Abstract
Dry mouth is a common complication of radiotherapy for head and neck cancers. This study compared the efficacy of an herbal compound containing Malva sylvestris and Alcea digitata (Boiss) with artificial saliva (Hypozalix) for improving the symptoms of dry mouth in head and neck cancer patients. The study examined a total of 62 subjects assigned to 2 groups. The herbal compound and Hypozalix were administered for 4 weeks. Efficacy was assessed using the visual analog scale and by grading the degree of dry mouth. Both groups showed a significant difference between visual analog scale before and following intervention. There was also a significant difference in visual analog scale between groups at 4 weeks after onset of intervention. The herbal group showed a significant difference between the grade of dry mouth before and after intervention, but no change was observed for grade of dry mouth in the Hypozalix group. This study supports the efficacy of the herbal compound for controlling symptoms of dry mouth in head and neck cancer patients.
Head and neck cancers comprise about 5% of cancers diagnosed worldwide and result in 350 000 deaths annually. 1,2 Radiotherapy plays an important role in the treatment of the majority of patients with head and neck cancer. 3 Dry mouth (xerostomia) is a common debilitating side effect of radiotherapy for head and neck cancer 4 that affects chewing, eating, tasting, swallowing, tooth decay, and speaking. 3 Despite advances in current medicine, there is no completely effective treatment for radiation-induced xerostomia and the treatments are often symptomatic. 3,4
Malva sylvestris L and Alcea digitata (Boiss) Alef have been used as herbal remedies in traditional Persian medicine for their antitussive, antioxidant, expectorant, anti-inflammatory, antimicrobial, and laxative therapeutic effects. 5,6 They are useful for lubrication of the throat and lungs and in respiratory disorders. Studies have shown these plants to be immune stimulants that are useful in mucositis. 6 Malva sylvestris L and Alcea digitata (Boiss) are mucilaginous plants that have long been used in traditional Persian medicine to treat disorders like dry mouth; however, to our knowledge, no clinical trial has examined the efficacy of Malva sylvestris and Alcea digitata on dry mouth. The present study evaluated the efficacy of this herbal compound (Malva sylvestris and Alcea digitata powder) on radiation-induced dry mouth in subjects with head and neck cancer.
Materials and Methods
In this randomized clinical trial, the subjects were 62 patients with radiation-induced xerostomia who came for follow-up treatment to Imam Hossein Hospital Oncology Clinic; they were recruited from September 2013 to May 2014. Eligible subjects were randomly allocated into 2 groups, those receiving the herbal compound containing Malva sylvestris and Alcea digitata powder (experimental group) or those containing Hypozalix artificial saliva (control group) 3 times per day for 4 weeks. Hypozalix is artificial saliva that is routinely used to treat dry mouth in Iranian oncology clinics.
The experiment was explained to potential subjects, and they were asked to provide written informed consent before participating in the study. The study protocol was approved by the ethics committee of Shahid Beheshti University of Medical Sciences and registered in the Iranian Registry of Clinical Trials. Inclusion criteria consisted of being 20 to 70 years of age and having been diagnosed with a minimum of grade 1 radiation-induced xerostomia at least 3 months prior to the beginning of the study. Subjects with connective tissue disorders, those taking medications affecting salivary gland secretions, and those with a chronic disease affecting body hydration were excluded.
The herbal compound was obtained and processed using standard methods under the supervision of a pharmacology professor at Tehran University of Medicinal Sciences. The powdered form of both plants were mixed and kept in 4 g sachets containing equal portions of Malva sylvestris and Alcea digitata.
The subjects were examined by the investigator at the beginning of the study, at 2 weeks, and at 4 weeks after onset of treatment. Personal data were collected by interview and physical examinations of subjects during their visits. In the first visit, the mode of application of each medication was explained to the subjects who met the inclusion criteria. The efficacy of each treatment was assessed by subjects using the visual analogue scale (VAS), and their dry mouth symptoms were graded by the investigator. The VAS was scored from 0 to 10, where 0 denotes the absence of dry mouth and 10 denotes severely dry mouth.
VAS scores were compared within each group and between groups prior to intervention and at 2 weeks and 4 weeks after onset of treatment using analysis of variance for repeated measures. If the results of Mauchly’s test showed sphericity to be significant (P < .05), a Greenhouse–Geisser test was applied; if the results of Mauchly’s test were not significant (P > .05), an assumption of sphericity test was administered. The χ2 test or Fisher’s exact test was used to compare the grade distribution between groups and within groups; the grade was assessed using the Friedman test.
Results
A total of 62 patients (41 male and 21 female) were enrolled as subjects in the study. Mean age was 51.5 in experimental group and 50.3 in the control group (which received Hypozalix). The experimental group received the herbal compound, and the control group received Hypozalix. In the experimental group, 42 subjects initially participated and 32 completed the study. In the control group, 33 subjects initially participated and 30 completed the study. The drop in participants resulted from complications such as nausea, vomiting, epigastric pain, or the subject’s assessment that the medication was not effective. Complications between groups were not statistically significant. There were no significant differences between groups for age distribution or other background variables.
The mean VAS was statistically significant at all test times for the experimental group (VAS1: before intervention; VAS2: at 2 weeks; VAS3: at 4 weeks). The difference between means for VAS1 and VAS2 and VAS1 and VAS3 differed significantly (P < .001). The means for VAS2 and VAS3 also differed significantly (P < .001). The differences between mean VAS at all test times for the control group were statistically significant (P < .05); the difference between mean VAS1 and VAS2 and VAS1 and VAS3 differed significantly (P < .001) and between VAS2 and VAS3 (P < .013). Differences between mean VAS1 and VAS2 for the experimental and control groups were not significant, but there was a significant difference between VAS3 for the experimental and control groups (P < .001) as shown by the results of the Greenhouse–Geisser test in Figure 1.

Visual analog scale score comparison between experimental and control groups.
There was a significant difference in the experimental group between the grade of dry mouth before intervention (grade A) and at 4 weeks after onset of intervention (grade C; P < .018) and at 2 weeks (grade B) and at 4 weeks after onset (grade C; P < .018). There was no significant difference between grade A and grade B. In the control group, there was a significant difference between grades. There was no significant difference between groups for grade A, B, or C as calculated by the χ2 or Fisher’s exact tests, as shown in Figure 2.
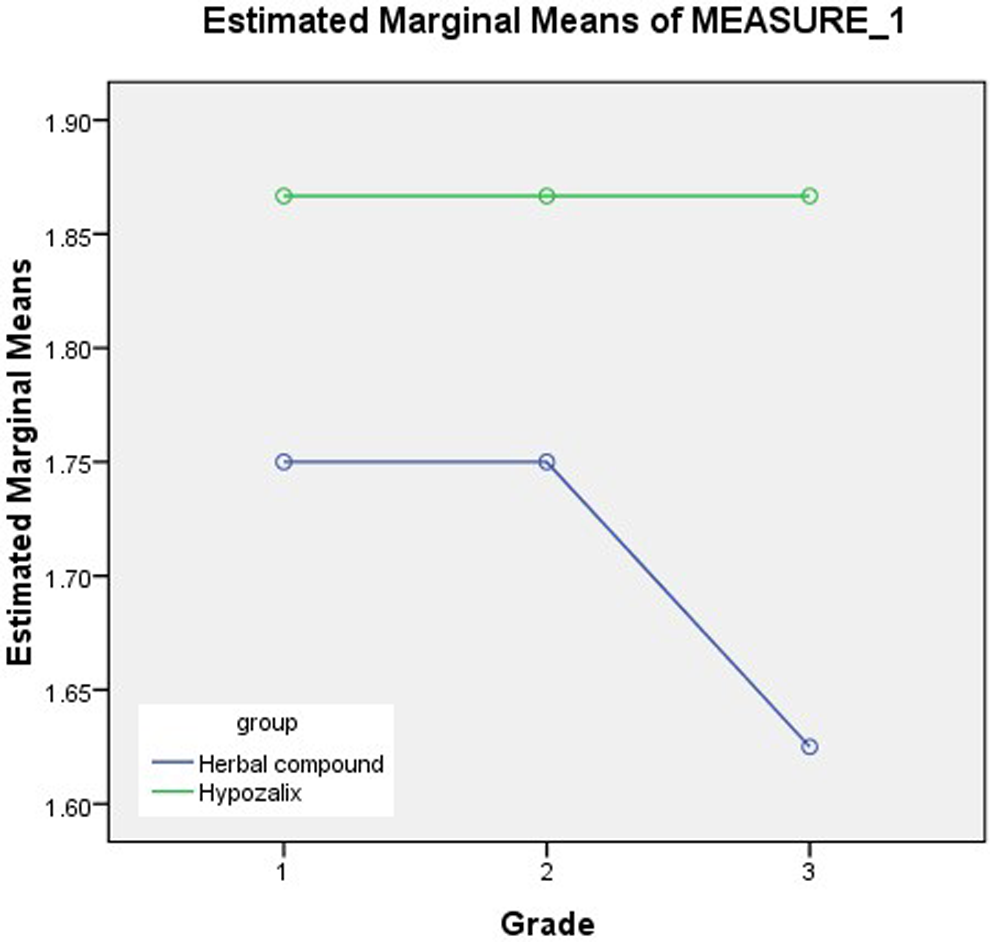
Comparison of grade of dry mouth in the experimental and control groups.
Discussion
Dry mouth is a common side effect of radiation therapy for head and neck cancer and has a negative effect on quality of life. 7 Dirix et al found that the majority of head and neck cancer patients (93%) suffer from dry mouth and 65% experience moderate to severe dry mouth. 7 In this study, 80.6% of subjects experienced moderate to severe dry mouth and required relief in the form of saliva replacement.
Traditional Persian medicine describes Malva sylvestris and Alcea digitata as mucilaginous plants that are used for their emollient and laxative properties. 5,6 Alcea digitata is a species that is sold in Iranian herbal markets. Alcea digitata is just like Althea officinalis, which is known as medicinal species in research studies. Studies have confirmed the antioxidant, 8 anti-inflammatory, 9,10 and antimicrobial 11,12 effects of Malva sylvestris and Althaea officinalis. Commission E (A scientific advisory board of the “Bundesinstitut für Arzneimittel und Medizinprodukte” (the German equivalent of the HYPERLINK “http://en.wikipedia.org/wiki/Food_and_Drug_Administration” \o “Food and Drug Administration” Food and Drug Administration (FDA)), formed in 1978. The commission gives scientific expertise for the approval of substances and products previously used in traditional, folk and herbal medicine) has approved the use of mallow to treat irritation of the mucosa of the mouth and throat and dry cough associated with this condition. 13 Deters et al found that Althaea officinalis can be used to treat irritated mucous membranes to help in tissue regeneration. 14 Rouhi et al found that the extract of Althaea officinalis and Zingiber officinalis Rosce decreased inflammation in patients with acute bronchitis and tracheitis and soothes coughs. 15
Medline research produced no previous studies on the use of Malva sylvestris and Alcea digitata (or Althaea officinalis) in the management of dry mouth in patients with cancer. The current study evaluated the efficacy of an herbal compound containing Malva sylvestris and Alcea digitata on radiation-induced xerostomia and compared the results to the efficacy of Hypozalix. Dry mouth symptoms were graded according to the common criteria for adverse events (version 4.0) and VAS was assessed. The duration of relief was not determined and salivary flow was not measured.
The herbal compound and Hypozalix showed beneficial effects on the symptoms of dry mouth. Although VAS decreased for both the experimental and control groups, a comparison between the 2 groups showed that, at 4 weeks, the VAS score for the experimental group decreased significantly when compared with that for the control group. This supports the observation by subjects of a preference for the herbal compound. Three subjects in the experimental group experienced a decrease in the grade of dry mouth, but there was no change in the grade of dry mouth in the control group. The change of grade was not statistically significant between groups. Changes in the grade of dryness at 2 weeks suggest that further study with a larger sample size of longer duration is needed to determine whether the herbal compound is effective for improving the grade of dry mouth.
There was large variation observed in the decrease in dry mouth between subjects in both groups. This response pattern may have been related to different types of structural damage to the salivary glands that resulted in varying degrees of saliva deficiency.
Studies have shown that medicinal mucilage used to treat dry mouth operates from 2 possible mechanisms. In the first, it may act like a salivary secretion to lubricate and protect mucus membranes. The second mechanism has been described from traditional Persian medicine as relating to the qualities of the plant; the medicinal mucilage is said to have wet and cold/moderate qualities that act against dryness. 16 It was suggested in this study that Malva sylvestris and Alcea digitata are mucilaginous plants that can be used to improve dry mouth.
Conclusion
The present study supports the efficacy of the experimental herbal compound containing Malva sylvestris and Alcea digitata for the control of symptoms of radiation-induced xerostomia.
Footnotes
Authors’ Note
This research was derived from a PhD thesis of Ghazaleh Heydarirad at the School of Traditional Medicine, Shahid Beheshti University of Medical Sciences.
Author Contributions
This work was performed by Heydarirad. Ameri and Choopani conceptualized the study, developed the inclusion criteria, collected the data, and reviewed the article. Rezaeizadeh suggested the idea of this study. Ghobadi prepared the experimental medicine. Gachkar analyzed the data, and Heydarirad wrote the first draft of the article.
Declaration of Conflicting Interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, or publication of this article.
Ethical Approval
The study protocol was approved by Ethics Committee of Shahid Beheshti University of Medical Sciences; number: 143.
