Abstract
Our study assessed the validity of self-reported restless sleep (SRRS) in measuring sleep efficiency and the degree to which these measures differed depending on obesity status in older women. Data were from 100 participants enrolled in the Kingston Senior Women Study. Participants recorded SRRS for 7 consecutive nights. Sleep efficiency measures were recorded nightly through actigraphy. Repeated-measures multilevel logistic analysis was used. Mean sleep efficiency was 87% (SE = 1.09), SRRS occurred in 37% (SE = 3) of nights. Obesity status moderated the association between sleep efficiency and SRRS (odds ratio [OR] = 1.08; 95% confidence interval [CI] = [1.02, 1.14]) when controlling for age, medication intake, and depressive symptoms. Higher sleep efficiency reduced the odds of SRRS in non-obese women, but no association was shown in obese women. The lack of correspondence between objective and subjective measures in obese women suggests that SRRS may not be as indicative of sleep quality in obese compared with non-obese older women.
Background
Sleep is a restorative process vital to overall health and well-being. Research has demonstrated that sleep duration and quality change with age (Crowley, 2011; Woodward, 2012). Sleep tends to be less intense, shorter, and more fragmented in older adults compared with younger adults (Vance, Heaton, Eaves, & Fazeli, 2011). Furthermore, studies find that sleep complaints are extremely common among older populations, particularly older women (Foley, Monjan, Brown, & Simonsick, 1995). Changing sleep patterns with aging can be attributed to age-related changes in circadian rhythms, the presence of primary sleep disorders, and secondary influences, including chronic diseases, cognitive disorders, and medications that directly or indirectly affect sleep (Cole & Richards, 2007; Crowley, 2011). Poor self-reported and objectively measured sleep quality in older adults is associated with various unfavorable health and quality of life outcomes, including reduced cognitive performance, diminished functional capabilities, increased likelihood of depressive symptoms, and increased use of the health care system and hypnotic drugs (Foley et al., 1999; Klink, Quan, Kaltenborn, & Lebowitz, 1992; Maglione et al., 2012; Nebes, Buysse, Halligan, Houck, & Monk, 2009; Redline & Newman, 2002). Of particular concern is the influence of obesity on sleep quality. The prevalence of obesity is increasing (Gotay et al., 2012), including among older women (Houston, Nicklas, & Zizza, 2009), and evidence suggests that obesity may be associated with several indicators of poor sleep quality (van den Berg, Neven, et al., 2008).
Despite the burgeoning research on sleep health and its relation to important health outcomes, less research has examined the degree to which self-reported restless sleep (SRRS) correlates with objectively measured sleep quality measures. Examining the degree to which subjective and objective measures correlate is important for advancing sleep research on a population-wide basis. Furthermore, research on whether the agreement between objective and subjective measures of sleep quality differs in obese and non-obese persons has been limited, particularly in older adult women. Identifying whether objective and subjective measures agree or disagree in different subpopulations can help lead to greater understanding of when subjective or objective measures of sleep can be used in research.
Measuring Sleep
There are multiple objective and subjective measurement tools used to assess sleep characteristics of older populations in non-laboratory settings (Buysse et al., 2008; van de Water, Holmes, & Hurley, 2011). Self-report questionnaires are often used to measure sleep in older populations because they are feasible and relatively inexpensive when conducting large-scale studies. The most commonly used questionnaires are the Pittsburgh Sleep Quality Index and the Pittsburgh Sleep Diary (Buysse, Reynolds, Monk, Berman, & Kupfer, 1989; Monk et al., 1994). As memory difficulties and cognitive impairments are prevalent in older adults, simplified surveys with questions relating to short time frames may be more optimal for collecting health-related data in older populations (Stewart & Mills, 2001). This study used a short sleep diary to avoid recall bias and reduce the burden on study participants.
Two commonly used objective sleep measurement tools are actigraphy and polysomnography (Blackwell et al., 2008). Polysomnography is known as the “gold standard” for measuring sleep. However, to collect sleep data using polysomnography, study participants are generally required to go to laboratory settings, making it an expensive, relatively invasive, and inconvenient option (Blackwell et al., 2008). Conversely, actigraphy is a measurement tool that can be used at home to measure and record levels of movement in sleep. Using actigraphy, a number of objective measures of sleep quality can be calculated. Sleep efficiency, sleep duration, and sleep fragmentation are commonly used indicators in older populations (Blackwell et al., 2008; Klink et al., 1992; van den Berg, Neven, et al., 2008). Sleep efficiency values provide a ratio of the time spent sleeping to time in bed. Poor sleep efficiency is linked to numerous unfavorable outcomes in older populations, including increased anxiety and mortality risk and decreased cognitive performance (Blackwell et al., 2006; Dew et al., 2003; Spira et al., 2012).
Explaining Self-Reported Sleep Quality
To be useful in research, SRRS reports should reflect whether a person experiences sleep problems. Yet, research examining the relationship between self-reported sleep quality and actigraphic measures of sleep has highlighted how socio-demographic, psychological, physical, and medical conditions may alter the relationship (van den Berg et al., 2009; van den Berg, Van Rooij, et al., 2008; Wells, Day, Carney, Freedland, & Duntley, 2004). Recent research has suggested that obesity status may alter the agreement between objective and subjective indicators of sleep. For example, studies defining obesity as a body mass index (BMI) of 30 kg/m2 or greater have found associations between obesity and subjective sleep measures, but not between obesity and objective measures (Vgontzas et al., 2013; Vgontzas et al., 2008). Obesity has been shown to modify the association between objective and subjective assessments of sleep duration among the general adult population. For example, Lauderdale et al. (2008) found that obese individuals reported less sleep than non-obese adults at the same level of measured sleep. More research is required to fully elucidate the impact of obesity on agreement between objective and subjective measure of sleep quality, particularly in older adult women.
Research Aims
This study aims to (a) provide the first examination of the correspondence between objective sleep efficiency and SRRS in older women and (b) examine whether this correspondence differs in obese and non-obese older women. To investigate these issues, we first examined whether nightly variations in actigraphically measured sleep efficiency was associated with daily SRRS. Second, we examined whether obesity status moderated the association between sleep efficiency and SRRS, while adjusting for age, depressive symptomology, obesity, and medication intake. Findings will contribute to our understanding of the factors that may influence the congruence between subjective and objective sleep measures and will assist in furthering research on the relationship between sleep and obesity in older women.
Method
Sample
Subjects for this study came from the Kingston Senior Women Study (KSWS) from July 2011 to December 2011. The aim of the KSWS was to examine the role of sleep, social connections, physical activity, and diet in the cardiovascular health of women aged 65 years and older. Participants consisted of a convenience sample of 100 women from Kingston, Ontario, Canada. Study participants were recruited through pamphlets, newspaper ads, television ads, posters, and via contact with seniors associations and retirement residences. Participants were required to come to the lab for anthropometric measurements, blood pressure and cholesterol readings, to complete the questionnaire and learn how to use the actigraphy equipment. Participants received CAN$35 compensation for completing the study to cover study-related costs such as transportation and parking. The KSWS was granted ethical clearance according to the recommended principles of Canadian ethics guidelines, and Queen’s policies. Based on agreements with study participants, external researchers should request access to data via collaborations with the KSWS group. Please direct initial enquiries to the corresponding author.
Measures
SRRS
For the restless sleep indicator, we asked participants to note in their KSWS-prepared sleep diary their answer to the question, “Did you feel rested when you woke up this morning?” To facilitate their completion of the diaries, response items were provided as yes/no options. Participants recorded their answers each morning when they awoke for a 1-week period.
Objective sleep efficiency
To measure objective sleep efficiency and duration, participants were instructed to wear a Philips Actiwatch 2 (Philips Healthcare, Andover, MA) on their wrist for 7 consecutive nights. Participants wore the Actiwatch over the same period in which they kept the sleep diary. Actigraphy has been shown to have a moderate to high level of agreement with polysomnography (the gold standard for measuring sleep) in elderly women (Blackwell et al., 2008). Based on previous research using actigraphy in elderly persons (van den Berg, Van Rooij, et al., 2008), each Actiwatch was set to the time above threshold setting so that activity counts above a certain intensity threshold over a period of time were coded as awake. Activity counts were collected in 30-s increments, also called epoch lengths, as previously used in validation studies measuring sleep with actigraphy in older populations (Blackwell et al., 2008). Participants were also instructed to press a small “event marker” button on the side of the watch when they began to fall asleep and on morning awakening to help validate sleep diary information.
After the 1-week wear period, sleep data were uploaded from the watch into Philips Respironics Actiware software. Nightly graphs of the participant’s movement and intensity of light exposure were generated. Using these graphs, we set “rest” and “excluded” intervals. Intervals were set to exclude non-bedtime wear of the Actiwatch. Excluded intervals consisted of the time during the day when the participant was not wearing the watch, whereas rest intervals represented times when the participant was wearing the watch during the night, either sleeping or resting in bed. Based on the activity counts during the set “rest intervals,” Actiware software identified specific “sleep intervals.” The interval setting process was carried out through consensus by two of the authors, who were trained by a Respironics software technician. The researchers independently set rest intervals and then compared the set intervals to ensure the defined intervals were within 10 min of one another. Any discrepancies were discussed to ensure that each set interval agreed with the predefined boundary rules. Criterion for setting rest intervals were defined based on previous research and recommendations from Respironics (van den Berg, Neven, et al., 2008; van den Berg, Van Rooij, et al., 2008). The Actiware software used a validated algorithm to categorize each epoch within a rest period as one in which the participant is either awake or asleep. Sleep start was defined as the first 10 min of a rest interval in which no more than one epoch was scored as wake. Similarly, sleep end was defined by the software as the last 10 min during which no more than one epoch was scored as wake.
Once data cleaning and coding was completed, Actiware software was used to calculate nightly “sleep efficiency” scores for each participant. Sleep efficiency was defined as the proportion of time in the rest period filled by sleep and was reported as a percentage value. “Total sleep time” was given in minutes and reflected the total time spent asleep during rest intervals.
Depressive symptoms
Depressive symptoms were measured with the 10-item Center for Epidemiological Studies Depression (CESD-10) Scale. The CESD-10 was included in a take-home questionnaire, which participants completed over the course of the weeklong study. Participants were asked to rate the extent to which they had experienced symptoms within the previous week on a 4-point Likert-type scale (1 = rarely or none of the time; 4 = most or all of the time). Examples of items include the following: “I felt everything I did was an effort” and “I felt depressed.” This scale has been previously tested for validity and reliability in older adult populations (Irwin, Haydari Artin, & Oxman, 1999; Radloff, 1977). The Cronbach’s alpha of the KSW Depression Scale was .69.
BMI
Anthropometric measures of height and weight were taken by trained technicians, using wall-mounted stadiometer, digital scale, and measuring tape. BMI was calculated as body weight (kg) / height (m2; World Health Organization, 2006). Participants were classified as obese if their BMI was greater than or equal to 30 kg/m2.
Medication intake
Participants were asked to name the physician-prescribed medications that they were currently taking for hypertension, high cholesterol, and other conditions. Medication intake is based on the number of unique medications prescribed, with higher medication intake indicative of more prescribed medications.
Statistical Analysis
Summary statistics and independent t tests were conducted to examine differences in the age, education, health, and sleep-related characteristics of obese and non-obese participants. To examine the association between SRRS and actigraphically measured sleep efficiency among older women, we used a multilevel logistic model. Level 1 consisted of the nightly SRRS and actigraphically measured sleep observations; Level 2 consisted of the individual variables, for example, age. Observations were dropped if they were missing information on either SRRS or sleep efficiency. Model 1 examined the association between SRRS and actigraphically measured sleep efficiency. Model 2 added individual-level variables to Model 1, including age, depressive symptomology, obesity status, and level of medication intake. In addition, the variable representing the interaction between obesity status and sleep efficiency was included in the second model.
The variance between individuals in SRRS was estimated and reported as the intraclass correlation coefficient (ICC) using the formula for dichotomous outcomes:
where ρ
I
is the ICC value,
Results
Table 1 shows the descriptive characteristics of the study sample. Missing sleep information resulted in the loss of 75 night observations and 7 participants for a final sample size of 625 night observations and 93 participants. Participants had an average age of 72.8 years with a range between 65 and 87 years. BMI ranged from 17.5 to 43.4 kg/m2 with 26% of the sample being classified as obese. The mean number of depressive symptoms was 4.7 out of a possible 10. Participants on average took one physician-prescribed medication. Average sleep efficiency for the 625 total nights of observation was 86.9%. Comparing the characteristics of obese and non-obese participants, independent t tests indicated that no differences were shown in age, number of medications taken for metabolic syndrome, depressive symptoms, or SRRS between obese and non-obese participants.
Descriptive Statistics, Kingston Senior Women Study, Sleep Quality Study, 625 Nightly Sleep Observations in 93 Women Overall and Stratified by Obesity Status.

Note. Level 1 consists of the nightly SRRS and actigraphic sleep observations; Level 2 consists of between-person variables. Sleep efficiency is the proportion of time during the rest period filled by sleep, as a percentage. Reported nights of restless sleep indicate the percentage of overall nights of restless sleep reported in the sample. SRRS = Self-Reported Restless Sleep.
Table 2 presents the results of the multilevel logistic regression. The ICC of SRRS was 0.37 in the null model, meaning that 37% of the variation in restless sleep reflected individual (i.e., between women) differences in SRRS. Conversely, roughly 63% of the variation was in participants’ night-to-night reporting of SRRS. The ICC increased slightly in Model 1 with the inclusion of nightly sleep efficiency. The ICC declined with the addition of the Level 2 variables in Model 2, indicating that these personal characteristics helped to explain between-person variation in SRRS. Model 1 showed that higher sleep efficiency decreased the odds of SRRS (odds ratio [OR] = 0.97; 95% confidence interval [CI] = [0.94, 0.99]). Model 2 showed that obesity status moderated the association between sleep efficiency and SRRS, while adjusting for age, depressive symptomology, and medication intake (OR = 1.08; 95% CI = [1.02, 1.14]). Using the coefficients from Model 2, Figure 1 graphed the predicted log odds of SRRS across sleep efficiency values of 0 to 100 in obese and non-obese women. Among non-obese women, as measured sleep efficiency increased, the predicted log odds of SRRS declined. Among obese women, there was no association between SRRS and sleep efficiency. Among the individual characteristics, depressive symptoms were associated with SRRS: the higher the level of depressive symptoms, the greater the chances of restless sleep (OR = 1.25; 95% CI = [1.14, 1.38]). The likelihood ratio tests showed that Model 1 was an improvement over the null model, and Model 2 represented an improvement over Model 1.
Multilevel Logistic Analyses Examining the Odds of SRRS, 625 Nightly Sleep Observations in 93 Women (ORs and 95% CIs).

Note. SRRS = Self-Reported Restless Sleep; CI = confidence interval; OR = odds ratio; ICC = intraclass correlation coefficient.
p < .01. ***p < .001.
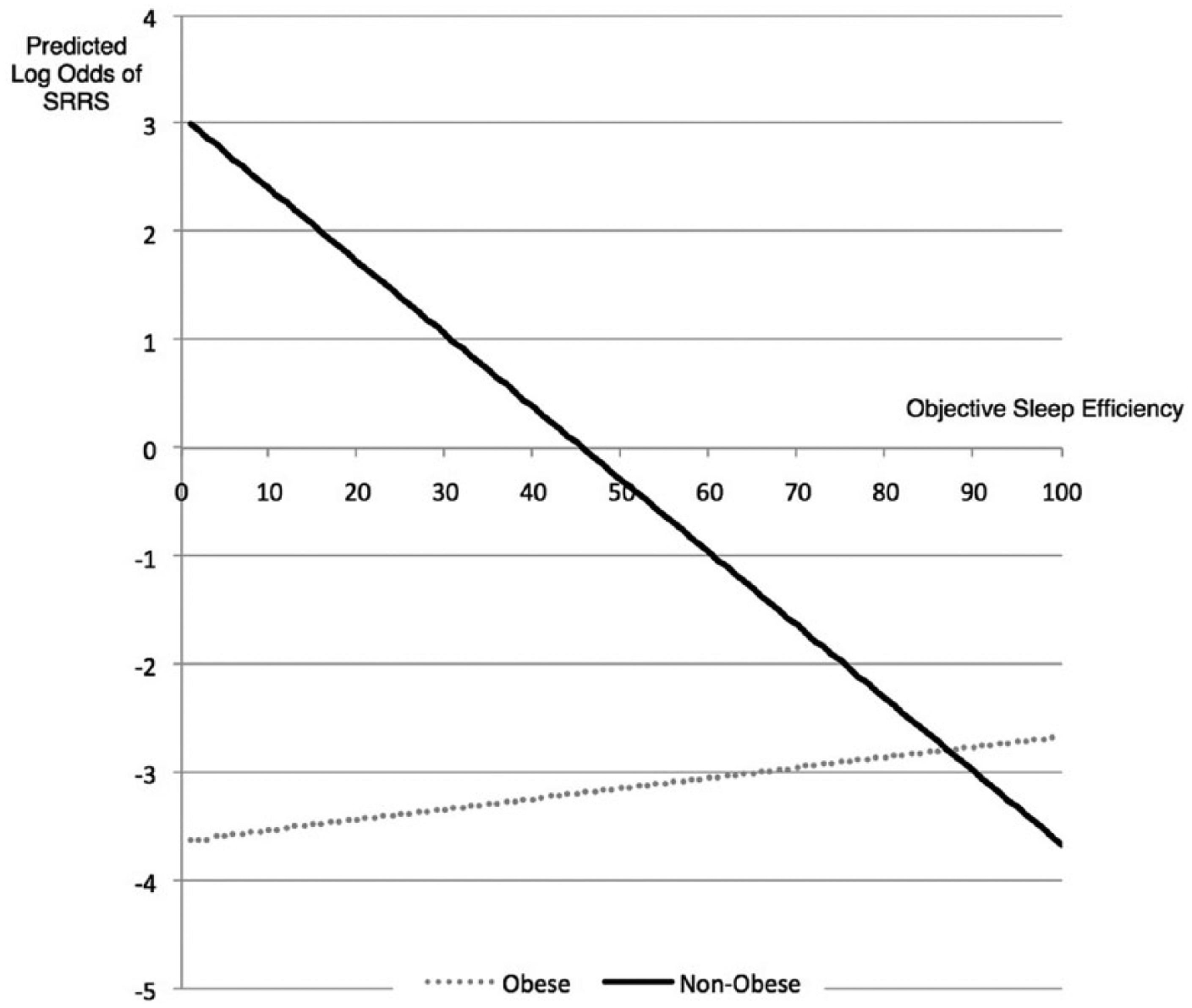
Predicted log odds of SRRS across objective sleep efficiency by obesity status.
Discussion
This study assessed the association between SRRS and objective sleep efficiency in a sample of women aged 65 years and older and examined whether obesity status moderated that association. Our findings highlight the possible impact of physiological factors on women’s subjective evaluations of sleep quality. Specifically, the study found that the association between sleep efficiency and SRRS differed in obese and non-obese women, whereby sleep efficiency did not correspond to SRRS in obese women, but it did in non-obese women. SRRS thus seems to be a poorer reflection of sleep efficiency in older obese women. These findings may have important implications for research on the relationship between sleep and obesity and health promotion programs seeking to improve the sleep of older women.
Firstly, the current study showed that the association between objectively measured sleep and SRRS differed in obese and non-obese women. Discrepancies between objective and subjective sleep assessments among obese individuals have also been reported previously (Lauderdale et al., 2008), but none to our knowledge have examined such associations in older adult women. In the general adult population, it has been found that obesity moderates the association between objectively and subjectively measured sleep duration (Lauderdale et al., 2008), such that individuals who are obese may report their sleep as shorter in duration, regardless of their objective sleep results (Lauderdale et al., 2008). Our finding that there was an association between sleep efficiency and SRRS in non-obese but not obese older women suggests that subjective sleep reports may provide a better reflection of overall sleep efficiency among non-obese individuals. Research has suggested that sleep may influence neuroendocrine function and metabolism, and the mechanisms linking sleep characteristics and obesity are likely complex and may involve metabolic disturbances and endocrine regulation, including an influence of appetite regulating hormones (e.g., ghrelin and leptin; Beccuti & Pannain, 2011). Yet, our study suggests that the possible effects of obesity on sleep quality may be underestimated if such research relies on self-reported sleep measures.
Second, in terms of potentially confounding correlates of SRRS, the study showed that women who reported more depressive symptoms over the previous week were more likely to report restless sleep. Previous research suggests there is a graded association between depressive symptoms and poor subjective sleep quality in older community-dwelling women (Maglione et al., 2012). Contrary to previous research, no associations with SRRS were found for age or medication prescribed for hypertension and cholesterol (Bloom et al., 2009; Lemola & Richter, 2013; Takada, Fujimoto, Yamazaki, Takamoto, & Hosomi, 2014).
Most studies examining the degree to which self-reported sleep quality agrees with actigraphically measured sleep have examined the mean values of sleep over a study. For example, van den Berg, Van Rooij, et al. (2008) examined the degree to which self-reported total sleep time over a 6-night period was associated with a person’s average sleep efficiency over the same period. However, this method may be limited as individual night-to-night variability in sleep patterns has been recognized as a fundamental component of disordered sleep and may offer further insight into sleep quality not obtainable through averaged data (Dzierzewski et al., 2010). Nightly variations in sleep quality and associations with health may be especially important in older adults, who are at greater risk of poor sleep and adverse health outcomes compared with younger adults (Dzierzewski et al., 2010). A multilevel design provided an opportunity to examine the association between SRRS and sleep efficiency on a nightly basis, and decompose the variation in poor sleep into within- and between-person components.
There are a number of limitations to the study worth considering. First, the KSWS consisted of a convenience sample of community-dwelling older women in Kingston, Ontario. Participants may have chosen to participate in the study due to its focus on sleep and cardiovascular health, and results may not be generalizable to other groups in other settings. Second, the KSWS was cross-sectional and therefore unable to examine potential recursive effects by which sleep quality may affect weight or depressive symptoms. Our study focused on obesity status as a possible predictor of sleep, given the overall focus of the KSWS. Third, we did not collect information on major medical illnesses. Finally, eight patients were excluded from the study because of missing sleep data that slightly decreased our sample size for analysis. Despite these limitations, our study is unique in its use of a community rather than clinical sample of older women and its analysis of a rich set of anthropometric and sleep data.
Conclusion
Findings suggest that the ability of subjective, single-item measures of restless sleep to capture variation in sleep efficiency in older women may differ depending on their obesity status. Specifically, SRRS corresponds to sleep efficiency in non-obese but not obese older women. Importantly, and in contrast to most previous work, this analysis included night-to-night variability which is considered a fundamental component of disordered sleep. The findings suggest that obesity influences sleep quality such that it results in a non-uniform impact on subjective assessment of restless sleep and objectively assessed efficiency. Further research is required to identify the mechanisms of this obesity-related dissociation. The study results also confirm that it is important to consider mental health when examining SRRS in older women, as higher levels of depressive symptoms were associated with restless sleep. This study offers a unique contribution to sleep and health research through its exploration of objective and subjective indicators of sleep on a night-by-night basis. Further research is needed to assess whether our findings are generalizable to other populations and investigate other possible individual-level characteristics that may influence SRRS.
Footnotes
Authors’ Contributions
A. W. contributed to the study conception and design, carried out the literature review, and helped with data analysis and interpretation, and drafting the manuscript. K.E.P. participated in the study conception and design, data interpretation, and manuscript revision. E.B. contributed to the study conception and design, data collection, analyses, and interpretation and helped to draft the manuscript. S.M. contributed to study conception and design, data collection, and analysis and interpretation, and helped to draft and revise the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
The KSWS was granted ethical clearance according to the recommended principles of Canadian ethics guidelines, and Queen’s policies. These principles accord with those of the Declaration of Helsinki.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This study was funded by Early Researcher Award program of the Ontario Ministry of Research and Innovation.

