Abstract
Background
Fabric lanyards are commonly worn by health care workers (HCWs) and are known to harbor infectious organisms and contribute to the transmission of infection to HCWs and patients. A diverse range of nosocomial pathogens have been found on lanyards, but there are very few studies describing how to successfully disinfect lanyards to break the chain of transmission. Recently, a steam disinfector-dryer device has come on the market, which performs rapid disinfection against nosocomial pathogens and also dries the contents of the device. It was the aim of this study to evaluate steam disinfection-drying as a method to eliminate pathogens from lanyards.
Methods
Thirty-eight strips of new, unused, and autoclaved polyester neck lanyards (4 × 2 cm) were inoculated with 30 (12 Gram-positive + 18 Gram-negative) bacteria and one yeast organism. The inoculated lanyard fabric (five organisms per lanyard strip) was placed into a steam disinfector-dryer device and disinfected for 5 minutes and dried for 30 minutes, in accordance with the manufacturer’s instructions. Following disinfection and drying, the presence of viable organisms on lanyard fabric was evaluated using enhanced microbiological broth culture methods for 48 hours. Control lanyard strips were treated with organisms and left at room temperature without undergoing disinfection and drying procedures.
Findings
Steam disinfection-drying eradicated all test organisms from treated lanyards, with no culturable organisms detected following disinfection-drying, even when employing enhanced bacteriological culture conditions. All test organisms remained viable on the control lanyards.
Conclusion/Application to Practice
Steam disinfection-drying offers a simple method of decontaminating lanyards, producing dry lanyards for immediate reuse. Occupational health practitioners and hospitals should consider assessing the feasibility of adopting this method in their settings to aid in breaking the chain of transmission of nosocomial pathogens via contaminated lanyards.
Background
Identification lanyards worn around the neck of health care workers (HCWs) have been shown to harbor a diverse range of bacterial and fungal pathogens, including skin flora (Alexander et al., 2008), such as methicillin-sensitive Staphylococcus aureus (MSSA; Alexander et al., 2008; Murphy et al., 2017) and methicillin-resistant S. aureus (MRSA; Hogue et al., 2017; Kotsanas et al., 2008); Micrococcus sp. (Cox et al., 2016); Clostridium perfringens (Pepper et al., 2014); Clostridium bifermentans (Pepper et al., 2014); and several Gram-negative organisms, including Proteus sp. (Alexander et al., 2008), Enterobacter cloacae (Kotsanas et al., 2008), Klebsiella sp. (Kotsanas et al., 2008), Acinetobacter lwoffii, and Acinetobacter radioresistens (Hogue et al., 2017; Ting et al., 2015). Furthermore, the emerging pathogenic yeast, Candida auris, has been isolated from a communal cloth lanyard containing the key to the controlled drugs locker, where the lanyard was determined to be involved in the transmission of C. auris in a small outbreak within two intensive care units (ICUs; Patterson et al., 2021). Table 1 provides the details of the studies that examined lanyards that are contaminated with infectious pathogens. A study that examined the presence of viruses found that lanyards were not significantly contaminated with common respiratory or gastrointestinal viruses and are unlikely to be a significant vector of nosocomial infection (Ting et al., 2015).
Previous Studies Examining Colonization of Health Care Workers’ Lanyards With Various Microbiology
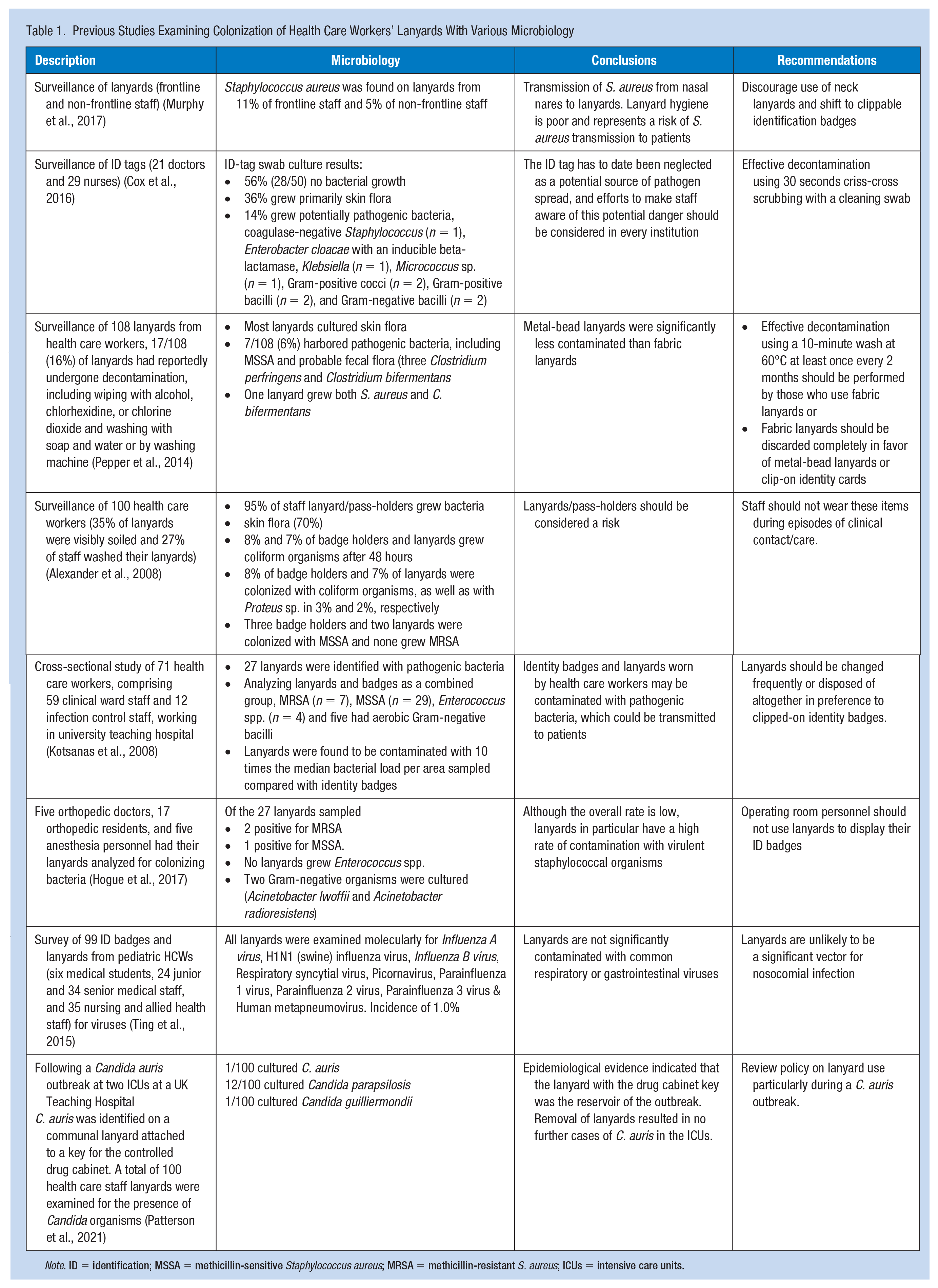
Note. ID = identification; MSSA = methicillin-sensitive Staphylococcus aureus; MRSA = methicillin-resistant S. aureus; ICUs = intensive care units.
Typically, lanyards have been used in health care settings to identify health care staff to patients. For example, the staff identification card with their name, photograph, and job title can be conveniently worn and viewed when using a lanyard. In addition, color-coding of lanyards can be used to differentiate between different types of health care professionals and different levels of seniority of clinicians (General Medical Council, 2017). More recently, the use of lanyards has been extended to patients to help identify those with hidden disabilities, such as those with low vision or hearing, and other disabilities that may not be obvious, such as autism, learning disabilities, dementia, Alzheimer’s disease, anxiety, and other mental health conditions (Anon, 2021a). The emergence of the COVID-19 pandemic has also served to change lanyard wearing practices by the prominent display of HCWs’ pictures on lanyards outside of their personal protection equipment (PPE) to improve staff engagement with residents and clients in care homes while dressed in full personal protective equipment (PPE) (Peart, 2020). Lanyards have also been used to hang face masks during the COVID-19 pandemic (Brickell, 2020). Finally, yellow lanyards were adopted by two health systems in the United Kingdom to identify specialist respiratory nurses who educate, train, and support health care staff during the pandemic (Kerslake et al., 2021).
Today’s health care professional may wear several plastic and fabric-based items in addition to the lanyard, including stethoscopes, pens, USB clips, and identification name badges, all of which require regular disinfection. Typically, plastic identification (ID) badges are disinfected using cold methods such as disinfectant wipes, including bleach and alcohol wipes (Caldwell et al., 2016). Most lanyards, however, are manufactured from woven polyester, ribbed polyester, tubular polyester, satin-like polyester, or braided polypropylene. Employment of wet steam disinfection has not been popular as a means of lanyard disinfection because of the time it takes to dry the fabric lanyard (Rachel Moore personal communication, 2021). Recently, household baby bottle steam disinfector manufacturers have developed a combined novel steam disinfector and drying device (Anon., 2017). Drying is included as an add-on function once the disinfector has gone through a complete cycle of disinfection, whereby internal fans switch on and dry the contents of the disinfector device, utilizing heat from the disinfection stage to aid with product drying. To date, there have been no reports of employment of domestic steam disinfector-dryer devices to disinfect and dry lanyards used by HCWs. Therefore, the aim of this study was to examine employment of a steam disinfector with dryer device to disinfect a wide range of Gram-positive, Gram-negative, and yeast organisms on contaminated lanyards and to subsequently dry lanyards to completeness.
Methods
An experimental design was employed to compare two groups of contaminated lanyards. The experimental group was subject to the steam disinfection-drying treatment and the control group was not treated (not disinfected). Bacteria and yeast organisms (n = 31) were added to 38 sterile strips of fabric lanyard material and these were treated using a steam disinfector-dryer device. Following the completion of disinfection and drying, the inoculated lanyard fabric was assessed for the survival of organisms. The lanyards were also weighed before and immediately post treatment, and at 15 and 30 minutes post treatment to assess the degree of dryness.
The Nuby Natural Touch Steriliser and Dryer (Model No. BB1022-GS; 220–240V; 50–60 Hz; 650W; Nuby UK Ltd. Tyne & Wear, UK) domestic steam disinfector-drier device was employed in this study. Thermal performance of this device has recently been described (Millar et al., 2021). The device consists of five components, namely, the base (containing in-built fans), main body, lower tray, upper tray, and lid. The device can be operated solely in disinfection mode and in disinfection plus drying mode (short drying time [15 minutes] or long drying time [30 minutes]), when in the latter mode, the fan dryer commences 2 minutes after the disinfection mode has ended, without any further heating. All steam disinfection studies were performed at GPS Co-ordinates 54588699, −5.942102 (54°35′19.3″N 5°56′31.6″W) at an altitude 12 m above sea level. The device was operated in accordance with the manufacturer’s instructions.
New and unused polyester neck lanyards (length = 46 cm; width = 2 cm) were cut into approx. 4 cm strips and sterilized by autoclaving at 121°C for 15 minutes and 15 psi. Sterilized lanyard strips were aseptically placed onto the plastic grid floor making up the upper layer of the disinfector-dryer unit and were subsequently inoculated with individual bacteria and yeast organisms (50 µL), equating to a 1.0 McFarland Standard (which corresponds to approx. 3 × 108 colony forming units (cfu)/mL). The lanyards were steamed using disinfector-dryer device for 5 minutes, followed by the 30-minute drying cycle (long drying program). When the disinfection-drying cycle was finished, lanyard strips were aseptically transferred to nutrient broth (10 mL; Oxoid CM0001, Oxoid Ltd., Basingstoke, UK) and incubated for 48 hours at 37°C to recover/enrich any remaining organisms. Broths were checked for microbial growth at 24 and 48 hours, as determined by the development of cloudiness. All broths irrespective of cloudiness were inoculated after 48 hours onto Columbia Blood Agar (CBA) and incubated as above for 48 hours. No growth (thermal inactivation) was recorded when both broths and broth subcultures failed to grow/recover any organisms. As a control, inoculated lanyards were left on the open bench, without steam disinfection-drying and were processed in a similar fashion to those above.
Thirty bacteria (including 12 Gram-positive isolates and 18 Gram-negative isolates) and one yeast were used in this study. Common non-spore-forming nosocomial bacterial and yeast pathogens were selected for employment in this study, which were representative of organisms commonly found on contaminated health care lanyards. These bacteria included two MRSA organisms (hospital-associated + livestock-associated), six MSSA, and a mucoid S. aureus. Bacterial and yeast isolates were subcultured on CBA (Oxoid CM0031), supplemented with 5% (v/v) defibrinated horse blood (Oxoid SR0050) and incubated aerobically for 24 hours at 37°C.
Lanyards were assessed for dryness by comparing their postdisinfectant treatment weight with their baseline weight in grams. Completely dry lanyards were weighed prior to steam disinfection, immediately following steam disinfection, at 15 and 30 minutes post drying. This process was replicated 4 times with one complete lanyard and the mean weights examined at each stage.
Results
Table 2 lists the organisms used for the experiment and the findings that none of the organisms could be recovered from the lanyards after using the steam disinfection for 5 minutes and drying for 30 minutes. This represents an approximate log108 reduction of the initial microbial inoculum, whereas all organisms were successfully recovered from control lanyards that were not subjected to steam disinfection-drying. Following steam disinfection of a lanyard on four occasions, the lanyard gained a mean 6.1% (1.1 g) of its original weight in moisture from steam, and after 15 minutes of drying (Figure 1), it contained 0.5 g (2.7% its original dry weight) in moisture. Drying for 30 minutes as recommended by the device manufacturer removed all moisture added to the lanyard during steam disinfection, returning its weight to predisinfection values.
Effect of Steam Disinfection-Drying on Bacterial and Yeast Pathogens Inoculated Onto Lanyard Fabric Material
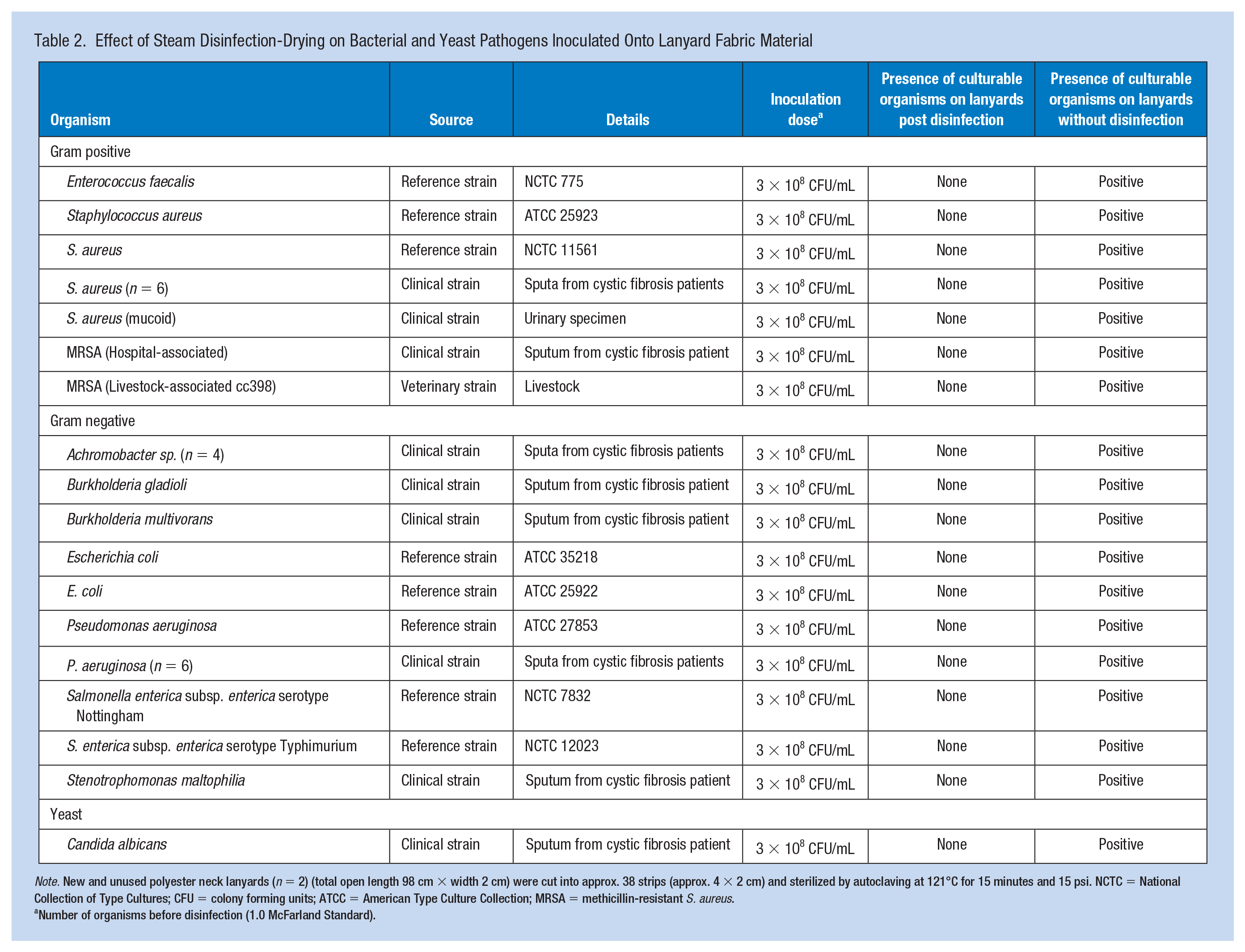
Note. New and unused polyester neck lanyards (n = 2) (total open length 98 cm × width 2 cm) were cut into approx. 38 strips (approx. 4 × 2 cm) and sterilized by autoclaving at 121°C for 15 minutes and 15 psi. NCTC = National Collection of Type Cultures; CFU = colony forming units; ATCC = American Type Culture Collection; MRSA = methicillin-resistant S. aureus.
Number of organisms before disinfection (1.0 McFarland Standard).
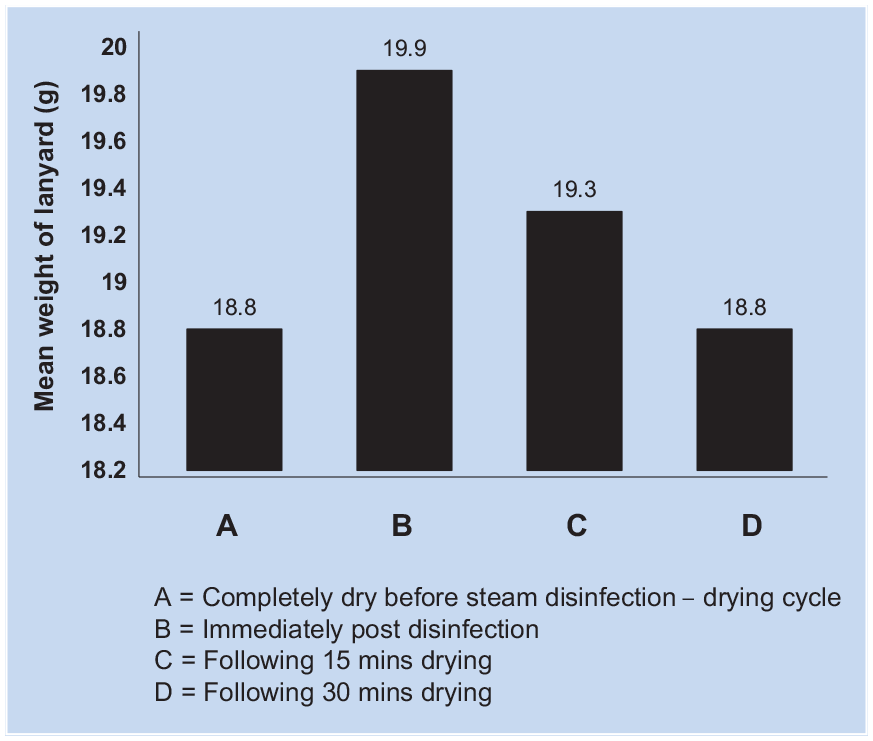
Weight profile of fabric neck lanyard during various stages of steam disinfection-drying.
Discussion
Health care lanyards are prone to contamination when touched by HCWs, patients, or the health care environment. Once contaminated, the lanyard may act as a source of nosocomial pathogens, which may then be transferred to the clean hands of HCWs, when they touch their lanyard (Lopez et al., 2013). The identification card and card holder components of identification badge assemblies may be successfully disinfected with wet chemical surface disinfection techniques such as bleach, but the challenge has been disinfecting the fabric neck lanyard component. Many reports recommend that lanyards be discarded and an alternative identity badge worn (Kotsanas et al., 2008; Murphy et al., 2017). In an unannounced observational study with medical students (n = 12) in a simulated patient examination involving a patient volunteer, 95% of medical students touched their lanyard during the patient examination, with a mean of 7.35 times ± 5.28 (SD; de Jager, 2015). Students touched their lanyards to identify themselves to the patient during introductions, to hold their lanyard to prevent the lanyard from touching the patients during the physical examination, while speaking with the patient or standing at ease in their default position, and when they were doing hand gestures, which also allowed for the collision of hands, stethoscopes, and lanyards (de Jager, 2015). In addition, 70% of students’ lanyards touched the patient at least once (M ± SD = 2.65 ± 2.99; de Jager, 2015).
Disinfection with wet disinfectant and also with wet steam disinfection creates a problem of the lanyard absorbing moisture. If the disinfected lanyard remains wet, either from wet chemical disinfection or from steam disinfection, this is a deterrent for health care staff who prefer to use a dry lanyard; therefore, effective disinfection method requires the final lanyard to be perfectly dry, for optimal health care staff compliance.
Until recently, baby bottle steam disinfectors have only provided a steam disinfection cycle, which has left the disinfected items wet from the steam condensate created during the disinfection cycle. Steam disinfection has become popular both in hospital and with patients at home and is recommended for the disinfection of nebulizer devices in patients with cystic fibrosis (Bell et al., 2020) and for the disinfection of toothbrushes (Millar et al., 2020). More recently, steam disinfector devices have been produced to dry excess moisture from devices and fomites following disinfection.
Best practice would provide HCWs with a sterile lanyard on a daily basis; therefore, the sterilized lanyard would not be a reservoir for any organism, whether in the vegetative state, or if they are a spore-former (e.g., Clostridium difficile). A previous study showed that the mean age of lanyards was 22 months and that 91% of lanyards had never been laundered from 102 health care staff (Murphy et al., 2017). Once contaminated, bacteria can survive on hospital fabrics and plastic surfaces, for 60 days with the Gram-negatives (Neely, 2000) and for 90 days with enterococci and staphylococci (Neely & Maley, 2000). Sterile and disinfected lanyards may quickly become recontaminated with pathogens, but this timeline, as yet, remains unclear. A longitudinal study is therefore needed to address this, but given that contamination is likely to occur from the first wearing of the disinfected lanyard, especially from the wearer’s nose (Raveenthiran, 2017), we therefore recommend that lanyards are disinfected at the first convenient stage after wearing, namely, after the completion of a HCW’s shift.
Steam offers disinfection of vegetative forms of bacterial and yeast organisms, as these organisms are vulnerable to heat in this physiological state. However, employment of steam should not be considered a sterilization technique, as resistant microbial physiological states, including the bacterial endospore, would be inherently refractive to killing by steam methods. For complete and absolute sterilization, autoclaving or ionizing radiation would be required, but these are impractical in a busy clinical setting (Raveenthiran, 2017).
There are several problems associated with the wearing of lanyards, including deep-seated contamination with nosocomial pathogens within the fabric; an inability to eradicate nosocomial pathogens with surface disinfection techniques; transfer of contaminating pathogens from lanyards to other worn items in contact with the lanyard, including stethoscopes worn around the neck; the transfer of nosocomial pathogens from lanyard to patient; the transfer of nosocomial pathogens to the hands of the health care worker wearing the lanyard, after performing adequate hand hygiene; the lack of footprint area in busy wards to adequately disinfect lanyards with cold disinfection methods, including sodium hypochlorite solution; corrosion of metal clips on lanyards due to exposure to potent oxidizing agents (bleach); the difficulty in drying lanyard fabric after cold liquid disinfection; the potential for skin irritation on the neck of health care professionals due to sodium hypochlorite carryover; and the risk of pediatric patients placing the lanyard/identification badge in their mouths while attending clinic.
Even with all of the above associated problems with lanyards, the wearing of these is still very common among health care staff in clinical settings. Many institutional guidelines ban the wearing of these within health care environments and have suggested alternatives to lanyards, including the clipping of identification badges high up on the chest (Kotsanas et al., 2008; Murphy et al., 2017). Therefore, in institutions where the wearing of lanyards is still common practice, it is important to introduce rigor into their disinfection, to protect patient and health care worker safety. Hospital administrators, financial administrators, and health care purchasing agents in collaboration with occupational health, infectious disease, and microbiology specialists need to assess whether to allow lanyards and, if they do, how to disinfect these optimally and safely, to protect HCWs and patients. Hence, to help make lanyard use safer, new evidence-based guidelines are urgently needed, including description of pragmatic disinfection methods and a schedule of planned disinfection.
This study has demonstrated that steam disinfection with drying can be performed using devices that are widely available, with excellent pathogen eradication. The next stage would be to perform an evaluation in the health care setting of this method, to assess logistics, practicalities, method compliance, and staff engagement.
Application to Practice
Domestic steam disinfection-drying, when performed properly, offers a simple, versatile, and widely available technology for the elimination of vegetative forms of common nosocomial pathogens, producing dry lanyards for immediate reuse, without the need to employ wet disinfection methods and wait for lanyards to dry. Therefore, our study recommends that lanyards are disinfected on a daily basis using a steam disinfector with dryer. Occupational health practitioners and hospitals should consider the adoption of such disinfection-drying methods to help break the transmission of nosocomial acquired infections, which is important in reducing the burden of disease in both patient and health care worker.
Supplemental Material
sj-doc-1-whs-10.1177_21650799211012653 – Supplemental material for Evaluation of a Domestic Steam Disinfector-Dryer Device for Disinfection of Health Care Workers’ Identification Lanyards
Supplemental material, sj-doc-1-whs-10.1177_21650799211012653 for Evaluation of a Domestic Steam Disinfector-Dryer Device for Disinfection of Health Care Workers’ Identification Lanyards by Beverley C. Millar and John E. Moore in Workplace Health & Safety
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Biographies
Beverley C. Millar is a healthcare microbiologists working at the Northern Ireland Public Health Laboratory, Belfast City Hospital, with an interest in the provision of robust and pragmatic disinfection methods to improve patient and staff safety within healthcare.
John E. Moore is a healthcare microbiologists working at the Northern Ireland Public Health Laboratory, Belfast City Hospital, with an interest in the provision of robust and pragmatic disinfection methods to improve patient and staff safety within healthcare.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
