Abstract
Diabetes is a life-threatening metabolic disorder. This study was undertaken to evaluate the antihyperglycemic and antioxidative potential of seed powder of Trigonella foenum-graecum L in alloxan (55 mg/kg) induced diabetic rats. The results obtained showed that extensive oxidative stress is generated in tissues of diabetic rats as evidenced by increased production of hydrogen peroxide, increased accumulation of malondialdehyde (MDA) and 4-hydroxynonanal (4HNE) and decreased activities of superoxide dismutase (SOD), glutathione peroxidase (GPx), and catalase (CAT) in tissues of diabetic rats. It was observed that the transcription of genes of SOD, GPx, and CAT was also significantly decreased when compared with control. Treatment of Trigonella for 15 days to diabetic rats showed hypoglycemic effect and improved the altered levels of H2O2, MDA, and 4HNE, the activities of SOD, GPx, and CAT as well as transcription of these genes in the liver and the brain of diabetic rats.
Keywords
Diabetes represents a spectrum of metabolic diseases that has become a major health challenge worldwide. The unprecedented economic development and rapid urbanization in Asian countries, particularly in India, has led to a shift in health problems from communicable to noncommunicable diseases. Among the noncommunicable diseases, diabetes leads the list. Diabetes is pandemic in both developed and developing countries. Diabetes mellitus is the most common endocrine disease with multiple etiologies and is a common disorder associated with increased morbidity and mortality rate and can be characterized by hyperglycemia due to defects in insulin secretion, insulin action, or both. Hyperglycemia is the important factor in the onset and progress of diabetic complications mainly by producing oxidative stress. 1,2 Some of the causes of development of long-term complications in persistent hyperglycemia are autooxidation of glucose, the formation of glycated proteins, and subsequent oxidative degradation of glycated proteins. 3 Several reports indicate that diabetic complications are associated with overproduction of free radicals including reactive oxygen species (ROS) and accumulation of lipid peroxidation by products. 4 ROS may also be involved in the initiation and development of vascular complications, both microvascular and cardiovascular, in diabetes. 1 Oxidative stress combined with mitochondrial dysfunction leads to the activation of inflammatory signaling pathways, which may damage insulin-producing cells and further aggravate the complications of diabetes. 5 ROS play an important role in the activation of stress responsive signaling pathways regulating the expression of genes responsible for cellular damage and other processes.
Cells have a wide range of defense mechanisms against ROS, still ROS bypass these defense systems and attack and modify subcellular components, including proteins, lipids, and nucleic acids. Cells are equipped with an elaborate antioxidant defense system. The first line of defense is constituted by antioxidant enzymes, namely, superoxide dismutase (SOD), glutathione peroxidase (GPx), and catalase (CAT), that directly scavenge free radicals and prevent cellular damage. Diabetes is known to induce alterations in the activities of these enzymes; however, the reports are contradictory. 6–8 The changes include decrease, increase, or no alterations in the activities of antioxidant enzymes in diabetic animals with wide variation depending on the age, duration of diabetes, or tissue studied. 9–13 Besides alterations in the activities of antioxidant enzymes, changes in mRNA expression of these enzymes have also been reported in various tissues. 14–17 The tissue antioxidant status seems to have an important role in the etiology of diabetic complications. Several reports are available in the literature on the perturbed levels of antioxidants in the plasma of diabetic patients, and most of the studies indicate that supplementation of antioxidants provide greater protection against oxidative stress–induced damage.
Plants have always been a very good source of drugs, and many of the currently available drugs have been derived directly from them. The added advantages of herbal drugs are safety, no side effects, and low cost. More than 400 plant species have been reported to have hypoglycemic and antidiabetic activity; however, search for new antidiabetic drugs is ongoing. Most of the plants contain glycosides, alkaloids, terpenoids, flavonoids, carotenoids, and so on, that are frequently implicated as having antidiabetic effect. Several plants, such as Azardica indica, Eugenia jambolana, Monordica charantia, Ocimum sanctum, Gymnema sylvestre, and Trigonella foenum graecum L, are being commonly used in the treatment of diabetes. Trigonella foenum graecum, commonly known as fenugreek, is well known for its pungent aromatic properties and is used both as a vegetable and as a spice and is consumed regularly in diet. Studies on different animal models have proved that fenugreek has strong antidiabetic properties. 18,19 Several studies have demonstrated that fenugreek seed extract, mucilage of seeds, and leaves can decrease blood glucose levels in human and experimental diabetic animals. 20,21 Several human intervention trials demonstrated that the antidiabetic effect of Trigonella seeds ameliorate most of the metabolic symptoms associated with type 1 and type 2 diabetes in both humans and relevant animal models. It is being used as folk medicine not only in diabetes but also a in number of other diseases. 20,22 However, the antioxidant potential of Trigonella foenum-graecum and its effects at the level of gene expression of antioxidant enzymes has not been clearly elucidated. Looking at the potent antidiabetic properties of Trigonella foenum-graecum, the present study was planned to elucidate the molecular mechanism of its antioxidant potential and to find out whether the effect of oral feeding of seed extract has any effect on the level of expression of mRNA of antioxidant enzymes in the tissues (liver and brain) of alloxan-induced diabetic rats.
Material and Methods
Chemicals
Chemicals used in the present study were of highest purity/analytical grade. Sodium chloride, potassium chloride, sodium carbonate, sodium bicarbonate, disodium hydrogen phosphate, sodium dihydrogen phosphate, triton X-100, sodium hydroxide, ethylenediamine tetraacetic acid, reduced glutathione, sodium azide, ethanol, 3-[N-morpholino] propane sulfonic acid (MOPS buffer), and magnesium chloride were purchased from HIMEDIA Chemicals (Mumbai, India). Glutathione, Folin-Ciocalteu’s reagent, glucose-6-phosphate dehydrogenase, glucose-6-phosphate, nicotinamide adenine dinucleotide phosphate (reduced and oxidized), TRI reagent, formamide, trisodium citrate, sodium acetate, and ethidium bromide were purchased from Sigma Aldrich Chemicals (St Louis, MO). One-step reverse transcriptase polymerase chain reaction (RT-PCR) kit and DNA molecular weight markers (100-600 bp) were obtained from Qiagen India Private Limited (New Delhi, India), and PCR primers were obtained from Eurofins Genomics India Private Limited (Bengaluru, India).
Experimental Animals
Adult male albino rats of Wistar strain (Rattus norvegicus) weighing about 120 ± 10 g were used in the present study. Rats were obtained from the animal facilities of Defence Research and Development Establishment, Gwalior, India, and were maintained in a light (light–dark cycle of 12 hours each) and temperature (25 ± 2°C) controlled animal room of our department on standard pellet diet (obtained from Amrut Rat & Mice Feed, New Delhi, India) and tap water ad libitum. Rats were acclimatized for 1 week prior to the start of the experiment.
Experimental Design
Twenty-four rats were randomly divided into 4 groups of 6 rats each. Animals were divided into 4 groups and were given following treatments:
Group 1: Control (healthy rats with normal blood glucose level)
Group 2: Treated control group (treated with Trigonella seed powder 1740 mg/kg body weight for 2 weeks)
Group 3: Diabetics (intravenous injection of alloxan 55 mg/kg body weight)
Group 4: Treated diabetic group (treated with Trigonella seed powder 1740 mg/kg body weight for 2 weeks)
Induction of Experimental Diabetes and Trigonella Treatment
Diabetes was induced in 12 overnight fasted adult rats by single intravenous injection of 55 mg/kg body weight of alloxan monohydrate dissolved in normal saline (0.85% NaCl). 23 Diabetes was confirmed by blood glucose estimation after 48 hours by an electronic glucometer. The rats with fasting glucose level above 300 mg/dL were considered diabetics and selected for the study.
Trigonella foenum-graecum (Fenugreek) seeds were purchased from the local herbal market, cleaned, dried, and finely powdered. The plant material was identified by the School of Studies in Botany, Jiwaji University, Gwalior, where the herbarium was submitted and the accession number given was 5310. Aqueous suspension of powdered Trigonella foenum-graecum seeds was prepared and 1740 mg/kg body weight was given orally to the rats of groups 2 and 4 with the help of cannula, daily for 2 weeks. 24
Rats were humanely killed 24 hours after the last treatment by cervical dislocation; different tissues were excised off, washed with 0.9% NaCl, and used for different estimations. Animals were handled, ethically treated, and humanely killed as per the rules and instructions of Ethical Committee of Animal Care of Jiwaji University, Gwalior, India, in accordance with the Indian national law on animal care and use. The study was approved by the academic ethical committee of Jiwaji University (Ethical Code IAEC/JU/2011/01).
Estimations
Estimation of Lipid Peroxidation
Malondialdehyde (MDA) and 4-hydroxynonanal (4HNE), the 2 major end products of lipid peroxidation, were estimated by the method of Jacobson et al 25 with minor modification. A 10% of tissue homogenate was prepared in Tris–HCl buffer (20 mM, pH 7.4). Prior to homogenization, 10 μL 0.5 M BHT in acetonitrile should be added per 1 mL of tissue homogenate. After homogenization, the homogenate was centrifuged at 3000 g at 4°C for 10 minutes, and the clear supernatant was used for the assay. Briefly, 200 μL of supernatant was transferred to 650 μL of 10.3 mM 1-methyl-2-phenylindole in acetonitrite and vortex mixed. To assay MDA + 4HNE, 150 μL of 15.4 M methanesulfonic acid (MSA) was added, vortexed, and incubated at 45°C for 40 minutes. To assay MDA alone, 150 μL of 37% HCl was added instead of MSA, vortexed, and incubated at 45°C for 60 minutes. After incubation the sample were kept on ice, centrifuged at 9500 g for 5 minutes, and absorbance was measured at 586 nm. The levels of MDA and 4HNE are expressed as nmol g−1 tissue using extinction coefficient 1.1 × 105 M−1 cm−1.
Estimation of Hydrogen Peroxide
Hydrogen peroxide (H2O2) levels were measured by the method of Pick. 26 For assay of H2O2 100 μL of tissue homogenate was prepared in Tris–HCl buffer (20 mM, pH 7.4) and 100 μL of assay solution (containing 0.2 mL phenol red, 0.2 g/L and 0.2 mL of horse radish peroxidase, 20 U/mL in potassium phosphate buffer, 0.05 M, pH 7.0, and 9.6 mL of 0.9% NaCl) was taken and reaction was started by the addition of 10 μL of 1.0 N NaOH. Absorbance was recorded at 600 nm in a microplate using ELISA reader. Hydrogen peroxide standard curve was plotted by taking different concentrations of H2O2, ranging from 20 to 100 μmol in a total volume of 100 μL and processed in the same way. Results are expressed as μmol H2O2 formed/mL homogenate.
Estimation of Enzyme Activities
Superoxide dismutase activity was assayed by estimation of inhibition of autooxidation of epinephrine. 27 Ten percent of tissue homogenate was prepared in 0.9% NaCl, centrifuged at 15 000 g for 15 minutes, and the corresponding supernatant was used for enzyme assay. Reaction mixture containing 0.5 mL sodium carbonate buffer (0.3 M, pH 10.2), 0.5 mL EDTA (0.6 mM), 0.5 mL homogenate, and 1.0 mL distilled water was incubated at room temperature for 10 minutes. The reaction was initiated by addition of 0.5 mL epinephrine (1.8 mM), and absorbance change per minute was recorded for 5 minutes at 480 nm. Specific activity is expressed as % inhibition of autooxidation of epinephrine by the enzyme min−1 mg−1 protein.
Glutathione peroxidase activity was estimated as described by Paglia and Valentine. 28 Ten percent of tissue homogenate was prepared in 1.15% KCl, centrifuged at 5000 g for 10 minutes, and the supernatant was used for enzyme assay. Reaction mixture containing 0.3 mL sodium phosphate buffer (0.1 M, pH 7.4), 0.1 mL GSH (0.15 M), 0.05 mL sodium azide (2.25 M), 0.05 mL homogenate, 0.1 mL NADPH (0.84 mM), and 0.05 mL glutathione reductase (2 U/mL) was incubated at room temperature for 10 minutes. Reaction was initiated by the addition of 0.05 mL H2O2 (0.0011 M), and absorbance change per minute was recorded for 5 minutes at 340 nm. Specific activity is expressed as nmole NADPH oxidized min−1 mg−1 protein.
Catalase activity was estimated by the method of Aebi. 29 Ten percent of tissue homogenate was prepared in 1.15% KCl, centrifuged at 5000 g for 10 minutes, and the supernatant thus obtained was used for enzyme assay. Reaction mixture containing 0.8 mL phosphate buffer (K2HPO4/NaH2PO4, 50 mM, pH 7.0), 0.1 mL homogenate, and 0.1 mL triton X-100 (0.02%) was incubated at room temperature for 10 minutes. Reaction was initiated by addition of 2.0 mL H2O2 (0.03 M prepared in potassium phosphate buffer, pH 7.0), and absorbance change per minute was recorded for 5 minutes at 240 nm. Specific activity is expressed as micromole H2O2 decomposed min−1 mg−1 protein.
Protein in the tissues samples was estimated by the method of Lowry et al using bovine serum albumin as standard. 30
Studies on Expression
Total RNA from the tissues was separated by the method described by Sambrook and Russell. 31 Briefly, 100 mg of fresh tissue was homogenized in 1.0 mL ice-cold monophasic lysis reagent (TRI reagent). The homogenate was incubated for 5 minutes at room temperature followed by addition of 0.2 mL chloroform with vigorous shaking. The mixture was centrifuged at 3360 g for 15 minutes in cold condition and the upper aqueous layer was separated. The RNA was precipitated by the addition of 0.25 mL isopropanol and 0.25 mL RNA precipitation solution (1.2 M NaCl and 0.8 M trisodium citrate), mixed thoroughly, and incubated at room temperature for 10 minutes. The solution was centrifuged at 3360 g for 10 minutes and the pellet containing RNA was separated. The pellet was washed twice with 75% ethanol and dissolved in 50 μL DEPC-treated water. RT-PCR was performed using Qiagen one-step RT-PCR kit. A master mix was prepared containing RT-PCR buffer, dNTP mix, Q solution, primers, reverse transcriptase enzyme, and template RNA as per manufacturer’s protocol. The primers used for superoxide dismutase, glutathione peroxidase, catalase, and 18 S RNA were designed using the Primer3 software.
Superoxide Dismutase
CCACTGCAGGACCTCATTTT
CACCTTTGCCCAAGTCATCT
Glutathione peroxidase
ATAGAAGCCCTGCTGTCCAA
GAAACCGCCTTTCTTTAGGC
Catalase
ACATGGTCTGGGACTTCTGG
CAAGTTTTTGATGCCCTGGT
18 S RNA
CGCGGTTCTATTTTGTTGGT
AGTCGGCATCGTTTATGGTC
18 S RNA was used as housekeeping gene. First strand of cDNA was synthesized using Omniscript reverse transcriptase at 50°C for 30 minutes. Thirty-five cycles of PCR were performed with a profile of 94°C for 1 minute, 59°C for 1 minute, and 72°C for 1 minute. The amplicons were analyzed on 1.5% TAE-Agarose gel. The bands were identified based on the product size using a 100 bp DNA ladder—documented using a gel documentation system. The bands were quantified densitometrically with the Quantity One software. The results were normalized to the levels obtained for the 18 S gene by taking a ratio of the value obtained for the gene of interest to that of 18 S RNA. The values of the diabetic samples were expressed as percentages with respect to the control.
Statistical Analyses
Results are expressed as mean ± SE of 6 sets of observations taken on different days. Statistical analyses were performed using Sigma Stat Statistical software version 2.0. All the statistical analyses were performed using one-way analysis of variance post hoc Bonferroni’s multiple comparison test applied across the treatment groups. Significance was based on P value <0.05.
Results
Effect of Oral Administration of Trigonella foenum-graecum L Seed Powder on the Blood Glucose Level in Control and Diabetic Rats
The blood glucose level of all the rats was tested by taking the blood from the tail vein and using an electronic glucometer. It was observed that oral administration of an aqueous extract of Trigonella seed powder significantly decreased the blood glucose levels in diabetic as well as control rats. The results of the present study showed that oral administration of 1740 mg/kg body weight Trigonella seed powder daily for 14 days to the diabetic rats caused 27.3% decrease on the 7th day and 50.9% decrease in the blood glucose level on the 14th day of the start of treatment. The same Trigonella treatment to the control rats showed 10.2% decrease in the blood glucose level on day 14 when compared with day 0 (Table 1). The results clearly showed the hypoglycemic potential of Trigonella seeds.
Effect of Oral Administration of Trigonella on the Blood Glucose Levels in Normal and Diabetic Ratsa.

aA drop of blood was taken from the tail vein and glucose level was measured using an electronic glucometer. Blood glucose levels are expressed as mg/dL. Results are mean ± SE of 6 sets of observation. *P < .05, **P < .001, ***P < .0001, and # P > .05 when compared with respective control; cdComparison between diabetic and diabetic + treatment group; Control and diabetic rats were given aqueous suspension of powdered Trigonella foenum graecum seed powder orally, 1740 mg/kg body weight, with the help of cannula, daily for 2 weeks.
Effect of Oral Administration of Trigonella for 2 Weeks on the Levels of MDA and 4HNE in the Liver and the Brain of Control and Diabetic Ratsa.

aMalondialdehyde (MDA) and 4-hydroxynonanal (4HNE) levels are expressed as nmol g−1 tissue; Results are mean ± SE of 6 sets of observation. *P < .05, **P < .001, ***P < .0001, and # P > .05 when compared with respective control, cdComparison between diabetic and diabetic + treatment group; Control and diabetic rats were given aqueous suspension of powdered Trigonella foenum graecum seed powder orally, 1740 mg/kg body weight, with the help of cannula, daily for 2 weeks.
Effect of Oral Administration of Trigonella for 2 Weeks on the Levels of Hydrogen Peroxide in the Liver and the Brain of Control and Diabetic Ratsa.
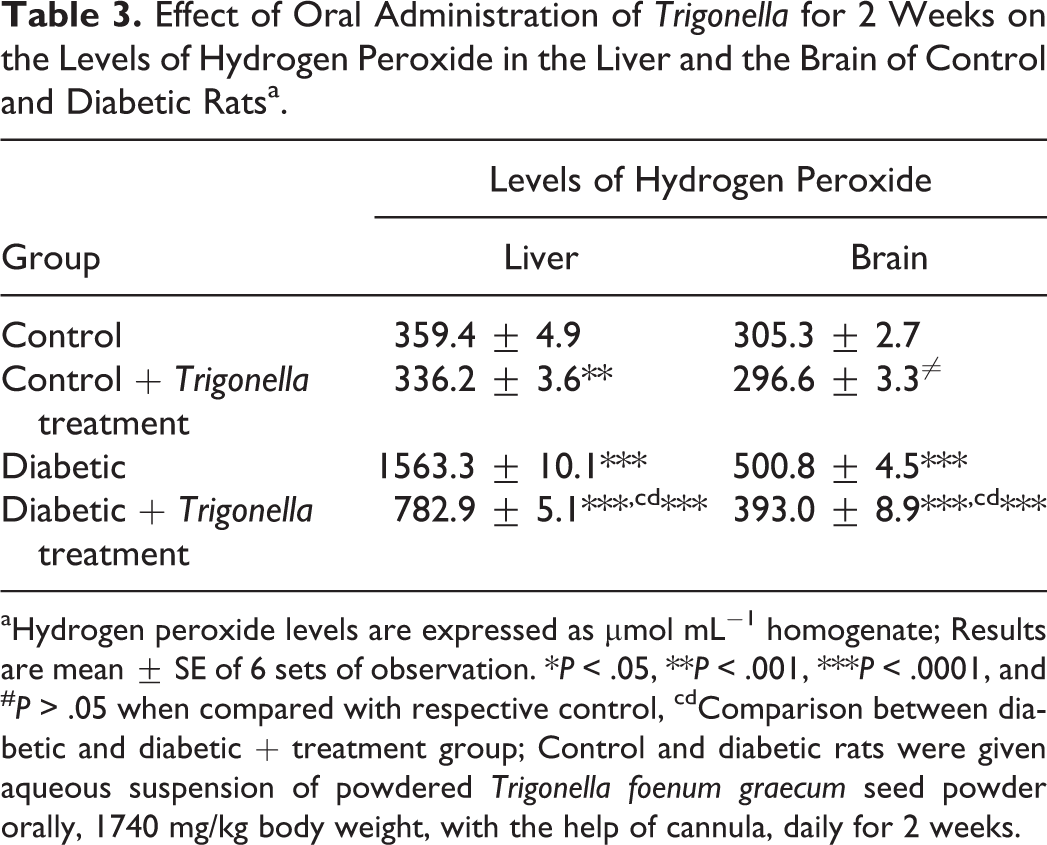
aHydrogen peroxide levels are expressed as μmol mL−1 homogenate; Results are mean ± SE of 6 sets of observation. *P < .05, **P < .001, ***P < .0001, and # P > .05 when compared with respective control, cdComparison between diabetic and diabetic + treatment group; Control and diabetic rats were given aqueous suspension of powdered Trigonella foenum graecum seed powder orally, 1740 mg/kg body weight, with the help of cannula, daily for 2 weeks.
Effect of Oral Administration of Trigonella for 2 Weeks on Antioxidant Enzyme Activities in the Liver and the Brain of Control and Diabetic Ratsa.

aGlutathione peroxidase (GPx) activity is expressed as nmol NADPH oxidized min−1 mg−1 protein; superoxide dismutase (SOD) activity is expressed as percentage inhibition of autooxidation of epinephrine by the enzyme min−1 mg−1 protein; and catalase (CAT) activity is expressed as micromole H2O2 decomposed min−1 mg−1 protein; Results are mean ± SE of 6 sets of observation. *P < .05, **P < .001, ***P < .0001, and # P > .05 when compared with respective control, cdComparison between diabetic and diabetic + treatment group; Control and diabetic rats were given aqueous suspension of powdered Trigonella foenum graecum seed powder orally, 1740 mg/kg body weight, with the help of cannula, daily for 2 weeks.
Effect of Oral Administration of Trigonella Seed Powder on the Levels of MDA and 4HNE in the Liver and Brain of Control and Diabetic Rats
The results of the present study clearly showed that alloxan administration in rats caused accumulation of MDA and 4HNE, the two major end products of lipid peroxidation, in the liver and the brain of rats when compared with control. MDA and 4HNE levels were increased 39% and 103% in the liver and 71.5% and 93% in the brain, respectively, when compared with control (Table 2). When the diabetic rats were given Trigonella treatment for 2 weeks, 15.8% and 25.2% decrease in MDA levels and 36.4% and 34.5% decrease in the 4HNE levels of the liver and the brain, respectively, were observed when compared with the control group (Table 2). When the Trigonella-treated diabetic rats were compared with the control group, percentage increase of MDA and 4HNE levels were 16.6% and 29.9% in the liver and 28.1% and 26.2% in the brain, respectively. The results clearly showed the antioxidative potential of Trigonella.
Administration of Trigonella showed antioxidative effect in the control group also by reducing the levels of MDA and 4HNE in both the liver and the brain. When the control rats were given Trigonella treatment for 2 weeks, 6.9% and 1.3% decrease in MDA levels and 0.22% and 7.2% decrease in the 4HNE levels of the liver and the brain, respectively, were observed when compared with the untreated control group (Table 2).
Effect of Oral Administration of Trigonella on the Levels of Hydrogen Peroxide in the Liver and the Brain of Control and Diabetic Rats
The presence of diabetes-induced oxidative stress was also confirmed by monitoring the levels of H2O2, the most stable ROS in rat tissues. The results showed that there was significantly high accumulation of H2O2 in the liver and the brain of diabetic rats. It was observed that hepatic H2O2 level was increased by 335% while 64% increase was observed in the brain of diabetic rats when compared with control. Trigonella treatment for 15 days caused a decrease in the levels of H2O2 in the control as well as diabetic rat tissues when compared with untreated group. Trigonella treatment caused 7% decrease in the liver and 3% decrease in the brain H2O2 levels of control rats when compared with the untreated group, while the same Trigonella treatment caused 118% decrease in the liver and 29% decrease in the brain H2O2 levels when compared with the control and 50% and 22% decrease in the liver and the brain, respectively, when compared with the untreated diabetic group (Table 3).
Effect of Oral Administration of Trigonella on the Levels of Antioxidant Enzymes in the Liver and Brain of Control and Diabetic Rats
Studies were carried out to monitor the effect of diabetes on the activities of antioxidant enzymes, namely, GPx, SOD, and CAT, in the tissues of rats. Experiments were also carried out to evaluate the protective effects of Trigonella treatment on the activities of these enzymes in tissues of control and diabetic rats. The results showed that the hyperglycemia induced by alloxan treatment decreased the activities of these antioxidant enzymes in the liver and the brain, when compared with control. The hepatic activities of GPx, SOD, and CAT were decreased by 30%, 23%, and 70% while 27%, 25%, and 73% decrease, respectively, was observed in the brain of diabetic rats when compared with control (Table 4).
Administration of Trigonella for 14 days showed some protection against diabetes-induced alterations in the activities of GPx, SOD, and CAT. The activities of GPx, SOD, and CAT were increased by 6%, 8%, and 17% in the liver and 16%, 10%, and 41% in the brain of control rats given Trigonella treatment for 2 weeks when compared with the untreated group. When the same Trigonella treatment was given to the diabetic rats, 28%, 13%, and 125% recovery was noted in the activities of hepatic GPx, SOD, and CAT, while the recovery in the brain was 29%, 30%, and 177%, respectively, when compared with untreated diabetic rats (Table 4).
Effect of Hyperglycemia and Trigonella Treatment on the mRNA Levels of Antioxidant Enzymes in the Liver and Brain of Control and Diabetic Rats
It was observed that experimental diabetes induced by alloxan altered the expression of GPx, SOD, and CAT genes in the liver and the brain of rats when compared with control. mRNA level of specific enzyme was monitored by first isolation of total RNA followed by RT-PCR, which consisted of (a) reverse transcription of specific mRNA using specific primer and (b) amplification of specific cDNA using specific primers. RT-PCR results revealed that experimental diabetes caused decrease in the gene expression of GPx, SOD, and CAT by 24.4%, 29.1%, and 23.4% in the liver and 24.3%, 41.1%, and 32.8% in the brain, respectively, compared with the control. The percentage of decrease in the gene expression of GPx, SOD, and CAT of diabetic rats was increased by 26.8%, 36.3%, and 22.9% in the liver and 23.1%, 55.7%, and 41.7% as a result of Trigonella administration (Figures 1 and 2).

Effect of the Trigonella treatment on (A) glutathione peroxidase (GPx), (B) superoxide dismutase (SOD), and (C) and catalase (CAT) mRNA in the liver and the brain of control and diabetic rats.

Densitometric scanning of expression pattern of mRNA of glutathione peroxidase (GPx), superoxide dismutase (SOD), and catalase (CAT) in the liver and the brain of control and diabetic rats with and without Trigonella treatment.
Administration of Trigonella seed powder showed some effect on control group also by reducing the expression level of GPx, SOD, and CAT in both the liver and the brain. When the control rats were given Trigonella treatment for 2 weeks, the expression of GPx, SOD, and CAT decreased by 4.9%, 3.3%, and 5.8% in the liver, while 6.7%, 8.3%, and 4.3% decrease in the brain, respectively, were observed when compared with the untreated control group (Figures 1 and 2).
Discussion
Oxidative stress plays a pivotal role in the development of diabetic complications, both microvascular and cardiovascular. In most diseases characterized by tissue damage, oxidative stress is either a cause or consequence of this damage. Cumulative or steady-state oxidative damage may increase in diabetes in response to an increase in oxidizable substrates, an increase in the rate of autooxidation of substrates, a decline in the antioxidant defense, or a combination of all of these processes. A precise understanding of oxidative stress in diabetes and the adaptive response to it may require the knowledge of the time of onset of manifestation of oxidative stress, their characterization in terms of oxidative damage to biomolecules like lipids, and the antioxidant enzymes affected and their regulation at the level of transcription or activity or both. Traditional plant remedies have been used for centuries in the treatment of diabetes, but only a few have been scientifically evaluated. 32
Trigonella foenum-graecum L, commonly known as fenugreek, is a plant that has been extensively used as an antidiabetic agent. 18,19,33 Both seeds and leaves of this plant are commonly used edibles as vegetable and flavoring agent to increase aroma and taste. Therefore, the present study was planned to evaluate the effect of oral feeding of seed powder of fenugreek on the level of diabetes-induced oxidative stress by monitoring the level of ROS and end products of lipid peroxidation, activities of antioxidant enzymes, and their expression as well as blood glucose levels. The results of the present study confirmed that fenugreek seeds demonstrated hypoglycemic potential in alloxan-induced experimental diabetes in rats. The blood glucose level of diabetic rats was decreased progressively and 27% and 57% decrease in the blood glucose levels was observed after 1 week and 2 weeks, respectively, of Trigonella seed treatment when compared with control. The antidiabetic potential of fenugreek may be due to the alkaloid, trigonelline or 4-hydroxyisoleucine, a unique amino acid present in fenugreek. 34,35 Generation of oxidative stress has also been evidenced by accumulation of lipid peroxidation products, MDA and 4HNE, in the tissue of diabetic rats.
Oxidative stress is known to exert varying effects on the activity of antioxidant enzymes. Various tissues show different sensitivity to oxidative stress and oxidative damage and could result in various complications in long-term diabetes, implying that the restoration of antioxidant status is an essential and important parameter to evaluate the effect of antidiabetic compounds. ROS-induced oxidative damage has been implicated in the pathogenesis of several disorders and degenerative diseases including Parkinson’s disease, Alzheimer’s disease, myocardial infarction atherosclerosis, bipolar disorder, fragile x-syndrome, and so on. 36–38 Oxidative stress is the outcome of either increased production of ROS or decreased scavenging ability of the organ, or both. The results of the present study clearly showed that the enzymatic antioxidant machinery comprising GPx, SOD, and CAT is severely inhibited in the liver and the brain of diabetic rats. Feeding of Trigonella seed power for 15 days improved the level of lipid peroxidation, levels of hydrogen peroxide, and activities of antioxidant enzymes. Several reports are available in literature on the antioxidant role of fenugreek. The formation of lipid peroxidation products, MDA and 4HNE, was measured in the tissues of diabetic rats as an index of oxidative stress and redox imbalance. A moderate increase in tissue MDA and 4HNE in alloxan-treated rats was found to be reduced after Trigonella treatment for 15 days. The observed increase in lipid peroxidation levels in the liver and the brain are in agreement with similar findings in other tissues. 9,16 Lipid peroxidation may bring about protein damage and inactivation of membrane-bound enzymes either through direct attack by free radicals or through chemical modification by its end products, MDA and 4HNE. 35 The decreased activities of SOD, CAT, and GPx may be a response to increased production of ROS, for example, hydrogen peroxide, by the autooxidation of the excess of glucose and nonenzymatic glycation of protein. 39 The decreased activities of antioxidant enzymes could also be due to the decreased protein expression levels in the diabetic conditions as seen in the present study. RT-PCR analysis showed a corresponding decrease in the level of transcription of these enzymes. In diabetic rats, however, the effect of oxidative stress on hepatic and brain CAT activities reported in literature varied significantly from no change, 9 decreased, 10,12,13 and increased 40 depending on the experimental conditions such as age of the animal and duration of diabetes.
The increased oxidative stress due to decreased activities of antioxidative enzymes and/or increased generation of ROS affects the transcription level of these enzymes. Elevated oxidative stress was found to be involved in the activation of NF-Kappa B and AP-1 transcription factors in tissue of diabetic rats, suggesting a possible mechanism for altered gene expression. 37 Trigonella treatment improved the oxidative stress and thus adverse effect on transcription of these enzymes was also decreased. Moreover, 4-hydroxyleucine, a novel amino acid from Trigonella seeds, is reported to have insulinotropic action and increases glucose-stimulated insulin release by isolated islet cells in rats, mice, and humans, 41–43 which may again be involved in restoration of deranged parameters.
In conclusion, the aforementioned findings indicate that in diabetic conditions, oxidative stress is induced that causes peroxidative damage to membrane lipids. Alloxan-induced diabetes has negative effects on the activities of antioxidant enzymes and inhibited their transcription in rats. Feeding of Trigonella foenum-graecum seed powder improved the activities of these enzymes and their transcription also. Further work is required to find out the active compound in the seeds of Trigonella foenum-graecum and molecular mechanism of its action.
Footnotes
Author Contributions
SS perform the experiments, collected the data, developed the figures, analyzed the data, and wrote the article. VM and SKJ performed the experiments and helped in statistical analysis. NS conceptualized the study, critically analyzed and discussed the data, and corrected and reviewed the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thankfully acknowledge the financial support of the Department of Science and Technology, New Delhi, India, in the form of FIST Grant to the School; and Madhya Pradesh Council of Science and Technology, Bhopal, India, in the form of individual research project to Nalini Srivastava.
Ethical Approval
The study was approved by the academic ethical committee of Jiwaji University (Ethical Code IAEC/JU/2011/01).
