Abstract
Malva sylvestris is an edible plant that is consumed as a herbal supplement for its antiulcer and colon cleansing properties in traditional Persian medicine. This study was designed to evaluate its effects on ulcerative colitis, which is a chronic gastrointestinal inflammation. Colitis was induced by rectal instillation of acetic acid solution. Rats in different groups received aqueous, n-hexane, or ethanolic fractions of the plant before induction of colitis. Isolated polysaccharide of plant was also tested in 2 groups before and after induction of colitis. Macroscopic and microscopic evaluation of colitis showed that the aqueous fraction was very effective in preventing the inflammation and efficacy was lower for ethanolic and n-hexane fractions. Polysaccharide was effective in reducing signs of inflammation, especially as pretreatment. These beneficial effects provide evidences that this plant can be suggested for patients with this disease to improve their health condition or to reduce adverse effects of their medication.
Idiopathic inflammatory bowel diseases, which include Crohn’s disease and ulcerative colitis, are chronic inflammation of the gastrointestinal tract with a growing prevalence especially in modern countries (150-200 cases per 100 000 individuals). 1 Although the highest incidence of inflammatory bowel disease has been reported in the populations of Northern Europe and North America and the lowest in Asia, the prevalence is increasing in developing countries as well. 2,3 Treatment of inflammatory bowel disease is still challenging because of poor understanding of the etiology of the disease. Choosing treatment regimens depends on the subtype and severity of the disease. These regimens consist of aminosalicylates 4 and glucocorticoids in mild to moderate cases 5 and immunosuppressants such as azathioprine and cyclosporine A in severe cases. 6 Severe cases may also require surgery. Inflammatory bowel disease may be associated with the risk of colorectal malignancy and cancers and may decrease the quality of life of patients. Poor treatment outcomes and high rate of side effects of the current medications used for this disease has encouraged researchers to examine alternative treatments for inflammatory bowel disease. Nowadays, tremendous attention is paid to medicinal plants, natural products, complementary and alternative medicine, and functional foods to find beneficial effects, drugs, or strategies for the treatment of inflammatory bowel disease as monotherapy or adjuvant therapy. 7 –10
Malva sylvestris L (Malvaceae), also known as cheeses, high mallow (in English), or panirak or khabbazi (in Persian), is a biennial or perennial plant that grows wild in farms and on waste ground. 11,12 Malva sylvestris is an edible plant wherein its boiled leaves are taken as wholesome vegetable in stews or marinated in yogurt as a side dish. In Persian folk medicine, the plant’s decoction and its cooked aerial parts are used for treatment of gastrointestinal lesions, as tonic for gastrointestinal tract, or as a side dish especially for its colon cleansing properties. 13 Also, in some Persian traditional and pharmaceutical manuscripts such as Qarabadin-e-azam (a lithograph manuscript written by Hakim Azamkhan in 1853 AD), Qarabadin-e-kabir (Aghili Shirazi, 1772), Qarabadin-e-salehi (Heravi, 1765), and Tohfat ol Moemenin (Tonekaboni, 1670 AD), 14 several preparations containing Malva sylvestris leaves, flowers, and fruits were explained as oral dosage forms, functional food, and rectal enema for treatment of gastrointestinal and rectal ulcers, pain, and/or inflammation. 15 Recently, different extracts of common mallow was reported to exhibit wound healing, antioxidant, anti-inflammatory, 12,16 –19 and immunomodulatory effects. 20 –22 Also, different polyphenols, polysaccharides, fatty acids, carotenoids, tocopherol, and ascorbic acid in different amounts have been isolated from the edible parts of the plant. 21,23
Although beneficial gastrointestinal effects of Malva sylvestris have been approved in Persian traditional medicine, healing properties of this plant on colitis and colon inflammation have not been investigated yet. With the aim of evaluating the anti-inflammatory effect of this plant against induced colitis in rats, aqueous, n-hexane, and ethanolic fractions and its isolated polysaccharide (as possible active constituent) of the plant were prepared and their efficacy on microscopic and macroscopic signs of colitis damage were studied and compared to the control groups receiving prednisolone.
Materials and Methods
Materials
Prednisolone was obtained from Iran Hormone Pharmaceutical Company (Iran). All other solvents and chemicals were of analytical grade and purchased from Merck (Darmstadt, Germany) or Sigma Aldrich (Sigma, St Louis, MO).
Malva sylvestris Fractions
Malva sylvestris was collected from farms around Marvdasht (Fars, Iran) in the flowering season (May 2012). The aerial parts were shadow dried, milled, and preserved in well-covered container at 4°C. Authentication was performed by Miss Sedigheh Khademyan, Taxonomist, Department of Pharmacognosy, Shiraz School of Pharmacy, and its herbarium was preserved with the code PM-380 in the department for further references.
A total of 200 g of powdered aerial parts was sequentially extracted with n-hexane, ethanol, and water (3 × 1 L and 48 hours for each) at room temperature. The fractions were concentrated by a rotary evaporator at 40°C and dried applying a speed vacuum dryer or a freeze dryer and kept at −20°C.
Polysaccharide Isolation and Characterization
Aqueous fraction of Malva sylvestris was mixed with 4 volumes of absolute ethanol, stirred vigorously, and left overnight at 4°C. The precipitated polysaccharide was collected after centrifugation (16 000 g, 20 minutes at 4°C). The sediment was freeze dried and applied to an ion exchange column (equilibrated Sephadex A-25) and eluted with gradient solutions of 0.02 to 2 M NaCl. Same fractions were pooled and applied to a size exclusion column (Sepharose CL-6B). The isolated polysaccharide was lyophilized and stored at −20°C.
To determine the monosaccharide content, 4 mg of the polysaccharide was first hydrolyzed with trifluoroacetic acid (2 M) at 110°C for 2 hours and then tetramethylsilane derivatizated with N,O-bis-(trimethylsilyl)acetamide. The monosaccharide content was analyzed using a gas chromatography–mass spectrometry instrument (7890A, Agilent). The gas chromatograph was equipped with a HP-5MS capillary column (5% phenyl methyl siloxane, 30 m × 250 μm × 0.25 μm). The carrier gas was helium with a flow rate of 1.0 mL/min. The mass spectrometer (5975C MSD, Agilent) operated in EI mode at 70 eV. The mass range was 30 to 600 m/z. Identification of components was based on a comparison of the retention index and mass spectra with Wiley (7 nL) and derivatized standards. The injection temperature was 150°C. Oven temperature was held at 150°C for 4.5 minutes, and then increased to 280°C (6°C/min). The temperature was held at this temperature for 5 minutes. The protein content was evaluated by Bradford assay and the uronic acids content was determined spectrophotometrically against standard galacturonic acid. Molecular weight of the polysaccharide was determined as compared with standard dextrans. A mixture of standard dextrans (667, 233, 80, and 1 KDa) was applied on the same size-exclusion column and chromatographed with a same condition for the sample. The average molecular weight of the isolated polysaccharide was calculated with Nishitani formula: MW = ∑MiWi /∑Wi , where MW is the average molecular weight of the sample, Wi is the carbohydrate content in the sample fraction, and Mi is the molecular weight of standard dextrans in the corresponding fraction. 24,25
Determination of Polyphenol Content of Fractions
A total of 50 mL of n-hexane was added to 2 g of dried fractions. Polyphenols were extracted with 60% methanol (3 × 100 mL). The collected methanolic phase was washed with 20 mL of n-hexane and dried with a rotary evaporator at 40°C. 26,27 Polyphenols were dissolved by adding adequate amount of distilled water. The solution was moved to a separation funnel and extracted with petroleum ether (60:80). The water phase was saturated with NaCl and extracted with 4 volumes of ethyl acetate (3 times). The ethyl acetate fractions were collected and dried by adding anhydrous Na2SO4 and dried with a rotary evaporator at 40°C. Folin-Ciocalteau reagent (5 mL) was added to 1 mL of different concentrations of gallic acid or sample tubes, and after 5 minutes, 4 mL of 7.9% sodium carbonate solution was added. Sample and standard tubes were covered for 2 hour at room temperature and away from strong light. Absorbance was read at 740 nm. All experiments were done in triplicate. 28
Animals
Male Wistar rats (190 ± 20 g) were purchased from Razi Vaccine and Serum Research Institute (Shiraz, Iran). The experiments were conducted in accordance with ethical guidelines for animal experimentation approved by the ethical committee of Animal Laboratories in Shiraz University of Medical Sciences. The rats (6 per group) were housed in the Shiraz School of Pharmacy Animal Laboratory under a 12-hour light–dark cycle, with room temperature maintained at 25°C, humidity at 55%, and food and water available ad libitum. The rats were deprived of food, but not water, for 24 hours prior to experimental procedures.
Induction of Colitis
Colitis was induced by rectal instillation of a 4% solution of acetic acid according to the method reported earlier. 29 Under light ether anesthesia, an 8-cm-long polyethylene tube was inserted in the rectum and through that acetic acid solution was instilled to the colon. After instillation of the solution, rats were kept in separate cages with free access to water and rat chow pellet.
Administration of Drugs
Animals were divided into 8 groups, each containing 6 rats. Drug administration was performed according to the following protocol:
Normal group: without induction of colitis that received normal saline solution rectally at the day of induction.
Negative control group: with induction of colitis that received distilled water orally.
Positive control group: with induction of colitis that received prednisolone at dose 5 mg/kg/day orally prior to induction of colitis.
Treatment group I: with induction of colitis that received aqueous fraction of Malva sylvestris at dose 200 mg/kg/day orally prior to induction of colitis.
Treatment group II: with induction of colitis that received ethanolic fraction of Malva sylvestris at dose 200 mg/kg/day orally prior to induction of colitis.
Treatment group III: with induction of colitis that received n-hexane fraction of Malva sylvestris at dose 200 mg/kg/day orally prior to induction of colitis.
Treatment group IV: with induction of colitis that received the isolated polysaccharide of Malva sylvestris at dose 200 mg/kg/day orally prior to induction of colitis. In groups I to IV treatment was performed for 5 days consecutively before induction of colitis.
Treatment group V: with induction of colitis that received the isolated polysaccharide of Malva sylvestris at dose 200 mg/kg/day orally for 5 days after induction of colitis.
The dosage regimen for animal study was selected based on our preliminary studies (data not shown).
Macroscopic and Microscopic Examination of Colon
Twenty-four hours after the last dose of the drug in treatment group V and 24 hours after induction of colitis in other groups, rats were euthanized with a high dose of ether and an incision was made to abdomen. Eight-centimeter length of the end part of the colon was removed and used for macroscopic and microscopic evaluations.
For macroscopic evaluation, ulcer area and colon weigh-to-length ratio (mg/cm) were measured. A macroscopic scoring was also used to present the severity of damage as follows 29 : normal appearance, 0; erythema without ulcer, 1; inflammation and ulcer, 2; and severe ulcer with necrosis, 3.
Colon samples were then fixed in 10% formalin solution, stained by hematoxylin and eosin, and observed by light microscope. Severity and extent of damage was examined and scored based on the parameters presented in Table 1. 30
Histopathologic Scoring of Colon Damage. 30

Statistical Analysis
Kolmogorov–Smirnov statistical test was used for controlling normality and homogeneity of the resulting data. Colon weight-to-length ratio and ulcer area in different groups was tested by one-way analysis of variance and Dunnett’s post hoc test. Nonparametric variables (ordinal variables of scoring) were compared by Mann–Whitney U test. Statistical analysis was performed by SPSS 16 software, and P value less than 0.05 was considered significant for all experiments.
Results
Phytochemical Analysis
The yield for polysaccharide extraction was 9.6% (w/w). The gas chromatography–mass spectrometry analysis revealed that the isolated polysaccharide has an acidic nature and consisted of galactose, glucose, uronic acids, arabinose, and rhamnose in the molar ratio of 4:5:14:6:1. The molecular weight of the polysaccharide was 1.3 × 106 kD.
Yields and phenolic contents of each fraction are shown in Table 2.
Yield and Phenolic Content of Fraction Obtained From Malva sylvestris.a
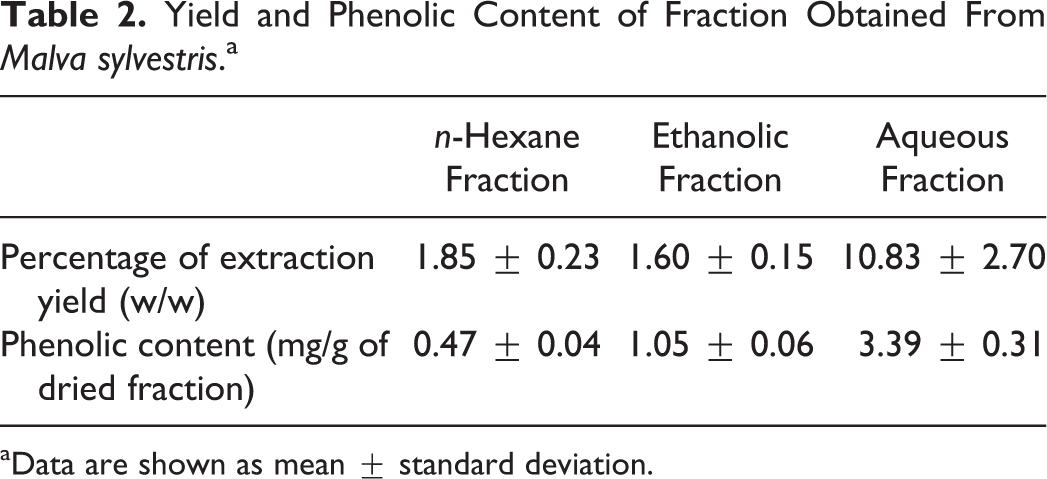
aData are shown as mean ± standard deviation.
Macroscopic Evaluation of Colon
Results of measuring colon weight-to-length ratio and ulcer area are presented in Table 3. Data of negative control group show that acetic acid instillation was an efficient method for inducing ulcerative colitis as reported in previous studies. In the positive control group, which received prednisolone 5 mg/kg/day for 5 days before induction of colitis, the macroscopic parameters were not changed significantly, which may be due to the pretreatment. Prednisolone cannot prevent the damage made by acetic acid in the form of pretreatment. Efficacy of treatment by herbal fractions was higher in treatment group I, which received aqueous fraction and decreased for the groups that received ethanolic and n-hexane fractions. In the groups that received the isolated polysaccharide as pretreatment, efficacy in decreasing macroscopic damage was very similar to treatment group I. However, the treatment plan of group V, which received the isolated polysaccharide after induction of colitis, was not as effective as the aqueous fraction.
Results of Macroscopic Damage of Colitis in Different Groups.
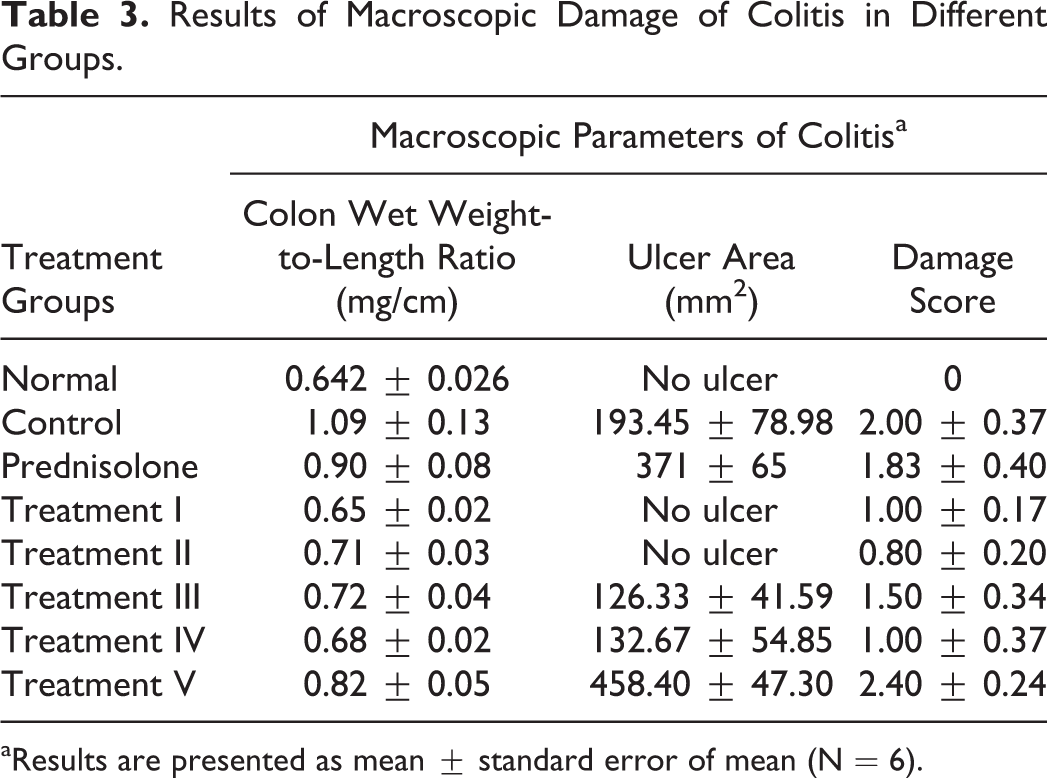
aResults are presented as mean ± standard error of mean (N = 6).
A same trend was observed for all the macroscopic damage parameters studied. Macroscopic appearance of colon specimens after treatment in different groups is presented in Figure 1.

Macroscopic appearance of colon in different groups: (a) Normal, (b) Control, (c) Prednisolone, (d) Treatment I, and (e) Treatment IV.
Microscopic Evaluation of Colon
Results of the histopathologic study confirmed the results of the macroscopic evaluation. As presented in Table 4, considerable inflammation and ulcer was observed in negative and positive control groups, and prednisolone did not decrease the microscopic parameters of inflammation by pretreatment. Between different groups treated with herbal fractions, the aqueous fraction provided the highest efficacy in preventing signs of inflammation and ulcer and the effect of n-hexane fraction was the lowest.
Results of Microscopic Damage of Colitis in Different Groups.

aResults are mean of 6 rats. The value in the parentheses is the mode of data.
Figure 2 shows the histological appearance of colon of rats in different groups. Pretreatment with polysaccharides of the plant also was effective in reducing the signs of inflammation, but posttreatment with the isolated polysaccharide was not as beneficial. Comparison between microscopic results of different groups is summarized in Figure 3.

Histopathologic appearance of colon in different groups: (a) Normal, (b) Control, (c) Prednisolone, (d) Treatment I, and (e) Treatment IV.
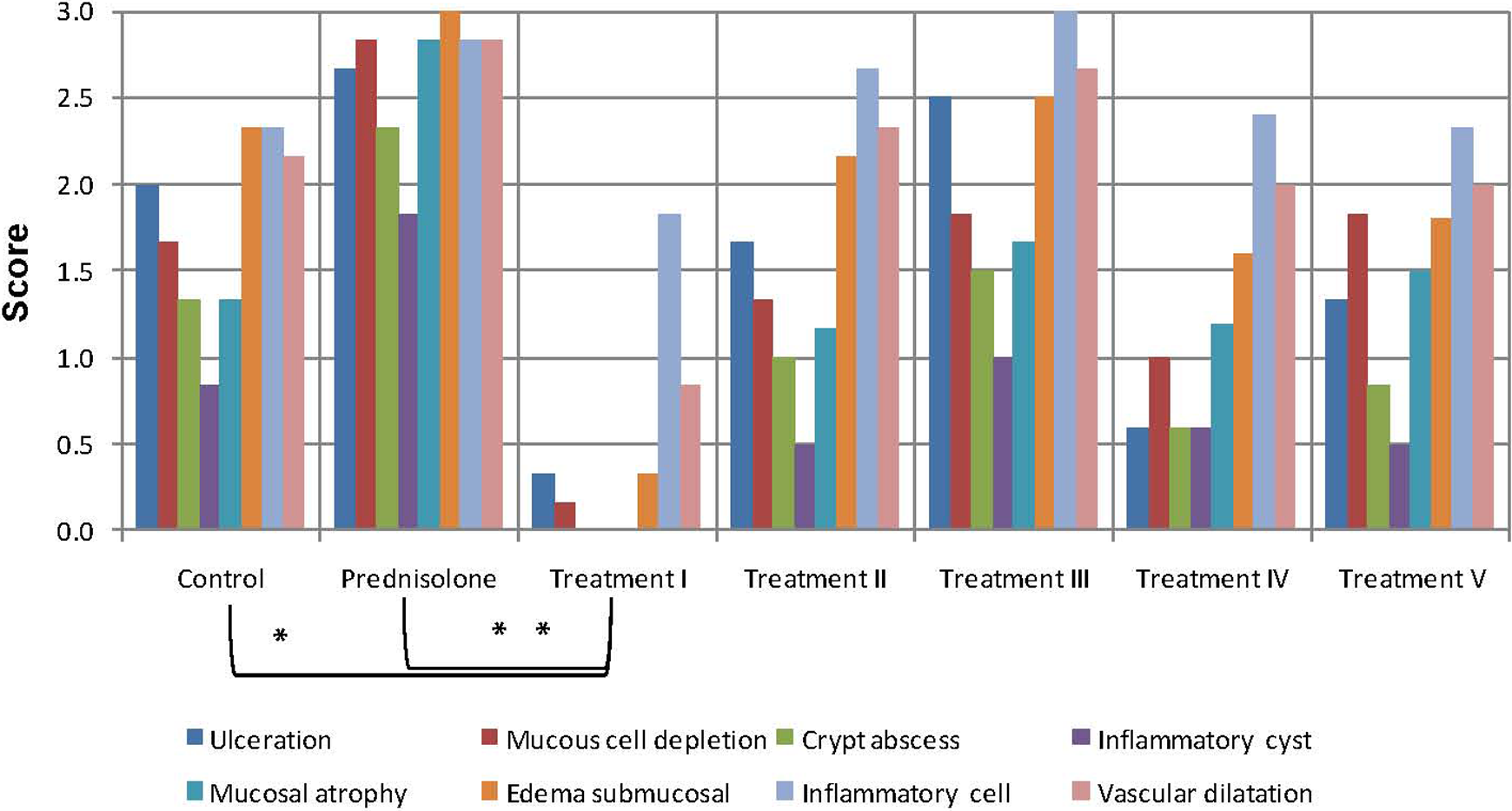
Comparison of results of microscopic evaluation of colitis in different groups.
Discussion
In this study, anti-inflammatory effects of different fractions of Malva sylvestris were evaluated against acetic acid–induced ulcerative colitis in rats. Anti-inflammatory effects of the tested fractions in this study seem to be related to their polysaccharide and polyphenol content. Therefore, the content of polyphenols in different obtained fractions was determined spectrophotometrically and used as the method for standardization of the fraction. Also, the structure of the isolated polysaccharide was characterized. Polyphenols including flavonoids, anthocyanins, and tannins are secondary plant metabolites with diverse biological activity such as antioxidant, anti-inflammatory, 12,16 –19 anticarcinogenic, antimicrobial, vasodilatory, and wound healing properties. 11,31,32 The highest phenolic content was found in the aqueous fraction (3.39 mg/g), which was lower than previous reports on this plant. 12,33 It may be due to different methods applied for total phenol determination and variation in plant growth condition. The yield of polysaccharide extraction in our study was higher than the optimized condition suggested by Samavati. 34 The characterized polysaccharide in this study had an acidic nature with a high molecular weight. These data are in accordance with previous reports on Malva sylvestris polysaccharides. 17,35,36
The protective effect of the aqueous fraction of Malva sylvestris on reducing microscopic and macroscopic parameters of colitis was significantly higher than prednisolone, which is a conventional medicine for the treatment of acute episodes of ulcerative colitis. Indeed, ulcers were not observed in rats treated with aqueous and ethanolic fractions of Malva sylvestris, and the damage scores in these 2 groups were significantly lower than the others. The aqueous extract of different parts (flowers, leaf, and roots) of this plant was reported to be rich in mucilaginous polysaccharides with monosaccharide contents of glucuronic acid, galacturonic acid, rhamnose, galactose, fructose, glucose, sucrose, and trehalose. 17,35 –38 The mucilaginous proteoglycans of this plant were reported to have anticomplementary activity, 17 which may be one of the probable mechanisms for the observed results. A considerable amount (12.8% dry weight) of arabinogalactans has been extracted from cultures of Malva sylvestris callus cells. 39 These polysaccharides are believed to be the main effective components of the aqueous fraction of the plant. The anti-inflammatory effect of extracted Malva sylvestris polysaccharides was also studied as pre- and post-treatment. The results showed that these polysaccharides have exhibited beneficial effects against acid-induced ulcerative colitis when used as pretreatment. To our knowledge, there is no report on effects of Malva sylvestris polysaccharides to compare the findings but polysaccharides of Rheum tanguticum, Angelica sinensis, and Vaccinium oxycoccos have been studied against this disease. Liu et al reported that treatment with Rheum tanguticum polysaccharides at dose 200 mg/kg significantly reduced mortality, colon mass, and ulcer areas. 40 Wong et al suggested that the protective effects of Angelica sinensis polysaccharides (in a pretreatment protocol) may be because of the prevention of oxidative stress in the pathological process of ulcerative colitis. 41 Beneficial effects of pectic polysaccharides from the common cranberry (Vaccinium oxycoccos) on acetic acid–induced colitis with the possible mechanism of reduction of neutrophil infiltration and polysaccharide antioxidant properties was also reported by Popov et al. 42 Polysaccharides of Malva sylvestris were reported to have antioxidant activity, 34 and this effect may be attributed to the observed activity in the present study. 34 Although we did not evaluate the mechanism of the observed results, our findings in macroscopic and microscopic studies are in agreement with the results of above-mentioned studies about other polysaccharides. This could be related to immunomodulatory, 43 anti-inflammatory, 44,45 and wound healing, 46 as well as antioxidant properties of polysaccharides.
The aqueous fraction of Malva sylvestris showed higher protective effects against ulcerative colitis than the isolated polysaccharide, which suggests that other constituents in the aqueous fraction provided synergistic effects with the polysaccharide. Magro et al reported that the aqueous extract of Malva sylvestris leaves was the only preparation of this plant that had significant antimicrobial properties against fungi, 47 and also it was reported to inhibit the prostaglandin synthesis by cyclooxygenase pathway.
The aqueous extract of fresh leaves of Malva sylvestris was previously reported to contain different terpenoids including linalool, linalool-1-oic acid, different megastigmene derivatives, blumenol A, dehydrovomifoliol, and malvone A. 23,48 Also, fructose, glucose, trehalose, ascorbic acid, and some glycosidic phenolics were identified in the aqueous extract of this plant. 12,49 Geerling et al reported that intake of fructose, magnesium, vitamin C, and fruit was associated with reduced ulcerative colitis risk. 50 On the other hand, some of these components were reported for exhibition of antioxidant, anti-inflammatory, and antioxidant properties that may have a role in the observed efficacy of this fraction. As far as we know, this is the first report on the beneficial effects of Malva sylvestris in the treatment or prevention of ulcerative colitis but the anti-inflammatory and anti-ulcerogenic properties of the aqueous extract of Malva sylvestris against gastric ulcers were previously reported in oral administration by Sleiman and Daher. 19 They showed that the aqueous extract of Malva sylvestris enhanced the mucous production in mucosal tissue and improved elastin promoter activity, 51 enhancing the elasticity of mucosal tissues by inhibition of elastase and trypsin. 52
The ethanolic fraction and to a lower extent the hexane fraction showed a reduction in evaluated macroscopic and microscopic parameters by pretreatment. The hexane extract of Malva sylvestris was previously reported to contain carotenoids, tocopherols, and different fatty acids with the considerable portion (50% to 80%) of polyunsaturated fatty acid. Although carotenoids and tocoferols have the potential of antioxidant properties and may be responsible for the efficacy of these fractions, Geerling et al reported that high intake of mono- and polyunsaturated fatty acids may increase the risk of developing ulcerative colitis and this may explain the lower beneficial results (compared to aqueous and ethanolic fractions) observed in present study. 50
The aqueous fraction of Malva sylvestris provided the highest extraction yield and the highest protective effects compared with the other fractions and isolated polysaccharide. Moreover, this plant has been known as an edible wild vegetable in many countries. The study by Sleiman and Daher 19 on the safety of oral administration of the plant showed that the oral intake of Malva sylvestris was not associated with serious side effects.
Conclusion
Overall, according to the result of this study, it can be concluded that fractions of Malva sylvestris have decreased the inflammatory symptoms of experimental colitis induced by acetic acid in rats. Further pharmacognostic and pharmacological studies on these fractions can result in finding new active constituents for treatment of inflammatory bowel disease. Considering that aqueous fraction of Malva sylvestris presented the highest extraction yield and protective effects compared to other fractions and the isolated polysaccharide, preparations of the aqueous fractions of this plant can be suggested as a safe remedy for patients with inflammatory bowel disease. Also, this vegetable can be suggested to be used as a functional food or a protective supplement for patients with ulcerative colitis to improve their health condition or to reduce the risks of adverse effects of their conventional medication; however, more in vivo and clinical studies are needed to support this hypothesis.
Footnotes
Author Contributions
Azadeh Hamedi (supervising pharmacognosy part and writing part of the article), Hossein Rezaei (conducting the animal study and preparing herbal extracts), Negar Azarpira (conducting the histopathological study), Mehrnaz Jafarpour (conducting mass spectroscopy and other experimental analysis and writing part of the article), and Fatemeh Ahmadi (supervising animal study and writing and finalizing the article).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was part of the PharmD thesis project of Hossein Rezaei and was financed by Shiraz University of Medical Sciences (Grant No. 90-01-36-3994).
Ethical Approval
The experiments were conducted in accordance with ethical guidelines for animal experimentation approved by ethical committee of Animal Laboratories in Shiraz University of Medical Sciences.
