Abstract
This study evaluates the safety and efficacy of Punica granatum var pleniflora mouthwash in treatment of diabetic gingivitis. In a double-blind randomized clinical trial 80 patients with diabetes mellitus and gingivitis were assigned to Golnaar and chlorhexidine 0.2% groups. After using mouthwashes for 2 weeks; participants underwent tooth scaling and the last visit was 2 weeks after scaling. The primary outcome measures were plaque, modified gingival and gingival bleeding indices, and pocket depth. Both interventions had significant improvement on all of the gingival and plaque indices (P < .001 for all indices). There were no significant differences between Golnaar and chlorhexidine in primary outcome measures except for modified gingival index for which Golnaar mouthwash had a superiority after 2 weeks when comparing with chlorhexidine (P = .039). Meanwhile, Golnaar mouthwash had no staining effect. Golnaar mouthwash is safe and effective in treatment of gingivitis in diabetic patients although further studies are recommended.
Gingivitis is a painless hyperemia of gum, which usually leads to spontaneous or easy bleeding after chewing of food or teeth brushing. 1,2 It is among the top general health problems in the world especially in developing countries. Dental plaques, gingival inflammation, and bacterial flora are the main factors in the pathogenesis of gingivitis and periodontal diseases. 1,2 Chronic gingivitis deepens the natural gingival sulcus, leads to gingival pockets, gradually loosens the periodontium and finally results in dental loss. 1 Diabetes mellitus is a chronic, systemic disease with multiorgan involvement with significant morbidity and mortality. 1 Periodontal diseases and diabetes mellitus are interrelated. 3 Diabetes is a predisposing factor for gingivitis 1,4 –8 and periodontal diseases interfere with control of blood sugar in diabetic patients. 7 –9 Furthermore, general health is affected by periodontal impairment and gingivitis may be associated with cardiovascular diseases. 1,9 –11
The best modalities for control of gingivitis are oral hygiene and continuous use of appropriate mouthwashes. 12,13 Chlorhexidine (CHX) mouthwash with acceptable influence on bacterial flora of the mouth is a standard mouth rinse for control of the gingival inflammation and plaque. 2,13 However, the main limitation of chronic use of the CHX is its staining effects on teeth. 13
The complementary and alternative medicine recommends numerous mouthwashes for control of periodontal diseases and gingivitis. But there is no adequate evidence to support their advantages and the long list of complementary and alternative medicine mouthwashes are limited to some essential oils.
2,13
–15
Traditional Persian medicine is a popular traditional medical system among many Iranian patients.
16,17
According to the traditional Persian medicine’s literatures digestive system has a close relation with many diseases such as cardiopulmonary impairments. The main medical books of traditional Persian medicine, such as al-Hāwī fī al-tibb by Rhazes (
The pomegranate, which has an Asian origin, is shown to have some benefits on oral hygiene and gingival health in several traditional Persian medicine medical and pharmaceutical literature. 35 –40
Punica granatum var. pleniflora (Golnaar in Persian) is a subspecies of pomegranate. The Golnaar is the male plant of pomegranate that only has the blooms with no fruit generation. 41 The blooms of the plant have been a royal symbol for productivity, abundance, peace, and friendship in Persian empire as shown on a petroglyph at Persepolis 42 (Figure 1).

Petroglyph at Persepolis (about 500
Also the vinegar, which is a famous vehicle for many pharmacologic compounds in traditional Persian medicine 19 –21 has been traditionally recommended for orodontal hygiene and diseases. 21,43 The combination of Golnaar and vinegar that is traditionally known as Golnaar mouthwash is a traditional Persian medicine formula for control of gingivitis. 19 –21
The aim of this study was to evaluate the safety and efficacy of the herbal Punica granatum (Golnaar) mouthwash in comparison with CHX (0.2%) in control of diabetic gingivitis.
Materials and Methods
Trial Design
The study was designed as a 2-arm, double-blind randomized comparative clinical trial using a parallel design with a 1:1 allocation ratio. There was no change in the methods after trial commencement.
Participants
All of the 3000 patients with diabetes mellitus attending the Shahid Motahhari Diabetes Clinics of the Shiraz University of Medical Sciences between July 2014 and October 2014 were invited by short message system via cellphone. The patients were asked to contact with a specific telephone number if they have any gingival problems such as gum bleeding. About 400 patients called back and almost 100 of them met the inclusion criteria and were invited for oral and dental examination by an expert dentist for confirmation of gingivitis. The inclusion criteria were the following: gingivitis, type 2 diabetes mellitus, and a controlled blood glucose levels defined as fasting blood sugar in the range of 80 to 130 mg/dL and hemoglobin A1c of ≤7.0%. Other eligibility criteria were the age of 20 to 65 years and having at least 20 teeth. The exclusion criteria were uncontrolled diabetes mellitus, to have a pocket depth of more than 5 mm, having any other systemic diseases, history of drug or food hypersensitivity, history of smoking or opium use, and consumption of antibiotics, corticosteroids, or nonsteroidal anti-inflammatory drugs in the past 3 months and during the study period. Finally, 80 patients fulfilled the eligible criteria and were allocated to intervention groups.
Sample Size
The sample size was calculated by statistician using Power SCC software. Considering 1-sided significance level of .05, a power of 0.90, the sample size was calculated to be 34 participants in each group. We considered an additional 15% probable dropout.
Interventions
A package were given to all of the patients that consists of a leaflet about dental care, a daily reminder note sheet, ordinary toothpaste, an average sized soft bristle toothbrush, a dental floss, and a 10 mL mini cup. All the participants were educated about the principles of oral hygiene and correct method of tooth brushing and flossing by a trained person. The participants were asked to gently brush and completely floss their teeth at least 2 times a day and record the time of brushing in the daily reminder sheet. Moreover, they were asked to avoid eating sticky sweet materials during the study period (Figure 2).
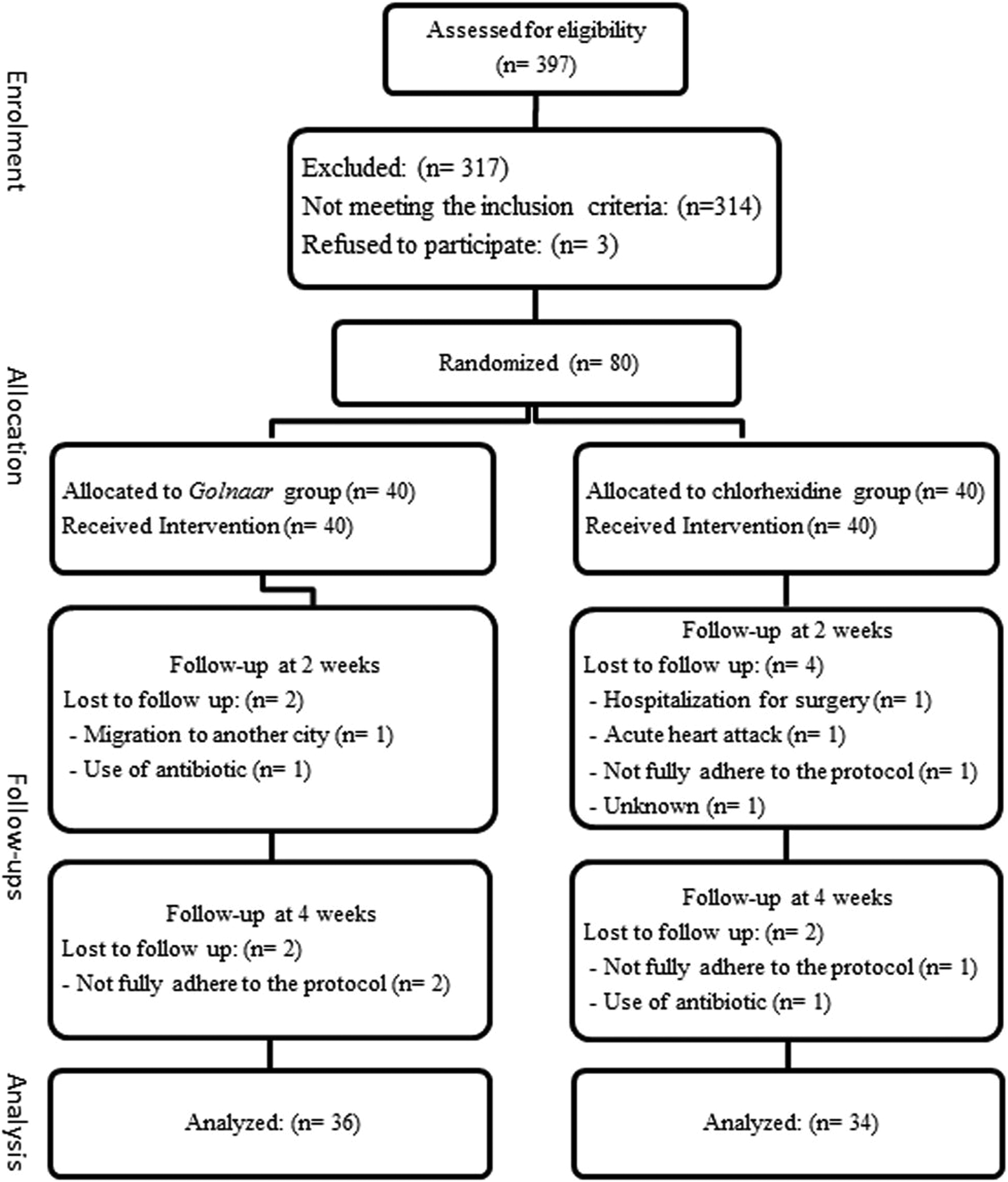
Flow diagram of the study, showing number of participants who were randomly assigned, received the interventions, and were analyzed for the outcomes.
Then a bottle of mouthwash was given to each patient and asked to use 10 mL of the liquid for 2 minutes every night for 2 weeks and bring back the bottle for the next visit for evaluation of the patient’s compliance. One group received CHX 0.2% mouthwash (the product of Iran Najo Co.) and the other received Golnaar mouthwash. At the next visits (after 2 weeks), the dental examination were done and questionnaire about the patient’s satisfaction and adverse events were filled. Dental scaling was performed for each patient by Cavitron instrument and 1 expert dentist. Each scaling lasted between 20 and 30 minutes. The patients were asked to discontinue the mouthwash and return after 2 weeks (postscaling) for the third visit and last dental examination.
After 4 weeks, all the patients were visited and examined for the third time and their fasting blood sugar levels were rechecked for reassurance about the control of blood sugar throughout the study period. All the history takings were done by 1 trained person and all of dental examinations were undertaken by 1 expert dentist.
Preparation of Golnaar Mouthwash
Punica granatum var pleniflora (Golnaar) flowers were provided from a medicinal plants market (Herbal Drugstore of Motahhari Clinic) and authenticated by a botanist and then a sample of the herb was deposited at the herbarium center of Department of Phytopharmaceuticals, School of Pharmacy, Shiraz University of Medical Sciences and a voucher number (PM 848) was specified for the medicament.
To prepare the mouthwash in an amber glass bottle (240 mL), 10 g of dried Golnaar powder was mixed with adequately 40 mL of vinegar sample (a certain brand with a determined acid value index to keep the batch-to-batch uniformity) and the mixture was diluted to 1:5 with boiling distilled water. The preparation was then cooled, filtered, and filled in the amber glass bottle. The remedy was then subjected for total tannin content determination and microbial evaluation before intervention. The Golnaar mouthwashes tannic acid content was 0.52 ± 0.03 mg/mL and regard to the microbial control results; none of the 5 main pathogens (Staphylococcus aureus, Shigella, Salmonella, Escherichia coli, and Candida) had grown on the related specific media. Golnaar mouthwash is not a product of pharmaceutical company and was just produced for use in this research.
Outcomes
All the participants were evaluated for outcome measures at first visit, after 2 and 4 weeks. The primary outcome measures were plaque index (PI), 44 modified gingival index (MGI), 45 gingival bleeding index (GBI), 46 and pocket depth (PD). Table 1 shows the details of the measurement of the quantitative and qualitative plaque and gingival indices. The secondary outcome measures were evaluation of the patients’ satisfaction and the number of participants with any adverse events. The patients’ satisfaction and tolerance were evaluated with the visual analogue scale and ranged from 1 (not satisfied at all) to 5 (fully satisfied). The scores were: 1 = not satisfied at all; 2 = not satisfied adequately; 3 = not good–not bad (so so); 4 = mostly satisfied; and 5 = fully satisfied.
The Quantitative and Qualitative Plaque and Gingival Indices.

aThe calculation of the mean score for each index was done by dividing the sum of scores to the number of teeth.
Randomization, Blinding, and Allocation Concealment
Eighty eligible patients were randomly allocated to 2 parallel groups by the secretary of the Dentistry Clinic who had been instructed to use a randomized list. The randomized list was generated using Microsoft Excel with a block randomization method. After obtaining a written informed consent the demographic measures such as age, gender, weight, and height for calculation of body mass index were recorded. The baseline quantitative and qualitative plaque and gingival indices were measured.
The bottle containers were same in regard to shape, size, and color of the bottle and its contents. Each bottle had a numerical label that only the pharmacist who produced them was aware of its content and all the data were recorded under the patients’ bottle number.
The trained person, dentist, researchers, and statistician were blinded to the allocation of patients.
Statistical Methods
The data were analyzed via the SPSS software (IBM Corp, Released 2013, IBM SPSS Statistics for Windows, version 22.0 Armonk, NY).The normal distribution assumption was checked by Kolmogorov-Smirnov test and Q-Q plot. The frequency (percent), mean ± SD (standard deviation), median and interquartile range were used for data description. The independent-samples t test, Mann-Whitney, chi-square, and Fisher’s exact tests were used for comparison of the difference between the 2 groups. Changes within an intervention group were evaluated with Wilcoxon signed rank test and paired sample t test. Analysis of covariance was used for comparison of both group adjusted for the baseline variables. A P value of <.05 was considered as statistically significant.
Results
General and Demographic Data
A total number of 80 patients (50 women and 30 men) with mean age of 51.33 ± 6.75 years, mean body mass index of 28.01 ± 4.47 kg/m2, and mean diabetes duration of 9.96 ± 6.23 years were entered into the initial study. Finally, 70 patients (36 in Golnaar group and 34 in CHX group) completed the study. The patients had between 20 and 32 teeth (24.86 ± 3.44 teeth). Ten participants (12.5%) withdrew from study; 6 of them did not come for the second visit (2 in Golnaar group and 4 in CHX group) and 4 patients did not come for the third visit (2 in Golnaar group and 2 in CHX group) (Figure 2).
The comparison of the baseline demographic criteria, Hemoglobin A1c and gingival indices between the 2 intervention groups shows no statistically significant difference (Tables 2 and 3).
The Baseline Demographic Criteria in Both Intervention Groups (n = 70).
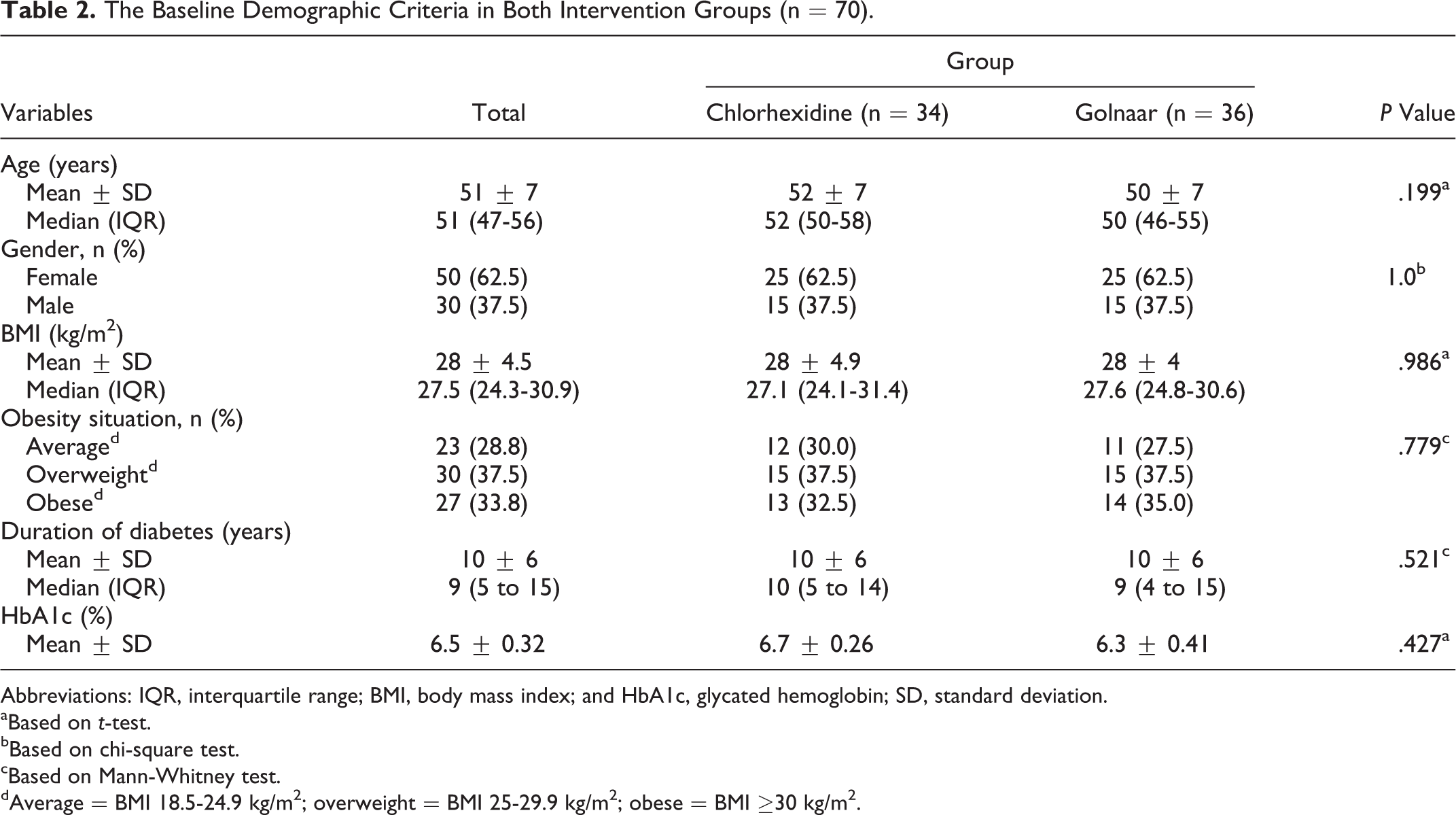
Abbreviations: IQR, interquartile range; BMI, body mass index; and HbA1c, glycated hemoglobin; SD, standard deviation.
aBased on t-test.
bBased on chi-square test.
cBased on Mann-Whitney test.
dAverage = BMI 18.5-24.9 kg/m2; overweight = BMI 25-29.9 kg/m2; obese = BMI ≥30 kg/m2.
The Differences of Primary and Secondary Outcome Measures Between 2 Intervention Groups (n = 70).

Abbreviations: CI, confidence interval; GBI, gingival bleeding index; MGI, mean gingival index; PD, pocket depth; PI, plaque index; Pre, at the starting of the study; After 2W, 2 weeks after using mouthrinse; After 4W, 4 weeks after starting the study (end point of the study).
aBased on Mann-Whitney test.
bAdjusted for the baseline, based on analysis of covariance.
cBased on Wilcoxon signed rank test.
dBased on t test.
eBased on paired t test.
Primary Outcome Measures
As shown in Table 3, after 2 weeks of using the mouthwashes, GBI improved dramatically from 13.3% ± 9.72% and 14.76% ± 12.25% at baseline to 3.87% ± 5.7% and 3.42% ± 5.63% in CHX and Golnaar groups, respectively (P < .001). Similarly, MGI, PD, and PI showed significant improvement after 2 weeks compared with baseline levels in both group (P < .001). At this juncture (after 2 weeks), Golnaar showed a better effect on MGI compared with CHX (P = .039).
At the end of the study (after 4 weeks) although GBI, MGI, PD, and PI showed significant regression in both intervention groups, there were no significant differences between Golnaar and CHX groups (Table 3).
Secondary Outcome Measures
Although there was more patients’ satisfaction in Golnaar group (94.7% vs 88.9%) and the overall adverse events were more in CHX group (42.1% vs 58.3%), the statistical analyses showed no significant difference between these groups (P = .263 and P = .349, respectively). On the other hand, the detailed adverse event data revealed significant difference between the 2 study groups (P < .001). Meanwhile, Golnaar mouthwash had no staining effects on teeth and seem to be more tolerable than CHX (97.4% vs 83.3%; P = .053) (Table 4).
The Comparison of the Patients’ Satisfaction and Number of Adverse Events Between Intervention Groups After 2 Weeks (n = 74).

aBased on Mann-Whitney test.
bBased on Fisher’s exact test.
cBased on chi-square test.
Power
The power for each outcome measurement of the study groups is shown in Table 5.
The Power of the Different Outcome Measures in Each Group After 2 Weeks (n = 74).

Discussion
This clinical trial shows that the traditional herbal Golnaar mouthwash is effective and safe in treatment of gingivitis in diabetic patients and its effects are comparable to CHX (0.2%) mouthwash in improvement of all the gingival and plaque indices (GBI, MGI, PI, and PD) after 2 weeks.
At the present time, the best method for preventing and controlling the gingivitis is recommending appropriate oral hygiene, which consists of treatment of dental decays, frequent teeth brushing and flossing, and a healthy food regimen; avoidance of confection and any sticky sweet materials, more consumption of fresh vegetables and fruits, and regular use of mouthwash solutions. 1,2,12,20,47,48 It had been shown that CHX and some other essential oils are effective mouthwashes. 2,12 –15 Though CHX is the commonly prescribed mouthwash for control of gingival and periodontal diseases, its bitter taste and staining effects on teeth are among the main obstacles against its general and public use. 49 Therefore there is a real demand for finding effective and safer alternative mouthwashes. Nowadays, complementary and alternative modalities have more usages in different medical fields. 17,50 According to the recommendation of the main traditional Persian medicine’s textbooks, amalgamation of the pomegranate (Punica granatum) and vinegar can be a good choice for prevention and treatment of gingival and periodontal diseases. 19 –22 Meanwhile, pomegranate has been shown to have antibacterial, 35,36,51 anti-inflammatory, 36 antioxidative, 36 and healing effects on gingival and periodontal impairments. 43 Also, no major adverse event or contraindication is reported for Punica granatum in its usual dosage. 52
To the best of our knowledge, there isn’t strong, well-designed, documented evidence about the use of pomegranate in a mouthwash solution, especially its subspecies; Punica granatum var pleniflora (Golnaar) in treatment of periodontal diseases and specifically in diabetic patients with gingivitis.
This study is a double-blind randomized comparative clinical trial that compared the Golnaar mouthwash with the conventional CHX mouthwash in diabetic patients with gingivitis and revealed that both are effective after 2 weeks and improved the gingival and plaque indices dramatically in comparison with their baseline situations. Meanwhile, the Golnaar group showed a better controlling power on MGI (P = .039) and the patients’ satisfaction was similar between both intervention groups. All of the patients had a controlled diabetes mellitus throughout the study. In this study, nearly all the required gingival and plaque indices were evaluated meticulously in each patient as outcome measures. All the patients were examined in 2 follow-up visits by 1 expert dentist who was blinded to the interventions. Also, the patients’ satisfaction and adverse events were evaluated concomitantly. Apparently, the overall adverse events in Golnaar group were fewer than CHX and there was more satisfaction with use of Golnaar mouthwash. Golnaar mouthwash had no staining effects on teeth and seems to be more tolerable.
Although it was shown that CHX and Golnaar have improved all the primary outcomes, as derived from Table 5; the powers of this study for PI and PD in both groups were low (between 7.3% and 55.2%) particularly in CHX group. By increasing the sample size of the study, such effects might not be seen. On the other hand, the powers of the study were very high for the outcomes such as GBI and MGI especially in the Golnaar group; 98.9% and 100%, respectively, compared with 95.5% and 88.1% in CHX, which emphasizes that apart from a significant P value (<.001), we had a very acceptable type-2 error in these outcomes.
To sum up, as mentioned before, a mouthwash that is a combination of pomegranate or its subspecies Punica granatum var pleniflora in vinegar, by different potential mechanisms such as providing better perfusion of the gingival tissue and hardening it, 41,43 affecting plaque bacterial flora, 35,36,51 having anti-inflammatory effect on gum, 36 exerting antioxidative influence, 36 and bringing back the swollen and edematous gingiva to its normal dry state, 41 may be an effective remedy for gingival and periodontal impairments.
Study Limitations
Our study had some limitations. We believe that the sample size was a bit small; although, all the 3000 registered diabetic patients in the major diabetic clinic of the city were invited for gingival problems, only 80 patients fulfilled the eligible criteria and entered initially into the study. Also, the study had a short-term follow up; the staining effect of the CHX mouthwash was a major restriction for longer administration of mouthwashes and follow-up. Another limitation was the absence of a placebo group to rule out placebo effects or the changes due to adherence to correct dental hygiene as potential reasons for the improvements achieved in both groups. Although all the patients were trained about the principles of oral hygiene and correct way of teeth brushing and flossing, and were asked to avoid sticky sweet materials, they were not under our direct daily observation and we were not sure about their careful attention to the study protocol. However, a daily reminder leaflet was given to them; in addition, they were asked to tick a daily checklist. Also a sticker with checkboxes was attached to the back of each mouth rinse bottle and participants were asked to tick them daily and bring back the used bottles for the second visit.
Accumulation of bacteria on dental plaque is an important etiological factor of gingivitis; thus teeth scaling is an effective therapy that usually performed at the beginning of gingivitis management. But in this study we performed teeth scaling 2 weeks after using mouth rinse. This is because, on one hand, we needed to reveal the effects of our traditional mouthwash on gingival indices, so performing the scaling from the beginning certainly interferes with the results and on the other hand, we ought to provide a complete gingival treatment for our participants, thus we performed the scaling after using mouthwashes, which is not a routine way.
Conclusion
This study showed that the use of traditional herbal Punica granatum var pleniflora (Golnaar) mouthwash is a safe and effective modality in treatment of gingivitis in diabetic patients as compared with the CHX (0.2%) mouthwash and may be considered as a good alternative. More randomized clinical trial studies with larger sample sizes and longer follow-ups are needed to confirm the effects of Golnaar mouthwash and for evaluation of its possible adverse effects.
Footnotes
Acknowledgments
The authors would like to express special thanks and appreciation to Mehdi Yaseri and Dr Mojtaba Heydari for their scientific support and guidance in writing the article.
Author Contributions
The work presented in this article was carried out through collaboration between all authors. MSR, MS and MMF made the initial hypothesis. All authors participated in defining the research theme and providing the proposal. MSR, MS, MMF, and MRC visited the patients, enrolled them, and followed-up on their progress. MMZ made the drugs. MSR, MMZ, MMF and MS wrote the article. MS supervised the work. All authors have contributed to, edited, and approved the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Shiraz University of Medical Sciences (Reference No. 93-7064).
