Abstract
B virus is endemic in macaque monkeys, which are frequently used for research studies. B virus is a risk for every employee working with macaque monkeys, their tissues or cells. Although the risk for B virus infection is low, the risk of death or permanent neurological deficit is high if an exposure is not promptly evaluated and treated. Researcher training, routine use of personal protective equipment, first aid protocols, and prompt reporting to a provider knowledgeable about B virus treatment are essential to prevent this 70% lethal infection in untreated humans. This article presents the history and pathogenesis of B virus, first aid, treatment, and prevention.
Keywords
Because of their genetic, anatomic, and physiologic similarities to humans, nonhuman primates are commonly used as models for biomedical research (Huff & Barry, 2003). Macaque primates are not endangered in the wild and can easily be bred and maintained in captivity (Cawthon, 2005). Research studies with macaque primates enable discoveries that can be applied to humans, bridging the gap between science and medicine. However, exposure to macaque monkeys poses potentially lethal risks to researchers and animal care workers through transmission of B virus.
The actual number of human B virus infections is unclear. In Centers for Disease Control and Prevention (CDC), National Institutes of Health (2009) publication,
Macacine herpes virus (i.e., B virus, formerly known as Cercopithecine herpes virus 1) is classified as a Biosafety Level 4 biologic agent, requiring the same research precautions for growth and propagation as the Ebola virus (Jeffrey et al., 2002). B virus, endemic in macaque monkeys, is closely related to the herpes simplex virus in humans, and most macaques carry B virus without overt signs of disease. B virus transmission between macaques occurs through contact with infected lesions of the skin, mucous membranes, or genitalia. As macaques age and become more socially and sexually active, viral exposure and infection increases markedly. The prevalence of anti-B virus antibodies in wild macaques as well as colonies where animals are not kept separate from one another can range from 70% to nearly 100% (Elmore & Eberle, 2008). Infection in macaques is lifelong with periodic, reactivation of the virus, which remains latent in the trigeminal and spinal ganglia serving the site of the infection (Ritz, Curtis, Buttery, & Babi, 2009). Reactivation of the virus from the latent state can result in shedding the virus in body secretions such as saliva, urine, and genital secretions (Estep, Lihem, & Wong, 2010). Most recurrent infections are asymptomatic, making it difficult to know whether the primate is actively shedding virus. Reactivation and possible viral shedding appear during the breeding season and when the primates are stressed during social challenges, transportation, or placement in new housing (Mitsunaga, Nakamura, Hayashi, & Eberle, 2007). However, the percentage of infected monkeys who shed virus at a given time is thought to be only 1% to 2% (Jones-Engel, Engel, Heidrich, Chalise, & Poudel, 2006).
Monkeys routinely test negative for the presence of B virus when acquired from a reputable vendor. However, attempts to maintain B virus-free colonies have failed because of difficulty in detecting seronegative latent infections. In one retrospective study, even with an aggressive surveillance program to establish a B virus pathogen-free colony, seropositive macaques were detected as late as 7 years into the program (Hilliard & Ward, 1999). Thus, risk for human infection must be considered for every potential macaque exposure.
The first documented case of human B virus occurred on October 22, 1932: A 29-year-old researcher, Dr. William Barlet Brebner, who was researching the poliomyelitis virus, was bitten by a macaque monkey. A neurological illness ensued and, 18 days later, he died of respiratory failure from acute ascending myelitis. A sample of his neural tissue revealed an unfilterable agent, similar to the herpes simplex virus, which was later named B virus after Dr. Brebner (Pimentel, 2008).
B virus is usually transmitted to humans via infected saliva through monkey bites or scratches, but cases have been documented from needlestick injuries or other laboratory-related accidents, cage scratches, or mucous membrane exposures to monkey body fluids (Jensen, Alvardo-Ramy, Gonzalez-Martinez, Kraiselburd, & Rullan, 2004). B virus is generally not found in monkey serum or blood, but if these products are obtained through venipuncture, they can be contaminated by infected monkey skin (CDC, National Institutes of Health, 2009). Several airborne B virus infections also have been reported (Tischer & Osterrieder, 2010). Only one case of human-to-human transmission has been documented after a woman applied hydrocortisone ointment to her husband’s infected skin lesions and then applied the same ointment to dermatitis on her finger (CDC, 1987). Deep wounds, especially bites, as well as wounds on the head, upper body, or neck, are considered high-risk exposures (Reme et al., 2009). However, transmission of the virus in past cases has often been associated with no more than a superficial scratch or puncture (Hilliard, 2007).
The incubation period varies from 3 days to 5 weeks. In some, but not all infections, a vesicular rash at the exposure site is associated with numbness, itching, tingling, pain, or local lymphadenopathy. Paresthesias may develop and travel proximally along the affected extremity. Other symptoms may include myalgias, inability to use the affected limb, fever, abdominal pain, sinusitis and conjunctivitis. With central nervous system (CNS) involvement, the virus may spread from the peripheral nerves to the spinal cord and brain; symptoms include meningismus, nausea, vomiting, headaches, confusion, cranial nerve palsies, ataxia, seizures, hemiplegia, hemiparesis, respiratory failure, and coma (Akhras & Blackwoods, 2013). Even with antiviral therapy and supportive care, most individuals with CNS involvement die, and those who survive often have serious neurologic sequelae (Huff & Barry, 2003).
First Aid
Immediate first aid is crucial during the first few minutes after injury or exposure to decrease the risk of infection. Herpes simplex virus can enter sensory nerve endings within 5 minutes of exposure, and B virus is likely to infect just as quickly (Huff & Barry, 2003). Wounds must be thoroughly washed and scrubbed with detergent soap, povidone-iodine, or chlorhexidine to destroy the virus lipid envelope and inactivate the virus. After a thorough cleansing, the wound should be irrigated with running water for 15 to 20 minutes. Only after cleansing should a wound culture be taken to avoid inoculation of the virus from the surface deeper into the wound (CDC, 2014).
Eye or mucous membrane exposures must be irrigated with water for 15 minutes as chemical agents are too harsh for use in these areas. After washing and irrigation for 15 minutes, an injured worker must immediately notify the supervisor and seek health care from the occupational health service, Emergency Department, or a provider familiar with treating B virus. No cases of B virus infection in humans have been reported when the wound was promptly cleaned or when the wound was cleaned followed by postexposure prophylaxis (CDC, National Institutes of Health, 2009).
Treatment
A detailed report of the injury should include the time and type of exposure, species of primate, personal protective equipment (PPE) in use at the time of the injury, and the time and adequacy of wound cleansing. The personal health history of the injured worker should be reviewed as well as the monkey’s health history, the research being conducted, and the monkey’s current health status.
A blood specimen should be collected from the employee on the day of the injury to establish a baseline antibody level. A blood specimen and immediate cultures from the involved monkey should also be obtained, if possible. The blood samples and wound cultures are sent to one of three laboratories in the United States that conducts diagnostic testing for herpes B virus. The B Virus Research and Source Laboratory at Georgia State University in Atlanta is available 7 days per week, 24 hours per day for phone consultation. Because of cross-reactivity with herpes simplex virus antibodies, a positive serological test result for B virus should be confirmed with a Western blot or enzyme-linked immunosorbent assay (ELISA; Akhras & Blackwoods, 2013).
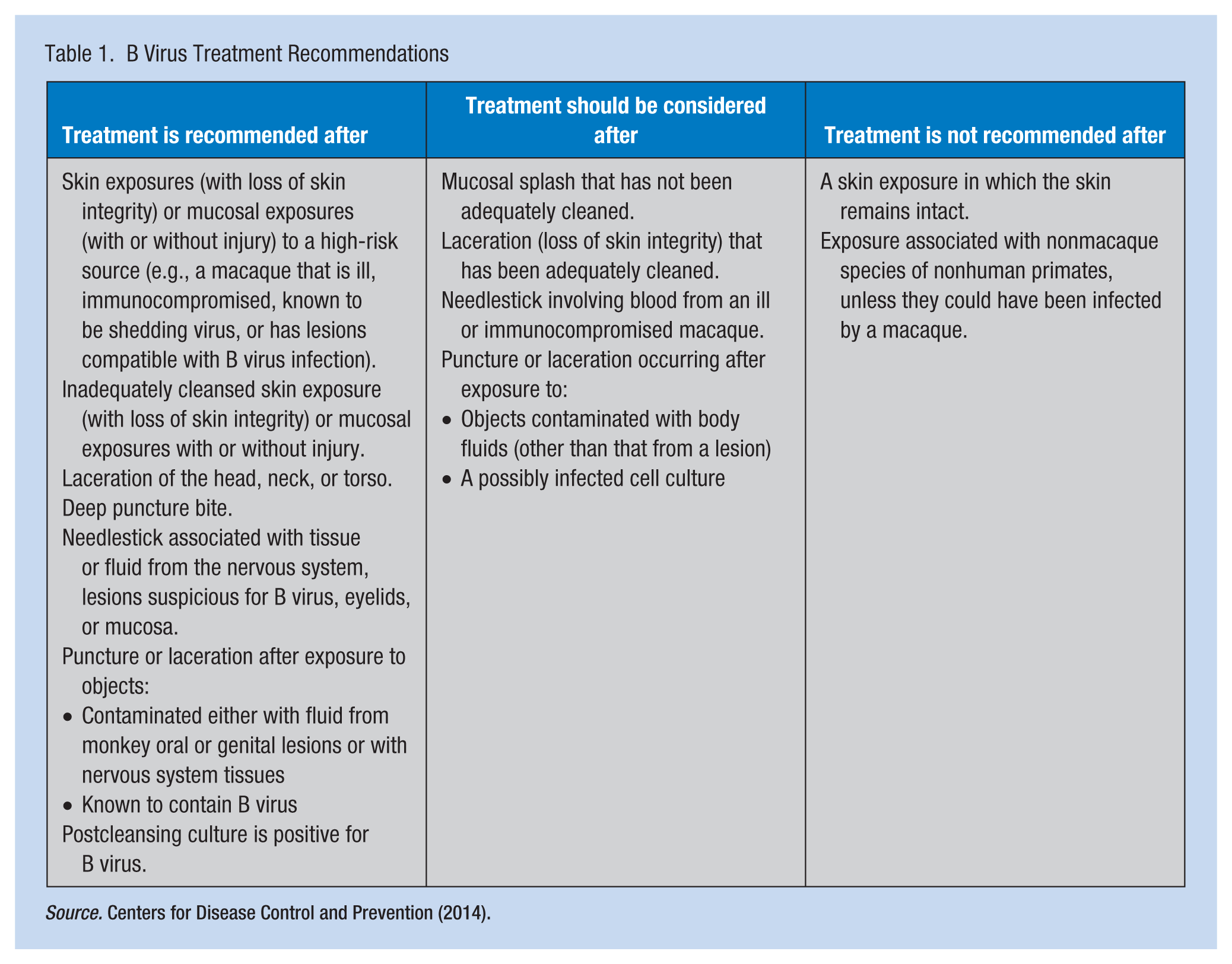
Treatment with antiviral medication, when necessary, should begin as soon as possible, preferably within the first few hours after exposure (Jeffrey et al., 2002). Treatment options outlined by the CDC for B virus (synonyms: herpes B, monkey B virus, herpes virus simiae, and herpes virus B), last updated in 2014, appear in Table 1. Treatment medications appear in Table 2.
Herpes B Medication Recommendations
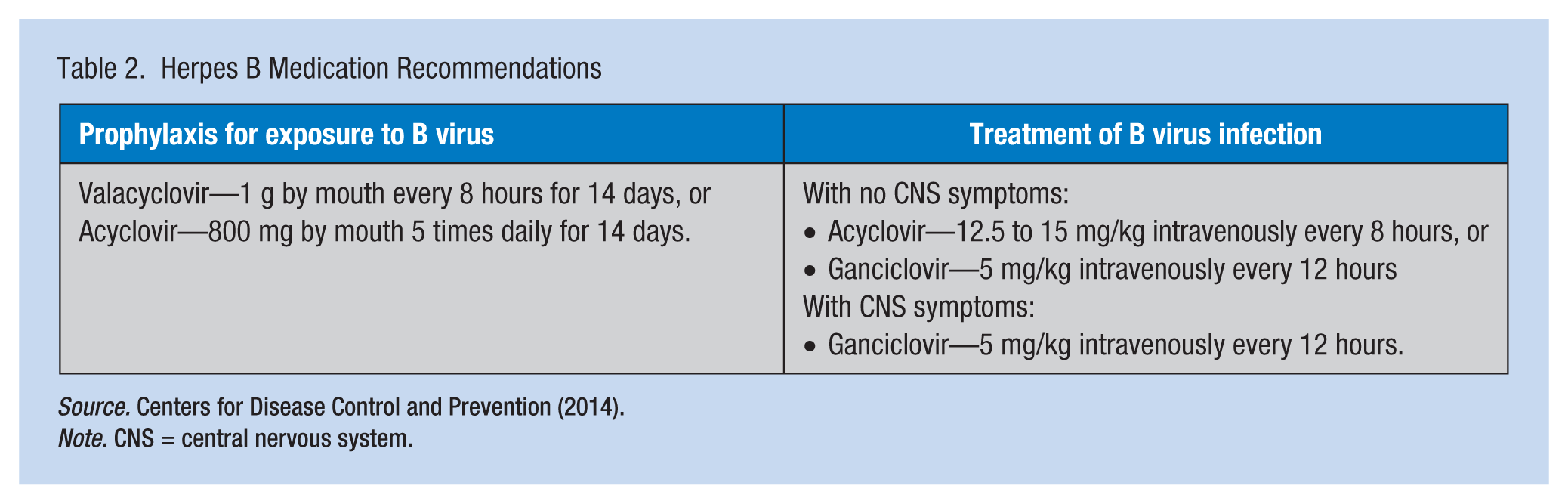
Antiviral agents are well tolerated, but counseling should include possible side effects and the importance of completing the medication prescribed. The exposure site is evaluated by an occupational health professional once weekly for 4 weeks. Antiviral medication compliance and signs or symptoms of B virus infection, such as flu-like illness, fever, severe headache, infection, or vesicles at the wound, are recorded. Repeat serologic testing should be initiated between 14 and 21 days after initial exposure to assess seroconversion. Employees who have received postexposure prophylaxis should have a third blood test 1 month after completing medication because the antiviral agent may delay the development of an antibody response to B virus (National B Virus Resource Center, n.d.). Those with an initial positive wound culture for B virus must have conjunctivae, oropharynx, and any unhealed skin lesion cultured 1 to 2 weeks after completing the prescribed course of antiviral medication to detect any viral shedding (Jeffrey et al., 2002). The presence of any sign of B virus infection necessitates intravenous IV therapy and evaluation by an infectious disease specialist. Early diagnosis and treatment with antiviral medication has reduced the death rate of 70% for untreated B virus infections (CDC, 2014). Survivors of B virus who are untreated may have B virus-specific antibodies for decades with periodic fluctuation, suggesting periodic reactivation of latent B virus. Two cases of morbidity related to reactivation of latent B virus have been documented (Vasireddi & Hilliard, 2012).
Prevention and Reporting
Prevention of B virus infection requires planning and education of personnel working with macaque primates, with updates at least annually. Equipment and supplies for first aid must be readily available, such as an eye wash station for eye exposures. Signage regarding first aid procedures should be visible in all work areas with macaque monkeys. Immediate first aid and barrier precautions are key to prevention of B virus infection. PPE is essential when working with macaques including protective gowns or coats, gloves, and face shields. In addition, work practices with macaque monkeys should include humane restraint methods that reduce the risk of bites and scratches (CDC, 2014).
Those who work with macaque monkey or their cells and tissues should carry a medical alert card in the case of an emergency to inform health care workers about their care. Any B virus infection must be reported to the local health department. A vaccine for B virus is currently not available.
Summary
Prevention of B virus infection requires rigorous adherence to institutional policies for handling macaque primates, wearing proper PPE, and immediate and thorough cleansing of any site exposed to macaque secretions or tissues. All macaque primates, despite negative serologic testing for B virus, must be considered potentially infected. When an exposure occurs, prompt reporting to an experienced clinician knowledgeable about B virus is essential. Timely treatment with antiviral medication can control B virus and prevent the development of severe infection and death.
Footnotes
Acknowledgements
The author acknowledges Cynthia LaFond, PhD, RN, CCRN-K, University of Chicago Medicine, for editorial assistance.
Conflict of Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Biography
Michele Rohrman, APN, BC, is a nurse practitioner in Occupational Medicine at the University of Chicago Medicine, Chicago, IL.