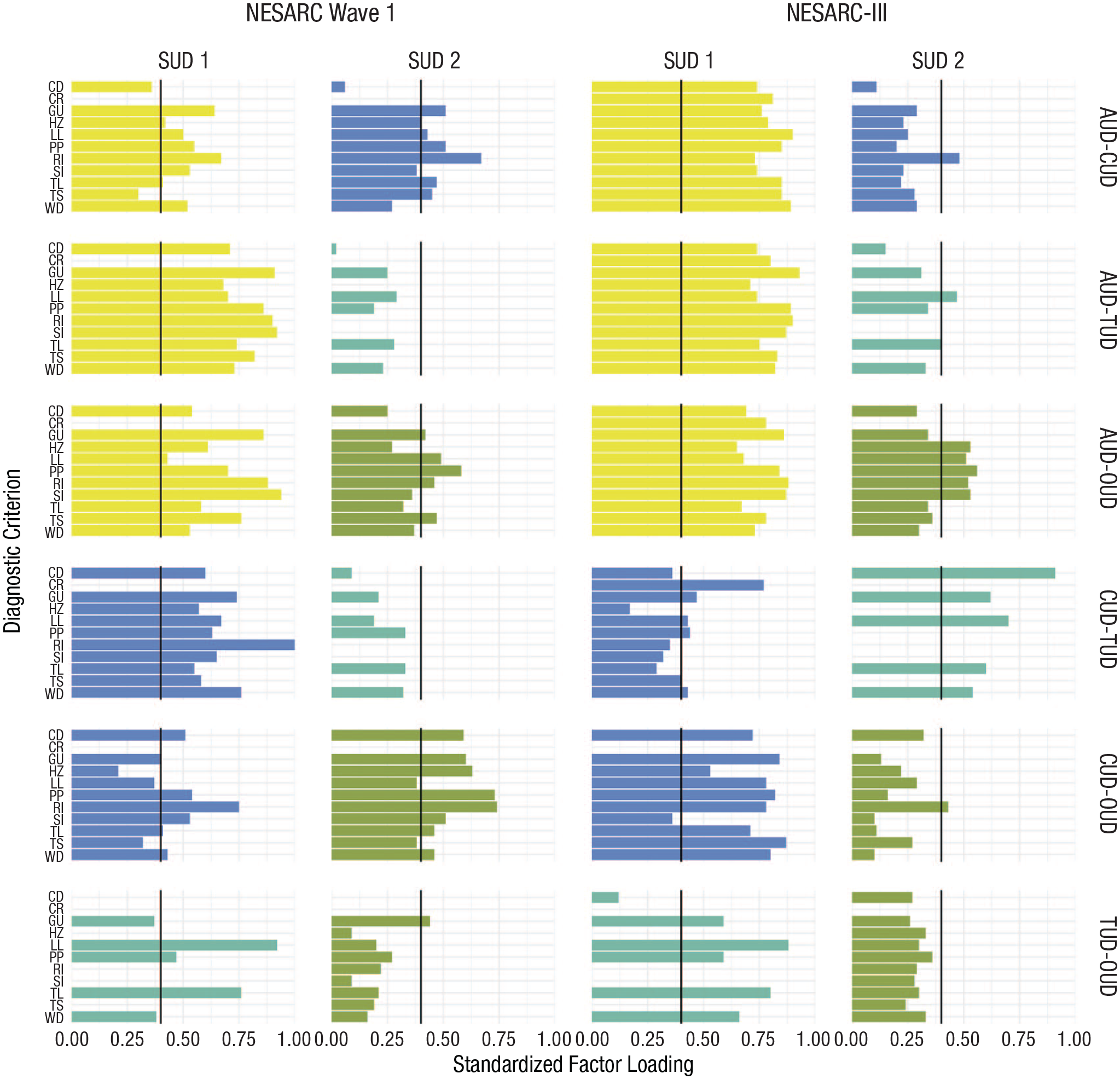
Standardized factor loadings for confirmatory bifactor models. AUD = alcohol use disorder; CUD = cannabis use disorder; OUD = opioid use disorder; TUD = tobacco use disorder; CD = desire or failed attempts to cut down/quit; CR = craving; GU = gives up activities; LL = use for larger/longer than intended; HZ = recurrent hazardous use while under the influence of the substance; PP = continued use despite physical/psychological harm; RI = use interferes with roles; SI = continued use despite social/interpersonal harm; TL = tolerance; TS = time spent obtaining, using, and getting over the effects of the substance; WD = withdrawal. SUD 1 refers to the first SUD listed in each figure, and SUD 2 refers to the second. For instance, in the top right figure, for National Epidemiologic Survey on Alcohol and Related Conditions-III, SUD 1 corresponds to AUD, and SUD 2 corresponds to CUD.
General factors for AUD-CUD, AUD-OUD, and CUD-OUD provided some support for general addiction liability. For AUD-CUD, in NESARC Wave 1, the general factor was relatively equally represented by all indicators, but the loadings were only moderate (AUD: Mdn λ = .51; CUD: Mdn λ = .44; ω = .48); in NESARC-III, the general factor was underrepresented by CUD (Mdn λ = .24). For AUD-OUD, general factors were better represented by AUD (NESARC Wave 1: Mdn λ = .66; NESARC-III: Mdn λ = .78) than they were OUD (NESARC Wave 1: Mdn λ = .39; NESARC-III: Mdn λ = .44). The AUD-OUD general factor was stronger in NESARC-III (ω = .93) than it was in NESARC Wave 1 (ω = .59), and continued use despite physical or psychological problems from OUD loaded strongly onto the general factor across both samples, whereas other OUD criteria did not load consistently strongly onto it. For CUD-OUD, the general factor was represented by all indicators but in a relatively weak manner in NESARC Wave 1 (CUD: Mdn λ = .42; OUD: Mdn λ = .55), whereas OUD indicators did not load strongly on the general factor in NESARC-III (Mdn λ = .19).
Step 4: Ising network models
We estimated psychometric network models to examine criteria-by-criteria connections (edges) across SUDs. In NESARC Wave 1, two networks (CUD-OUD, TUD-OUD) had estimation problems because of having too few observations in binomial or multinomial tables. Thus, we focus our interpretation of these two networks on the results from NESARC-III (for the results for both solutions, see Tables S10 and S11 in the Supplemental Material). Otherwise, we focus on cross-SUD edges that replicated across both samples. The networks depicted in Figure 4 reflect the average of edge weights across the methods and samples, with edge-weight estimates weighted by sample size.
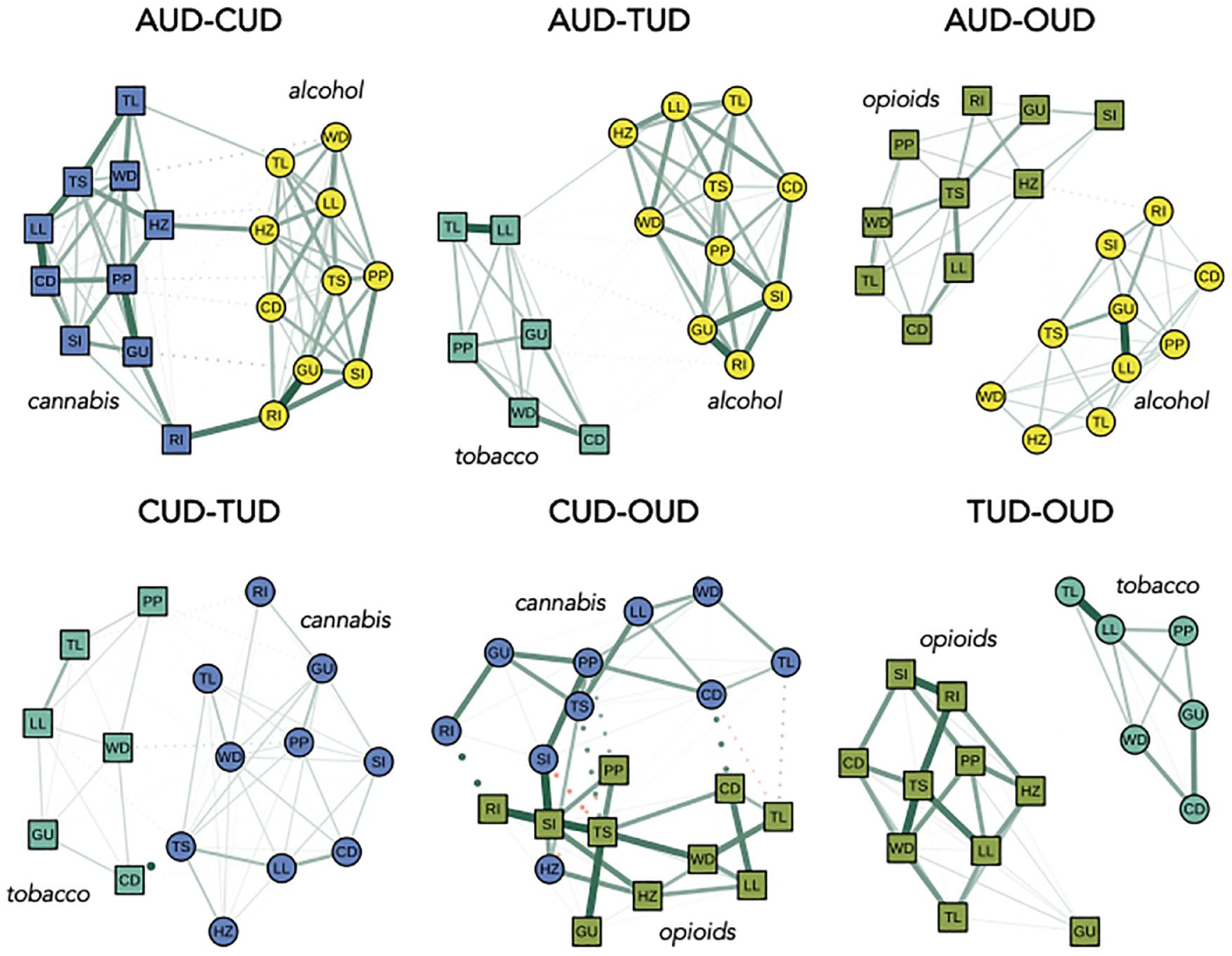
Averaged Ising networks across samples and methods. AUD = alcohol use disorder; CUD = cannabis use disorder; OUD = opioid use disorder; TUD = tobacco use disorder; CD = desire or failed attempts to cut down/quit; CR = craving; GU = gives up activities; LL = use for larger/longer than intended; HZ = recurrent hazardous use while under the influence of the substance; PP = continued use despite physical/psychological harm; RI = use interferes with roles; SI = continued use despite social/interpersonal harm; TL = tolerance; TS = time spent obtaining, using, and getting over the effects of the substance; WD = withdrawal. Here, we present averaged networks in which we averaged the edge weight matrices across samples and methods while weighting all edges by sample size. Nonzero edges that replicated across sample (i.e., National Epidemiologic Survey on Alcohol and Related Conditions Wave 1, National Epidemiologic Survey on Alcohol and Related Conditions-III) are depicted as solid lines. Nonzero edges that did not replicate are depicted as dotted lines. Edges with positive weights are depicted in green, and negative weights are depicted in red. All edges are summarized in Tables S7 through S12 in the Supplemental Material available online.
Networks between pairs of SUD criteria sets revealed dense connections within SUD but weak connections across SUDs, at least on balance. That is, there were relatively few instances of criteria connections across SUDs (Fig. 4). Specifically, of all possible cross-SUD connections (e.g., tolerance from AUD with hazardous use from CUD), networks contained anywhere from 0% to 25% of edges, indicating weak support for general addiction liability. For AUD-OUD, CUD-TUD, and TUD-OUD networks, no cross-SUD edges replicated. In the case of all other networks—AUD-CUD, AUD-TUD, CUD-OUD—each pair of nodes connected by the edge were for the same symptom across the SUDs. For the AUD-CUD network, three cross-SUD edges emerged: tolerance, role interference, and hazardous use. For the AUD-TUD network, one cross-SUD edge emerged: using larger amounts or for longer than intended. Finally, for the CUD-OUD network, two cross-SUD edges emerged: hazardous use and social/interpersonal harm.
Step 5: two-mode block modeling
We used two-mode block modeling to simultaneously cluster SUD criteria and participants, which more directly informs comorbidity. With respect to SUD criteria clusters, as with the three-factor EFA solutions, there were no cross-SUD clusters for the AUD-CUD, AUD-OUD, CUD-TUD, and CUD-OUD models (see Table S14 in the Supplemental Material). In contrast, in the AUD-TUD models, tolerance and giving up activities to consume tobacco from TUD formed a cluster with giving up activities to drink, role interference, and social/interpersonal harm from AUD. In addition, giving up activities to consume tobacco from TUD formed a cluster with all OUD criteria.
With respect to clusters of participants (see Table S15 in the Supplemental Material), there was no evidence of general addiction liability for two sets of SUD pairs: TUD-OUD, CUD-OUD. That is, no clusters of participants with moderate or high degrees of (a) TUD and OUD and (b) CUD and OUD emerged. For instance, in NESARC Wave 1, there were five clusters of tobacco and opioid users: (a) no TUD and OUD (6% of the subsample), (b) moderate TUD and no OUD (22%), (c) moderately high TUD and no OUD (31%), (d) moderately high TUD and high OUD (4%), and (e) high TUD and no OUD (37%).
In contrast, there was some support for general addiction liability for the other pairings of SUDs. Specifically, there were clusters of participants with high degrees of (a) AUD-CUD, (b) AUD-TUD, and (c) AUD-OUD. Nevertheless, these cases constituted a minority of polysubstance users. For AUD-CUD, these clusters contained 6% and 15% of the subsample of alcohol and cannabis users (for NESARC-III and NESARC Wave 1, respectively), which is 1.01% and 0.74% of the entire NESARC sample. Likewise, the clusters of alcohol and cannabis users with high degrees of AUD and CUD comprised 4% of the subsample and between 0.12% and 0.25% of the entire NESARC samples, respectively, whereas the clusters of alcohol and opioid users comprised 3% of the subsample and between 0.03% and 0.13% of the NESARC samples, respectively. For TUD-CUD, NESARC Wave 1 contained a cluster of tobacco and cannabis users with high degrees of TUD and CUD (7% of the subsample, 0.6% of NESARC), but NESARC-III did not.
Discussion
The concept of a liability to experience addiction broadly construed is provocative, intriguing, and worthy of exploration. Using data from two large epidemiologic samples, we revisited the notion of general addiction liability by placing it at greater theoretical and empirical risk. To do so, we extended Hatoum and colleagues’ (2022) analytic approach in three critical ways. First, considering evidence that SUDs are multidimensional as opposed to unidimensional (Kendler et al., 2012; Muthén et al., 1993; Watts et al., 2021; cf. Hasin et al., 2013), we modeled diagnostic criteria rather than disorders. Second, in the spirit of establishing dual phenomenology, we exploited varied quantitative methods instead of relying exclusively on CFA. Third, we focused solely on the polysubstance users, as opposed to abstainers and polysubstance users (Watts, Brusco, et al., 2023).
Synthesis of findings
In short, we did not find strong evidence of a general-addiction-liability dimension. Across the methods we used, some revealed tepid support for a general-addiction-liability dimension, although no methods achieved consensus in terms of either the degree of support for general addiction liability or the nature of it. That is, particular sources of support for general addiction liability, where indicated, did not replicate across methods and/or samples. For instance, when we focused on the relations among TUD and OUD criteria, two-mode block modeling revealed that the criterion probing giving up activities to smoke assorted with OUD broadly construed, but giving up activities to smoke did not have appreciable connections with any nodes in the network models, and it did not assort with OUD in any other factor models.
Still, where there were inklings of support for general addiction liability, they warrant further discussion here. Of the methods we used, bifactor CFA models tended to reveal the strongest support for general addiction liability. Nevertheless, where bifactor models were apparently supportive of a general addiction liability, their general factors were weak and diffuse at best (e.g., AUD-CUD in NESARC Wave 1, CUD-OUD in NESARC Wave 1); some general factors were represented by all SUD criteria, but the magnitudes of the factor loadings were far lower than expected for strong general factors (Revelle & Wilt, 2013; Watts et al., 2019). Moreover, these general factors tended not to replicate in an independent data set (e.g., NESARC-III).
One possible explanation for a lack of strong general addiction factors might be that general addiction liability is narrower and not general to all SUD criteria, necessitating the use of methods that model cross-SUD criteria connections directly. Along these lines, network models did reveal some limited support for general addiction liability, whereby the same criterion across SUDs (e.g., AUD-CUD) formed a strong edge: We found that the hazardous-use criteria for AUD-CUD and CUD-OUD had strong edges, as did the role interference criteria in AUD-CUD, continued use despite social/interpersonal harm in CUD-OUD, and tolerance in AUD-CUD. There are several possible interpretations of these findings.
Most directly, it is possible that nearly identical wording in the criteria across SUD types is causing the same criterion to correlate strongly across substances, producing a method effect (Kam, 2018). If this interpretation were strongly supported by data, we would expect to find a larger number of edges between the same SUD criterion across substances. Instead, we observed that only a limited subset of criteria (i.e., hazardous use, role interference, continued use despite social/interpersonal harm, tolerance) tended to form strong edges across substances.
Furthermore, these network edges do make some theoretical and empirical sense. First, tolerance to alcohol formed a strong edge with tolerance to cannabis in the network models. That tolerance, a substance-specific neuroadaptation that develops in response to repeated, high-dose exposures, would be associated across drug classes is slightly counterintuitive. But acute cross-tolerance, where tolerance to the effects of one drug produces tolerance to another, occurs for certain pairs of drugs. In particular, there is evidence in nonhuman animals that alcohol cross-tolerates with cannabis (Pava & Woodward, 2012) and nicotine (Funk et al., 2006). Although we did not find a strong connection between tolerance in AUD-TUD, we did find that using larger amounts of a drug or for longer periods of time than intended (i.e., larger/longer) formed a strong edge. This criterion is conceptually overlapping with tolerance; the development of tolerance facilitates consumption of larger and larger amounts of alcohol over time. All told, our findings support either the notion of acute cross-tolerance or heavy consumption for certain pairings of substances.
Setting aside tolerance and larger/longer, most of the criteria that emerged as having strong cross-SUD edges—hazardous use, role interference, and continued use despite social/interpersonal harm—reflect consequences, or sequelae, of substance use. That is, substance use that leads to risky behaviors (e.g., driving, operating heavy machinery), an inability to perform or complete routine tasks (i.e., going to work, taking care of one’s family), or social/interpersonal harm arguably captures negative consequences associated with substance use.
If the consequence-based cross-SUD criteria edges are not reflective of a method effect and indeed reflect some shared, substantive mechanism, our findings raise another important question regarding SUD diagnosis and comorbidity. Consider a common operationalization of hazardous use: intoxicated driving. With intoxicated driving, it is unclear whether use of one drug causes the disinhibited decision-making that might lead to drinking and driving, the other, or both. Use of alcohol appears associated with increased disinhibition while under the influence (Allen et al., 2022; Giancola, 2000; Winograd et al., 2014), particularly for some people (Giancola, 2002), which might lead to the decision to drive while intoxicated. Tobacco, in contrast, is not known to cause increases in disinhibition. Although simultaneous alcohol and tobacco users might endorse intoxicated driving, it is plausible that the cause of such behavior is alcohol and not tobacco. Thus, counting the same behavior (e.g., driving under the influence) twice increases the likelihood of someone receiving multiple SUD diagnoses even though only one substance appears to cause hazardous use, in turn inflating comorbidity estimates.
To return to the nature of general addiction liability, if it is reflective of some substantive process, it is certainly possible that it exerts an influence on particular subsets of SUD symptoms. But if this interpretation were entirely accurate, we would expect that the same edges (e.g., hazardous use-hazardous use) would emerge across all pairings of SUDs (e.g., AUD-CUD, OUD-TUD) and not just subsets of them (e.g., AUD-CUD). Hence, the cross-SUD criteria connections, although thought provoking, should not necessarily be taken as evidence of general addiction liability.
Take two instances in which general addiction liability might be indicated in our data. In the bifactor CFA models, the AUD-OUD general factor in NESARC Wave 1 was weak and diffuse, but there was little to no evidence of a TUD-OUD general factor or an AUD-TUD general factor. If what is captured in the AUD-OUD general factor is general addiction liability, we would expect that any SUD would form a strong general factor with any other SUDs that are modeled with it. Likewise, in the network models, hazardous use from one SUD formed strong edges with hazardous use from other SUDs only in some instances. It is unclear why a general-addiction-liability-like dimension, if truly general, would more strongly influence certain substances and not others.
Furthermore, should general addiction liability be robustly empirically supported, evidence for it should arise across numerous methods and not just factor analysis. We should have seen cross-SUD factors form (either in an EFA or CFA), strong cross-SUD edges in networks, and appreciable clusters of people with moderate to high degrees of multiple SUDs. We did not. In summary, although we concluded that there was some evidence of general addiction liability across methods and samples, such a conclusion is somewhat liberal given that we did not observe a single instance in which aspects of SUD appeared to cohere across substances, methods, and samples.
Competing explanations of general-addiction-liability dimensions
Given the stark differences in our conclusions regarding the robustness of general addiction liability and those of the existing literature (e.g., Hatoum et al., 2022; Hicks et al., 2012; Palmer et al., 2012, 2015; Vanyukov et al., 2009; Vanyukov & Ridenour, 2012), we considered what might generate evidence (either substantive or artifactual) for it in other studies. We offer and contextualize three potential explanations here, none of which are necessarily mutually exclusive. In fact, each explanation may be true to some extent, depending on the data used to model a general-addiction-liability dimension.
Where there is evidence of general addiction liability, the dimension reflects (a) absence of drug consumption and addiction, (b) a severity index, or (c) impairment, externalizing, or some combination of the two.
Absence of drug consumption and addiction
We focused on people who endorsed past-year substance use as opposed to the entire sample of people who participated in the NESARC studies. We did so intentionally, and we view this aspect of the study design as a feature, not a bug (Watts, Brusco, et al., 2023). As in Hatoum and colleagues (2022), the inclusion of people who do not endorse substance use (i.e., abstainers) in an analysis of general addiction liability raises important theoretical and statistical challenges that are underappreciated in the field.
By including abstainers while modeling general addiction liability, we make a major assumption: that liability toward SUD and addiction grades along the same dimension as substance consumption. Furthermore, we make a second, related assumption: The mechanisms involved in the absence of substance consumption are the same as those involved in the presence, chronicity, and severity of addiction. We are unlikely to conclude that a pig and a giraffe are highly phenotypically and genetically similar simply because neither have wings. Among abstainers, the absence of addiction is necessarily conflated with the absence of drug consumption, whereas the two are dissociable among people who use drugs. Some people who use drugs develop addiction, but most do not. Even among daily binge drinkers, only 50% diagnose with an AUD (Vergés et al., 2018). Hence, substance consumption is necessary but not sufficient to describe addiction (e.g., Sartor et al., 2007).
A severity index
Because general addiction liability is modeled with factor analysis, which is historically associated with a common-cause model whereby the latent variable is assumed to cause shared variation in its indicators, researchers tend to assume that general addiction liability reflects some causal mechanism or set of mechanisms. Such a conclusion is at least implied by statements such as “Common genetic liability undergirds distinct SUDs,” with “undergirding” meaning that shared genetic liability forms the foundation of SUDs (Hatoum et al., 2022). It is further implied by naming a statistical product “risk” or “liability,” particularly in the context of examining its genetic substrates, with “risk” implying that what is captured in the general addiction liability dimension precedes addiction. But this discussion raises an important question: Why use the term “risk” when what is being modeled are shared outcomes associated with addiction, that is, disorders?
Although it is tempting to reify latent variables, factor analysis cannot possibly inform the nature of an extracted general-addiction-liability dimension. Latent factors are, quite literally, the sum of their parts (Cattell, 1973). Recall that our general-addiction-liability dimension was perfectly correlated with a count variable capturing one’s number of SUD diagnoses, and few would argue a count of diagnoses necessarily reflects some underlying, causal process. Thus, general-addiction-liability-dimension scores should be interpreted literally, as indices of severity or comorbidity, with the particular causal associations among the indicators unclear and unresolvable in cross-sectional data (e.g., van Bork et al., 2017).
Impairment, externalizing, or some combination of the two
It is equally as likely that general addiction liability reflects the shared outcome or outcomes associated with addiction, not its shared cause or causes (Tomarken & Waller, 2003). Such a conceptualization is consistent with a formative variable, as opposed to a reflective latent variable, which is fit equivalent (e.g., Hershberger & Marcoulides, 2013; Raykov et al., 2019). That shared outcome could reflect impairment, or problems in living, associated with drug consumption.
The impairment hypothesis is tenable because SUD diagnoses have impairment baked directly into them, with criteria that require poor life functioning (e.g., social/interpersonal harm, putting oneself in harm’s way, inability to perform necessary activities related to work, school, and home; Martin et al., 2011, 2014; McDowell et al., 2019; Watts, Watson, et al., 2023). If what is captured in general-addiction-liability factors is something akin to impairment, it would suggest that general addiction liability is not a latent entity given that what SUDs appear to share are sequelae. Furthermore, the contents of impairment-laden general-addiction-liability factors are inconsistent with core addiction mechanisms emphasized in modern models of addiction (e.g., compulsivity, negative affectivity, reward, cognitive control; Bickel et al., 2019; Boness et al., 2021; Kramer et al., 2020), further distancing general addiction liability from its plausible etiologic mechanisms.
What complicates matters more is that the consequence-based SUD criteria are also associated with externalizing (i.e., poor behavioral and emotional control; Martin et al., 2011, 2014; McDowell et al., 2019; Watts, Watson, et al., 2023). It is possible that latent general-addiction-liability factors do capture a shared cause by way of latent tendencies toward disinhibition and/or antagonism. But again, that cause is not necessarily central to addiction per se.
In fact, the role of externalizing in SUD is fraught. Because some people do not view externalizing as central or core to addiction, they have argued that the DSM criteria for SUD should be revised to de-emphasize consequences (Martin et al., 2011, 2014), whose inclusion might lead to overdiagnosis of SUD among highly externalizing individuals. Externalizers who drink alcohol may indeed engage in hazardous use and role interference, but they might not be addicted per se to alcohol. Our supposition reminds us of Cleckley’s (1976) description of the classic psychopath, who is prone to “fantastic and uninviting behavior with drink and sometimes without” (p. 355). If externalizing is not core or central to addiction, externalizing may entail a dimension that is separate from those more central to addiction, and it may simply modify the addiction’s clinical presentation. For instance, people with tendencies toward externalizing who are simultaneously addicted to a drug may be especially prone to heedless behavior while intoxicated (Giancola, 2002; Martin et al., 2011, 2014; Watts, Watson, et al., 2023).
If our supposition is correct, it bears implications for research on the Transmissible Liability Index to addiction (Vanyukov et al., 2003, 2009), which effectively captures “risk” for SUD without including SUD symptoms. 3 Vanyukov and colleagues (2003, 2009) modeled phenomena ostensibly antecedent to substance use, mostly including symptoms of childhood disruptive behavioral disorders (e.g., conduct disorder, attention-deficit/hyperactivity disorder) and ability to adopt a structured lifestyle (i.e., maintain routines for sleep and eating). Hicks and colleagues (2012) expanded on this index to include a broader set of externalizing-relevant features, such as truancy, attention problems, negative affect, and suicidality. Both research groups found that the Transmissible Liability Indices modeled in childhood forecasted risk for SUD in adolescence and adulthood (Hicks et al., 2012; Vanyukov et al., 2009). Still, such forecasting of risk may be due to predictor-criterion overlap: Transmissible Liability Indices may predict later SUD because SUD contains allied problem behaviors that are included in the Transmissible Liability Index composite.
Limitations and future directions
Here, we focused on the very type of data analyzed by Hatoum and colleagues (2022)—cross-sectional, largely DSM-based conceptualizations of SUD—with several noteworthy exceptions worth expanding on before addressing more general limitations of our study design.
First and foremost, we modeled SUD criteria as opposed to disorders. We encourage researchers to abandon modeling of phenotypes (e.g., AUD) that are widely accepted as heterogeneous (e.g., Strain, 2021) and whose symptoms have causal orderings (Jellinek, 1952; Koob & Le Moal, 2001) that are incompatible with a unitary-latent-variable model (Kendler et al., 2012; Muthén et al., 1993; Watts et al., 2021). SUDs and all diagnostic categories are teleological, or tools of convenience; they do not carve nature at its joints, and they are insufficiently equipped to inform etiology. Therefore, investigations of general addiction liability’s genetic substrates that start with disorders are, at best, an inefficient means of detecting the causes of addiction. At worst, if symptoms themselves have different causes, modeling disorders obscures the ability to detect addiction’s etiology.
Second, we focused on phenotypic associations, whereas Hatoum and colleagues (2022) used genomic data. Our design extends easily to genomic investigations because all investigations rely on variance in a phenotype that is associated with certain polymorphisms, and a genomic structural equation model of SUDs will always rely on the covariance among the phenotypes; highly correlated phenotypes are more likely to share genetic substrates. Third, as we mentioned earlier, we focused on subsets of participants who endorsed past-year use of two substances, purely for practical reasons. The base rates of polysubstance use were too low to consider participants who endorsed using all four classes of drugs (see Table S1 in the Supplemental Material), underscoring the low base rate of polysubstance use in the general population. Still, we do not view this aspect of our study design as particularly concern-worthy: If general addiction liability is truly general, we should have seen evidence for it no matter whether we focused on polysubstance use of two drugs, three drugs, and so on.
Beyond deviations from Hatoum and colleagues’ (2022) work that might be perceived as limitations, we relied on cross-sectional data, which, of course, cannot inform causality. We used cross-sectional data to maintain compatibility with the lion’s share of investigations of general addiction liability (e.g., Hatoum et al., 2022). It is possible that longitudinal designs could be used to further inform general addiction liability. For instance, perhaps general addiction liability is a latent trait that predisposes to varied types of disordered drug use across the life span but not necessarily polysubstance use. Someone with a high degree of general addiction liability may switch between drugs such that they experience tobacco problems in early adolescence, cannabis problems in emerging adulthood, and alcohol problems later in life. We look forward to extensions of our study to novel designs, which will necessarily require large-scale (i.e., N > 40,000) longitudinal data.
In addition, it is important to emphasize that our data cannot resolve time course of use in these data. We selected participants who reported use of two or more substances in the past year, but it is unclear whether those substances were used simultaneously or at different times over the year. Relatedly, because of the relatively small sample sizes for certain pairings of substances, we could not take patterns of consumption and context of consumption into account. For instance, it would be interesting to consider whether each substance was used chronically or sporadically and exclusively in social settings as opposed to while home alone.
Another important factor to consider among polysubstance users is the primary versus secondary (and tertiary, etc.) substance. Waddell (2021) showed that alcohol and cannabis co-users were at increased risk of an AUD diagnosis but not CUD diagnosis, suggesting that co-use does not necessarily equivalently indicate comorbidity risk for both substances. Waddell also found that primary use of alcohol increased risk for AUD and not CUD, whereas primary use of cannabis did not increase risk for AUD. Hence, the connections among SUDs may be weighted as opposed to unweighted, with Disorder A having a stronger directional tie with Disorder B than Disorder B has with Disorder A. We look forward to finer-grained analyses that can differentiate primary and secondary substances.
Conclusion
Addiction is a complex, multifaceted condition with myriad posited etiologic mechanisms (Bickel et al., 2019; Boness et al., 2021; Sher, 1991). The statistical model of general addiction liability—which assumes that tendencies toward addiction can be described or quantified in terms of a single, unitary dimension—is at odds with that statement. All told, our findings suggest that general addiction liability should be investigated with increased scrutiny and promoted with increased caution, even skepticism. Indeed, among polysubstance users, SUDs appear more varied than uniform: Psychoactive substances have unique heritable influences, environmental influences, withdrawal syndromes, and different patterns of correlates (Crump et al., 2021; Kendler et al., 2007; Kendler, Jacobson, et al., 2003). Addiction is not a monolith.
Supplemental Material
sj-xlsx-1-cpx-10.1177_21677026241245070 – Supplemental material for “General Addiction Liability” Revisited
Supplemental material, sj-xlsx-1-cpx-10.1177_21677026241245070 for “General Addiction Liability” Revisited by Ashley L. Watts, Kenneth J. Sher, Andrew C. Heath, Douglas Steinley and Michael Brusco in Clinical Psychological Science
Footnotes
Transparency
Action Editor: Tamika C. Zapolski
Editor: Jennifer L. Tackett
Author Contributions
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
