Abstract
This study investigated various chemical and antioxidant properties of Anatolian royal jelly samples. Moisture, pH, total protein, 10-hydroxy-2-decenoic acid (10-HDA) and sugars were analyzed from 18 samples. Total phenolic contents, ferric reducing antioxidant capacity and 2,2-diphenyl-1-picryhydrazyl (DPPH) free radical scavenging activity were measured as antioxidant determinants. 10-HDA contents and total protein content of fresh weight ranged between 1.0% and 3.9%, and 11.4% and 15.8%, respectively. The main sugars detected were glucose and fructose. Maltose, trehalose, and melibiose were detected at less than 1.0% in all samples. Lactose, a milk sugar, was detected in only 3 samples, at values between 0.8% and 1.4%. Total henolic content ranged from 91.0 to 301.0 mg gallic acid equivalents/kg fresh weight. Antioxidant activity is due to both to the total phenolic content, proteins and fatty acids of royal jelly. Anatolian royal jelly samples were not different from other royal jelly samples from across the world.
The use of royal jelly has increased considerably in recent years because of its numerous attractive properties. This completely natural product is secreted from special glands of young worker bees (Apis mellifera L.) and is used to feed the larvae. Queen bees are fed on royal jelly throughout the larval period, associated with the fertility and longevity of the queen, while nurse bees are fed royal jelly for only 3 days. 1,2 Humans today believe they can extend their own life spans by consuming royal jelly, similarly to queen bees. Studies have shown that royal jelly exhibits many beneficial properties, such as anti-inflammatory, antibacterial, antioxidant, vasodilative and hypotensive, immune activating, and antitumoral activities. 1,3,4 Pharmacological and experimental animal studies have proved the beneficial properties of royal jelly, including vasodilative, antifatigue and hypotensive activities, 5 cell growth enhancement, 6 antibacterial effects, 7 antitumoral 4 and anti-inflammatory activities, 8 antiaging effects, 9,10 hepatoprotective effects against toxic agents, 11 and immunomodulatory and antihypercholesterolemic activities. 12
The chemical composition and biological active properties of royal jelly is affected by factors such as floral variety and geographical and environmental conditions. 1,3 Generally, lyophilized royal jelly contains less than 5% water, 27% to 41% proteins, 22% to 31% carbohydrates, and 15% to 30% lipids. 1,13 Unlike most animal fatty acids, royal jelly contains short-chain fatty acids, 8 to 10 carbon atoms, and mostly either hydroxy- or dicarboxylic fatty acids, such as 8-hydroxyoctanoic, 3-hydroxydecanoic, 9-hydroxydecanoic, 9-hydroxy-2-decenoic, 10-hydroxydecanoic, 10-hydroxy-2-decenoic (10-HDA), 3,10-dihydroxydecanoic, 2-octene-1,8-dioic, and 2- decene-1,10-dioic acids. 1,14 –16 10-HDA is thought to be present only in royal jelly and is known to have many pharmacological properties. 1,15,17,18 10-HDA is regarded as the most important compound in evaluating royal jelly quality criteria.
Because of its many beneficial properties, royal jelly is becoming increasingly used as a dietary supplement for children and athletes, and in cosmetic supplements. The Food and Agriculture Organization describes 100 to 300 mg of royal jelly as the most commonly recommended daily dosage for human use. 19 Different production styles and geographical, regional, climatic, and floral differences can also change the composition and biologically active properties of royal jelly. Turkey is one of the richest regions of the world in terms of bee production and variety, because there are some conducive features such as geographical position and climatic conditions. So, it is suitable for honey production during 3 seasons of the year. While there has been some previous investigation of Turkish royal jelly, studies have not assessed its composition and antioxidant properties. 20 The purpose of this study was to reveal the composition and antioxidant properties of Turkish royal jelly.
Materials and Methods
Chemicals
All chemicals and solvents were analytical grade standards. Sugars (fructose, glucose, sucrose, lactose, maltose, trehalose, melibiose, and melezitose), acetonitrile and methanol were supplied by Sigma-Aldrich Co. (St. Louis, MO, USA). High-performance liquid chromatography (HPLC) syringe filters (RC-membrane, 0.45 µm) were purchased from Sartorius Minisart RC 15, Sartorius (Germany). 2,4,6-Tripyridyl-s-triazine (TPTZ), Folin-Ciocalteu’s phenol reagent, 2,2-diphenyl-1-picrylhydrazyl (DPPH), butylated hydroxytoluene (BHT), Trolox (6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid), 10-hydroxy-2-decenoic acid (10-HDA), methyl 4-hydroxybenzoate (MHB), K4Fe(CN)6·3H2O and Zn(CH3COOH)2·2H2O were purchased from Sigma Chemical Co. (St Louis, MO, USA).
Samples
Eighteen fresh royal jelly samples were collected from local Anatolian beekeepers in the 2014 harvest season. These were transferred to our laboratory packed in dry ice. The samples were lyophilized using CHRIST Alpha 1-4 LD plus (Germany). The lyophilized samples were stored at −20°C until use.
Analyses
Water, pH, and crude protein contents of the fresh royal jelly samples were determined using the AOAC method. 21 Total protein contents of the lyophilized samples were determined using the Kjeldahl method with distillation parameter optimization (Digester K-424, Distiller KjelFlex K-360 and titrator SchottTitro Line). pH was measured with samples diluted with bidistilled water (1:5, g/mL) using a pH meter (Mettler Toledo 8603 Schwerzenbach, China).
Royal jelly sugar analysis was performed using a Hitachi LaChrom HPLC system, equipped with an RID detector Nucleosil (EC 200/4.6 Nucleosil 100-5 NH2) column adjusted to 45°C as column temperature. The mobile phase was a mixture of acetonitrile and ultrapure water (79:21, v/v) and injection volume 1.5 mL/min. The total run time of each injection volume was 15 minutes, and volume of each injection was 20 µL. The method makes it possible to measure fructose, glucose, lactose, sucrose, maltose, melibiose, and melezitose in royal jelly. First, 0.2 g from each sugar standard was accurately weighed and dissolved in 10 mL of solvent water for a final concentration of 20 mg/mL. For calibrations of the sugars, the stock solutions were diluted at concentrations of 2.5, 5, 10, and 20 mg/mL. 22
Exactly 0.25 g of lyophilized royal jelly was dissolved in 1.5 mL acetonitrile/ultrapure water solution (50:50 v/v). Flask contents were mixed, and the solution was then centrifuged at 12 000 rpm for 10 minutes to remove the protein fraction. The supernatant was filtered through a 0.45-μm filter and then used as solutions for HPLC injection.
10-Hydroxy-2-Decenoic Acid Analysis
10-Hydroxy-2-decenoic acid analysis was performed using HPLC on a Hitachi LaChrom HPLC system, equipped with a variable wavelength ultraviolet absorbance detector and column Lichrosper 100 RP-18 (10 µm) adjusted to 25°C as column temperature. The maximum absorbance of 10-HDA was confirmed at 215 nm through ultraviolet-visible absorption measurements. The mobile phase was a mixture of methanol, ultrapure water, and phosphoric acid (55:45:2.7, v/v/v) with a flow rate of 0.75 mL/min. The total run time of each injection volume was 15 minutes. The injection volume was 20 µL for each sample. 23
Briefly, 16 mg of 10-HDA was completely dissolved in solvent (methanol and ultrapure water, 50:50, v/v) to prepare 100 mL of stock solution with a concentration of 160 µg/mL. By diluting this stock solution, a series of 10-HDA solutions were prepared. Next, 0.1 g of MHB as internal standard was dissolved in the same solvent mixture. Finally, solutions for the construction of a standard curve were prepared by mixing an equal volume of MHB and 10-HDA solutions. 23
Approximately 25 mg of dried royal jelly sample was dissolved in 12.5 mL of solvent (methanol and ultrapure water, 50:50, v/v), and mixed for at least 20 minutes. The sample was then filtered at 0.45 µm and the filtrate was injected with an equal volume of MHB.
Total Phenolic Content
Total phenolic content of the methanolic extracts was determined with the Folin-Ciocalteu method using gallic acid as standard. 24 First, 680 µL distilled water and 20 µL methanolic royal jelly extracts and 400 µL of 0.5 mol/L Folin-Ciocalteu reagents were mixed in a tube and vortexed for 2 minutes. Next, 400 µL Na2CO3 (10%) was added and incubated for 2 hours at room temperature. The absorbance was measured at 760 nm at the end of the incubation period. All the measurements were performed in triplicate. Total phenolic content was expressed as milligrams of gallic acid equivalents per kilogram of royal jelly samples (mg GAE/kg royal jelly).
Ferric Reducing Ability of Plasma
The reducing ability of ferric tripyridyltriazine (Fe-III-TPTZ) complex was used for total antioxidant capacity assay. 25 Working ferric reducing ability of plasma (FRAP) reagent was prepared as required by mixing 25 mL of 300 mmol/L acetate buffer, pH 3.6 with 2.5 mL of 10 mmol/L TPTZ solution in 40 mmol/L HCl and 2.5 mL of 20 mmol/L FeCl3ċ6H2O solution. Three milliliters freshly prepared FRAP reagent and 100 µL of aquatic sample was mixed and incubated in 4 minutes at 37°C, and the absorbance was read at 595 nm against reagent blank containing distilled water. Trolox was used a positive control to construct a reference curve (62.5-1000 µmol/L), FRAP values were expressed as µmol/L Trolox equivalent of kg.
Determination of Free Radial Scavenging Activity
The scavenging of 2,2-diphenyl-1-picrylhydrazyl (DPPH) radicals was used to determine radical scavenging activity of the aquatic royal jelly samples. The colorimetric test was assayed using the Molyneux method. 26 DPPH radical has a purple color, and this color decays in the presence of antioxidant agents, and the change in the absorbance can be measured at 517 nm. For each sample, 1.5 mL of the extract solution was mixed with 1.5 mL of 0.1 mmol/L DPPH (dissolved in methanol), vortexed, and incubated for 50 minutes in the dark at room temperature. The blank contained DPPH solution with no sample. The results were expressed as scavenging capacity (SC50; mg/mL), which was calculated from the curves by plotting absorbance values. The SC50 values represent the concentration of the extract (mg/mL) required to inhibit 50% of the radicals.
For determination of the radical scavenging activity of royal jelly total fatty acids, total fatty content was extracted from lyophilized royal jelly samples. First, 1 g dried royal jelly was dissolved in saline solution (0.9%, w/v) and vortexed. Next, 5 mL hexane was added mixed well, after which 5 mL Carrez I (10.56 g K4Fe(CN)6·3H2O was dissolved in 100 mL bidistilled water) and 5 mL Carrez II (21.94 g Zn(CH3COOH)2·2H2O was dissolved in 100 mL bidistilled water) reagents were added. After centrifugation at 3000 × g for 10 minutes, the solution was decanted. The upper organic phase was removed and evaporated to evaluate DPPH radical scavenging ability.
Statistical Analyses
All the results were calculated as mean values with standard deviations using Microsoft Office Excel 2013 (Microsoft Corporation, Redmond, WA, USA) software. Regression and correlation analysis were performed with Pearson correlation analysis. Significance was set at P < .05.
Results and Discussion
The pH and water contents of the royal jellys investigated are shown in Table 1. The pH values of royal jellys ranged between 3.6 and 5.1. The mean pH value was 4.2. The pH values determined were compatible with those in the literature, with values between 3.4 and 4.5 being reported in previous studies. 1,14,27 One study reported a pH value of 3.4 for royal jelly, 3.6 for honey, and 6.7 for cow’s milk. 27 The pH value for royal jelly derives from the various fatty acids, phenolic acids, organic acids, and proteins in the composition of a substance such as honey, with its acidic character. 14,15,28 Total fatty acids of the samples ranged from 2.3% to 7.3% with a mean of 4.6%.
Some Analysis of Anatolian Royal Jelly (RJ) Samples From Turkey.

Abbreviations: 10-HDA, 10-hydroxy-2-decenoic acid; SD, standard deviation.
The mean water content of the royal jellys investigated was 66.8%, ranging between 61.6% and 73.0%. One study of French royal jellys reported a mean value of 65.3%, similar to our own finding. 18 The water content of royal jelly is higher than that of honey. 27,28
Turkish royal jelly sugars were analyzed using HPLC-RID, and the percentage values for mono-, di- and trisaccharides are shown in Table 2. The main sugar components of royal jelly were glucose, fructose, and sucrose, and total amount of these 3 sugars ranged from 8.2% to 19.0%, with a mean of 13.9%. Glucose levels were higher than fructose levels in all royal jelly samples. Sucrose is always present, but often in highly variable concentrations. This may be attributed to production techniques, or the way the bees are fed. In agreement with our research, other studies have reported that glucose and fructose are the main sugars in royal jelly, while sucrose and maltose are present in smaller quantities. 22,29,30 Melezitose and maltose were detected in all the samples, but lactose was detected in only 3. Melibiose was detected in 7 of the 15 royal jelly samples, ranging from 0.1% to 1.0%. Lactose was detected only 3 samples, the concentrations ranging between 0.8% and 1.4%. On the basis of these results, the major sugar compounds in royal jelly are glucose and fructose, with lower concentrations of sucrose, melibiose, maltose, and trehalose. The fact that lactose was detected in only 3 samples indicates possible contamination in these. However, lactose is an important milk sugar, the presence of which in some royal jelly samples revealed adulteration with milk or milk products, such as yogurt, cream, milk powder. 31 We were unable to compare our results with the literature due to the lack of studies involving lactose in royal jelly samples. The pH values of royal jellys with high lactose contents (between 4.8% and 5.1%) were relatively high, while 10-HDA values were lower (between 1.0% and 2.1%). No statistical analysis was possible because of the small number of samples. Small quantities of milk or milk products (yoghurt, cream, milk powder) may have been added to the 3 royal jellys in which lactose was detected. Milk is the only source of lactose. Royal jelly is known as “bee milk” in Turkey. Beekeepers wishing to adulterate their products are thought to add milk to royal jelly, due to the similarities between these products’ names and colors. The fact that the pH of cow’s milk is higher than that of royal jelly and that the pH values of royal jellys in which lactose was detected were higher than those of other royal jellys corroborates this thesis. 27
Sugar Composition of Lyophilized Royal Jelly (RJ) in the Anatolian Samples From Turkey.

Abbreviations: F, fructose; G, glucose; S, sucrose; SD, standard deviation.
Royal jelly is a protein-rich compound. Total protein contents in this study were calculated using the Kjeldahl method to measure nitrogen. The protein content of fresh royal jelly ranged between 11.4% and 15.1%, with a mean protein value of 14.1%. Total protein content of royal jelly in the literature has been reported at 12% to 15% in fresh royal jelly and 27% to 41% in dried royal jelly. Our percentages are compatible with those in the literature. 1,14,18 Soluble proteins are reported to constitute 80% of the proteins in royal jelly, the great majority of which belong to the major royal jelly proteins family (MRJP1). 32 Royal jelly MRJP3 protein, with its glycoprotein structure, is reported to strengthen the immune system. 17
10-hydroxy-2-decenoic acid, or 10-HDA), a fatty acid in royal jelly, was also measured using HPLC. 10-HDA levels determined in lyophilized royal jellys varied between 1.0% and 3.9%, with a mean value of 2.7%. In contrast to other animal fats, the fatty acid composition of royal jelly is relatively rich in short-chain (C8 and C10) hydroxy and dicarboxylic fatty acids. 10-HDA was the fatty acid determined at the highest level, and is also known as the royal jelly fingerprint. 1 The bioefficacy of royal jelly derives from the fatty acids and protein compounds in its structure. 1 Previous studies have reported that the amount of 10-HDA in royal jelly ranges from 1.0% to 4.0%. French royal jelly samples have been reported to contain a mean 10-HDA level of 2.3%, ranging between 1.4% and 3.7%. 18 Three antioxidant determination methods were used to calculate the antioxidant capacity of royal jelly; the FRAP method that reflects total antioxidant capacity, the DPPH method that shows total radical scavenging capacity and total phenolic content, which shows the total concentration of antioxidant compounds in royal jelly. All antioxidant results are shown in Table 3. The total phenolic contents of royal jelly samples were determined to vary widely, between 91.0 and 301.0 mg GAE/kg. Honey exhibits much higher total phenolic contents than royal jelly. The phenolic content of royal jelly will inevitably be low since is secreted by young worker bees, and phenolic contents in royal jelly can only derive from young bees mixing small quantities of honey, propolis, and pollen in with royal jelly. 3 One study reported the presence in royal jelly of flavonoids such as chrysin (0.8%), pinocembrin (1.8%), and pinostrobin chalcone (0.7%). 3 A positive correlation was determined between total phenolic content in royal jelly and FRAP values (R 2 = 0.53, P < .05). A negative correlation was observed between royal jelly DPPH radical scavenging capacity (SC50) values and total polyphenol contents (R 2 = −0.41, P > .05) although this was not of statistical significance. Previous studies of ours involving honey, propolis and pollen have determined a high negative correlation between total polyphenolic contents and DPPH radical scavenging activity, but only a weak relation emerged in royal jelly. This suggests that the antioxidant potential of royal jelly may derive more from other factors than from polyphenols.
Some Antioxidant Properties of Anatolian Royal Jelly (RJ) Samples From Turkey.
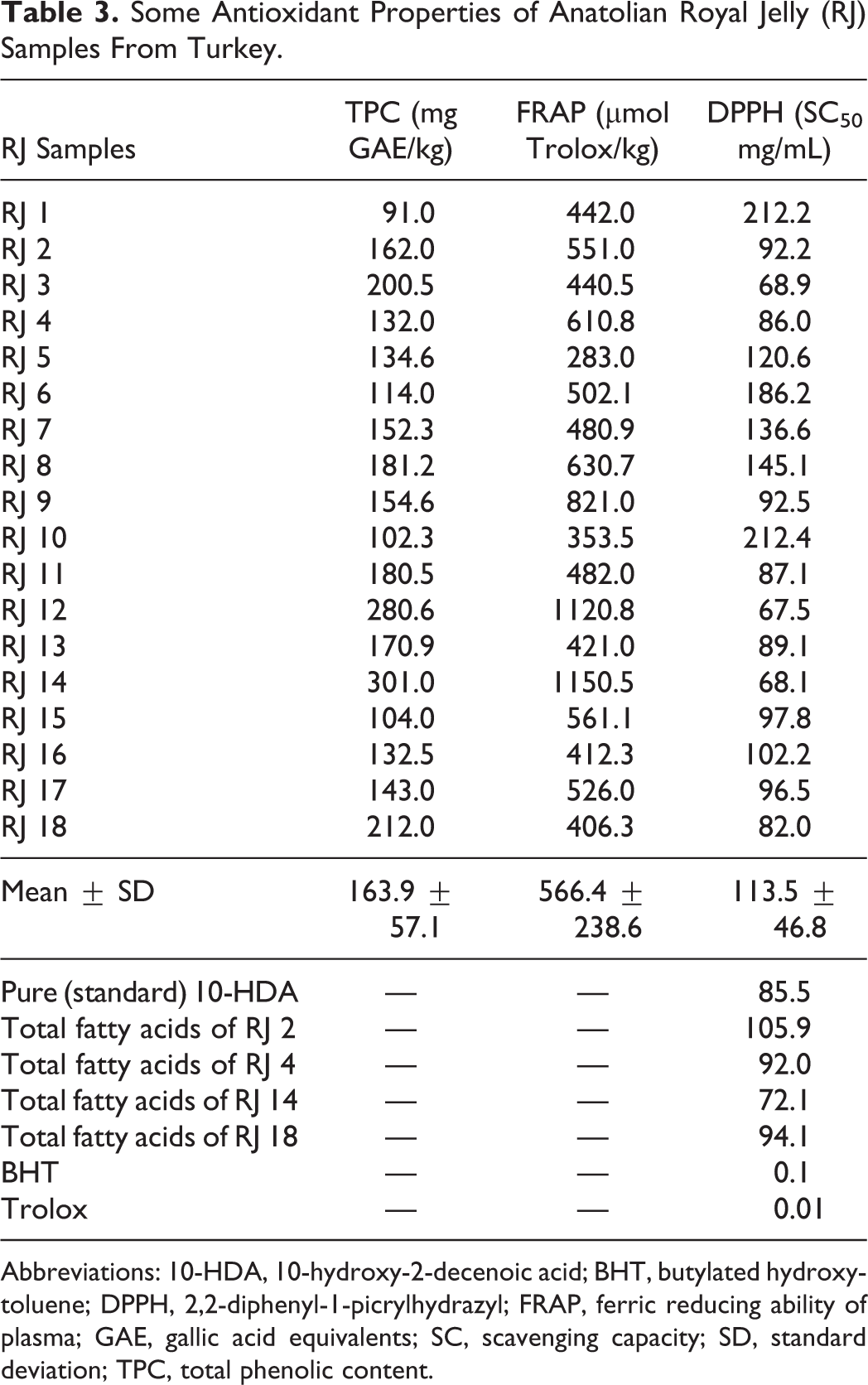
Abbreviations: 10-HDA, 10-hydroxy-2-decenoic acid; BHT, butylated hydroxytoluene; DPPH, 2,2-diphenyl-1-picrylhydrazyl; FRAP, ferric reducing ability of plasma; GAE, gallic acid equivalents; SC, scavenging capacity; SD, standard deviation; TPC, total phenolic content.
The antioxidant character of royal jelly may derive from the phenolic compounds and short peptides in its structures, various antioxidant type vitamins (A and E) and short-chain hydroxyl and carboxylic fatty acids. 33,34 The short peptides in royal jelly have already been shown to have antioxidant activities. These peptides have been shown to exhibit strong hydroxyl radical scavenging activity, but neither metal-chelating activity nor superoxide-anion radical scavenging activity differed significantly among these peptides. These peptides containing tyrosine residues at the C-terminal HDA strong hydroxyl radical and hydrogen peroxide scavenging activity. 33 In addition, 10-HDA and other fatty acids have been reported to exhibit radical scavenging effects. In our study, the DPPH radical scavenging capacities of both pure 10-HDA and royal jelly fatty acids were detected in only 3 royal jelly samples. The radical scavenging activity of 10-HDA was lower than that of total fatty acids. This means that other fatty acids apart from 10-HDA possess antioxidant activities.
Conclusions
In conclusion, while royal jelly contents vary, water constitutes 60% to 70% of fresh weight, sugar 10% to 20%, raw protein 11% to 18%, and 10-HDA 1% to 4%, with very low levels of phenolic compounds. Sugar contents consist mainly of glucose, fructose, and sucrose, together with low levels of maltose, melibiose, melezitose, and trehalose. The presence of lactose in royal jelly was the subject of suspicion and needs to be clarified with further research. The antioxidant capacity of royal jelly derives from its fatty acids, short peptides, and phenolic compounds.
Footnotes
Acknowledgments
The authors are grateful to all beekeepers and to those who assisted with the collection of honey specimens.
Author Contributions
All authors designed the study and directed its implementation, including quality assurance and control. They helped supervise the field activities and designed the study’s analytic strategy, and also helped conduct the literature review.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded a part of TUBITAK projects (370T114) and a part of Turkish Ministry of Food and Agriculture (TAGEM-AR-GE/15).
