Abstract
Objective:
This post hoc analysis determined the correlation between wound bed preparation (WBP), defined as complete debridement of nonviable tissue and complete granulation tissue coverage, and wound closure, using data from a published, Consolidated Standards of Reporting Trials (CONSORT)-compliant randomized controlled trial that evaluated bromelain-based enzymatic debridement (BBD) compared with a placebo gel vehicle (GV) or nonsurgical standard of care (NSSOC) in patients with chronic venous leg ulcers (VLUs).
Approach:
Patients with chronic VLUs were randomized (3:3:2 ratio) to daily treatment with BBD, GV, or NSSOC for up to 2 weeks and followed up weekly with NSSOC for 12 weeks. Wound closure incidence was compared between those who did and did not achieve WBP by 14 days or anytime during the study.
Results:
Data were analyzed from 119 VLUs. Among 80 wounds that achieved WBP anytime during the study, 42% healed; among 39 wounds without WBP, only 10.3% healed (relative risk [RR] = 4.1, p = 0.0004, negative predictive value [NPV] = 90%). Among 37 wounds that achieved WBP by 14 days, 54% healed; among 78 wounds that did not achieve WBP by 14 days, only 22% healed (NPV = 78%). Wounds were 2.4 times more likely to achieve closure anytime during the study, if they achieved WBP by 14 days (RR = 2.4, p = 0.0005).
Innovation:
This landmark analysis confirms that WBP status is an early predictive variable of wound closure.
Conclusion:
WBP of chronic VLUs significantly increased the likelihood of wound closure and is a critical, though not sufficient, condition for healing.
INTRODUCTION
Normal wound healing occurs in four overlapping, coordinated stages to restore tissue integrity and function: hemostasis, inflammation, proliferation, and remodeling/maturation.1–3 During hemostasis, fibrin clots are formed, and growth factors are released to initiate repair. During inflammation, neutrophils and macrophages clear debris and microorganisms and release prohealing cytokines. During proliferation, fibroblasts produce collagen, endothelial cells form new blood vessels, and keratinocytes migrate to close the wound. During remodeling/maturation, type I collagen replaces type III collagen, and scar tissue reorganizes.1,3 Unfortunately, wounds often have a nonlinear healing trajectory that stalls, often due to persistent inflammation or proliferation characterized by increased levels of inflammatory cytokines and proteases, inadequate blood supply, reduced cellular activity, and senescence.1–4 Within 72 h of wound onset, autolytic debridement to remove nonviable tissue and foreign bodies from the wound bed is supposed to naturally occur by enzymes derived from neutrophils. 2 In hard-to-heal wounds, alternative debridement is needed to clear the wound bed of persistently accumulating necrotic and nonviable tissue and biofilm, and excessive exudate levels also must be addressed.
Comprehensive wound bed preparation (WBP) involves debridement and management of microorganisms and exudate to allow for granulation tissue to form and facilitate wound closure.2,3 In hard-to-heal wounds, the TIMERS (Tissue, Infection/Inflammation, Moisture/Exudate, Regeneration/Repair, Social) approach to systematic WBP should be implemented to transition the wound to the acute healing state.5,6 This involves debridement to remove nonviable “Tissue”; “Infection” control to reduce “Inflammation”; the use of dressings to manage “Moisture” and exudates; assessment of the wound “Edge” to monitor callus formation, determine the need for debridement, and ensure that re-epithelialization from a healthy wound edge can occur; appropriate wound therapies for “Repair and regeneration”; and addressing patient “Social factors” and adherence barriers that may impede wound healing. 6
Wound care providers have long considered that any wound that does not reduce in size by 40 − 50% after 4 weeks of standard of care is chronic and hard to heal.6,7 However, recently, some authors have proposed to de-emphasize chronicity and instead focus on the “hard-to-heal” characteristics of wounds. Hard-to-heal wounds do not undergo natural, autolytic debridement; increase in size; and have exudate and slough 3 days after onset. 7
The immediate end result of effective debridement is granulation tissue formation.2,8 Granulation is essential to wound healing, by providing a vascular framework for angiogenesis and for cellular proliferation (including for fibroblast migration, which contracts the wound and promotes closure), and serves as a physical barrier against infection.1,8,9
Alternative methods to autolytic debridement include the following: surgical/sharp debridement, mechanical debridement, enzymatic debridement, and biological debridement. Surgical and sharp debridement methods are the gold standard for skilled health care professionals, who may not be available in some settings. Furthermore, these methods are often associated with intense pain, and they may not be appropriate in immunocompromised patients, for poorly vascularized wounds, or in patients on anticoagulants or with bleeding disorders.2,5,10 Mechanical debridement has similar limitations. 9
Recently, the ChronEx randomized controlled trial (RCT) evaluated the effect of a novel bromelain-based enzymatic debridement (BBD, EscharEx®,
Marissa J. Carter, PhD, MA

CLINICAL PROBLEM ADDRESSED
Debridement of nonviable tissue and promotion of granulation tissue are key components of WBP that can transition a hard-to-heal wound from the chronic state to the acute healing trajectory. Until now, definitive evidence demonstrating association between WBP and wound closure has been lacking. This post hoc, exploratory analysis of the ChronEx RCT provides preliminary evidence of a correlation between WBP, based on complete debridement of nonviable tissue and complete granulation tissue coverage, and VLU wound closure.
MATERIALS AND METHODS
The ChronEx study was a multicenter, randomized, controlled, phase 2 trial that compared 2 weeks of daily BBD treatment followed by 12 weeks standard of care in 46 VLUs to 43 VLUs treated with 2-week daily treatment of GV followed by 12 weeks of standard of care and to 30 VLUs treated with 14 weeks of NSSOC. The complete methodology has been previously published in compliance with the Consolidated Standards of Reporting Trials (CONSORT) reporting guidelines for RCTs. 11 The respective institutional review boards of the 20 study sites in the United States, Switzerland, and Israel approved the study protocol, which adhered to the Declaration of Helsinki as revised in 2013. Electronic Case Report Forms for data collection were used in this study. Complete study data are available from the corresponding author upon request.
In the ChronEx study, patients with chronic VLUs provided their written informed consent and were randomized (3:3:2 ratio) to daily treatment with BBD, placebo GV (hydrogel), or NSSOC, for up to 2 weeks or until reaching complete debridement. They were then followed up for additional 12 weeks. Major inclusion criteria were as follows: having a wound duration of 4 weeks to 2 years, a wound area of 2–100 cm2, and having at least 50% of the wound bed covered with nonviable tissue. Major exclusion criteria were wound size reduction by at least 20% within the screening period, clinically significant infection, sinus tracts, or impaired arterial flow (Toe–Brachial Index ≤0.50, Ankle–Brachial Index ≤0.70, skin perfusion pressure ≤40 mm Hg, or transcutaneous oximetry ≤40 mm Hg). 11
Subjects randomized to the BBD or GV group received daily (every 24 h) applications of a thin layer (∼3 mm) of topical BBD or GV. Zinc oxide ointment (Calmoseptine, Calmoseptine Inc., Huntington Beach, CA) was applied to protect the periwound skin, and an occlusive dressing (3M Tegaderm, 3M, Saint Paul, MN) helped secure the BBD or GV in place. An absorbent dressing was placed over the occlusive layer, followed by a two-layer compression system (Coban™ 2, 3M).
Daily treatment continued until either complete debridement was achieved, defined as the presence of a fully viable wound bed with removal of all nonviable tissue, or the subject received a maximum of eight daily applications within a 2-week period. Surgical, mechanical, biological, and enzymatic debridement or honey-based products were not permitted during this phase.
Following the initial 2-week daily treatment phase, BBD and GV treatments were discontinued in the respective study groups. Subjects were subsequently followed twice weekly for 2 weeks and then weekly for an additional 10 weeks. During this follow-up period, subjects received standardized nonactive dressings, selected at the investigator’s discretion (e.g., alginates, foams, highly absorbent dressings, medical-grade honey, hydrocolloids, hydrofibers, hydrogels, or silver-containing products), whereas the two-layer compression system was maintained throughout the study.
In the NSSOC group, treatment consisted of standardized wound dressings, including enzymatic agents (collagenase ointment), alginates, foams, highly absorbent dressings, medical-grade honey, hydrocolloids, hydrofibers, hydrogels, and silver products, applied in accordance with approved labeling, instructions for use, and investigator discretion. These dressings continued throughout the study. All NSSOC treatments were similarly covered with the two-layer compression system. Surgical, mechanical, or biological debridement was not permitted at any point during the study.
Wound infection was monitored throughout the study using established clinical criteria (non-healing, exudate, red friable tissue, debris (discoloration) and smell [NERDS] and size increasing, temperature elevation, os (probes to bone), new breakdown, erythema/edema, exudate and smell [STONEES]). After removing dressings, the wound was cleansed with sterile saline or a mild soap solution, qualitative wound culture was done in case of suspected clinical infection, and antimicrobial treatment was permitted at the investigator’s discretion when warranted.
WBP was defined as complete debridement of nonviable tissue and a wound bed completely covered with granulation tissue. Percent debridement and cover with granulation tissue were clinically and visually assessed, as recommended by regulatory authorities (the United States Food and Drug Administration and European Medicines Agency). To enhance consistency, the same investigator was asked to perform repeated assessments of the wound whenever feasible.
Wound closure was defined as complete re-epithelialization of the wound surface without drainage or dressing, confirmed at two visits.
All statistical analyses were performed using SAS® version 9.4 (SAS, Cary, NC). Categorical variables were summarized by counts and percentages. Means and standard deviations (SDs) summarized continuous variables with normal distributions; medians and interquartile ranges (IQRs) summarized non-normal distributions.
This post hoc analysis used several methods to analyze the correlation between WBP (as defined above) and wound closure.
One method analyzed the incidence of wound closure among those wounds that achieved WBP anytime throughout the study compared with those that did not achieve WBP. A diagnostic table was prepared to determine the probability of a wound achieving closure, by wound bed prepared status (Yes/No). The negative predictive value (NPV) was calculated to determine the probability that a wound would not heal, if its wound bed was not prepared. The positive predictive value (PPV) was calculated to determine the probability that the wound would heal, if its wound bed was prepared. Odds ratios (ORs) and relative risks (RRs) for achieving wound closure and their respective 95% confidence intervals (CIs) were calculated. Forest plots of wound closure by WBP status were created to visually compare the ORs and RRs. Chi-square test was used to assess statistical significance.
In addition, a landmark analysis was performed, in which the incidence of wound closure during the 12-week follow-up period was compared between wounds that achieved WBP by 14 days with those that did not achieve WBP by that time, to understand whether WBP within 2 weeks could be predictive of wound closure. Diagnostic tables were prepared. The median time to wound closure in each group was estimated based on the Kaplan-Meier (KM) method. A log rank test was utilized to compare the KM curves between these groups. This landmark analysis excluded subjects that already achieved wound closure before day 14.
A Spearman correlation between WBP anytime throughout the study to wound closure within this time frame was assessed.
Finally, a time-dependent proportional hazards Cox regression model was used to assess the association between time to WBP and time to wound closure. In this analysis, WBP status served as the time-dependent variable. The wound was considered to have achieved WBP only once from the time of WBP and was handled as not achieving WBP before that timepoint.
Missing data for subjects who did not achieve complete WBP or wound closure at any of the analysis timepoints were imputed as “failure.”
RESULTS
Baseline characteristics were comparable across the three treatment arms. For all randomized subjects (n = 119), their mean (SD) age was 64 (12) years, and 55% were female. Their VLUs ranged in size from 1.9 to 100 cm2 (mean [SD]: 15.5 [19.4] cm2; median [IQR]: 8.7 [4.4, 16.4] cm2) and had a duration of 4 weeks to 2 years (mean [SD]: 31.1 [24.0] weeks). The mean (SD) percentage of nonviable VLU tissue was 73% (15.2).
Overall, 80 out of 119 wounds (67%) achieved WBP anytime during the study, and 39 (33%) did not achieve WBP. There were 115 subjects evaluated for wound closure during the 12 weeks follow-up period; four wounds were excluded (one wound was excluded for having closed during the initial 2-week daily treatment period, and three wounds did not have wound closure assessments reported following this period). Among the 115 wounds evaluated, 37 wounds (32%) achieved WBP during the initial 2-week daily treatment period; 78 (68%) did not achieve WBP.
Figure 1 depicts the diagnostic table showing the probability of wounds reaching wound closure during the study based on their WBP status at 14 days. Among the 37 wounds that achieved WBP by 14 days, 20 (54%) went on to closure. Among the 78 wounds that did not achieve WBP by 14 days, 17 (22%) achieved closure and 61 (78%) did not achieve closure during the study. Therefore, the NPV was 78%, and the PPV was 54%. Wounds were 2.4 times more likely to achieve closure by end of study, if they achieved WBP by 14 days (RR: 2.4; 95% CI: 1.5–4.2; p = 0.0005).
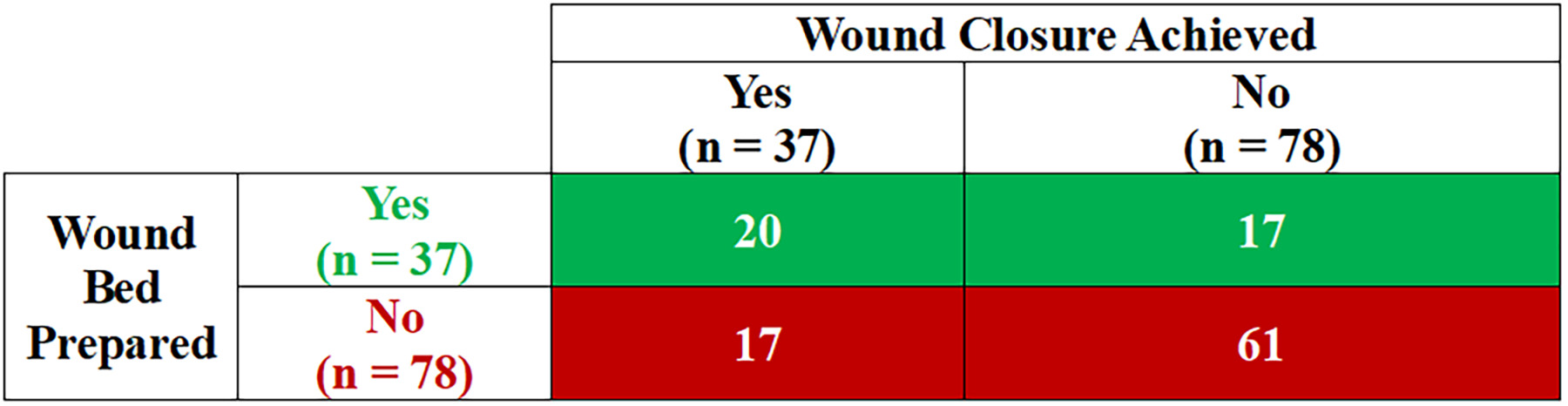
Diagnostic table depicting the probability of wounds reaching wound closure based on their wound bed preparation status at 14 days. The positive predictive value was 54% (20/37); the negative predictive value was 78% (61/78). The odds ratio (95% confidence interval) was 4.2 (1.8–9.8). The relative risk (95% confidence interval) of wound closure in wounds with wound bed prepared versus wound closure in wounds with wound bed not prepared was 2.4 (1.5–4.2), p = 0.0005.
Figure 2 depicts the diagnostic table showing the probability of wounds reaching wound closure during the study based on their WBP status. Among the 80 wounds achieving WBP, 34 (42%) achieved wound closure, whereas among the 39 wounds that did not achieve WBP, 4 (10%) achieved closure and 35 (90%) did not close. The high NPV was 90%, whereas the PPV was 42%. Wounds were 4.1 times more likely to close anytime during the study if they achieved WBP during the study compared with wounds that did not have their wound bed prepared (RR: 4.1; 95% CI: 1.6–10.9; p = 0.0004).
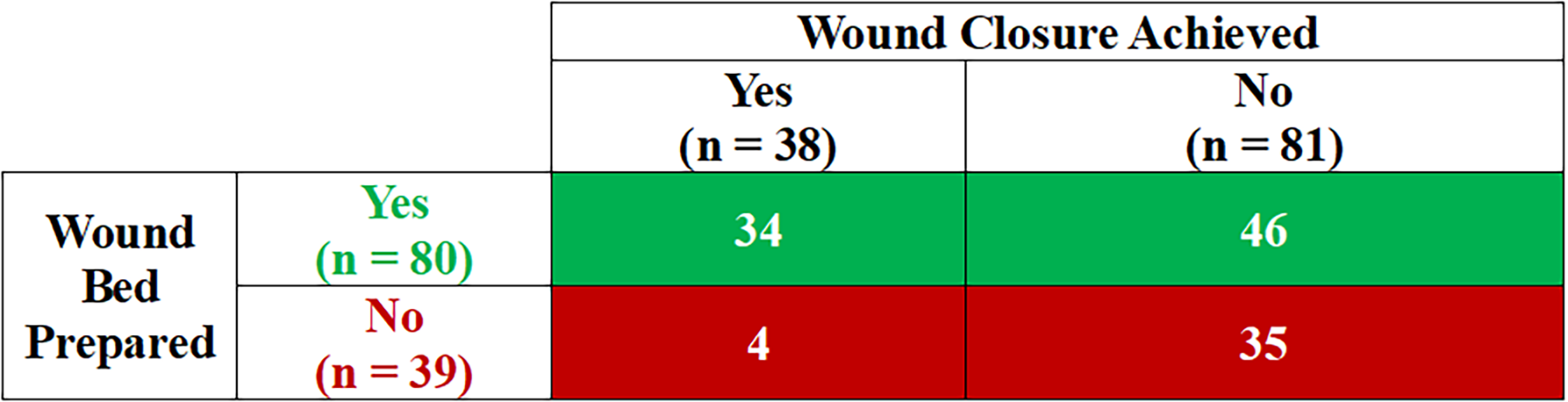
Probability of wounds reaching wound closure based on their wound bed preparation status at any time during the study. The positive predictive value was 42% (34/80); the negative predictive value was 90% (35/39). The odds ratio (95% confidence interval) was 6.5 (2.1–19.9). The relative risk (95% confidence interval) of wound closure in wounds with wound bed prepared versus wound closure in wounds with wound bed not prepared was 4.1 (1.6–10.9), p = 0.0004.
Figure 3 depicts forest plots of wound closure by WBP status to visually compare the RRs (Fig. 3A) and ORs (Fig. 3B).
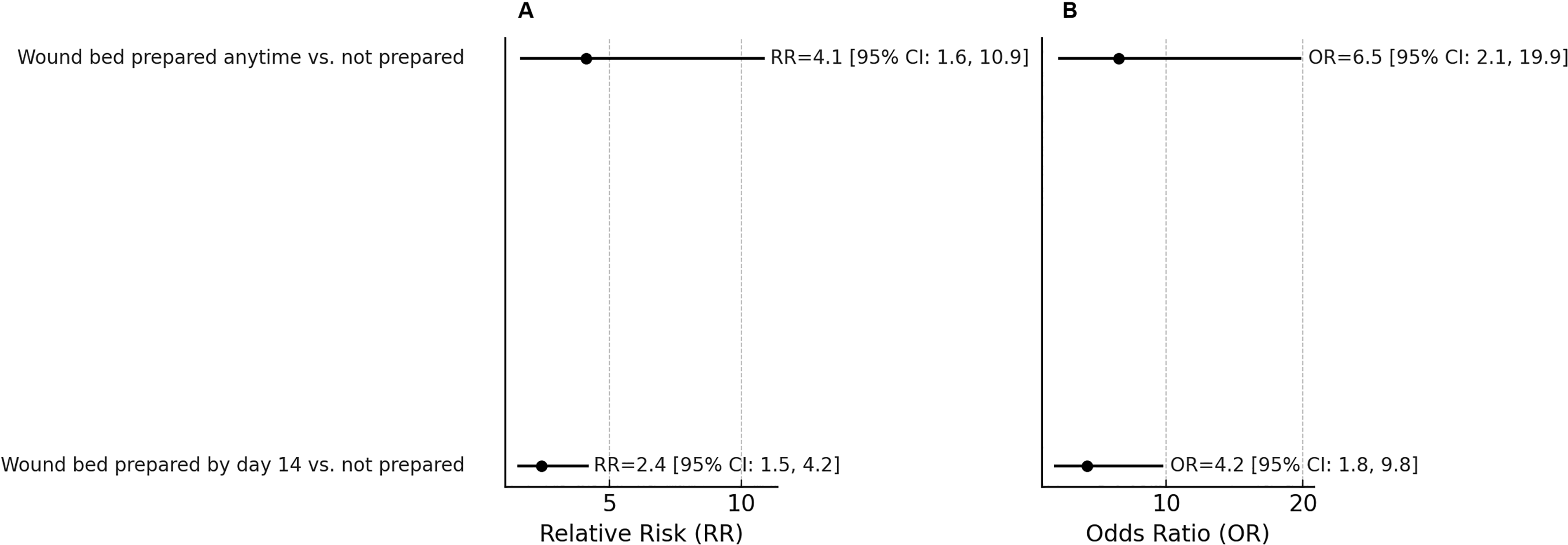
Forest plots of wound closure by wound bed preparation status to visually compare the relative risks (RRs;
In Fig. 4, the time to wound bed prepared was positively correlated with time to wound closure among wounds that achieved wound closure (Spearman correlation: 0.66, 95% CI: 0.4–0.8, p < 0.0001).
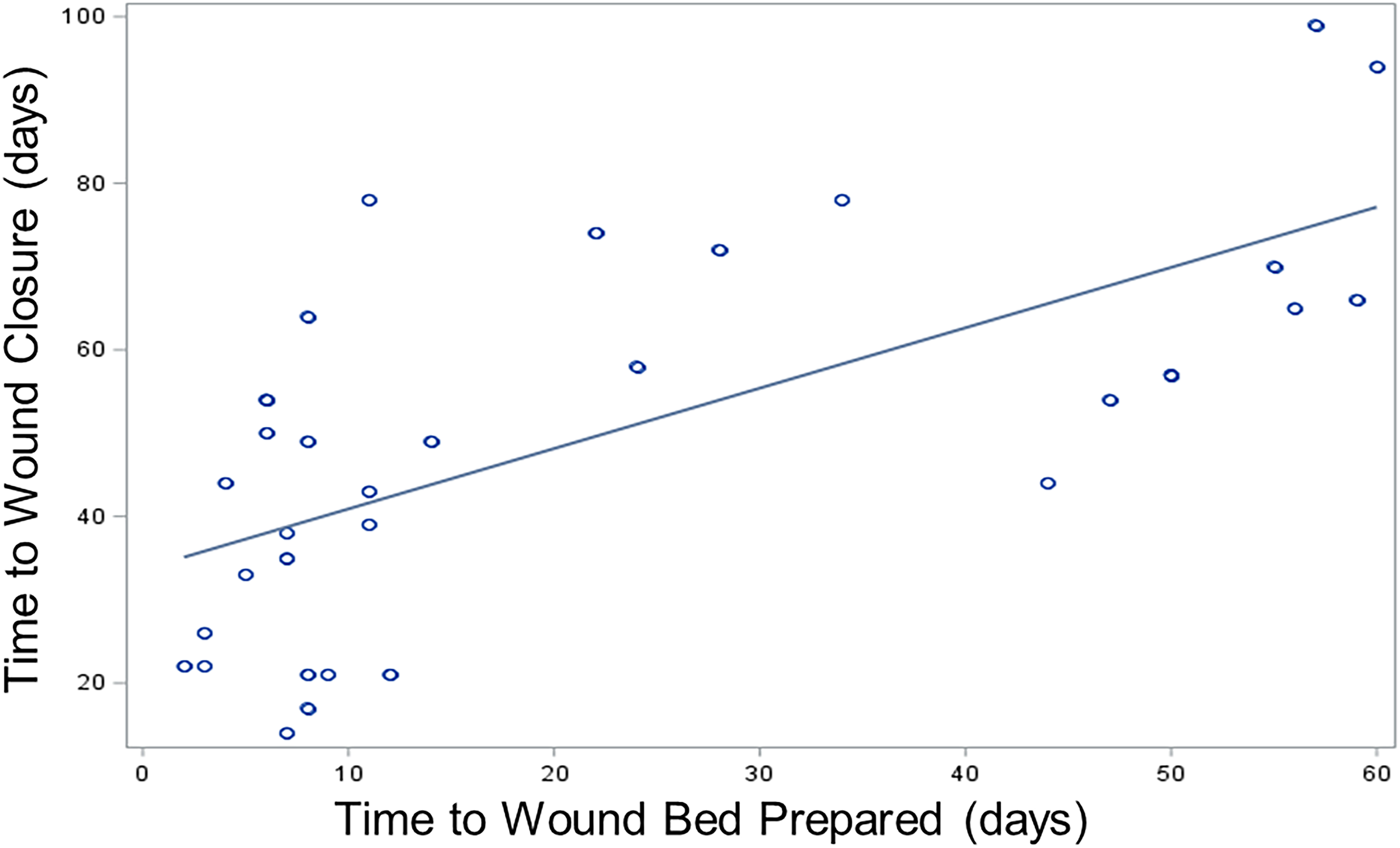
The correlation between time to wound closure and time to wound bed prepared among venous leg ulcers that achieved wound closure (Spearman correlation: 0.66, p < 0.0001).
Figure 5 depicts the KM curve of the probability of VLUs achieving closure associated with whether they reached WBP within 14 days (green line) compared with those that did not achieve WBP within 14 days (red line). Wounds that had their wound bed prepared within 14 days had a KM median time to wound closure of 64 days (95% CI: 43–not available), whereas for VLUs that did not have their wound bed prepared within 14 days, the median time for wound closure was not reached (p < 0.0001, log rank test), as only 22% of these subjects closed their wounds. When analyzed as a time-dependent variable, the hazard ratio (HR) of WBP achieving wound closure is 12 (95% CI: 4.2–33.8, p < 0.0001), suggesting a strong time-dependent correlation between WBP status and time to wound closure. These findings demonstrate that WBP was significantly associated with wound closure and faster wound closure rates.
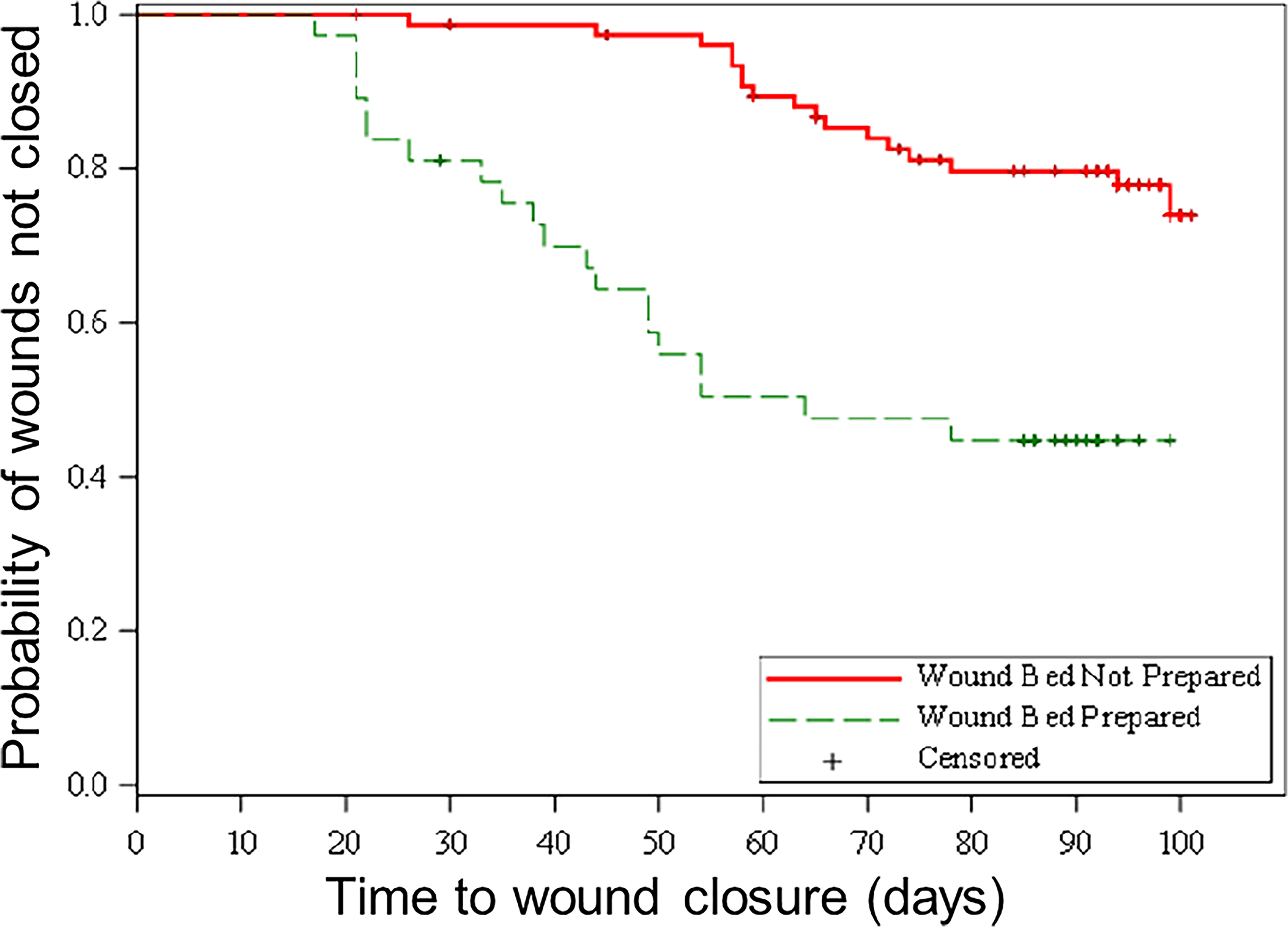
Kaplan–Meier curve of the probability of wounds achieving closure associated with whether they reached wound bed preparation within 14 days (green line) compared with those who did not achieve wound bed preparation within 14 days (red line).
DISCUSSION
Upon enrollment in the ChronEx RCT, nonviable tissue covered nearly three-quarters of the wound beds of the chronic VLUs evaluated in this post hoc analysis. Among VLUs that did not achieve WBP by 14 days, the overwhelming majority (78%) remained unhealed at the end of the study. Early WBP by 14 days was positively correlated with faster closure rates (Fig. 4; p < 0.0001), further demonstrating the pivotal role of WBP on healing rates. The fact that the NPV of healing was nearly 80% for wounds that did not achieve WBP by 14 days indicates that not achieving WBP is a strong, early predictor of a wound not healing (Fig. 1). The NPV increased to 90% for wounds that did not achieve WBP at any time during the study (Fig. 2), confirming that WBP is critical to healing.
Among wounds that achieved WBP by 14 days or during the study, the PPV of healing was 54% or 42%, respectively. The lower PPV (compared with NPV) merely indicates that achieving WBP is important to heal the wound, but there are other relevant variables that also affect wound closure. Moreover, based on the RR in Fig. 2, wounds that achieved WBP during the study were 4.1 times more likely to achieve wound closure. The HR determined that WBP was a time-dependent variable for wound closure; if the wound bed was not prepared during the study, then the likelihood that it did not heal was 12 times higher than if the wound bed was prepared.
Previously, granulation rates alone were determined to be early predictors of healing diabetic foot ulcers (DFUs). In a small study of 48 DFUs, wounds that failed to achieve granulation coverage of at least 50% after 8 weeks and those with granulation rates less than 75% at 6 months were unhealed or recurred at 1 year. 12 Another post hoc analysis of two RCTs also found that a granulation rate of more than 75% at the end of treatment predicted the healing of severe Wagner 3 and 4 DFUs, with a similarly high NPV of 94% compared with our 90% NPV calculated for VLUs. 13 Complete debridement and complete granulation were used to determine WBP status in VLUs in the ChronEx RCT. 11 BBD was applied daily for up to eight applications followed by nonactive dressings for 12 weeks, a protocol which implemented the repetitive principles of wound hygiene and TIMERS by focusing on early, frequent debridement, following wound cleansing and preceding dressing the wounds.
The limitations of the ChronEx RCT were discussed in the original publication. 11 The primary limitation of the current study is its exploratory, post hoc design aimed at generating preliminary evidence on the correlation between WBP and wound closure, using predefined and widely accepted definitions. While the results are supported by the 95% CIs, they should be interpreted with caution pending confirmation in a prospective, controlled study. The lower RR reported in Fig. 1 for WBP at 14 days compared with WBP at any time during the study in Fig. 2 could be explained by the smaller sample size, as supported by the 95% CIs of the ORs reported with these figures.
On a final note, while WBP is a foundational concept in modern wound care, several real-world barriers may limit its consistent implementation across clinical settings. These include human resource and workflow barriers (limited clinician time, expertise, and training), WBP access barriers (limited resources, reimbursement for debridement procedures, and access to advanced debridement techniques), patient-related barriers (lost to follow-up, inability to tolerate sharp or mechanical debridement techniques, or having comorbidities that may inhibit the achievement of WBP), and the lack of standardized, validated tools to adequately assess WBP.2–6,9,10
This landmark, exploratory post hoc analysis of the ChronEx RCT confirms that WBP, defined here as complete debridement and complete granulation, is an early predictive variable that is strongly correlated with wound closure in chronic VLUs. WBP within 2 weeks has very high NPV of nearly 80% in healing VLUs, meaning that VLUs that do not achieve WBP are very unlikely to achieve wound closure and suggesting that WBP is critical for wound closure, underscoring its pivotal role in healing. Among wounds in the ChronEx RCT that never achieved WBP during the study, their likelihood to remain unhealed was 12 times more than among wounds that achieved WBP. These preliminary findings support the positive effects of complete debridement, achieved through frequent applications, and complete granulation on healing outcome. A confirmatory, prospective study is needed to confirm this correlation between WBP and wound closure.
INNOVATION
Treatment guidelines emphasize the importance of WBP in wound healing.5–8,10 This landmark exploratory analysis provides preliminary evidence demonstrating a strong correlation between WBP and wound closure, with WBP being highly predictive of wound closure. Wounds that reach WBP within 14 days are 2.4 times more likely to achieve closure, as exemplified by the chronic VLU (wound age of 10 weeks) pictured in Fig. 6. The VLU achieved WBP within 14 days (following seven daily treatments with BBD) and healed following split-thickness skin grafting. Moreover, the high NPV suggests that WBP is critical for wound closure, underscoring its pivotal role in healing.
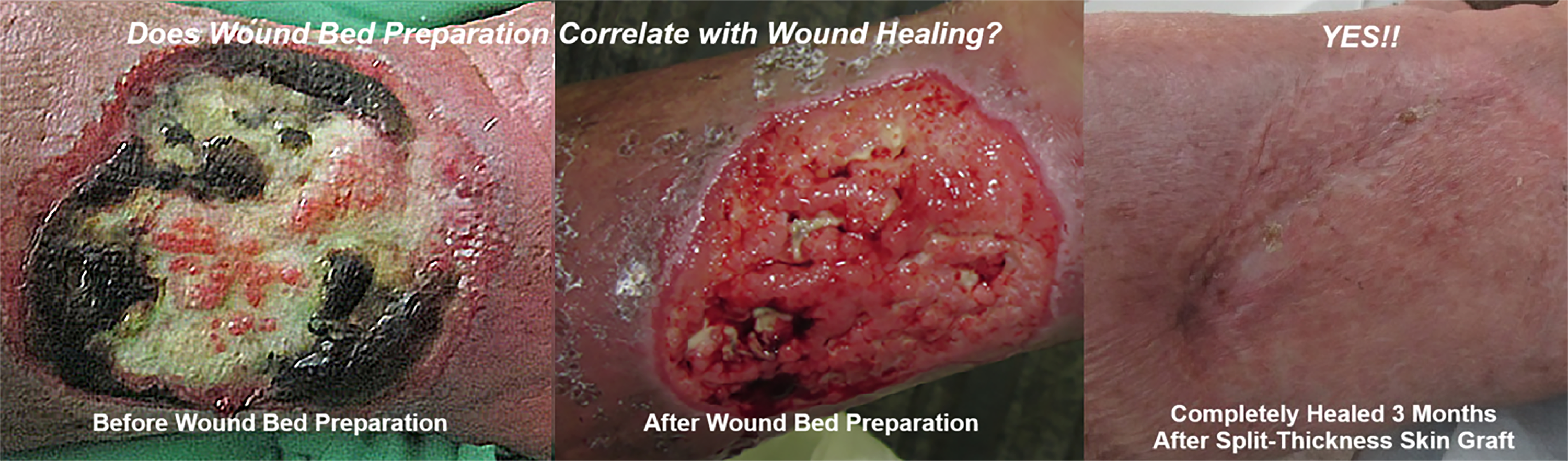
This landmark analysis provides evidence that wound bed preparation (WBP) is positively correlated with wound healing. Wounds that undergo WBP are 4.1 times more likely to achieve wound closure, as exemplified by the hard-to-heal venous leg ulcer (VLU) pictured here that was unable to heal after 10 weeks of standard of care. After 14 days of daily application of a novel bromelain-based enzymatic debridement agent, the wound achieved WBP, with healthy, highly vascularized granulation tissue visible in the wound bed. Once WBP was achieved, split-thickness skin grafting occurred, and the wound healed. The first two images pictured here were previously published by the same author of this landmark analysis, and permission to reuse is granted by the publisher.
KEY FINDINGS
In this landmark, exploratory analysis, not achieving WBP during the study had a very high NPV of 90% in VLUs; that means that the probability that these wounds would heal is very low and that WBP is essential but not sufficient for healing. VLUs that achieved WBP were 4.1 times more likely to achieve wound closure than those that did not achieve WBP. This post hoc analysis provides preliminary evidence that WBP is an early predictive variable of wound closure; wounds that did not achieve WBP as early as 14 days also had a very low healing probability, with a high NPV of 78%. A confirmatory, prospective study is needed to support these initial experimental findings. At any timepoint during the study, if the wound bed was not prepared, then the likelihood that it did not heal was 12 times higher than if the wound bed was prepared.
AUTHORS’ CONTRIBUTIONS
M.J.C.: Conceptualization, formal analysis, and writing—review and editing; R.J.S.: Writing—review and editing; K.D.Z.: Conceptualization; Y.K.-L.: Investigation; A.H.: Project administration and writing—review and editing; E.K.: Project administration and writing—review and editing; O.B.-G.: Formal analysis and writing—review and editing; C.P.-R.: Formal analysis and writing—review and editing; Y.S.: Writing—review and editing; and J.C.L.: Writing—review and editing.
Footnotes
ACKNOWLEDGMENTS
The authors thank Kristen Eckert (Strategic Solutions, Inc.) for her assistance in writing and editing the article. All authors of the original ChronEx RCT publication are aware of this post hoc analysis.
FUNDING INFORMATION
AUTHOR DISCLOSURE AND GHOSTWRITING
M.J.C.: Paid consultant of
