Abstract
Antenatal depression is of utmost concern to public health due to its potential to negatively impact both mother and child health. Less research has been done on it, and it is not receiving enough attention in the study area. This study aimed to assess the magnitude of antenatal depressive symptoms and its associated factors among pregnant mothers in Dessie City administration, Ethiopia. From March 1 to 30, 2021, a cross-sectional study was conducted among 451 pregnant mothers in Dessie City Administration. Edinburgh Postnatal Depression Scale (EPND) was used to assess antenatal depression. A multivariable logistic regression analysis was used to assess the determinants of depressive symptoms among the women. The proportion of antenatal depressive symptoms was 18.6% with 95% CI [15.1%, 22.2%]. Age greater than 30, monthly income less than 1,500 Ethiopian Birr, history of mental illness family member, a recent intimate partner violence history, death of a close family in recent year, and insomnia were significant factors affecting antenatal depression among pregnant mothers. Depression during pregnancy are high in the study area. Age >30 years, monthly income <1,500 Ethiopian Birr, history of mental illness in the family, experiencing recent intimate partner violence, death of a close family in recent year, and insomnia were independent predictors of depressive symptoms. Considering the integration of mental health services with maternal health care and providing attention to pregnant women during follow-up are recommended actions to reduce depressive symptoms.
Background
Depression is a common mental disorder that manifests as poor energy, guilt or emotions of self-worth, interrupted sleep or appetite, and difficulty concentrating (Sadock, 2007). It greatly affects people in all societies around the world and dramatically increases the burden of disease on the entire population. In fact, depressive symptoms are the main contributor to the disease burden for women in both high- and low-income countries (Federal Democratic Republic of Ethiopia, Ministry of Health, 2012). Expectant moms undergo considerable physical and psychological changes during pregnancy and the adjustment to motherhood. The emergence of depression may be linked to these modifications (Ghaffar et al., 2017). Due to its chronic nature, intensity, and recurrence as well as its detrimental effects on women’s general health and children’s development, it is now acknowledged as a global public health issue (Srivastava et al., 2016). The prevalence of gestational depressive symptoms in developed and developing countries was 5% to 30% and 20% respectively (Pereira et al., 2011).
Antenatal depressive symptoms can occur at any time during pregnancy and symptoms may gradually increase over a period of many weeks or they might start suddenly and unexpectedly but are not taken into consideration by medical professionals during antenatal evaluation since they are probably caused by emotional changes in pregnancy hormones (Sadock, 2007). It is estimated that one in seven women will experience persistent symptoms of depressive symptoms during pregnancy (Underwood et al., 2016). Numerous conceptual modes like the stress-vulnerability and stress-coping approach were used to describe perinatal depression. However, this paradigm was criticized since it ignored other social and behavioral factors that affect women’s health in favor of concentrating only on marital dissatisfaction. Depression in pregnant women has a controversial and not fully known etiology. Different studies found the Biopsychosocial model, created by George L. Engel in 1977, to provide the most comprehensive explanation of the causes of prenatal depression (Fricchione et al., 2012). According to the Biopsychosocial paradigm, anxiety and generalized stress are two intermediate variables that indirectly cause depression during pregnancy rather than biological causes directly causing it (Ross et al., 2004). Researchers modified the Biopsychosocial model later in 2008. They also found evidence that low sociodemographic characteristics, a lack of personal resources, and prenatal stressors directly influenced antenatal depression (Leigh & Milgrom, 2008). Additionally, a recent study from 2018 added to the Biopsychosocial model by highlighting the important role that an unhealthy lifestyle has as a covariate promoting prenatal depression (English et al., 2018).
Antenatal depressive symptoms can have disastrous effects on the mother, the unborn child, and the entire family (Sadock, 2007). Pregnant women have many possibilities to receive treatment since they frequently interact with universal services. Despite the fact that pregnant women with depressive symptoms sometimes forgo treatment because they are afraid to talk to their doctors about their mental health issues or because they lack knowledge about depressive symptoms (Glover, 2014).
Antenatal depressive symptoms can lead to maternal complications including increased risk of obstetric complications like hyperemesis gravidarum, pregnancy-induced hypertension, preeclampsia, postpartum bleeding, preterm delivery, gestational diabetes, preterm labor, planned cesarean delivery, epidural analgesia during labor (Ali et al., 2017; Mohamad Yusuff et al., 2016; Weobong et al., 2014). Depressive symptoms in pregnancy may persist into the postpartum period and disrupt the parenting behavior, the attachment process between the mother and baby, as well as the relationship with the partner and any other children (Shakeel et al., 2015).
Different studies across the world have reported different prevalence rates for depressive symptoms. A study in the United States reported that the overall pooled prevalence of depressive symptoms in pregnant women was USA 9.9% (Melville et al., 2010). Similarly, the studies conducted in China were 13.7% (Mohamad Yusuff et al., 2016), Malaysia 13.8% (Wang et al., 2016), Pakistan 29.7% (Ali et al., 2017), and Saudi Arabia 54.5% (Alotaibe et al., 2016). A systematic review conducted in low- and middle-income countries (LMIC) reported that the overall pooled prevalence of depressive symptoms among pregnant women was 16% (Fisher et al., 2012). Furthermore, high rates of depressive symptoms among pregnant women have been reported in many countries in Africa such as Nigeria 24.5% (Thompson & Ajayi, 2016), Tanzania 33.8% (Rwakarema et al., 2015), Maichew Town, Ethiopia 31.1% (Mossie et al., 2017), Gondar University Hospital 23% (Ayele et al., 2016), and Addis Ababa was 24.94% (Biratu & Haile, 2015). There are several risk factors for depressive symptoms among pregnant women. Some of the factors associated with depressive symptoms are being a housewife, being single (Wang et al., 2016; Pereira et al., 2011; Coll et al., 2017), younger age (Pajulo et al., 2001), lower educational level (Biratu & Haile, 2015), being unmarried (Pajulo et al., 2001), low level of income (Mossie et al., 2017), rural residents (Fisher et al., 2012), unplanned pregnancy (Gourounti et al., 2015), Brazil primigravida (de Jesus Silva et al., 2016), increasing trimester (Thompson & Ajayi, 2016), history of stillbirth (Moawed et al., 2015), previous obstetric complications (Moawed et al., 2015), previous history of mental illness (Biratu & Haile, 2015; Bisetegn et al., 2016), medical illness (Thompson & Ajayi, 2016), intimate partner violence (van Heyningen et al., 2016), lower marital satisfaction (Gourounti et al., 2015), lower spousal support (Biratu & Haile, 2015) and substance use (Pajulo et al., 2001).
Even if there are opportunities for pregnant women to be identified and treated during an antenatal visit, pregnant women and new mothers experiencing depressive symptoms often do not get the treatment they need due to fear of discussing mental health concerns with their providers or a lack of education about depressive symptoms (Santoro & Peabody, 2010). Additionally, Ethiopia does not routinely screen for prenatal depression symptoms, and no information is available in the research area. Therefore, the aim of this study was to assess the severity and contributing causes of antenatal depressive symptoms in pregnant women who get antenatal treatment in medical facilities run by the Dessie City Administration in northeastern Ethiopia. Utilizing the findings from this study, policymakers may create a strategic plan that will prevent and treat pregnant women’s depressive symptoms and improve maternal and neonatal outcomes.
Methodology
Study Design, Area, and Period
This cross-sectional study design study was conducted in Dessie city administration; located in Amhara National Regional State, Northeast, Ethiopia, 401 km far from, Addis Ababa, the capital city of Ethiopia. Dessie city administration has 18 urban kebeles and 8 rural kebeles. It has 18 kebeles and a 350,000 population. According to data from the South Wollo Zone statistics office for the years 2016 to 2017, there were 186,571 men and 163,429 women among these populations. There are two governmental hospitals, eight health centers, five private hospitals, four specialty private clinics, 13 private medium clinics, two Non-Governmental Origination (NGO) medium clinics, 16 private junior clinics, five gross distributor pharmacies, 18 pharmacies, 33 drug stores, one rural drug venders, six health posts in Dessie city. This study was done from March 1 to 30, 2021.
Source Population
All pregnant women attending ANC among health institutions in Dessie city administration.
Study Population
All pregnant mothers attending ANC among health institutions in the study period.
Inclusion and Exclusion Criteria
All pregnant mothers aged 18 and above were included in the study, while pregnant women who were critically ill and had difficulty communicating were excluded from the study.
Sample Size Determination and Sampling Technique
Sample Size Determination
The sample size was calculated using a single population proportion formula with the assumption of a prevalence (P) of depressive symptoms in a previous study 24.94% (Biratu & Haile, 2015) with a confidence limit of 5%.
The sample size is calculated as follows;
Where; n denotes sample size, P is the prevalence of prior study (24.94%), d denotes the degree of precision (assumed to be 5%), and (z α/2) denotes the value of a standard normal variable that corresponds to be 95% confidence levels (1.96).
Since we used a multi-stage sampling method to consider the designing effect, we increased the calculated sample size by 1.5 to account for sampling error. Ultimately, a 10% non-response rate resulted in a sample size of 474.
Sampling Procedure
Initially, all health institutions found in Dessie city administration were stratified into public and private health institutions. From all 10 public health institutions, five health institutions (Dessie comprehensive specialized hospital, Tita Health Center, Buanbua wuha health center, Hotie health center, Boru Sellasie Health Center), and from all six private health institutions, three health institutions (Ethio General Hospital, Dr. Misganaw Specialty Clinic, and Selam Hospital) were selected by using lottery method. The previous one-year total ANC uptake of pregnant women for each health institution was taken to get the average 2 months follow up. Then study participants were allocated depending on the average number of antenatal care attendants in each health institution. Finally, the sample fraction was calculated to choose each study unit (pregnant women) using systematic random sampling (K = N/n = 1,729/474 = 3.6~3). The second woman who visited ANC OPD during the data collection time was considered as the random start. Then the selection of study participants was continued every three intervals until the calculated sample size (474) become achieved (Figure 1).

Schematic representation of sampling technique 2021 G.C.
Where: DCSH = Dessie Comprehensive Specialized Hospital, THC = Tita Health center, BWHC = Buanbua Wuha Health Center, HHC = Hotie Health Center, BSHC = Boru Sellasie Health Center, EGH = Ethio General Hospital, Dr, MSC = Dr. Misganaw Specialty Clinic, and SGH = Selam General Hospital
Data Collection Tools and Techniques
A structured interviewer-administered questionnaire was used which had comprised different subsections; questionnaires to assess socio-demographic, obstetrical, clinical, substance-related, and psychosocial factors. Edinburgh Postnatal Depression Scale (EPDS) was used to assess symptoms of maternal depression (Cox et al., 1987). EPDS is a common tool for screening depressive symptoms; initially, for use during the postnatal periods, it is also additionally validated for use during the postnatal periods in different contexts and nations (Murray & Cox, 1990; National Collaborating Centre for Mental Health, 2007; Anbesaw, Negash, et al., 2021). Additionally, it was validated in Ethiopia’s perinatal population, with a sensitivity and specificity of 84.6% and 77.0%, respectively (Hanlon et al., 2008). It consists of 10 questions that look at your emotional condition over the last week or more. There are four potential answers for each question score, with scores ranging from 0 to 3, and 30 is the maximum score. Similar to the previous study, the most significantly depressed women are taken into consideration if the score is 13 or higher, which is utilized to identify likely instances (Ola et al., 2011; Wang et al., 2016).
The Insomnia Severity Index, which comprises seven items, was used to evaluate sleep patterns. The seven answers are added up to obtain a final score. Scores 0 to 7 indicate no clinically significant insomnia, 8 to 14 indicate sub-threshold insomnia, 15 to 21 moderate clinical insomnia, and 22 to 28 indicate severe clinical insomnia (68). Data on the level of social support was collected using the Social Support Scale (The Oslo 3-items) (OSSS-3). It was categorized into three main categories of social support; 3 to 8 poor social support, 9 to 11 moderate social support, and 12 to 14 good social support (Kocalevent et al., 2018). The internal consistency (Cronbach alpha) of Oslo-3 social support was 0.79. The IPV was evaluated using the Abuse Assessment Scale (AAS). It is the method most frequently utilized in clinical settings to assess the abuse of expectant mothers. Women who responded positively to questions 2, 3, or 4 were regarded as having experienced abuse (McFarlane et al., 1992).
The level of marital satisfaction is assessed by three items Kansas marital satisfaction scale each rated on a 7-Likert scale ranging from one (extremely dissatisfied) to 7 (extremely satisfied). The scale contains a minimum of three points and a total of 21 cumulative scores. Those pregnant women who scored 17 and above from 21 were considered satisfied with their current marital relation (Nichols et al., 1983) and score less than 17 from 21 were considered as not satisfied with their current marital relation. Socio-demographic factors, clinical factors obstetrical factors, substance use history, and psychosocial factors were used on “yes/no” response questionnaires and were operationalized in accordance with various works of literature.
Data Collection Procedures
Data was collected through face-to-face interviews with skilled six BSc nurses and two supervisors (MPH graduates). For the sake of clarity and consistency, the questionnaire was first written in English, then translated into Amharic, and then returned to English by language experts. The data collectors and supervisors received training for 2 days duration on the aims of the study, instruments, data collection methods, sampling techniques, and how to handle ethical issues including confidentiality. In the Haik district hospital, 5% (n = 24) of the participants took part in the pre-test, which intended to find any potential problems with the data collection procedure and suggestions for improvement. The principal investigator and supervisors regularly supervise and assist the data collectors. Editing and entering the data from a paper into the computer allowed for timely processing after they had been double-checked.
Data Processing and Analysis
Data was coded and entered into Epi-Data version 3.1 and then exported to SPSS version 26 for analysis. It was done using descriptive statistics like frequencies and proportions The multivariable regression model included independent variables with a bivariable model p-value of less than .25 to account for potential confounding effects. An odds ratio with a 95% confidence interval (CI) for each variable in the multivariable model that had a p-value <.05 indicated the strength of association. The Hosmer and Lemeshow goodness of fitness test was used to assess the model’s fitness (p-value = .456). In the end, the findings were presented as text, a table, or a graph. Multicollinearity was tested using tolerance and variance inflation factors (maximum VIF = 2.3).
Results
Socio-Demographic Characteristics Participants
A total of 451 individuals were involved in this study and the response rate was 95.75%. The mean age (SD) of the participants was 26.01 (±4.85 years) with an age range of 18 to 41 years. The majority of participants, 380 (84.3%) lived in urban, 433 (96%) were married, 195 (43.2%) were government employees, 229 (50.8%) attended college and above, and 421 (93.3%) earned above 1,500 ETB per month (Table 1).
Socio-Demographic Factors of Pregnant Mothers Attending Health Institutions in Dessie, Northeast, Ethiopia, 2021 (N = 451).
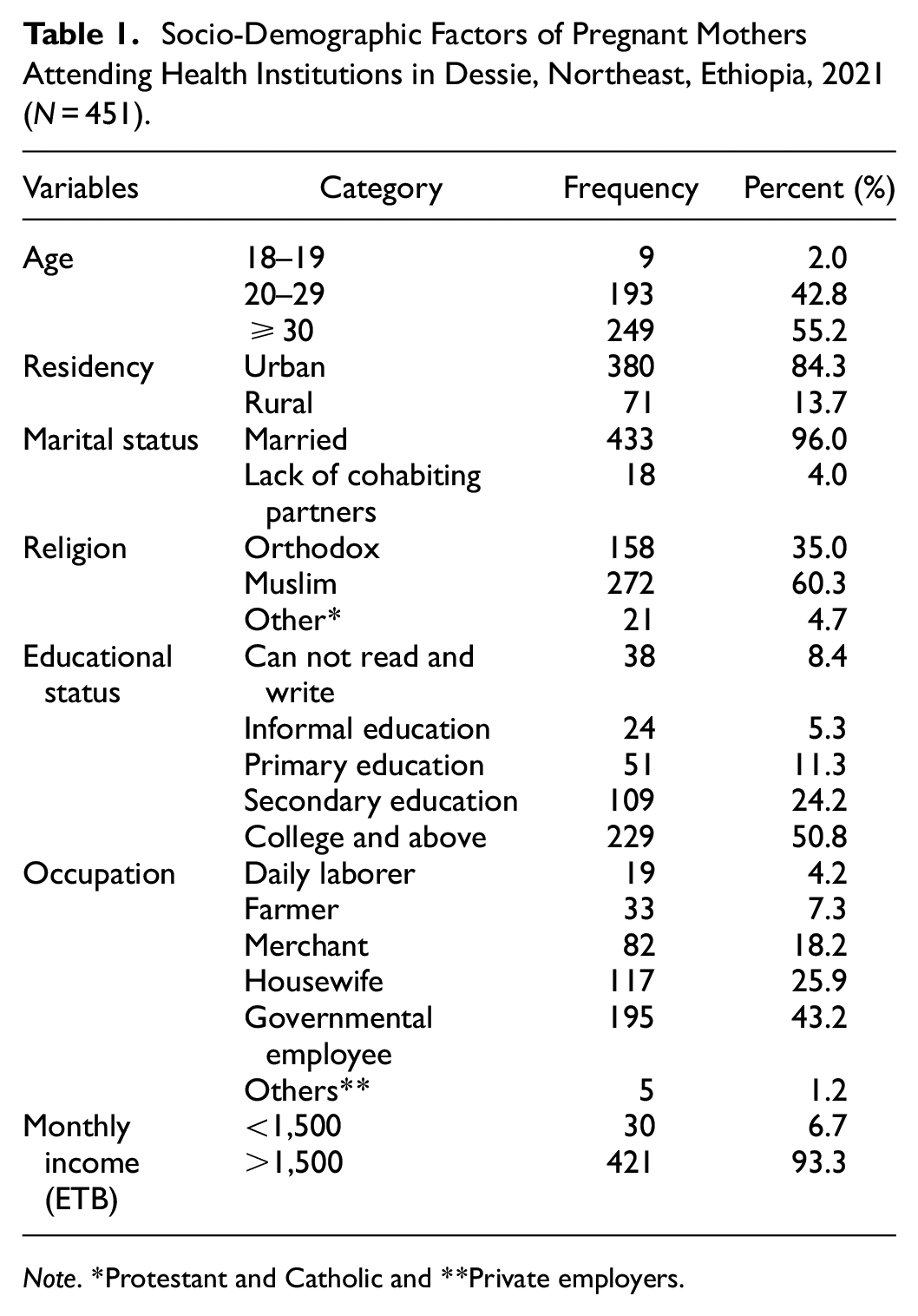
Note. *Protestant and Catholic and **Private employers.
Obstetrics-Related Characteristics of the Participants
More than half of the study respondents (54.8%) were in third-trimester gestation followed by second-trimester 191 (42.4%) in gestational age. Nearly three fourth (73.2%) of the participants were multigravida. From the total participants, 29 (6.4%) mothers had a previous abortion history, and 43 (9.5%) had a history of stillbirth. Nearly one-third of respondents (28.1%) had a history of complications in the previous obstetric period. Also, more than half (55%) of the mothers had a planned pregnancy. Among the participants, (47%) had two times ANC visits during the current pregnancy (Table 2).
Obstetrics History of Pregnant Women Who Attended Health Institutions in Dessie Northeast, Ethiopia, 2021 (N = 451).

Clinical and Substance-Related Characteristics of the Participants
Among pregnant women, 28 (6.2%) participants had reported a previous history of depression, 34 (7.5%) participants had reported mental illness in a family member, 41 (9.1) were taken drugs for treating medical illnesses, and 145 (32.2%) participants had a history of mild insomnia during the current pregnancy. Of the participants, 31 (6.87%) consumed any substance during the current pregnancy and 65 (14.4%) partners used any substance as reported (Table 3).
Clinical and Substance-Related Factors of Pregnant Women Who Attended Health Institutions in Dessie, Northeast, Ethiopia, 2021 (N = 451).

Psycho-Social Characteristics of the Respondents
From the total respondents, 24 (5.3%) of the participants experienced violence from their intimate partner and 45 (10%) of the mothers reported the death of close family members this year. Among respondents, 32 (7.1%) had reported sickness of family members currently, and 277 (61.4%) were satisfied with their marital relationship. Regarding social support, half (51.2%), 118 (26.2%), and 102 (22.6%) of the pregnant mothers had received low, medium, and high social support respectively (Table 4).
Psychosocial of Pregnant Women Who Attended Health Institutions in Dessie, Northeast, Ethiopia, 2021 (N = 451).

Prevalence of Depressive Symptoms Among Pregnant Mothers
This study showed the overall prevalence of depressive symptoms among pregnant women attending health institutions in Dessie city administration was 18.6% (95% CI: [15.1, 22.2]) (Figure 2).

Magnitude of depression among pregnant mothers attending health institutions in Dessie city administration, Northeast Ethiopia, 2021 (N = 451).
Factors Associated with Antenatal Depressive Symptoms Among Pregnant Women
Factors such as age, residence, educational level, marital status, monthly income, gestational age, a complication in this pregnancy, current pregnancy planned, marital satisfaction, history of mental illness in the family, intimate partner violence, family death in recent year and insomnia were significantly correlated (p < .25) in bi-variable analysis. Among these, variables such as age greater than 30, monthly income <1,500 ETB, history of mental illness in the family, intimate partner violence, family death in recent years, mild insomnia, and moderate insomnia were significantly associated with depression in multivariable analysis.
Those pregnant mothers age >30 were 96% less to be depressed as compared with respondents with the age less than 19 years (AOR = 0.04; 95% CI [0.01, 0.87]). The odds of depressive symptoms among participants who had income <1,500 ETB was 8.71 times higher as compared to income greater than 1,500 (AOR = 8.71; 95% CI [1.76, 18.12]). Those women who had a mental illness history in the family of were about 4 times more likely to be depressed than their counterparts (AOR = 4.18; 95% CI [1.39, 12.5]). Pregnant mothers who reported intimate partner violence were nearly 8.00 times more likely to be depressed as compared with those who did not experience intimate partner violence (AOR = 7.90; 95% CI [3.12, 15.60]). Furthermore, the odds of having depressive symptoms among women who had family death in recent year was about 4 times higher as compared with the referent groups (AOR = 4.00; 95% CI [1.54, 10.53)]. Finally, pregnant women who had mild insomnia were nearly 5 times (AOR: 4.80, 95% CI: [2.13, 11.43]), and moderate insomnia was 5.56 times (AOR: 5.56, 95% CI: [2.22, 13.10]) more likely to develop depressive symptoms compared to good sleep pattern (Table 5).
Logistic Regression Analysis Results of Depression Among Pregnant Women Attending Health Institutions in Dessie, Northeast, Ethiopia, 2021 (N = 451).

Note. 1-Reference category.
Statistically significant value *p < .05 and **p < .01.
Discussion
The study looked at the magnitude and contributing causes of prenatal depressive symptoms among pregnant mothers receiving antenatal follow-up at public and private health facilities in Dessie city administration. The proportion of antenatal depression among pregnant women who attend ANC in the health institution of Dessie city administration was 18.6%, (95% CI [15.1%, 22%]). This finding is higher than 13.7% in the result done in China (Mohamad Yusuff et al., 2016), 14.8% in Alfenasto (Pereira et al., 2009), 13.8% in Malaysia (Wang et al., 2016), and 9.9% in Ghana (Weobong et al., 2014). This might be a result of these countries’ providing better health services compared to Ethiopia. Pregnant women who have good economic capacity and who lived in countries that can provide appropriate services will be satisfied with their life.
Conversely, this study’s finding is lower than those of some studies conducted in Nigeria 24.5% (Thompson & Ajayi, 2016), Pakistan 29.7% (Ali et al., 2017), Saudi Arabia which was 54.5% (Alotaibe et al., 2016), and Tanzania 33.8% (Rwakarema et al., 2015). In Ethiopia a study in Maichew town 31.1% (Mossie et al., 2017), Gondar Hospital 23% (Ayele et al., 2016), and Addis Ababa 24.94% (Biratu & Haile, 2015). This disparity may be clarified by the disparity in the socio-economic, and cultural status of nations, as well as by variations in the study method, cutoff points, and sample size.
Pregnant women with an age greater than 30 years were 96% less likely to be depressed as compared to pregnant women with an age less than 19 years. This is similar to other studies done in South Africa (Hartley et al., 2011) and Nigeria (Thompson & Ajayi, 2016). This might be supported by the fact that depressive symptoms are commonly beginning in the early 20’s of life. Additionally, this may reduce the risk of being distressed due to the new physiological cycle of pregnancy and labor; that is “as the age increases, the more exposure to pregnancy and labor, then the more exposure leads the less being frustrated to pregnancy” (Sadock, 2007).
A pregnant woman who gets a monthly income level of <1,500 ETB was 8.71 times more likely to be depressed than compared to an income greater than 1,500. This result was supported by a study conducted in Maichew, Ethiopia (Mossie et al., 2017). This is due to low monthly income has been associated with impaired physical, psychological, and social functioning of pregnant women. Those pregnant women do have not enough monthly income, and they can not get enough antenatal care services, and other important services to maintain their quality of life. Then this makes them depressed about her life including pregnancy. Also in fact having low socioeconomic status by itself is one of the risk factors for depression (Sadock, 2007).
Those mothers who had a family history of mental illness were about 4 times more likely to be depressed than their counterparts. This finding is in line with a study conducted in West, South Ethiopia (Lodebo et al., 2020), Jimma town, Ethiopia (Tesfaye & Agenagnew, 2021). This may be clarified by the potential contribution of genetics, the psychological support system within the family, and the dynamics of the pregnancy to the development of depressive symptoms (Sadock, 2007). Theoretical studies predicted that people with a family history of the disorder would be “more susceptible from a genetic point of view” and, as a result, “would require more, or more severe events to precipitate depression than the more vulnerable” individuals (English et al., 2018).
Pregnant women who reported intimate partner violence were nearly 8.00 times more likely to be depressed as compared with those who did not experience intimate partner violence. This result was supported by a study done in Brazil (de Jesus Silva et al., 2016) and Malawi (Stewart et al., 2014). Similarly, a study conducted in North West Ethiopia showed that major depressive symptoms were significantly associated with experience of intimate partner violence (Habtamu Belete et al., 2019). This may indicate that violence by partner intimate, particularly during gestation, including psychological, sexual, and physical abuse, can result in depressive symptoms (Anbesaw, Negash, et al., 2021).
The odds of having depressive symptoms among mothers who had a family death in recent year was about four times higher as compared with their counterparts. This might be due to the death or loss of close family causing sadness and grief to the individuals. Such disturbing emotional feelings bring serious depressive symptoms. On the other side, one of the complications of losing a family is developing depressive symptoms (van Heyningen et al., 2016).
Finally, pregnant women who had mild insomnia were nearly 5 times, and moderate insomnia was 5.56 times more likely to develop depression as compared to good sleep patterns. Consistent with other findings done in China (Yu et al., 2020). This might be due to the fact that sleep duration and quality decreased as a result of nighttime urination, fetal movement, leg cramps, and other symptoms experienced by pregnant women. As a result, depression symptoms were associated with sleep difficulties during pregnancy. In addition, when women can not sleep as obvious, they often become anxious about not sleeping. Thus, anxiety increases the potential for becoming depressed as well (Mindell et al., 2015; Anbesaw, Abebe, et al., 2021).
Strengths and Limitations of the Study
The research employed standardized assessment scales and potential factors. Additionally, both governmental and public health institutions which provide ANC services found in Dessie city administration were included in the study. However, the cross-sectional nature of the design, can not determine the temporal relationship between predictor factors and the outcome variables. Even though, an individual was interviewed in separate and private areas, using a structured interviewer-administered questionnaire by itself may lead to social desirability bias.
Implications
The results of our findings have improved indulgent of prenatal depressive symptoms and the value of mental clarity well-being and health during gestation. The results reported in additionally, this study suggest that regular efforts should be made to the ability to recognize antenatal women who are depressed and the information to help those affected by the situation’s mental health population. Prenatal depression is identified, along with its risk factors aid mental health professionals in keeping track of high-risk mothers and administering preventive measures with caution and targeting women’s treatment. Clinical research and clinical initiatives should focus on an effective prenatal depression intervention that is cost-effective.
Conclusion
This research finding showed that one out of five pregnant mothers attending ANC in health institutions had experienced antenatal depressive symptoms. Age greater than 30, monthly income <1,500 ETB, mental illness history in the family member, violence by an intimate partner, death of a close family member in a recent year, mild insomnia, and moderate insomnia were significantly associated with antenatal depressive symptoms. Antenatal clinics should develop instruments for depressive symptoms screening as part of standard antenatal care in order to enable early detection and appropriate intervention.
Footnotes
Abbreviations
AD = Antenatal Depression, CMHS = College of Medicine and Health Science, CI = Confidence Interval, EDPS = Edinburgh Postnatal Depression Scale, ETB = Ethiopian Birr, LMIC = Low and Middle-Income Countries, MPH = Master of public health, OSS = Oslo Social Support Scale, PHQ-9 = Patient Health Questionnaire item nine, SD = Standard Deviation, SPSS = Statistical Package for Social Sciences, VIF = Variance Inflation Factor, and WHO = World Health Organization.
Authors’ Contributions
TA, HE, ZA, and MA were involved in designing, supervising the study, carrying out the analysis, and interpreting the data. TA wrote the manuscript for publication. The final manuscript was read and approved by all of the authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Wollo University. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Ethical Approval
Ethical clearance and a letter of cooperation were obtained from the Research Ethical Review Committee of Wollo University CMHS with an ethical review number (RCSPG-505/13). Written consent was obtained from the study participants after explaining the purpose, confidential nature, and right of withdrawal to fill the questionnaires before data collection. For confidentiality, the participant’s name and identification number were not recorded on the questionnaire. For the participants who had a history of depressive symptoms, we provide psychological counseling and link them to the psychiatrist of the institution.
Data Availability Statement
All data generated and analyzed during the current study are included in this published article. The data of the current study are available from [Tamrat Anbesaw], Email:


