Abstract
Background
We previously reported on a pilot study to assess the incorporation of a novel wellness assessment device, the Preventiometer (iPEx5 GmbH, Greifswald, Germany), into an academic medical practice. The present follow-up study expands on those data and evaluates the acceptability of the assessment process in a larger sample population.
Objective
The aim of this study was to evaluate participant satisfaction with the Preventiometer wellness assessment.
Methods
A total of 60 healthy volunteers participated. Each participant underwent a comprehensive wellness assessment with the Preventiometer and received data from more than 30 diagnostic tests. A 32-question survey (with a numeric rating scale from 0 to 10) was used to rate the wellness assessment tests and participants’ impressions of the wellness assessment.
Results
Each assessment had a significantly higher rating than 7 (P < .001), and the majority of participants agreed or strongly agreed that they were satisfied (98.3%), and they strongly agreed that they were engaged the entire time (93.2%), and liked the instant test results feature of the Preventiometer device (93.2%).
Conclusion
This study confirms findings from our previous pilot study regarding the feasibility of the Preventiometer as a wellness assessment tool. The study further demonstrated that 98% of participants were satisfied with the assessment and that all of them would recommend it to others.
Introduction
During the past decade, the importance of periodic wellness assessments, health screening, and healthy habits has become apparent since they not only reduce the risks of various diseases by allowing improved surveillance1,2 but also decrease associated health care costs.3-11 Wellness, preventive health care,12,13 and healthy lifestyles are associated with improving various diseases and underlying conditions,14-18 and the rate of morbidity may be decreased by embracing healthy lifestyles as recommended by health care providers.19-25 In 2011, the value of wellness promotion was emphasized when Medicare created the annual wellness visit for health assessments and personal prevention plans.26-28 Similarly, Medicaid has initiated Healthy Behavior Incentive Programs in several states, 29 and the Centers for Disease Control and Prevention has developed the Workplace Health Promotion website. 30 Many employers foster an awareness of healthy lifestyles by providing workplace wellness programs.31-34 Some large employers and corporations also offer multidimensional wellness programs for employees with incentives to enhance a healthy lifestyle, reduce health care costs, and optimize productivity levels. 30
Although an increasing number of employers offer workplace wellness programs, the participation rate is relatively low. 30 A survey study by the Centers for Medicare & Medicaid Services reported that annual wellness visits have a low participation rate (<10%). 35 Similarly, wellness tools such as self-managed apps are being innovated and used, but the participation rate is still less than expected.36,37 Reasons for underuse may include the following: (1) physicians’ time constraints for asymptomatic patients; (2) lack of awareness about the benefits of wellness, lifestyle management, and wellness resources 35 ; (3) patients’ fear of learning about abnormal findings from wellness visits and screening tests; (4) uncertainty about insurance coverage for wellness visits; and (5) multiple appointments for laboratory testing and discussions about wellness assessment results. 38 Some of these issues could be addressed with an innovative approach that uses a multitesting medical device with supportive technologies and provides a one-stop health assessment with recurrent follow-up support to maintain a healthy lifestyle.39-42
We assessed a wellness evaluation device, the Preventiometer (iPEx5 GmbH, Greifswald, Germany), that has the unique capability to generate comprehensive data from several tests. 38 The Preventiometer is a one-stop device that measures several health parameters and produces a broad-ranging health evaluation quickly. We previously conducted a pilot study with 10 healthy volunteers to determine the feasibility of using the Preventiometer in an academic medical center. That phase 1 pilot study reported excellent operational performance and a high rate of satisfaction among the study participants. 38 The findings from that pilot study prompted us to conduct a larger study with 60 healthy volunteers. The purpose of this large-scale phase 2 study was to gather data from a larger number of participants on their acceptance of the Preventiometer health assessment and their satisfaction with it.
Methods
Study Design and Participants
The protocol was approved by the Mayo Clinic Institutional Review Board, and the same methodology was followed as described in the first study. 38 Healthy volunteers were recruited with the use of a flyer and advertisements in the institution’s intranet site.
Sample Size and Power Calculation
The sample number for this phase 2 study was determined from the results of the pilot study. According to that study, 38 a sample size of 60 would achieve a power of more than 90% for detecting a difference of .7 from an estimated numeric rating score (NRS) of 7 (with an SD of 1.5 and α = .05 with a 2-sided 1-sample t test).
Preventiometer Description
The Preventiometer (Figure 1), an innovative combination of other devices, offers multiple tests and assessments in 1 visit. The entire process is conducted with various integrated diagnostic devices, and the participant is directed by a virtual person on the large parabolic screen of the Preventiometer who reviews the upcoming tests and their importance. A live Preventiometer technician is present during the entire wellness assessment process to answer questions and to assist with various examinations (e.g., apply electrocardiographic leads, collect blood samples, and operate the equipment for ultrasonography). Individual functions of the Preventiometer can be included or excluded for different populations and desired outcomes. Preventiometer wellness assessment device (iPEx5 GmbH, Greifswald, Germany). A) A technician (in white clothes) is helping the person seated inside the Preventiometer. B) The person seated inside the Preventiometer is facing the screen, which shows a virtual person. A technician is seated in front of the screen to provide assistance. Used with permission from https://vilua.com/competencies/healthcare-competency/.
For this large-scale Preventiometer phase 2 study, we completed all the tests described in our previous study, 38 which included measurements of vital signs, pulmonary function, cardiovascular status, body muscle and fat, hearing and vision, bone density and spine analysis, and posture. In addition, a complete blood cell count was performed. A hard copy of the test results was available for the participant at the end of the assessment.
Survey Instruments
Our previous 25-question survey
38
was expanded into a 32-item survey to collect participants’ feedback in the following areas: 1. Ratings of different tests with an 11-point NRS, where 0 indicated not satisfied at all and 10 indicated the best possible satisfaction (for 13 questions) 2. Ratings of the Preventiometer screening wellness assessment with a Likert scale from 1 (strongly disagree) to 5 (strongly agree) (for 6 questions) 3. Perception of outcomes with a Likert scale from 1 (strongly disagree) to 5 (strongly agree) (for 6 questions) 4. Utility of the Preventiometer with a Likert scale from 1 (strongly disagree) to 5 (strongly agree) (for 7 questions)
After participants rated their satisfaction on the 13 test assessments with the NRS, the average score was calculated for each assessment and compared to a score of 7 and to a score of 9. The study participants also provided their demographic information, and they were encouraged to give additional comments about the entire assessment process and their willingness to pay a fee for the assessment.
Data Collection
After the Preventiometer wellness assessment, all study participants were requested to complete a hard copy of the survey. Completed survey questionnaires were collected and entered into the secure electronic Research Electronic Data Capture (REDCap) database hosted by Mayo Clinic.
Statistical Methods
Descriptive characteristics and results from the Preventiometer assessment were reported with mean (SD) and minimum and maximum values for continuous variables and with frequency (percentage) for categorical variables. Results from each section of the wellness assessment were reported as frequency (percentage). Test satisfaction on the 11-point NRS scale was reported as mean (median) for each of the 13 sections. The median test satisfaction scores were compared against a score of 7 and also against a score of 9 with a 1-sample median Wilcoxon signed rank test. P values less than .05 were considered significant. All analysis was conducted with SAS software (SAS Institute Inc) and with Microsoft EXCEL (Microsoft Corp).
Results
Demographics of Participants
A total of 60 healthy volunteers were recruited for this study and completed the Preventiometer assessment; 59 of them completed the study (1 participant did not complete any of the follow-up surveys). Most participants were female (74.1%), all were White (100%), and most had completed at least some college-level education (98.1%). The average age of the participants was 47 years, 89.3% were employed, and 70.9% were married.
Preventiometer assessment results for study participants.

Abbreviations: ARI, autonomic regulation index; BMI, body mass index (calculated as weight in kilograms divided by height in meters squared); bpm, beats per minute; CBC, complete blood cell count; ECG, electrocardiogram; HRV, heart rate variability; IMT, intima-media thickness; NA, not applicable; VRT, venous refilling time; WHtR, waist-to-height ratio.
aData are presented as mean (SD) unless indicated otherwise.
Preventiometer wellness assessment time for each participant ranged from 56 to 79 minutes; the average SD duration was 68.1 (4.7) minutes.
Participants’ Perceptions About the Preventiometer Assessment
Perceptions About the Preventiometer Assessment From 59 Participants. a
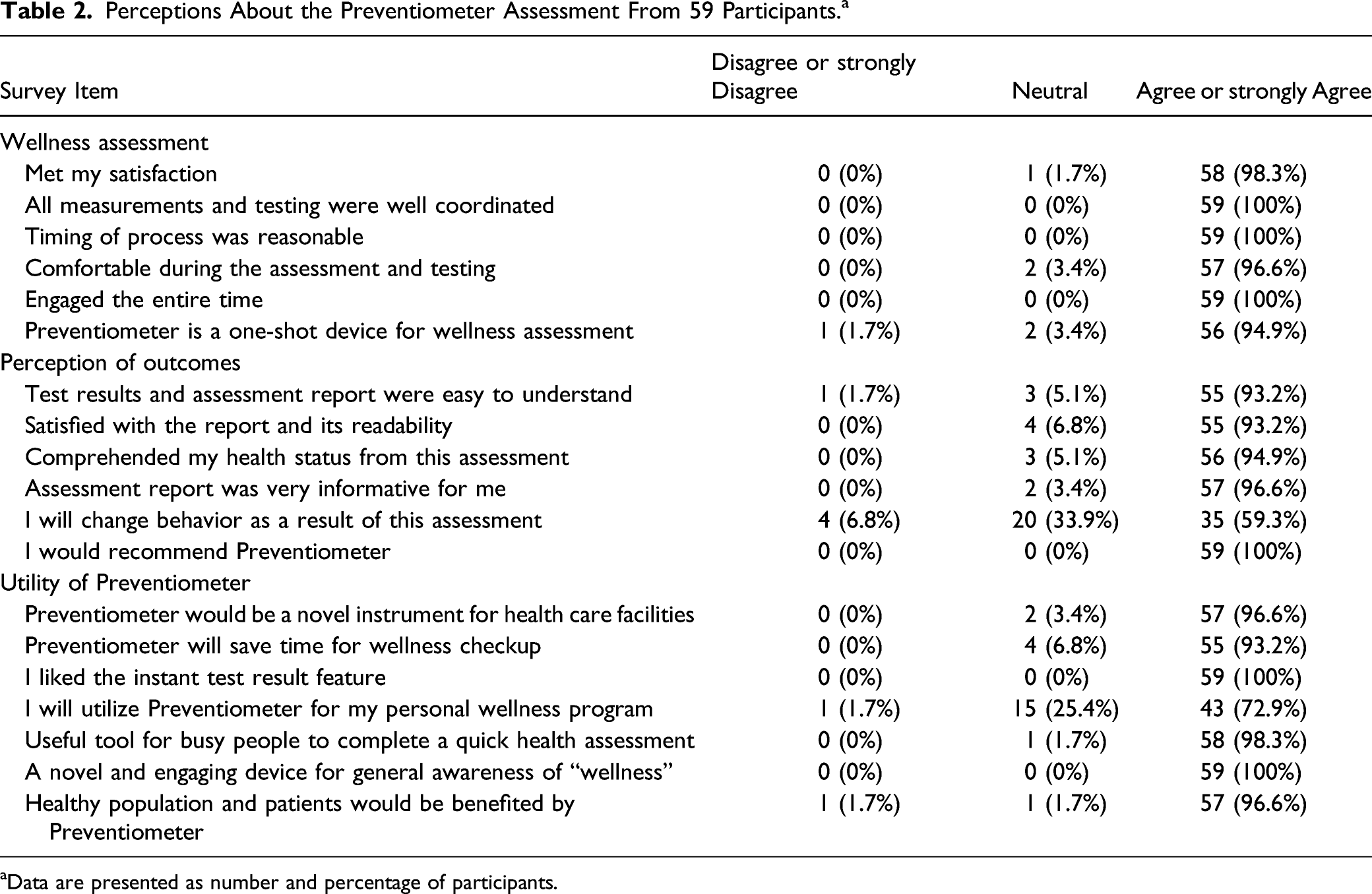
aData are presented as number and percentage of participants.
Ratings of Wellness Assessment Tests
Satisfaction Scores for Tests.

Abbreviations: CBC, complete blood cell count; ECG, electrocardiogram; HRV, heart rate variability; Q, quartile.
aOne-sample Wilcoxon signed rank test compared against a score of 7.
bOne-sample Wilcoxon signed rank test compared against a score of 9.
Participants’ Opinions and Comments
When participants were asked to provide qualitative feedback and comments about the Preventiometer assessment
38
and any features they liked or did not like about the entire assessment, 51 participants provided specific comments about the assessment. Many participants provided positive comments (Figure 2A). Multiple participants mentioned that they would like a more extensive explanation of the results, especially for the heart rate variability test. Two participants requested reference ranges for the blood tests. Other comments about improvements for the test experience included making the chair more comfortable, having a smaller straw for the lung test, and improving the annunciation of the virtual person. Participants’ comments about the wellness assessment with the preventiometer (iPEx5 GmbH, Greifswald, Germany). A) Comments from 51 participants. B) Responses from 38 participants when asked how much they would be willing to pay for a Preventiometer assessment.
The survey also included questions relating to participants’ willingness to pay for the Preventiometer assessment. Of the 56 who answered the question, 52 (92.9%) said that they would be willing to pay a fee. 38 When participants were asked to provide a dollar amount for how much they would pay, 38 answered, with amounts ranging from $25 to $600 (Figure 2B), and 14 did not specify the amount. The median amount participants were willing to pay was $150, and the mean was $193.
Discussion
The Preventiometer, a novel wellness device, was developed in Germany and is being evaluated in several clinical studies in Germany. It was designed with the objective of promoting wellness among corporate employees by providing health risk data in a timely fashion and, with the support of a coach, empowering the participants to make lifestyle changes that would improve their health.
The present study confirmed findings from our previous small pilot study 38 and demonstrated that 98% of participants agreed that the Preventiometer met their satisfaction and that they would recommend it to others (100%); 93% of the study participants felt engaged and 80% felt comfortable during the entire evaluation process. Participants’ willingness to pay for the assessment confirmed that they found value in it. This acceptance is important because multiple studies have shown that trust in new novel devices is essential for success.43,44
Importantly, a majority of participants indicated that the results from the Preventiometer would change their behavior. This finding may reflect the fact that the Preventiometer provides a comprehensive assessment and includes results not usually provided in conventional wellness assessments (e.g., heart rate variability). Future studies are needed to help determine whether this perception of behavioral change is driven by the aggregate results or by specific metrics. An innovative approach, such as use of the Preventiometer, may be a suitable alternative to conventional assessments as a low-cost, one-stop option.
Investigators from the University of Greifswald have evaluated the reliability of the Preventiometer for 4 procedures (blood pressure measurement, pulse oximetry, body fat measurement, and spirometry) and the validity for 5 procedures (somatometry, blood pressure measurement, pulse oximetry, body fat measurement, and spirometry) (Junge M, unpublished data, 2020). Overall, reliability was found to be mostly good to excellent and allowed for detection of differences between and within participants at different measurement points, which is crucial for scientific applications. However, when validity was evaluated, the limits of agreement for most examinations were very large compared to results from similar method comparison studies. The investigators recommended that better measurement procedures from relevant guidelines should be adopted to improve the validity, and these procedures are being evaluated.
The Preventiometer assessment as outlined by the company includes coaching sessions and discussion of test results by a physician; those portions of the assessment were not evaluated in this study. Multiple reports have shown that involving a wellness coach can help to improve health outcomes and maintain participants’ motivation.45,46 Understanding test results is important for practicing healthy behavior and maintaining healthy lifestyles. This is corroborated by the fact that some study participants indicated that they would have appreciated more detailed explanations of the tests, such as heart rate variability.
In conclusion, the Preventiometer was well accepted by our study population consisting of healthy Mayo Clinic employees. Providing employees with novel health data that are accessible at the workplace and available with a 60- to 75-minute time commitment appears to have the potential to change behavior and allow large numbers of individuals to be screened. Further investigations are necessary to improve the validity of the various tests performed during the Preventiometer assessment and to document the effects of coaching and interpreting and explaining test results by a provider.
Supplemental Material
sj-tif-1-gam-10.1177_21649561211045016 – Supplemental Material for Preventiometer, a Novel Wellness Assessment Device, Used With Healthy Volunteers: A Phase 2 Study*
Supplemental Material, sj-tif-1-gam-10.1177_21649561211045016 for Preventiometer, a Novel Wellness Assessment Device, Used With Healthy Volunteers: A Phase 2 Study* by Sanjeev Nanda, Tony Y. Chon, Saswati Mahapatra, Stephanie A. Lindeen, Karen M. Fischer, Markus Krüger, Bernd Schierwater, Carsten O. Schmidt, Dietlind L. Wahner-Roedler and Brent A. Bauer in Global Advances in Health and Medicine
Supplemental Material
sj-tif-2-gam-10.1177_21649561211045016 – Supplemental Material for Preventiometer, a Novel Wellness Assessment Device, Used With Healthy Volunteers: A Phase 2 Study*
Supplemental Material, sj-tif-2-gam-10.1177_21649561211045016 for Preventiometer, a Novel Wellness Assessment Device, Used With Healthy Volunteers: A Phase 2 Study* by Sanjeev Nanda, Tony Y. Chon, Saswati Mahapatra, Stephanie A. Lindeen, Karen M. Fischer, Markus Krüger, Bernd Schierwater, Carsten O. Schmidt, Dietlind L. Wahner-Roedler and Brent A. Bauer in Global Advances in Health and Medicine
Supplemental Material
sj-tif-3-gam-10.1177_21649561211045016 – Supplemental Material for Preventiometer, a Novel Wellness Assessment Device, Used With Healthy Volunteers: A Phase 2 Study*
Supplemental Material, sj-tif-3-gam-10.1177_21649561211045016 for Preventiometer, a Novel Wellness Assessment Device, Used With Healthy Volunteers: A Phase 2 Study* by Sanjeev Nanda, Tony Y. Chon, Saswati Mahapatra, Stephanie A. Lindeen, Karen M. Fischer, Markus Krüger, Bernd Schierwater, Carsten O. Schmidt, Dietlind L. Wahner-Roedler and Brent A. Bauer in Global Advances in Health and Medicine
Supplemental Material
sj-tif-4-gam-10.1177_21649561211045016 – Supplemental Material for Preventiometer, a Novel Wellness Assessment Device, Used With Healthy Volunteers: A Phase 2 Study*
Supplemental Material, sj-tif-4-gam-10.1177_21649561211045016 for Preventiometer, a Novel Wellness Assessment Device, Used With Healthy Volunteers: A Phase 2 Study* by Sanjeev Nanda, Tony Y. Chon, Saswati Mahapatra, Stephanie A. Lindeen, Karen M. Fischer, Markus Krüger, Bernd Schierwater, Carsten O. Schmidt, Dietlind L. Wahner-Roedler and Brent A. Bauer in Global Advances in Health and Medicine
Footnotes
Acknowledgments
We express our sincerest gratitude to all the healthy volunteers who participated in this large-scale study for their time, engagement in wellness assessment, and willingness to complete all the survey questions.
Declaration of conflicting interests
Vilua Healthcare and the University Medicine Greifswald cooperate on a mobile health promotion project with the Preventiometer. Markus Krüger is employed at the University Medicine Greifswald with partial financial support from Vilua. The other authors declare that there are no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental Material for the article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
