Abstract
Ocimum basilicum L has been traditionally used for the treatment of inflammatory bowel disease in Iran. This study investigates the ameliorative effect of Ocimum basilicum essential oil on an acetic acid–induced colitis model in rats. Ocimum basilicum essential oil with 2 doses (200 and 400 μL/kg) significantly ameliorated wet weight/length ratio of colonic tissue compared to the control group. Higher doses of essential oil (200 and 400 μL/kg) significantly reduced ulcer severity, ulcer area, and ulcer index. On the other hand, histological examination revealed the diminution of total colitis index as a marker for inflammatory cell infiltration in the colonic segments of rats treated with Ocimum basilicum essential oil (200 and 400 μL/kg). The increased level of myeloperoxidase was significantly decreased after the treatment with the essential oil (200 and 400 μL/kg). These results suggest that Ocimum basilicum exhibits protective effect against acetic acid–induced colitis.
Inflammatory bowel diseases (IBDs), including ulcerative colitis and Crohn’s disease, are immune-mediated chronic relapsing intestinal disorders characterized by the presence of the chronic inflammatory process of the bowel. Although the etiopathogenesis of IBD remains unclear, the current literature suggests that genetic, microbial, environmental, and immune factors contribute to the disease. 1 The main physical symptoms include abdominal pain, diarrhea, mucous, bloody stools, and psychosocial consequences such as loss of control, energy, feeling a burden, isolation, and fear. 2,3 The pathological characteristics of ulcerative colitis consist of transmural inflammation of neutrophils, macrophages, and lymphocytes, ultimately leading to nonspecific intestinal inflammation. 4 Moreover, it has been proposed that the inflammatory cascade in the pathological process of colitis results from the overexpression of pro-inflammatory mediators such as cytokines, eicosaniods, reactive oxygen species, and neutrophil infiltration. 5 Available current therapy for IBD including corticosteroids, 5-aminosalicylates, and immunosuppressants, is not entirely effective and both short-term and long-term adverse effects confine their use. 6 Nowadays, there is an increasing interest for traditional, complementary and alternative medicines. 7 It may have originated from their easy accessibility, lower costs, and possibly their lower adverse effects. 8 Traditional Persian medicine as one of the most ancient schools is a welcomed system in Iran. 9,10 Ocimum basilicum has been mentioned in different traditional Persian medicine resources such as Avicenna (980-1037 AD) Canon of Medicine for a variety of diseases. 11,12 In addition, Aghili Shirazi, in his famous book, The Storehouse of Medicaments, which was written about 1772 AD, has recommended it for symptoms presenting in ulcerative colitis. 13,14 In recent decades, medicinal plants have been used as an important source for the search of new compounds with anti-inflammatory potential. 15 Ocimum basilicum L (Lamiaceae), popularly known as basil, is a culinary plant extensively utilized as a flavoring agent and has been applied for the treatment of gastrointestinal disorders including diarrhea, vomiting, gastrodynia, dyspepsia, flatulence, and gastritis. 16,17 Relevant studies have identified anti-inflammatory, antioxidant, antiulcerogenic, immunomodulatory, antitumor, antibacterial, antiviral, and antifungal activities of the Ocimum genus. 16,18 –24 Sajjadi demonstrated 20 compounds of essential oil being obtained from Ocimum basilicum cultivated in Isfahan, Iran, namely methyl-chavicol (52.4%), linalool (20.1%), epi-α-cadinol (5.9%), and trans-α-bergamotene (5.2%). 25 In addition, it has been shown that natural monoterpenes act as anti-inflammatory drugs. 26 Hence, the present study aimed to assess the anti-inflammatory potential of Ocimum basilicum essential oil as a source of monoterpenes in the rat model of acetic acid–induced colitis. We have investigated the inflammatory response by macroscopic and histopathological analysis as well as the determination of myeloperoxidase as an inflammation marker.
Methods
Plant Material and Preparation of Essential Oil
The aerial parts of the plant were collected from Najwan district, Isfahan, Iran. A voucher specimen (No. RD-112) was deposited by Dr Ghannadi, a plant taxonomist at the Herbarium of Faculty of Pharmacy and Pharmaceutical Sciences, Isfahan University of Medical Sciences, Isfahan, Iran. The essential oil was prepared by hydrodistillation of the air-dried, powdered, aerial parts of the plant for 3 hours according to the method recommended in European Pharmacopoeia. 27
Analysis of the Essential Oil
The essential oil was analyzed by gas chromatography–mass spectrometry (GC/MS) on a Hewlett Packard 6890 MS selective detector coupled with Hewlett Packard 6890 gas chromatograph equipped with a cross-linked 5% PHME siloxane HP-5MS capillary column (30 m × 0.25 mm; film thickness 0.25 μm) operated under the same conditions as follows: carrier gas, helium with a flow rate of 2 mL/min; column temperature, 60°C to 275°C at 4°C/min; injector and detector temperature, 280°C; volume injected, 0.1 μL of the oil; split ratio, 1:50. The MS operating parameters were as follows: ionization potential 70 eV, ionization current 2 A, ion source temperature 200°C, resolution 1000. The identification of oil components was based on the computer matching with the Wiley 275L library as well as comparison of the fragmentation patterns of mass spectra with those reported in the literature. 28 –30 The relative percentage of the oil constituents was calculated from peak areas.
Animals
Thirty-six male Wistar rats (180-220 g) acquired from the animal house of the School of Pharmacy, Isfahan University of Medical Sciences, Isfahan, Iran, were used in this study. Animals were maintained at controlled environmental conditions where the temperature was maintained at 20°C to 23°C, relative humidity at 50% to 60%, with a 12:12 hour light/dark cycle. All animals were given access to a standard pellet diet and water ad libitum. Animals were housed individually in standard cages and acclimatized for 7 days before the initiation of the treatment. The animal study was approved by the guideline of the Ethical Committee of Isfahan University of Medical Sciences.
Chemicals
Dexamethasone was purchased from Iran Hormone Pharmaceutical Company (Tehran, Iran). Hexadecyl trimethyl-ammonium bromide (HTAB) and O-dianisidine dihydrochloride were purchased from Sigma Chemical Co (St Louis, MO). Formalin solution 35% w/w, glacial acetic acid, and diethyl ether oxide were purchased form Merck (Darmstadt, Germany). All other solvents and chemicals were of analytical grade. Test samples including the suspension of reference drug and the emulsion of Ocimum basilicum essential oil were freshly prepared using 0.2% Tween 80 in normal saline as a vehicle for oral (po) administration.
Animal Groups
Animals were randomly divided into following groups of 6 rats in each: sham group, received normal saline (1 mL, po) without induction of colitis; control group, received vehicle (1 mL, 0.2% Tween 80, po) 2 hours before induction of colitis; dexamethasone group, dexamethasone (2 mg/kg, po) was given 2 hours prior to induction of colitis. Test groups received Ocimum basilicum essential oil at doses of (100, 200, 400 μL/kg, po) 2 hours before induction of colitis. All treatments were performed for the following 5 days. The test plant samples were freshly prepared prior to administration.
Induction of Experimental Colitis
The animals were fasted for 24 hours with free access to water before the induction of colitis. Acute colitis was induced using a modification of the method described by Mascolo et al. 31 The rats were lightly anesthetized with diethyl ether, and a flexible plastic catheter was inserted into the anus up to a length of 8 cm, and then 2 mL of acetic acid (4% v/v in 0.9% saline) was slowly infused. The rats were then maintained in a head down position for 1 minute to prevent solution leakage.
Evaluation of Colon Macroscopic Damage
The rats were killed 24 hours after the last treatment (day 6) by an overdose of ether inhalation. The last 8 cm of the colon was excised, opened longitudinally, rinsed with normal saline, and the wet weight was measured. 32 Then the tissue specimens of colon were used for macroscopic scoring and histopathology damage and the measurement of tissue myeloperoxidase activity. Macroscopic damage appearance was scored by an independent observer according to the following scale: 0 = no macroscopic changes; 1 = mucosal erythema only; 2 = mild mucosal edema, slight bleeding, or slight erosion; 3 = moderate edema, bleeding ulcers, or erosions; and 4 = severe ulceration, erosions, edema, and tissue necrosis. 33 Tissue specimens were cut into 2 pieces, one piece for histopathology assessment (maintained in 5 mL formalin 10% as fixator) and the other for measuring myeloperoxidase enzyme activity. The pieces for measuring the myeloperoxidase enzyme activity were frozen in liquid nitrogen and kept in a freezer (−70°C). Furthermore, ulcer area was assessed by Fiji-Win 32 software (NIH Image for the Macintosh, 2004). For each specimen, ulcer index was measured with summing ulcer score and ulcer area using the following formula: Ulcer index = Ulcer area (cm2) + Macroscopic damage score. 34
Assessment of Colon Histopathological Damage
Colon tissues were separately fixed in 10% formalin, dehydrated, paraffin embedded, processed, sectioned as 4-μm-thick sections, deparaffinized with xylene, hydrated, and stained with hematoxylin and eosin. Inflammation severity (0 = none, 1 = slight, 2 = moderate, 3 = severe), extent (0 = none, 1 = mucosa, 2 = mucosa and submucosa, 3 = transmural), as well as crypt damage (0 = none, 1 = basal 1/3 damaged, 2 = basal 2/3 damaged, 3 = surface epithelium intact only) were assessed in hematoxylin and eosin–stained, coded sections using a modification of a validated scoring scheme described by Cooper et al 35 and Dieleman et al. 36 Total colitis index was measured by summing the scores of inflammation severity, inflammation extent, and crypt damage. Histological damages were investigated using a Zeiss microscope equipped with a Sony color video camera for digital imaging.
Determination of Myeloperoxidase Activity
Tissue myeloperoxidase activity was measured according to the method described by Bradley et al 37 with some modifications. A portion of colon tissue (0.1 g) was homogenized in 1 mL of 50 mM potassium phosphate (pH 6) with 0.5% HTAB in an ice bath using a polytron homogenizer. More buffers were added to obtain a concentration equivalent to 5 mL per 0.1 g of colon tissue. The resultant homogenate was sonicated in an ice bath for 10 seconds, then subjected to a sequence of freezing and thawing for 3 times, sonicated again for 10 seconds and centrifuged for 15 minutes at 15 000 rpm at 4°C. A total of 0.1 mL of the supernatant was mixed with 2.9 mL of 50 mM phosphate buffer (pH 6) containing 0.167 mg/mL O-dianisidine dihydrochloride and 0.0005% hydrogen peroxide. The change in absorbance at 460 nm was measured using a UV/VIS spectrophotometer (LSI Model Alfa-1502). Myeloperoxidase activity was reported as units (U) per gram (g) weight of wet colon tissue.
Statistical Analysis
The results are reported as mean ± SEM or median (range). All statistical analyses were performed using GraphPad Prism 5.0 software. Group differences were calculated by parametric one-way analysis of variance with Tukey’s HSD as post hoc test. Nonparametric data were analyzed using Kruskal-Wallis followed by Mann-Whitney U test. For all tests, differences with values of P < .05 were considered significant.
Results
Analysis of the Essential Oil
Thirty-two constituents were characterized accounting for 90.91% of total oil components detected, which are listed in Table 1 with their percentage composition and retention indices. The principal constituents of Ocimum basilicum essential oil was linalool (39.73%), followed by β-pinene (6.65%), trans-verbenol (6.39%), and α-terpinolene (5.03%).
Volatile Constituents of the Essential Oil of Ocimum Basilicum.

Macroscopic Results
No mortality was observed in animals receiving intracolonic 4% acetic acid (2 mL) solution. Acetic acid caused severe ulceration and hemorrhage of colonic mucosa in control group (Table 2 and Figure 1B); dexamethasone significantly reduced macroscopic damage score, ulcer area, ulcer index, and weight/length ratio of distal colon (8 cm) compared to the control group (P < .001; Table 2 and Figure 1C). On the other hand, Ocimum basilicum essential oil (200 and 400 μL/kg) showed significant decrease in macroscopic damage, ulcer area, ulcer index, and weight/length ratio of distal colon compared to the control group (P < .001; Table 2, Figure 1E and F). In addition, treatment with 100 μL/kg dose of Ocimum basilicum essential oil was not effective to reduce weight/length ratio as well as ulcer index (ulcer area + ulcer severity) in colon samples compared to control group (Table 2 and Figure 1D). No significant differences were found between dexamethasone and Ocimum basilicum essential oil (200 and 400 μL/kg) groups (P > .05).

Macroscopic photographs of colon tissue 5 days after acetic acid–induced colitis in rats: (A) sham group treated with saline; (B) colitis in control group treated with vehicle; (C) colitis in dexamethasone-treated group; (D, E, and F) Colitis in Ocimum basilicum essential oil–treated group, 100, 200, and 400 μL/kg, respectively.
Effects of Ocimum basilicum Essential Oil on the Macroscopic Parameters of Colitis Induced by Acetic Acid in Rats.a
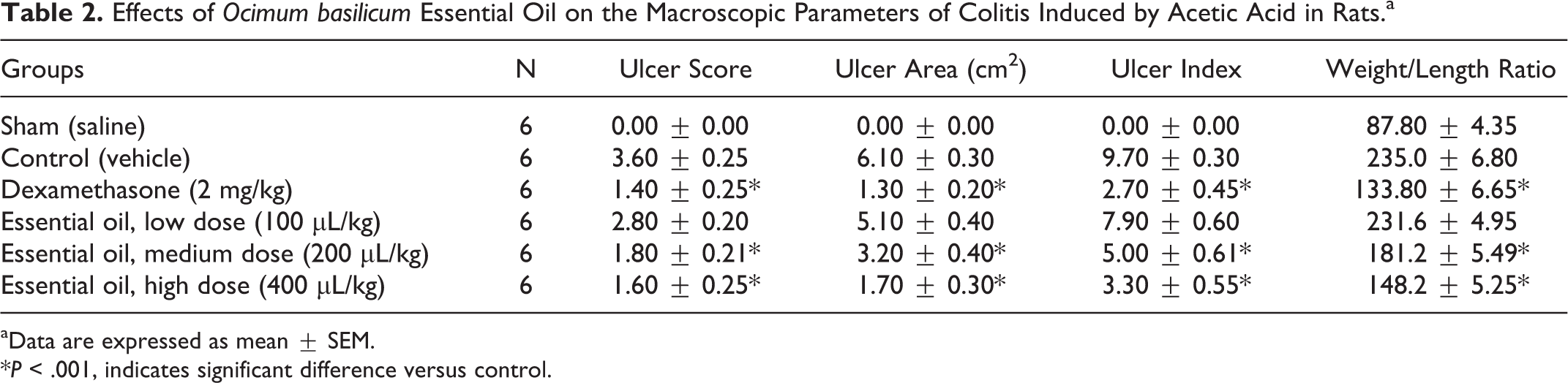
aData are expressed as mean ± SEM.
*P < .001, indicates significant difference versus control.
Microscopic Results
In the sham group, histological evaluation of the colon mucosa showed no inflammation or necrosis (Table 3 and Figure 2A). On the other hand, acetic acid–induced colitis displayed destruction of epithelium, transmural necrosis, edema, areas of hemorrhage, and submucosal inflammatory cellular infiltration (Table 3 and Figure 2B). Dexamethasone (2 mg/kg) and Ocimum basilicum essential oil at doses (200 and 400 μL/kg) showed significant reduction of neutrophil infiltration, edema, and extent or severity of tissue damage, as evaluated by total colitis index (Table 2 and Figure 2C, D, and E, respectively; P < .001).

Microscopic presentation of acetic acid–induced colitis in rats stained by hematoxylin and eosin (light microscopy, 20×): (A) normal colon treated with saline, mucus layer, and crypts are normal and leucocyte infiltration is absent; (B) colitis in control group treated with vehicle, mucosal and submucosal inflammation, as well as crypt damage and leucocyte infiltration are evident (arrow); (C) dexamethasone-treated colitis; (D) Ocimum basilicum essential oil (200 μL/kg) treated colitis; (E) Ocimum basilicum essential oil (400 μL/kg) treated colitis.
Effects of Ocimum basilicum Essential Oil on the Microscopic Parameters of Colitis Induced by Acetic Acid in Rats.a

aData are expressed as median (range).
*P < .001, indicates significant difference versus control (Mann-Whitney U test).
Myeloperoxidase Activity
Intrarectal administration of acetic acid caused significant increase in the level of myeloperoxidase in colonic tissue when compared to the sham group (P < .001). In contrast, myeloperoxidase activity was decreased (P < .001) in groups treated with Ocimum basilicum essential oil (200 and 400 μL/kg) as well as dexamethasone (2 mg/kg) (Figure 3).

Effect of Ocimum basilicum essential oil (100, 200, and 400 μL/kg) on myeloperoxidase (MPO) enzyme activity in the colonic tissue. Dexa = dexamethasone. Results are presented as mean ± SEM (N = 6). ***P < .001 compared to control, one-way analysis of variance followed by Tukey test.
Discussion
The present study demonstrates that Ocimum basilicum essential oil reduces tissue damage in experimental model of acetic acid–induced colitis as established by macroscopic, histological, and biochemical changes. The anti-inflammatory effects of this substance observed in this study is in accordance with previous investigations of its ability to prevent carrageenan-induced paw edema and turpentine oil–induced acute inflammation in rats. 18,38 Intrarectal administration of acetic acid is one of the validated methods to induce an experimental model of IBD. This method is widely adopted to screen potential drugs due to its similarity to human IBD. Destruction of mucosal barrier and colon structure by chemical stimulation, enhanced vasopermeability, and increased inflammatory mediators are major contributing factors in the induction of this animal model. 39 –41 The decrease in the wet weight of the inflamed colon tissue can be directly correlated with the severity and extent of inflammation. 42 Our finding showed that treatment with essential oil could reduce the wet weight/length ratio of distal colon segments and macroscopic damage score compared with colitis control group. In addition, tested substances significantly decreased the histological signs of inflammation such as infiltration of lymphocytes and macrophages into the mucosa, edema, and tissue damage. Neutrophil accumulation in the inflamed colonic mucosa is one of the most noticeable histopathological aspects of IBD. Myeloperoxidase is a proteolytic heme enzyme existing in neutrophil granulocytes that combat against bacteria. These enzymes are released from activated neutrophils and catalyze the formation of hypochlorous acid from hydrogen peroxide and chloride ions that lead to the oxidative injury associated with induction of colitis. 41,43 The activity of myeloperoxidase from the damaged tissue is directly proportional to the number of neutrophils observed in histological fields. 44 Therefore, the reduction in the activity of myeloperoxidase can be explained as a manifestation of the anti-inflammatory effect of a given drug. Our research showed that the administration of Ocimum basilicum essential oil decreased the myeloperoxidase level during acetic acid–induced colitis indicating an excellent anti-inflammatory activity on experimental colitis model. Phytochemical composition analysis indicated that Ocimum basilicum essential oil possessed compounds such as linalool, cis-verbenol, β-pinene, and α-terpinolene.
Cis-verbenol, one of the active constituents present in Ocimum basilicum essential oil, has been shown to have anti-inflammatory activity by the reduction of the expression levels of pro-inflammatory cytokines in ischemic brain and immunostimulated glial cells. 45 Another compound linalool has been shown to exhibit protective effect against lipopolysaccharide (LPS)/D-galactosamine (GalN)-induced liver injury in mice through inhibition of caspase-3 and caspase-8 expression and the reduction of inflammatory response via NF-κB suppression. 46 Linalool has been reported to reduce LPS-induced tumor necrosis factor-α and interleukin-6 production both in vitro and in vivo, block phosphorylation of IκBα protein, p38, c-Jun terminal kinase, and extracellular signal-regulated kinase in LPS-stimulated RAW 264.7 cells and could attenuate histological changes in lipopolysaccharide-induced lung injury mouse model. 47 Deepa and Venkatraman Anuradha 48 had shown that linalool could be useful for the treatment of renal damage in diabetic subjects through the attenuation of TGF-β1 and NF-κB expression.
In conclusion, the present study showed that Ocimum basilicum essential oil could inhibit acetic acid–induced colitis in rats by reducing leukocytes accumulation and inhibiting myeloperoxidase production. This anti-inflammatory effect may, at least in part, be due to the presence of monoterpenes, which are known to inhibit the inflammatory signaling through NF-κB suppression. 26 The results of this study show that Ocimum basilicum essential oil could potentially be useful as a supplemental remedy for the treatment of IBD. However, further investigations are needed to elucidate the entire mechanism of action of Ocimum basilicum essential oil and its isolated compounds on the cell receptors and signaling pathways associated with colitis.
Footnotes
Acknowledgments
The authors thank Isfahan University of Medical Sciences, which supported this work, and Dr Parvin Mahzooni for her help.
Author Contributions
The work presented in this article was carried out through collaboration between all authors. MM and ARG made the initial hypothesis and supervision. All authors participate in defining the research theme and providing the proposal. PR and AR performed the experiments, collected the data, analyzed the data, and wrote the article. PR and SM conceptualized the study, critically analyzed and discussed the data, and corrected and reviewed the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for theresearch, authorship, and/or publication of this article: This study was a part of a Pharm. D thesis by Parnia Roohi which was supported by council of research, Isfahan University of Medical Sciences (Project Number: 393321).
Ethical Approval
This study was performed according to the guidelines of the US National Institute of Health (NIH Publication No. 85.23, Revised 1985) guides for the care of laboratory animals.
