Abstract
Children who experience environmental adversities are at increased risk of both internalizing and externalizing disorders. Epigenetic mechanisms may regulate the influence of environmental adversities on mental health. We examined the hypothesis that salivary DNA-methylation patterns of pace of biological aging (Dunedin pace of biological aging [DunedinPoAm]) and inflammation (DNA-methylation C-reactive protein [DNAm-CRP]) are socially stratified and associated with mental health in 1,183 children (609 female; age: M = 13.6 years) from the Texas Twin Project. Analyses were preregistered. Participants’ DNA-methylation algorithms and psychiatric symptoms differed by socioeconomic contexts and race/ethnicity. Children with more parent-reported internalizing symptoms had higher DunedinPoAm and DNAm-CRP scores, and children with more aggression problems had higher DNAm-CRP. DunedinPoAm partially mediated advantage of White racial identity on internalizing. Likewise, DNAm-CRP partially mediated advantage of higher family socioeconomic contexts and, in a separate model, White racial identity on reduced internalizing symptoms. Children’s epigenetic measures of pace of biological aging and inflammation are associated with social inequalities and mental health.
Children who experience environmental adversities, including financial scarcity and the discrimination, prejudice, and oppression associated with racial and ethnic marginalization, are at increased risk of both internalizing and externalizing disorders (Kinge et al., 2021; Peverill et al., 2021; Vaid & Lansing, 2020). In contrast, White racial identity may confer protective effects on children’s mental health because of the effects of White privilege (McIntosh, 1988; Priest et al., 2013). 1 Exposure to toxicants, opportunities for restorative leisure and exercise, physical and psychological safety, social support, and access to nutritious food and health care, among other factors, differ across these social positions (Goosby et al., 2018; Williams et al., 2016).
The developing brain is especially sensitive to social adversity, which can increase chronic low-grade inflammation and consequently affect the development of neural structures known to be involved in later-life psychopathology (Danese & Baldwin, 2017; Snyder-Mackler et al., 2020). DNA methylation is one epigenetic mechanism thought to be involved in the biological embedding of environmental adversities and to differ by chronic inflammatory levels (Ligthart et al., 2016). Inflammation-related changes to patterns of DNA methylation are also considered key mechanisms of multisystem aging processes (i.e., “inflamm-aging”; Aristizabal et al., 2019; Peters et al., 2021). Thus, DNA-methylation patterns of inflammation and aging may partially originate in molecular processes during childhood and adolescence, which in turn contribute to social disparities in mental well-being across the life course.
Here we test whether DNA-methylation alterations of inflammation and aging discovered in studies of adults are sensitive to socioeconomic and racial/ethnic disparities in childhood and are associated with child and adolescent mental health. Following a preregistered analysis plan (https://osf.io/ta29g), we examined DNA-methylation algorithms associated with (a) a faster pace of biological aging (i.e., Dunedin pace of biological aging [DunedinPoAm]; Belsky et al., 2020) and (b) higher levels of C-reactive protein (DNA-methylation C-reactive protein [DNAm-CRP]), a peripheral proxy for systemic low-grade inflammation (Ligthart et al., 2016). We previously reported, using a prior subset of the current analytic sample, that socioeconomic disadvantage and Latinx identity (compared with White identity) were associated with a faster DunedinPoAm (Raffington et al., 2021). In contrast, other epigenetic clocks, including Horvath and GrimAge, were not sensitive to social inequality in this sample and were therefore not included in the present analyses. Likewise, recent work has found that DunedinPoAm, but not other epigenetic clocks, are sensitive to a randomized controlled calorie-restriction intervention designed to slow the pace of biological aging (Waziry et al., 2023).
One previous study of children found that DNAm-CRP, measured using children’s blood samples, was associated with internalizing and externalizing symptoms (Barker et al., 2018). It is not yet known, however, whether DNAm-CRP is associated with children’s mental-health symptoms when DNA methylation is measured from saliva, which is less invasive and more feasible to collect than blood in large-scale epidemiological and intervention research. Neither is it known whether DNAm-CRP and DunedinPoAm algorithms reflect the social disparities relevant for mental health. In particular, if DNA-methylation measures that were originally developed in adults are shown to be related to childhood exposures and well-being, this result is evidence of a molecular continuity between childhood social inequalities and adulthood health disparities.
Transparency and Openness
Because of the high potential for deductive identification in this special population from a geographically circumscribed area and the sensitive nature of information collected, data from the Texas Twin Project are not shared with individuals outside of the research team. Code to compute DNA-methylation scores will be shared by the first author on request. For Mplus code, see the Supplemental Material available online. Preregistration can be found at https://osf.io/t3vnp/. We report how we determined our sample size, all data exclusions, all manipulations, and all measures in the study.
Method
Sample
The Texas Twin Project is a population-based study of twins and multiples recruited from public-school rosters in the central Texas region (Harden et al., 2013). Participants in the current study were 1,183 unique individuals (609 female) who had at least one DNA-methylation sample that passed quality control (from initially enrolled sample of 1,213 children). They came from 617 unique families, including 426 monozygotic and 757 dizygotic twins (see zygosity measure), ages 8 to 19 years (M = 13.6 years, SD = 3.05). One hundred ninety-five participants contributed two DNA-methylation samples (time between repeated samples: M = 22 months, SD = 6.5; range = 3–38 months), and 16 samples were assayed in duplicate for reliability analyses (total observations at enrollment: n = 1,424). See Table 1 for descriptive statistics of the analytic sample. The University of Texas Institutional Review Board granted ethical approval. Informed consent to participate in the study was obtained from all participants and their parent or legal guardian.
Descriptive Statistics of the Analytic Sample (N = 1,183 Unique Individuals)
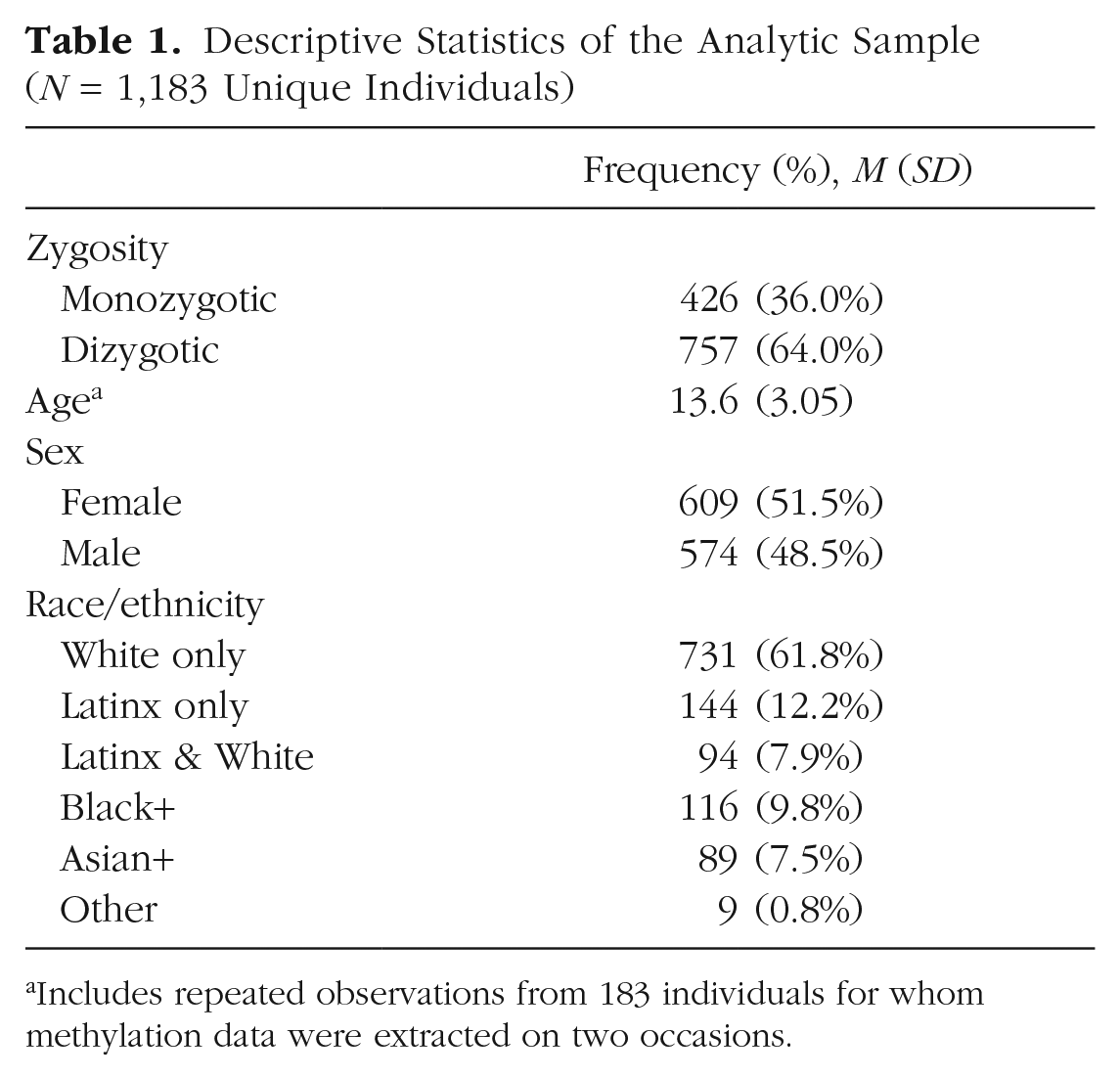
Includes repeated observations from 183 individuals for whom methylation data were extracted on two occasions.
Associations Between DNA-Methylation Algorithms With Parent-Reported and Self-Reported Mental Health
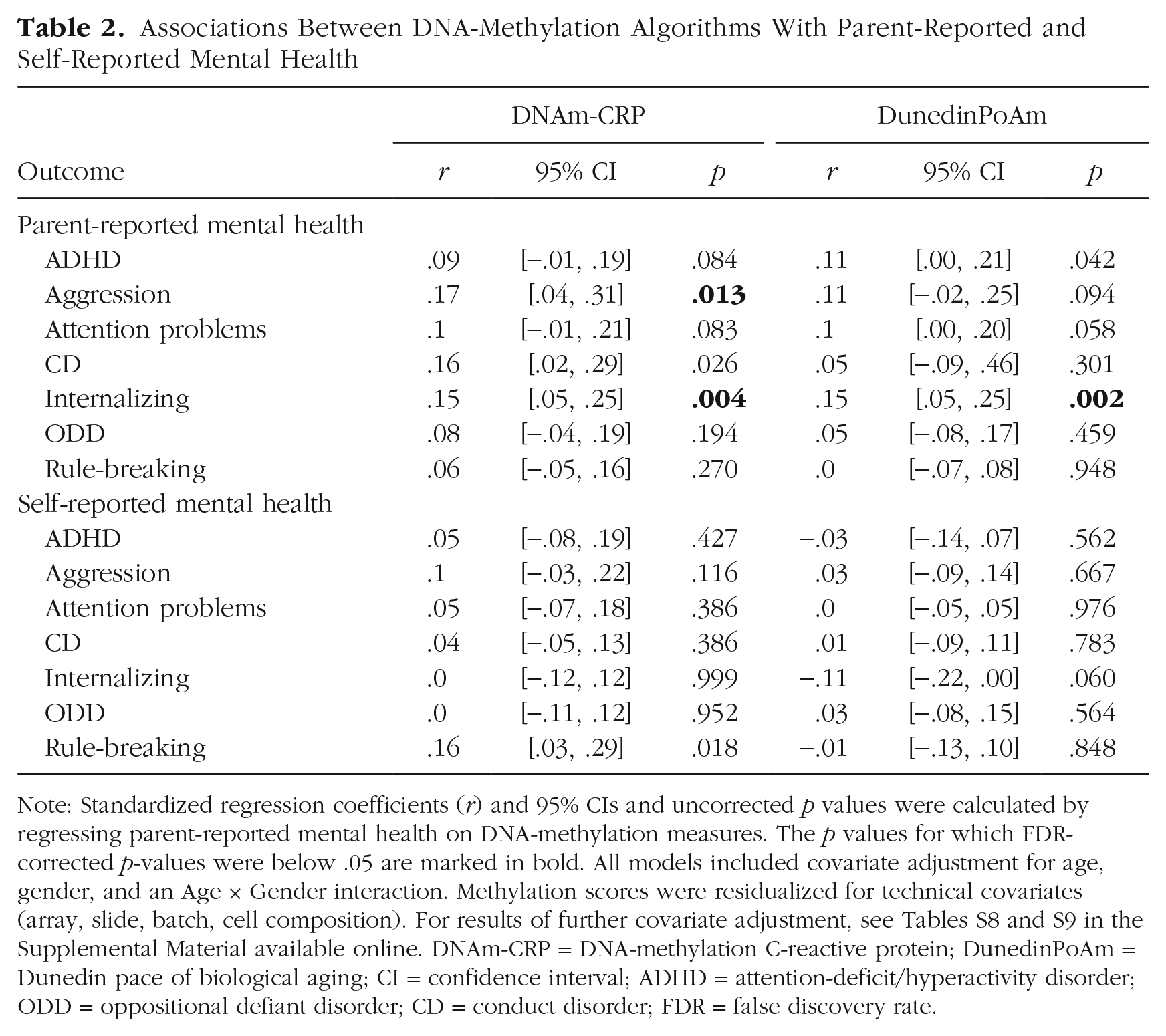
Note: Standardized regression coefficients (r) and 95% CIs and uncorrected p values were calculated by regressing parent-reported mental health on DNA-methylation measures. The p values for which FDR-corrected p-values were below .05 are marked in bold. All models included covariate adjustment for age, gender, and an Age × Gender interaction. Methylation scores were residualized for technical covariates (array, slide, batch, cell composition). For results of further covariate adjustment, see Tables S8 and S9 in the Supplemental Material available online. DNAm-CRP = DNA-methylation C-reactive protein; DunedinPoAm = Dunedin pace of biological aging; CI = confidence interval; ADHD = attention-deficit/hyperactivity disorder; ODD = oppositional defiant disorder; CD = conduct disorder; FDR = false discovery rate.
Measures
DNA-methylation algorithms of C-reactive protein and pace of biological aging
Saliva samples were collected during a laboratory visit using Oragene kits (DNA Genotek, Canada). The Infinium MethylationEPIC BeadChip kit (Illumina, Inc., USA) was used to assess methylation levels at 850,000 methylation sites. Information on preprocessing is available in the Supplemental Material. Using the summary statistics of the associations between CpG sites measured in blood and adult C-reactive protein (CRP) from Ligthart et al. (2016), we created the DNAm-CRP score in each person by summing the product of the weight and the individual beta estimate for each individual at each of the 218 CpG sites previously found to be significantly associated with CRP (epigenome-wide significant threshold: p < 1.15 × 10−7). Previous independent target studies have validated both saliva- and blood-derived DNAm-CRP in association with serum CRP levels in children, adolescents, and young adults (Barker et al., 2018; Mrug et al., 2022). Specifically, the correlation between saliva DNAm-CRP and serum CRP was r = .27, p < .001, in the Birmingham Youth Violence Study (Mrug et al., 2022).
DunedinPoAm was developed from blood DNA-methylation analysis of “pace of aging” in the Dunedin Study birth cohort (Belsky et al., 2020). Briefly, pace of aging is a composite phenotype derived from analysis of longitudinal change in 18 biomarkers of organ-system integrity measured when Dunedin Study members were all 26, 32, and 38 years old. Elastic-net regression machine-learning analysis was used to fit pace of aging to Illumina 450k DNA-methylation data generated from blood samples collected when participants were 38 years old. The elastic-net regression produced a 46-CpG algorithm. Increments of DunedinPoAm correspond to “years” of physiological change occurring per 12 months of chronological time. The Dunedin Study mean was 1 (i.e., the typical pace of aging among 38-year-olds in that birth cohort). Thus, a 0.01 increment of DunedinPoAm corresponds to a percentage-point increase or decrease in an individual’s pace of aging relative to the Dunedin birth cohort at midlife. DunedinPoAm was calculated based on the published algorithm using code available at https://github.com/danbelsky/DunedinPoAm38.
All methylation scores were residualized for technical covariates (array, slide, batch, cell composition; see Supplemental Methods in the Supplemental Material).
Mental health
Mental health was measured using self-reports and parent reports on abbreviated versions of the Achenbach Child Behavior Checklist (CBCL; Achenbach, 1991; Lizotte et al., 1992), and the symptom-count scales from the fourth edition of the Diagnostic and Statistical Manual of Mental Disorders of the Conners 3 (Conners et al., 1998). Following prior published work in this sample (Harden et al., 2020), we calculated sum scores for seven domains of psychopathology: (a) internalizing (CBCL anxiety and depression), (b) CBCL aggression, (c) rule-breaking, (d) CBCL attention problems, (e) Conner’s attention-deficit/hyperactivity disorder (ADHD) symptoms, (f) Conner’s conduct disorder (CD) symptoms, and (g) Conner’s oppositional defiant disorder (ODD) symptoms. For a full list of items in each measure, see Table S1 in the Supplemental Material.
Socioeconomic context
We measured children’s socioeconomic disadvantage at the family and neighborhood levels of analysis. Family-level disadvantage was computed from parent reports of household income, parental education, occupation, history of financial problems, food insecurity (based on the U.S. Household Food Security Survey Module; Economic Research Service, n.d.), father absence, residential instability (changes in home address), and family receipt of public assistance. These were aggregated to form a composite measure of household-level cumulative socioeconomic disadvantage described in Engelhardt et al. (2019) and coded such that higher scores reflect greater disadvantage.
Neighborhood-level disadvantage was calculated from tract-level U.S. census data according to the method described in Engelhardt et al. (2019). Briefly, participant addresses were linked to tract-level data from the U.S. Census Bureau American Community Survey averaged over 5 years (available at https://www.census.gov/programs-surveys/acs). A composite score of neighborhood-level socioeconomic disadvantage was computed from tract-level proportions of residents reported as unemployed, living below the federal poverty threshold, having fewer than 12 years of education, not being employed in a management position, and single mothers. These were aggregated to form a neighborhood-level socioeconomic-disadvantage composite measure described in Engelhardt et al. (2019) and coded such that higher scores reflect greater disadvantage.
The neighborhood-opportunity measure indexed the intergenerational economic mobility of children of low-income parents and was based on analysis of the average annual household income in 2014–2015 of offspring (born between 1978 and 1983, who are now in their mid-30s) of low-income parents (defined as mean pretax income at the household level across 5 years, 1994, 1995, 1998–2000, at the 25th percentile of the national income distribution, or $27,000/year) within each census tract. These data were compiled by and obtained from the Opportunity Atlas (available at https://opportunityatlas.org; Chetty et al., 2018). Household income was obtained from federal tax-return records between 1989 and 2015, the 2000 and 2010 decennial censuses (U.S. Census Bureau, 2000, 2010; https://data2.nhgis.org/main), and 2005–2015 American Community Surveys.
Racial and ethnic identities
Participants self-identified current racial/ethnic identities in response to the question, “What best describes your racial and ethnic background?”; they selected one or multiple categories of White, Latinx/Hispanic, Black, Asian, Indigenous American, Pacific Islander, and other. Respondents identified as White only (n = 752, 62%); Latinx only (n = 147, 12%); Latinx and White (n = 97, 8%); Black and potentially another race/ethnicity (n = 120, Black = 10%); Asian and potentially another race/ethnicity but not Latinx or Black (n = 90, Asian = 7.5%); and Indigenous American, Pacific Islander, or other but not Latinx, Black, or Asian (n = 7, 0.6%).
Covariates
We measured body mass index (BMI) from in-laboratory measurements of height and weight transformed to gender- and age-normed z scores according to the method published by the U.S. Centers for Disease Control and Prevention (2009).
Pubertal development was measured using children’s self-reports on the Pubertal Development Scale (Petersen et al., 1988). The scale assesses the extent of development across five sex-specific domains (for boys and girls: height, body-hair growth, skin changes; for girls: onset of menses, breast development; for boys: growth in body hair, deepening of voice). A total pubertal-status score was computed as the average response (1 = not yet begun, 4 = has finished changing) across all items. Pubertal development was residualized for age, gender, and an Age × Gender interaction. Finally, we indexed tobacco exposure using a DNA-methylation smoking (DNAm-smoke) score created by summing the product of the weight and the individual beta estimate for each individual at each CpG site significantly associated with smoking in the discovery epigenome-wide association study (Joehanes et al., 2016). Excluding self-reported tobacco users (n = 58) did not significantly alter results. For descriptive statistics of all study measures, see Table S2 in the Supplemental Material.
Statistical analyses
Fourteen mental-health outcomes were examined in each model, including seven self-reported measures and seven parent-reported measures. We performed multilevel, multivariate regression models fit with full information maximum likelihood in Mplus (Version 8.2; Muthén & Muthén, 1998–2022) software (for sample code, see the Supplemental Material). To account for nesting of repeated epigenetic measures within individuals and multiple twin pairs within families, we used the Mplus “TYPE=twolevel complex” analysis options. Under this specification, Mplus accounts for clustering due to the lowest level of clustering (repeated measures within individuals) using a two-level model and accounts for the clustering due to the highest level of clustering (individuals within families) using a sandwich correction to produce cluster-robust standard errors (for technical information, see MPlus user guide; Muthén & Muthén, 1998–2022, Chapter 9). We specified the two-level portion of the models to include a random intercept to account for the nonindependence of repeated measures within individuals. Because repeated measures data were available for only approximately 15% of individuals, we did not attempt to model random slopes. Results can be conceived of having been derived from a cross-sectional analysis in which different individuals are observed at different ages clustered within different families without the inferential statistics being biased by the inclusion of multiple individuals from the same family. All models included age, gender, and an Age × Gender interaction as covariates.
Thus, the multilevel model for the key dependent variable, mental health (Y) for observation t for person n, can be written as follows:
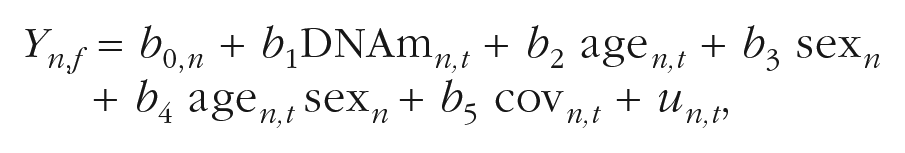
where b0,n is a random intercept representing the person-level intercept of the dependent variable, DNAm is the DNA-methylation predictor, age is the age of the individual at the time of measurement, sex is the sex of the individual, cov is any additional covariate included in the model, and u is a residual representing unexplained variance. Because the nested nature of the family data is not explicitly modeled in the multilevel model (and instead corrected for using cluster-robust standard errors), we do not include subscripts for family in the equation above. Note that when we seek to explicitly examine associations within family, we apply a bivariate biometric ACE (additive genetic variance [A], shared environmental factors [C], and nonshared environmental factors plus measurement error [E]) decomposition within a structural-equation-modeling framework (Neale & Maes, 2004).
Regression models were grouped according to substantive hypothesis (as summarized in Table S13 in the Supplemental Material) and for each regression model. The p values for each group were corrected using the Benjamini–Hochberg method for controlling the false discovery rate (FDR), where the FDR was selected to be less than 5% (Benjamini & Hochberg, 1995). For a list of preregistered regression models by grouping for FDR correction, see Table S13 in the Supplemental Material. Effect sizes are reported as standardized regression coefficients that are interpretable as Pearson r, and nominal p values are reported as significant when the FDR-corrected p values were below α < .05.
We also performed several supplementary analyses. First, we quantified the reliability of the methylation algorithms by fitting a biometric twin model in which the additive genetic factor (A) and shared environmental factor (C) are summed to represent the lower bound of reliability. Second, we tested the robustness of the main results to complete genetic and environmental control by estimating a twin fixed-effects model. Third, using a subsample of European-ancestry twins, we estimated the impact of adjusting for genetic indices associated with inflammation (for more details see, the Supplemental Material).
Results
Our main analyses examined whether (a) mental health is socially stratified and (b) salivary DNA-methylation patterns of higher inflammation (DNAm-CRP) and faster pace of biological aging (DunedinPoAm) are associated with child mental health.
Mental health is socially stratified in children
Results for parent-reported psychopathology
For parent-reported psychopathology (Fig. 1a; also see Table S3 in the Supplemental Material), family-level disadvantage was associated with worse mental health across all domains except CD symptoms (standardized regression coefficients range: rs = .11–.26). Neighborhood-level disadvantage and lack of neighborhood opportunity were also associated with higher internalizing and attention problems (rs = .12–.19).
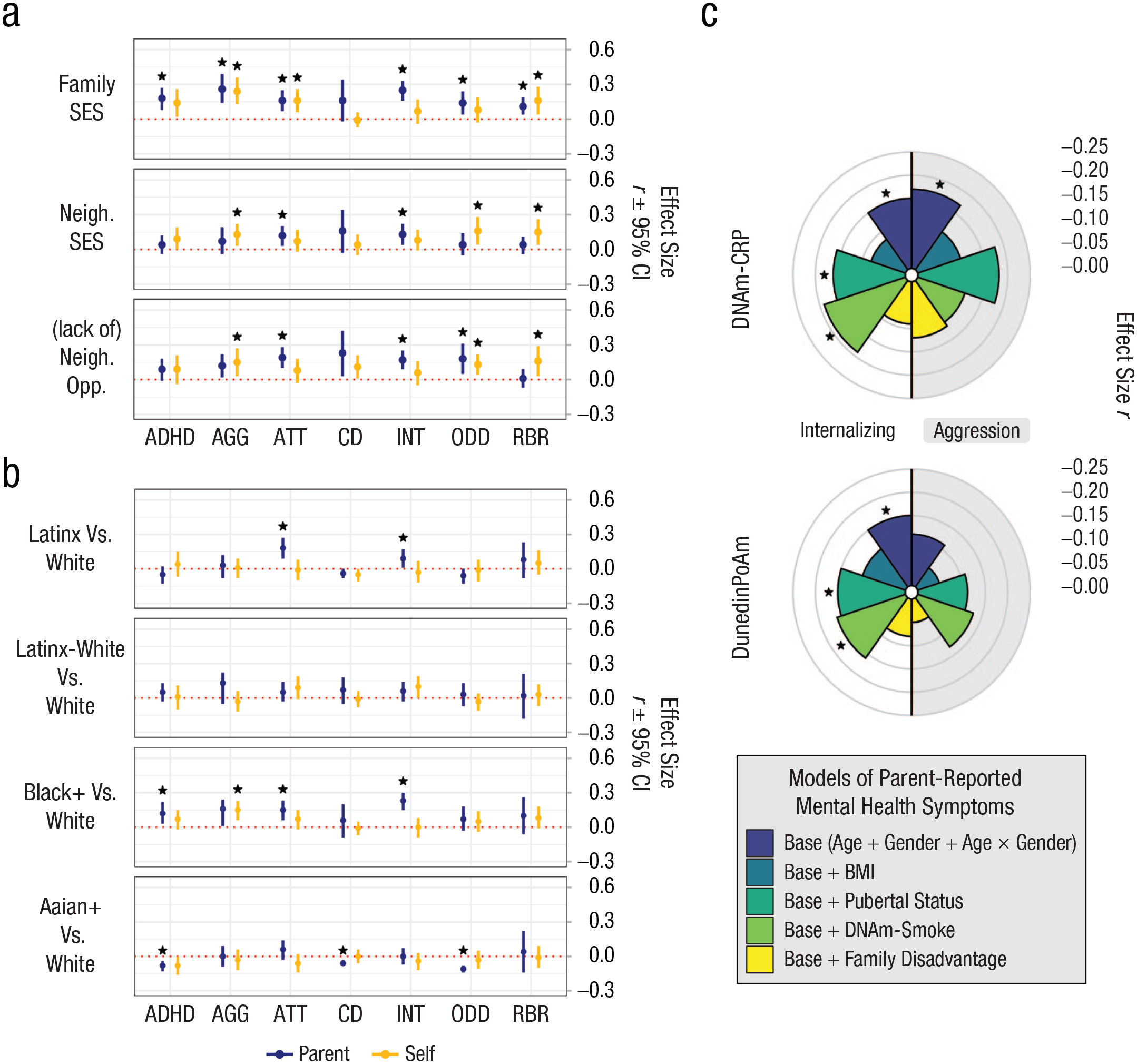
Associations between measures of socioeconomic inequality, racial and ethnic identity, and DNA-methylation with parent- and self-reported mental health in children and adolescents. Depicted are standardized effect size estimates For coefficients, see Tables S2, S3, and S7 in the Supplemental Material available online. Asterisks indicate significant false-discovery-rate-adjusted values (p < .05).
Children identifying as Latinx-only or Black+ had higher parent-reported internalizing and attention problems than White-only identifying children (effect sizes: rs = .09–.23; Fig. 1b; also see Table S3 in the Supplemental Material). Black+-identifying children also showed more parent-reported symptoms of ADHD. Asian+-identifying children had lower parent-reported ADHD, ODD, and CD than White-only children (effect sizes: rs = –.06 to –.11).
Results for child-reported psychopathology
Socioeconomic disadvantage was less ubiquitously associated with children’s self-reported psychopathology (Fig. 1a; also see Table S4 in the Supplemental Material). Family-level disadvantage, neighborhood-level disadvantage, and lack of neighborhood opportunity were all associated with higher child-reported aggression and rule-breaking; the neighborhood variables were additionally associated with ODD symptoms (rs = .13–.24). Child-reported mental health was largely unrelated to race/ethnicity (Fig. 1b), except that Black+-identifying children self-reported higher rates of aggression than White-only children (r = .15, 95% confidence interval [CI] = [.06, .23], p = .001).
Intersection of race/ethnicity with socioeconomic disadvantage
Children reporting Black+ and Latinx-only identities lived in substantially more socioeconomically disadvantaged families and neighborhoods compared with children reporting White-only identity (effect sizes: rs = .25–.43; see Table S5 in the Supplemental Material). Family-level disadvantage statistically accounted for differences between Latinx-only and White-only children’s parent-reported internalizing but not attention problems (see Table S3 in the Supplemental Material). Family-level disadvantage accounted for differences between Black+ and White-only children’s parent-reported ADHD and child-reported aggression but not differences in parent-reported internalizing or attention problems (see Table S3 and S4 in the Supplemental Material). After accounting for family-level disadvantage, Latinx-only children self-reported lower rates of CD than White-only children.
Salivary DNA-methylation algorithms are associated with internalizing and aggression
In contrast to the widespread associations between socioeconomic and racial inequality with mental health, DNA-methylation algorithms demonstrated associations with two specific expressions of psychopathology (see Table 2). Higher DNAm-CRP and faster DunedinPoAm were associated with higher parent-reported internalizing (both: r = .15, 95% CI = [.05, .25], p = .002), and faster DunedinPoAm was also associated with higher parent-reported aggression (r = .17, 95% CI = [.04, .31], p = .013; Fig. 1c; also see Table S8 in the Supplemental Material). DNA-methylation algorithms were not associated with children’s self-reported mental health (see Table S9 in the Supplemental Material). For effect-size estimates between DNA-methylation algorithms and mental health reported separately for each racial/ethnic group (not preregistered), see Tables S10 and S11 in the Supplemental Material. Although some of these parameters were estimated in the opposite direction, they were not reliably different from zero and thus not interpretable as showing associations in the opposite direction. This is most likely because of the small sample sizes within some of the racial/ethnic subgroups.
Finally, we explored the possibility of confounding in our results because of genetic algorithms specifically associated with inflammation. Using a polygenic score for CRP (PGS-CRP; see Supplemental Methods in the Supplemental Material) and a subsample of European-ancestry dizygotic twins, we found that PGS-CRP was associated with parent-reported internalizing and attention problems (see Table S8 in the Supplemental Material; for child-reported mental-health results, see Table S9 in the Supplemental Material).
Within-families analyses indicate family-level stratification of DNA-methylation
We further assessed the extent to which DNA-methylation associations with mental health are robust to complete genetic and family-level environmental control in a bivariate ACE model that used the twin family structure. We found no evidence to suggest that monozygotic twins who differ from their co-twins in DNA methylation show corresponding differences in their mental health (see Table S12 in the Supplemental Material). This is consistent with the hypothesis that DNA-methylation associations with mental health represent (potentially partially unmeasured) effects of family-level stratification.
Effect-size estimates of the association between family-level and neighborhood-level socioeconomic disadvantage with pace of aging were slightly larger in the present study compared with a previous publication that used a subsample of the present sample (see Table S6 in the Supplemental Material; e.g., family-level disadvantage: r = .28, 95% CI = [.18, .37] vs. r = .18, 95% CI = [.08, .27], p = .001 for both; Raffington et al., 2021). This may be related to the fact that the present sample is more sociodemographically diverse.
Unique and overlapping predictive value of DNAm-CRP for mental health
Higher DNAm-CRP was correlated with higher DunedinPoAm (r = .89, 95% CI = [.81, .96], p < .001, accounting for age and gender). This is presumably due to CRP levels being one of the 18 biomarkers that the pace-of-aging algorithm was trained on and seven CpG sites overlapping across measures (Belsky et al., 2020). Given this overlap, we performed commonality analyses to examine the proportion of overlapping and unique variation explained in parent-reported internalizing. DNAm-CRP and DunedinPoAm explained largely unique variation in parent-reported internalizing (DNAm-CRP alone = 2.5%, DunedinPoAm alone = 2.2%, combined = 4.5%).
We next examined the role of BMI, pubertal status, and DNAm-smoke in associations of DNA methylation with mental health (see Table S8 in the Supplemental Material). Associations of DNAm-CRP and DunedinPoAm with parent-reported internalizing were not attenuated when controlling for pubertal status but were attenuated when controlling for BMI (for the association of DNAm-CRP with internalizing, rs range from .15 to .08; for the association of DunedinPoAm with internalizing, rs range from .15 to .10). In addition, associations of DNAm-CRP with aggression were attenuated when controlling for DNAm-smoke (rs range from .17 to .11). Finally, using a subset of 364 dizygotic twins, we found that adjusting for a polygenic score associated with CRP did not account for the associations between DNAm-CRP or DunedinPoAm with internalizing or attention problems (for details, see the Supplemental Material). Dizygotic-twin differences in PGS-CRP were weakly correlated with differences in DNAm-CRP (r = .14, 95% CI = [.01, .27], p = .043; not preregistered).
We observed that DNAm-CRP and DunedinPoAm partially accounted for associations of family-level disadvantage and racial/ethnic disparities in mental health. DNAm-CRP, but not DunedinPoAm, partially mediated family-level disadvantage on internalizing (indirect effect 16% of total effect) but not on aggression. Both DNAm-CRP and DunedinPoAm partially mediated the impact of being racially marginalized on internalizing (indirect DNAm-CRP effect 12% of total effect; indirect DunedinPoAm effect 19% of total effect in separate model). (Note that given cross-sectional data, a model with mental health regressed on both social inequalities and DNAm is akin to a cross-sectional mediation model.)
Discussion
We analyzed epigenetic data from 1,183 children and adolescents participating in the Texas Twin Project to examine whether salivary DNA-methylation patterns of inflammation (DNAm-CRP) and pace of aging (DunedinPoAm) discovered in studies of adults are sensitive to socially stratified mental-health problems in children and adolescents. As hypothesized, we found that children from marginalized racial/ethnic groups and who experience socioeconomic disadvantage show worse mental health across internalizing and externalizing domains. These social disparities are most clearly evident for parent-reported psychopathology compared with child-reported psychopathology. We furthermore found that salivary DNA-methylation algorithms of inflammation and pace of biological aging were associated with some dimensions of worse mental health, specifically, parent-reported internalizing problems and aggression problems. Moreover, salivary DNA-methylation algorithms partially accounted for child mental-health disparities related to family-level disadvantage, racial, and ethnic identities. Because these DNA-methylation measures were originally developed in adults, our findings suggest a molecular continuity between childhood social conditions and adult inflammation and biological aging that is relevant to mental-health stratification.
Our findings linking environmental inequality to epigenetic algorithms and mental health were most consistent for internalizing compared with externalizing symptoms, which is in line with previous research on peripheral inflammation (Danese & Baldwin, 2017) and a blood-based study on DNA methylation of inflammation in children (Barker et al., 2018). For instance, Barker et al. (2018) found that higher postnatal adversity exposure in the first 7 years of life was associated with more internalizing, but not externalizing, problems at ages 7 to 15, which was mediated by higher blood DNA-methylation algorithms of CRP at age 7.
DNA methylation is tissue specific and has different patterns in saliva, blood, and brain. The original estimates on which our salivary algorithms were based were estimated from DNA methylation in blood. There are several factors that, when taken together, increase our confidence in our findings with salivary DNA methylation. First, we made statistical corrections for cell composition by implementing a pediatric saliva-reference panel (Middleton et al., 2022). Second, we have seen good correspondence of pace-of-aging measures across blood and saliva (r = .85; Raffington et al., 2021), and a previous study reported modest correspondence between saliva-derived DNA-methylation measures of CRP with serum CRP levels in children and young adults (r = .27; Mrug et al., 2022). Studies in adults suggest that DNA-methylation measures of CRP show greater longitudinal stability and stronger associations with brain outcomes compared with serum CRP and may provide more stable reflections of cumulative inflammatory burden than traditional serum approaches (Conole et al., 2021; Stevenson et al., 2020). Third, methylation predicts smoking status in both saliva and blood DNA in adolescents (Dawes et al., 2019). Fourth, epigenetic age clocks were highly correlated with age in a subsample of the present cohort (Raffington et al., 2021). Nevertheless, we caution that there is still considerable uncertainty about what DNA-methylation measures generated from saliva samples reflect.
Our results add to a growing body of research in animals and humans that links exposure to social adversity, DNA-methylation changes, and gene expression markers associated with inflammation (Cole et al., 2020; McDade et al., 2019; Snyder-Mackler et al., 2020). Animal research suggests that inflammation affects synaptogenesis, synaptic survival, and myelination, for instance, in neural circuitries subserving executive control and threat and reward processing that are pertinent to emotional and behavioral regulation (Hostinar et al., 2018). Furthermore, inflammation and DNA-methylation alterations are environmentally sensitive hallmarks of biological aging (Emeny et al., 2021; Epel, 2020; Peters et al., 2021). We found that controlling for BMI attenuated associations of DNAm-CRP and DunedinPoAm with mental health. This observation corresponds to new insights casting obesity as an inflammatory disease of accelerated biological aging (Santos & Sinha, 2021) and also suggests that BMI measurements are capturing overlapping information as measurements of the epigenome in relation to mental health. Our findings open up an interesting avenue of inquiry for future research exploring the ways through which DNA methylation, BMI, and mental health become associated over development.
Highlighting the distinctly harmful role of social and institutional racism, we found that even after accounting for the lower rates of socioeconomic disadvantage experienced by White children, White-identifying children sustained a lower burden of mental-health symptoms. Thus, socioeconomic inequality captures some but not all of the pathways through which social and institutional racism affects children’s mental health (Williams et al., 2016). Moreover, our categorization of children’s complex racial and ethnic identities that, for instance, grouped Black and Black-White-identifying children together is likely to dilute and underestimate the biopsychological effects of racial marginalization because the proximity to White privilege and experiences of marginalization are not the same between these identities.
Further research is needed to contextualize the biological and mental-health effects of race-based discrimination, prejudice, and oppression experienced by marginalized youths compared with the favoritism and privilege experienced by White youths (Card & Giuliano, 2016; Goosby et al., 2018; Vaid & Lansing, 2020). Although our findings are in line with the hypothesis that DNA-methylation alterations are one candidate mechanism for how psychosocial stress is biologically embedded to injure children’s psychiatric health, our observational design is, of course, not well suited to evaluate mechanistic hypotheses. Randomized controlled trials and natural experiments that involve exogenous changes to various aspects of social inequality (e.g., child tax credits) are necessary to establish whether children’s salivary DNA-methylation algorithms change in response to intervention in a way that is tied to mental health. More work also needs to be done to validate DNA-methylation measures across racial/ethnic groups. One recent well-powered study found that DNA-methylation levels of circulating leukocytes show robust and consistent association with blood lipid levels across multiple racial/ethnic groups (Jhun et al., 2021).
In contrast to results seen for parent-reported psychiatric symptoms, child-reported mental health was generally not associated with DNA methylation. Previous research in this sample and by others has found that children and parents agree minimally on specific symptoms scales (De Los Reyes et al., 2015; Harden et al., 2020). In addition, both children and parent self-reports can be influenced by their level of insight of their specific psychopathological burden and the degree to which they have internalized narratives associated with being racially marginalized (i.e., internalized racism; Williams, 2018).
In conclusion, our findings suggest that salivary DNA-methylation patterns of higher inflammation and faster pace of biological aging originally developed in adults are economically and racially stratified and are associated with child mental health. Because saliva can easily be collected in large-scale pediatric epidemiological studies and may indicate emerging health conditions, these DNA-methylation algorithms may be employed in research seeking to understand and prevent economic and racial disparities in childhood mental health.
Supplemental Material
sj-docx-1-cpx-10.1177_21677026231186802 – Supplemental material for Associations of DNA-Methylation Measures of Biological Aging With Social Disparities in Child and Adolescent Mental Health
Supplemental material, sj-docx-1-cpx-10.1177_21677026231186802 for Associations of DNA-Methylation Measures of Biological Aging With Social Disparities in Child and Adolescent Mental Health by Laurel Raffington, Peter T. Tanksley, Liza Vinnik, Aditi Sabhlok, Megan W. Patterson, Travis Mallard, Margherita Malanchini, Ziada Ayorech, Elliot M. Tucker-Drob and Kathryn Paige Harden in Clinical Psychological Science
Footnotes
Transparency
Action Editor: Aidan G. C. Wright
Editor: Jennifer L. Tackett
Author Contributions
Elliot M. Tucker-Drob and Kathryn Paige Harden contributed equally.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
