Abstract
In the United States, there are more than 120,000 nail salons in which workers could be potentially exposed to a number of volatile organic compounds (VOCs) used in various procedures. Measuring workers exposure in the field is time-consuming and could be very expensive. The purpose of this study was to estimate the VOC levels in the proximity of workers in nail salons through simulating the application process of some popular nail polishes in a laboratory chamber. The worst-case scenario was defined as a worker’s exposure during nail polish application to one set of fingernails every 15 minutes for an 8-hour shift (total nail sets = 32). Nail polish was applied on paper plates in a flow-controlled test chamber. Air was sampled during the application of five different nail polishes for 8 hours using passive air samplers and the experiment was triplicated. Passive samplers were used for VOCs and formaldehyde. In this worst-case scenario setting, a total of 17 VOCs were detected, with eight that were found in all the samples. The mean concentration of butyl acetate (161-330 ppm, parts per million) and ethyl acetate (440 ppm) exceeded the threshold limit value (TLV) of 150 ppm and 400 ppm, respectively. Formaldehyde was analyzed separately and the mean concentrations exceeded the TLV of 0.10 ppm in all types of nail polish, ranging from 0.12 ppm to 0.22 ppm. Occupational safety and health professionals could use these data to increase awareness of workers’ potential exposure to high levels of VOCs in nail salons and recommend practical measures to reduce potential exposures.
Introduction
With the rise in popularity of nail care services, the number of nail salons has dramatically grown in recent years (Goldin et al., 2014). It is estimated that in the United States, there are more than 120,000 nail salons employing a high proportion of Vietnamese immigrants (Quach et al., 2013). Nail products such as nail polish, nail polish remover, nail hardener, and artificial nails can contain a variety of potentially hazardous chemicals (U.S. Environmental Protection Agency [EPA], 2007; Roelofs & Do, 2012). Hazardous chemical ingredients in nail polishes include volatile or semivolatile compounds such as formaldehyde, toluene, dibutyl phthalates (DBPs), ethyl acetate, butyl acetate, and methyl ethyl ketone (Goldin et al., 2014; Roelofs & Do, 2012; U.S. EPA, 2007; U.S. Food and Drug Administration, 2016). Due to short-term and long-term adverse health effects of these chemicals, ranging from sensory irritations to cancer, a number of occupational exposure studies have been conducted related to the nail care industry. However, there is limited evidence that the exposures are above occupational exposure limits (OELs; Alaves, Sleeth, Thiese, & Larson, 2013; U.S. EPA, 2007; Quach et al., 2013; Roelofs & Do, 2012). Most studies have been focused on the sampling of such hazardous compounds in nail salons (Alaves et al., 2013; Gjølstad, Thorud, & Molander, 2006; Goldin et al., 2014; McNary & Jackson, 2007; Park, Gwak, & Choi, 2014; Quach et al., 2011; Tsigonia et al., 2010). However, large variations between studies, as well as differences between nail salons have been frequently reported. Most often reported variations were due to the use of different types of nail products and services, the operation (type of task performed) and type of ventilation, study design, sampling methods, and reluctance of participation, which makes it difficult to determine the true exposure to the hazardous compounds. The measurement of the hazardous chemical emissions/concentrations in a controlled setting would thus help to better understand field exposure by identifying the most common chemicals and their predicted maximum levels. It would also allow occupational safety and health professionals to establish guidelines to reduce exposures. To the best of our knowledge, there are very few laboratory and computational simulation studies on exposure to the hazardous compounds in nail salons. Computational modeling of health risk from toluene and DBP in nail lacquers was performed in which toluene air concentration was computed using the two-zone model for salon patrons and nail technicians, and the well-mixed room model for home users; findings indicated approximately 3 ppm and 6 ppm, respectively, when applying the maximum concentration of 190,000 mg/kg of nail products (Kopelovich, Perez, Jacobs, Mendelsohn, & Keenan, 2015).
We aimed to advance this body of research by directly measuring the volatile organic compound (VOC) emissions from nail polishes by simulating the application process and then comparing the potential exposures resulting from this simulation to the corresponding OELs. We employed a controlled-air flow chamber and quantified the VOCs emitted into the test chamber by applying nail polishes to determine the VOC emissions directly emitted from the nail polishes.
Method
Brands and colors of five nail polishes were selected based on observations made at a local nail salon: Big Apple Red and Miami Beet from O·P·I (OPI Products Inc., N. Hollywood, CA), Red Pearl and Seduce Me from China Glaze (American International Industries, Los Angeles, CA), and Framed in Red from FingerPaints (High Intensity Products Inc., Santa Monica, CA). The three nail polish brands used in our simulation were all solvent-based and had similar composition based on the list of ingredients provided by the manufacturers. The main categories of compounds in each of them included solvents (e.g., ethyl acetate, butyl acetate, propyl acetate, isopropyl alcohol, diacetone alcohol), film formers (e.g., nitrocellulose, tosylamide/formaldehyde resin, dimethicone), plasticizers (e.g., trimethyl pentanyl diisobutyrate, triphenyl phosphate, ethyl tosylamide, camphor), suspending agents (e.g., stearalkonium bentonite, stearalkonium hectorite), and colorants (e.g., titanium dioxide, FD&C red 6, FD&C yellow 5, aluminum lake). One of the brands, FingerPaints, listed fewer ingredients, but all of the listed compounds were found in the other two brands.
A 114.5-liter plastic chamber was purchased, and rubber gloves were attached on the front side of the chamber similarly to a glove box (Figure 1). A mass flow controller (Smart-Trak Sierra Instruments, Inc., Monterey, CA) was set to 1.9 liter/minute to generate one air exchange per hour in the chamber. A compact fan was placed in the chamber to provide air mixing. The chamber was place on the lab benchtop and simulation was performed at standing position. Nail polish was applied to a paper plate to simulate the act of painting a full set of fingernails every 15 minutes (approximately 2 mins application plus 13 mins resting) based on the observations made at a nail salon. A formaldehyde vapor monitor (F-50 Advanced Chemical Sensors, Inc., Boca Raton, FL) and an organic vapor monitor (OV-00 F-50 Advanced Chemical Sensors, Inc., Boca Raton, FL), which was analyzed for 81 compounds were used. Whenever VOC is mentioned hereafter, formaldehyde is excluded because it was treated as a separate compound. Monitors were hung from the top of the chamber with a string where the height of the samplers would be matched to the height of the breathing zone of a nail salon employee, assuming that the worker would be hunched forward during the nail polish application process.
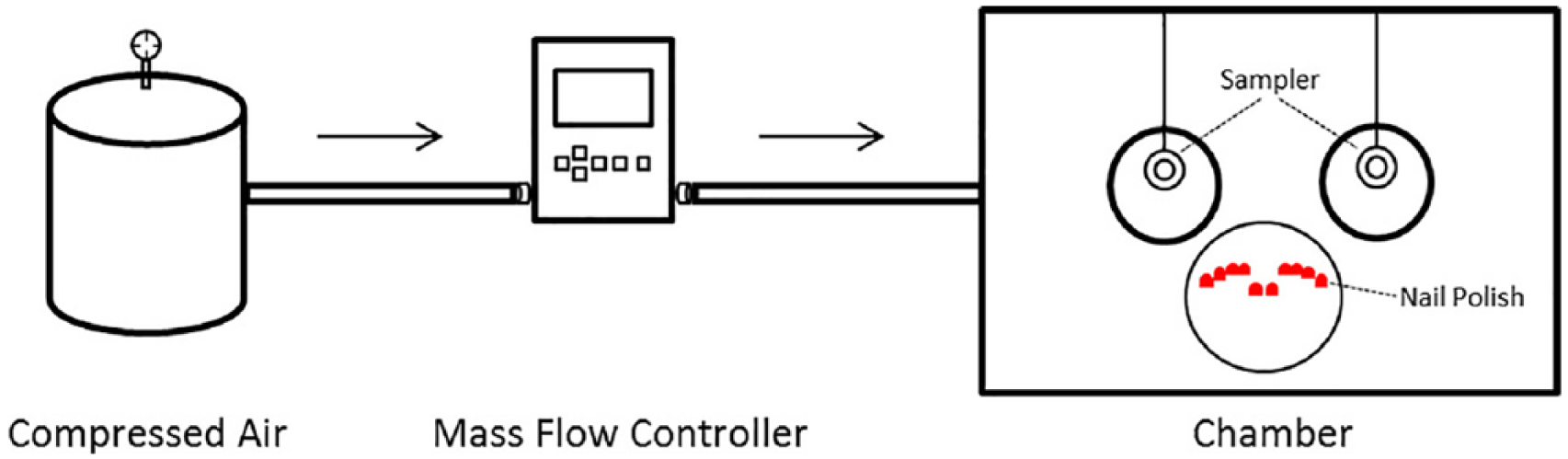
Diagram of the laboratory simulation set-up.
A total of 32 sets of fingernail paintings were simulated to achieve the 8-hour exposure without any breaks (for a worst-case scenario). Each nail polish was tested over three replicates: with a total of 15 samples for formaldehyde and 15 samples for VOCs. The first and second sampling rounds were conducted using the same bottle of a nail polish, whereas the last round was conducted using a new bottle to determine if there is difference between using a fresh bottle and an already opened one. Approximately half of a bottle (14-15 milliliter) was used in each round. To account for the background and potential traces of compounds already existing in the samplers, “blank” samples are usually collected. In this case, the blank samples were collected from unopened badges employed in the same environment as the opened badges. Blank samples for each (i.e., formaldehyde and organic vapor monitors) were collected in the same manner as the other samples for each round. After each sampling round, the chamber was flushed with air overnight to prevent contamination of further sampling. At the completion of each round, samples were sent to Advanced Chemical Sensors (#102047) for analysis. For formaldehyde analysis, NIOSH Manual of Analytical Methods (Eller, 1994) was used. VOCs were analyzed using EPA Method TO-15 in SCAN mode. Reporting limits (for these methods) were 10 parts per billion (ppb) for formaldehyde and 0.1 ppb for most VOCs scanned for an 8-hour sampling period. The results were reported in ppm and arithmetic mean concentration to be comparable with the currently available literature.
Results
Formaldehyde was detected in all samples except one, the FingerPaints Framed in Red from the second sampling round. The formaldehyde concentration was up to 0.35 ppm in one of the samples. From the VOC SCAN, a total of 17 compounds were detected in at least one of the 15 samples. Among those reported, eight VOCs including 1-butyl alcohol (1.87-22.32 ppm), acetone (0.56-8.07 ppm), butyl acetate (4.46-444.09 ppm), ethyl acetate (157.40-563.00), ethyl alcohol (2.79-14.66 ppm), isopropyl acetate (3.67-28.34), isopropyl alcohol (36.56-99.79 ppm), and n-propyl acetate (0.05-122.90 ppm) were found in all samples regardless of the batch and color of the nail polish. Methyl acetate was also found in all samples except one, which was sampled for the FingerPaints Framed in Red from the second sampling round. Except that sample, methyl acetate concentration range was 0.02 ppm to 0.06 ppm. In addition, we found no conclusive pattern when we examined if differences in concentration between the already opened versus fresh bottle batches.
Table 1 shows the arithmetic mean concentrations in ppm and standard deviations for the three sampling rounds for those 10 compounds mentioned above. In addition, the table shows the OELs: permissible exposure limit (PEL), recommended exposure limit (REL), and threshold limit value (TLV) for the same compounds in ppm. Formaldehyde, butyl acetate, and ethyl acetate exceeded at least one of the OELs in at least one brand/color. The average formaldehyde level was over the REL and TLV (0.016 and 0.10 ppm, respectively) in every nail polish, with the O·P·I Big Apple Red being the highest, 0.22 ± 0.04 ppm. The average concentration of butyl acetate exceeded all OELs for all tested nail polishes, FingerPaints Framed in Red being the highest, 330 ± 99 ppm. The average concentration of one nail polish product, China Glaze Seduce Me, exceeded all OELs (400 ppm) for ethyl acetate. The relative high standard deviation, due to variations in concentration between sampling within the same nail polish could be explained by the variance in the amount of nail polish applied. Although we tried to keep similar application times, there was no quantification process involved. This, however, could be more realistic because the professionals would not have the same amount applied to different customers. In fact, high standard deviations have been often reported in the literature from on-site nail salon measurements (Gjølstad et al., 2006; McNary & Jackson, 2007; Park et al., 2014; Quach et al., 2011). In addition, ethyl acetate, isopropyl alcohol, and methyl acetate had a lower concentration in the second round than in the first and third rounds, indicating that those compounds may have evaporated from the old bottles. However, no supporting argument could be made for this phenomenon as they were similarly volatile as other compounds.
Average Concentration of the Volatile Organic Compounds Found in Nail Polish Samples.
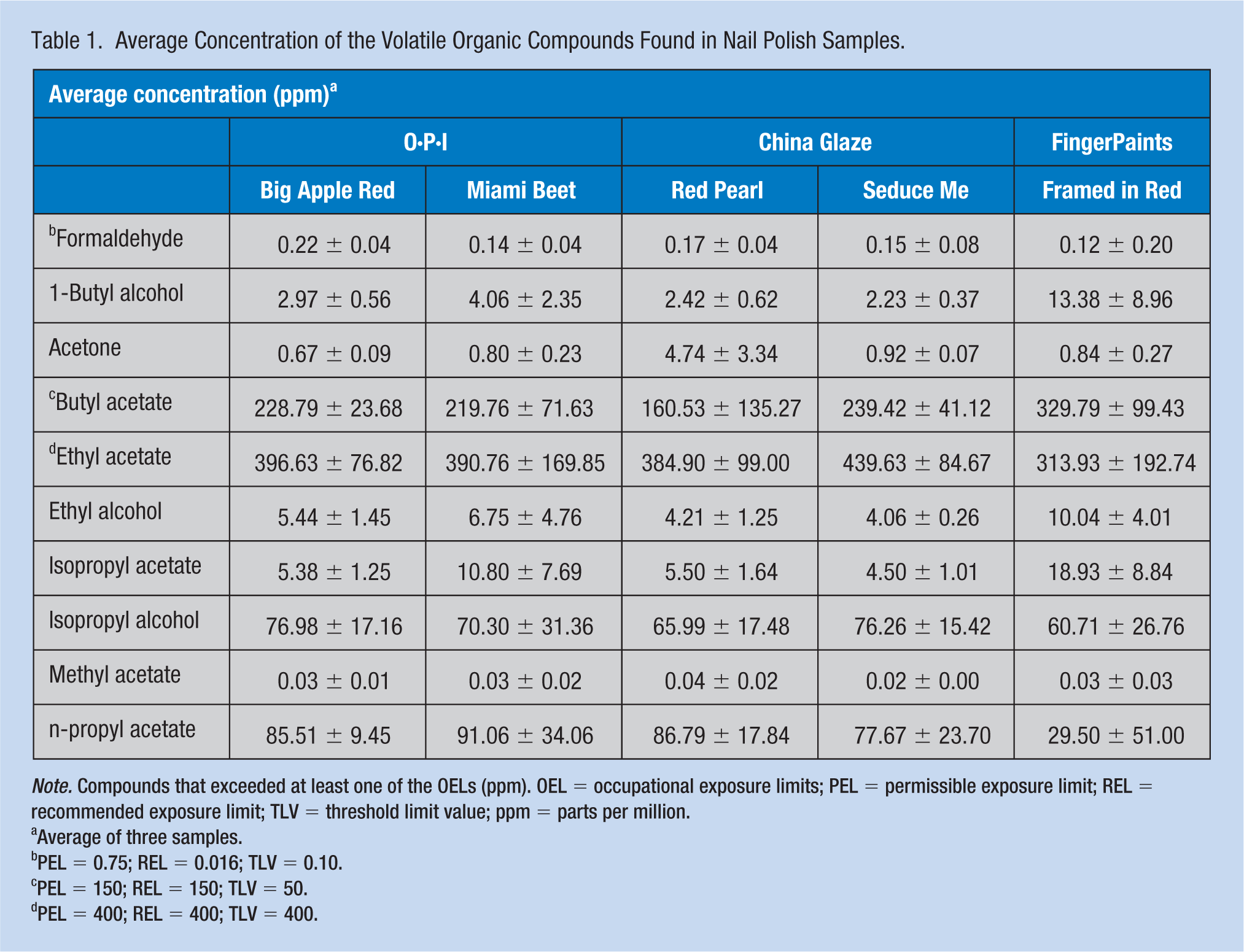
Note. Compounds that exceeded at least one of the OELs (ppm). OEL = occupational exposure limits; PEL = permissible exposure limit; REL = recommended exposure limit; TLV = threshold limit value; ppm = parts per million.
Average of three samples.
PEL = 0.75; REL = 0.016; TLV = 0.10.
PEL = 150; REL = 150; TLV = 50.
PEL = 400; REL = 400; TLV = 400.
Discussion
There are health effects associated with the three compounds, which exceeded the OELs. For example, exposure to formaldehyde can irritate the skin, throat, lungs, and eyes. Repeated exposure to formaldehyde can possibly lead to myeloid leukemia and rare cancers, including cancers of the paranasal sinuses, nasal cavity, and nasopharynx (Main & Hogan, 1983). Butyl acetate is a synthetic solvent, irritant to lung, eyes, and skin (Iregren, Löf, Toomingas, & Wang, 1993). Ethyl acetate is also a synthetic solvent, irritant to lung, eye, and skin (Clayton & Clayton, 1993-1994).
Our findings are similar to those reported in other studies with regard to elevated exposures to formaldehyde and VOCs. For example, area air sampling at breathing zone height was performed randomly at selected 12 nail salons in Utah (Alaves et al., 2013), which reported that formaldehyde measures ranging from 0.09 to 0.032 ppm (arithmetic mean of 0.017 ± 0.007 ppm) with seven salons found to be above the NIOSH REL (0.016 ppm). Possible nonconformity by the nail salons such as unusual ventilation, different ventilation systems, and the effect of building age were addressed as limitations. In a separate study, of 30 California nail salons, the arithmetic mean of formaldehyde was initially 0.022 ± 0.012 ppm and after correction for outside concentration, the reported range was from 0.0012 to 0.0038 ppm, with an average toluene concentration of 0.236 ± 0.258 ppm (McNary & Jackson, 2007). These authors concluded that neither nail technicians nor clients were at risk from the exposure to formaldehyde and toluene in nail products. In another study conducted in 21 Boston salons, total VOC (TVOC) concentration varied across the nail salons, ranging from 0.061 to 38 ppm; however, results were generally consistent within each salon (Goldin et al., 2014). This study might not reflect the true exposure as sampling was conducted at a few feet distance from workers and customers. In a study including eighty Vietnamese workers from 20 California nail salons, (Quach et al., 2011) personal air sampling during work shifts for each employee with two to three measurements on nonconsecutive days, reported the arithmetic means of ethyl acetate, isopropyl acetate, and toluene concentration of 0.53 ± 0.71, 0.04 ± 0.02, and 0.15 ± 0.15 ppm, respectively. Although the results were much less than the OELs from the California OSHA or U.S. federal OSHA standards, the toluene level was higher than the California EPA recommendation for ambient air of 0.08 ppm. Table top ventilators used for certain nail services lowered toluene and ethyl acetate concentrations.
Our study had some limitations worth noting. First, the sampling results were not corrected using blanks because a single blank was collected for each round of measurements, and eight out of 10 compounds were found in at least one blank sample. Because the amount found in blanks was within 3% of the highest concentration measured for that compound and blanks were taken only when starting each batch (i.e., every fifth nail polish), the results were not corrected with blanks. Furthermore, the context of studies like this in a completely controlled experimental set-up should be considered, as the experiment was set up in a shared laboratory with other researchers where cross-contamination, even if unlikely, is possible. Second, our analytical results showed that similar compounds and comparable amounts of each compound were found in the selected nail polishes from different manufacturers. However, we examined only a limited number of nail polishes and thus, more examinations of a variety of brands and colors would be needed to make the results applicable to a variety of nail salons. In addition, the concentration of our samples may have been slightly higher than what we reported as the conversion temperature applied was 24°C; whereas, the OELs use 25°C. However, considering only 1°C difference, the results should not have been considerably affected. Finally, the results were inevitably higher than possible “real life” exposures in a salon because the small chamber size limited the dilution of the compounds.
Conclusion
A laboratory simulation of VOC emission by nail polish was performed to investigate the potential exposure level of nail salon workers. An airflow-controlled chamber was used to obtain the true concentrations of VOCs that would be emitted. A worst-case scenario was assumed and five different nail polish colors from three different manufacturers were examined. Eight VOCs were found in all samples including 1-butyl alcohol, acetone, butyl acetate, ethyl acetate, ethyl alcohol, isopropyl acetate, isopropyl alcohol, and n-propyl acetate. Formaldehyde, butyl acetate, and ethyl acetate exceeded at least one of the OELs (i.e., PEL, REL, or TLV) in at least one of the batches from all nail colors. Assuming little variation in the ingredients of the nail polishes between manufacturers, our results indicate that there would be a potential for overexposure to those VOCs, especially when working in a confined nail salon. Preventive measures to lower exposure levels should be considered by owners, managers, and occupational safety and health professionals. Simple measures, such as installing desktop fans to increase ventilation and limitation of time when nail polishes are applied by workers could be recommended and easily implemented to reduce potential exposures.
Implications for Occupational Health Nursing Practice
The occupational health nurses could use our material to raise awareness of the potential exposures encountered in the nail salon industry and recommend owners and workers to use specific control methods, such as increased ventilation. Occupational health nurses can also use these results found here to educate the providers on the potential exposures, health risks, and controls.
Applying Research to Practice
Nail salon workers could be potentially exposed to harmful chemicals that are part of nail care products such as nail polishes. Measuring personal exposure in the field could be expensive and intrusive, so to identify the chemicals potentially emitted during nail polish application and the range of the concentrations, a laboratory simulation was performed. Based on the results obtained here, considering the worst case scenario, formaldehyde, butyl acetate and ethyl acetate were found at levels that exceeded some occupational exposure limits. It is important to raise awareness of potential exposures and inform owners and workers of some simple practical control methods such as installing a benchtop fan or increasing the air ventilation in the room. These practices could significantly reduce the concentrations of the emitted chemicals, reducing the risk of exposure.
Footnotes
Authors’ Note
This study does not represent the views of the authors’ affiliations; and its contents are solely the responsibility of the authors and do not necessarily represent the official views of NIOSH (National Institute for Occupational Safety and Health).
Conflict of Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the University of Alabama System Collaborative Research Initiation (Grant No. 3116656.000.213116656) and in part by The Deep South Center for Occupational Health and Safety (Grant No. 5T5T42OH008436 from National Institute for Occupational Safety and Health, NIOSH).
Author Biographies
Tasha Heaton is an Associate Environmental Health Specialist at Wisconsin State Laboratory of Hygiene, University of Wisconsin-Madison. She earned a MPH degree at the University of Alabama at Birmingham with a focus on Industrial Hygiene in 2015, and is an experienced industrial hygiene practitioner.
Laura K. Hurst graduated from the University of Alabama at Birmingham in 2017 with a MSPH degree in Industrial Hygiene. She is an experienced industrial hygienist with a demonstrated history in the profession, her experience including work stages at CSX and Amerex Co. Currently, Laura is an industrial hygienist at Georgia Power Company in Atlanta.
Azita Amiri is an assistant professor in the College of Nursing at University of Alabama in Huntsville. She obtained her PhD from the University of Alabama at Birmingham, and her dissertation research focused on formaldehyde exposure during pregnancy. Her current main research area is on the chronic effects of indoor air pollution.
Claudiu T. Lungu is an associate professor in the department of Environmental Health Sciences at UAB. His research interest is in industrial hygiene, particularly in developing new sampling methods for improved exposure assessment and development of lite respiratory protection devices for dual, particular matter and gas use.
Jonghwa Oh is an assistant professor in the Department of Environmental Health Sciences at University of Alabama at Birmingham (UAB). She earned a PhD in Environmental Health Sciences (Industrial Hygiene track) from UAB. Her main research area is on improving the current occupational exposure assessment and control methods.
