Abstract
Background
In December 2019, an outbreak of coronavirus disease 2019 (COVID-19) began in Wuhan, China, and led to a global epidemic. We aimed to compare the clinical and serological features of COVID-19 patients with positive and negative reverse transcriptase polymerase chain reaction (RT-PCR) tests.
Methods
This was a retrospective cohort study conducted from 9 February to 4 April 2020. COVID-19 patients at Leishenshan Hospital in Wuhan, China (125 total cases; 87 RT-PCR positive and 38 RT-PCR negative) were included. COVID-19 serology was assessed by colloidal gold assay. All cases were analyzed for demographic, clinical, and serological features.
Results
There were no significant differences in most demographic features, clinical symptoms, complications or treatments of RT-PCR positive and negative COVID-19 patients. Serum IgM/IgG was positive in 82 (94%) and 33 (87%) RT-PCR positive and negative cases, respectively. IgM was detectable as early as 3 days after symptom onset and was undetectable 60 days after symptom onset. By contrast, IgG could be detected only 10 days after symptom onset and reached its peak 60 days after symptom onset.
Conclusions
Serological tests performed during the appropriate time window of disease progression could be valuable auxiliary methods to RT-PCR in COVID-19 patients.
Keywords
Introduction
Coronaviruses are large, enveloped viruses with linear positive-strand RNA genomes 1 that are major causes of respiratory tract infections in humans. Two previous pandemics caused by severe acute respiratory syndrome coronavirus (SARS-CoV)2–4 and Middle East respiratory syndrome coronavirus5,6 demonstrated the serious threat posed by coronaviruses to global public health.
In December 2019, an outbreak of coronavirus disease 2019 (COVID-19) began in Wuhan, China. Whole genome sequencing of viruses in lower respiratory tract samples revealed that the outbreak was caused by a novel beta-coronavirus4,7,8 with phylogenetic similarity to SARS-CoV. The novel coronavirus was named severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) by the International Committee on Taxonomy of Viruses. SARS-CoV-2 causes severe respiratory tract infection and rapid person-to-person transmission led to a global pandemic.9–11
The clinical features of COVID-19 are complex and similar to those of other diseases including influenza virus infection. Symptoms include fever, cough, sore throat and fatigue.12,13 Therefore, rapid diagnosis of COVID-19 in suspected cases is essential to curb viral transmission and to initiate appropriate therapies. At present, definite diagnosis of COVID-19 is mainly based on positive results reverse transcriptase polymerase chain reaction (RT-PCR) amplification of viral nucleic acids from respiratory tract specimens.12,14,15 However, RT-PCR is restricted to certified laboratories with specialized equipment, expensive kits and trained technicians. Furthermore, RT-PCR requires respiratory tract specimens, such as throat swabs, nose swabs or sputum samples, which could delay diagnosis and treatment. 16 Thus, serological tests are increasingly applied for diagnosis of SARS-CoV-2 infection.17,18 In this study, we comprehensively analyzed the demographic and clinical features of 125 COVID-19 patients at a mobile hospital in Wuhan, China. We assessed seropositivity rates and analyzed the dynamics of IgM and IgG against COVID-19. Our data demonstrate the clinical value of serological tests for diagnosis of COVID-19, especially in patients with negative RT-PCR tests.
Materials and methods
Patients
The patients enrolled in this study were transferred to Leishenshan Hospital in Wuhan, China, from 9 February to 4 April 2020. Patients were first tested in other hospitals in Wuhan within 20 days of the onset of clinical symptoms. Leishenshan Hospital uniformly treated patients with severe clinical symptoms transferred from other hospitals in Wuhan. After transfer to Leishenshan Hospital, RT-PCR testing was performed every week for most patients following weekly treatments. Testing dates were extended or shortened according to each patient’s condition. RT-PCR was conducted from 15 January to 1 April 2020; this period included the first test performed in Wuhan hospitals and all tests performed in Leishenshan Hospital until discharge. The patients included cases with detectable SARS-CoV-2 RNA in respiratory samples since disease onset (defined as RT-PCR positive cases) and cases with clinical manifestations characteristic of COVID-19 but no detectable viral RNA in upper respiratory tract samples (defined as RT-PCR negative cases). After transfer to Leishenshan Hospital, all COVID-19 patients were treated uniformly. For RT-PCR positive patients, at least one RT-PCR test had to be positive; by contrast, in RT-PCR negative patients RT-PCR tests were consistently negative. Leishenshan Hospital was a designated mobile hospital and mainly admitted COVID-19 patients with severe clinical symptoms transferred from other hospitals in Wuhan. This study was approved by the Leishenshan Hospital ethics committee and written informed consent was obtained from patients. We followed the most up‐to‐date version of the Coronavirus Pneumonia Prevention and Control Program published by the National Health Commission of China.
Serological tests
Serological tests were performed using the SARS-CoV-2 IgM/IgG Antibody Combined Test Kit (Wondfo, Guangzhou, China; INNOVITA, Tangshan, China) as per the manufacturer’s instructions. Serum samples were collected and treated with heparin or sodium citrate to prevent coagulation. Serum/plasma (10 µL) was added to the specimen well of the test cassette and then two drops of sample buffer were immediately added. The result was read and recorded after 15 minutes.
Data collection
Demographic, clinical, serological and outcome data were obtained using data collection forms from electronic medical records. The data included demographic characteristics, results of serological tests, comorbidities, symptoms and signs (i.e. fever and cough), complications, treatments received (i.e. antiviral therapy, antibiotics, oxygen therapy, corticosteroids, and dialysis) and outcome data during hospitalization. The antiviral treatments administered included arbidol (200 mg thrice daily, orally), ritonavir (600 mg twice daily, orally), ganciclovir (250 mg every 12 hours, intravenously), and ribavirin (500 mg every 12 hours, intravenously). The duration of antiviral treatment was 4 to 14 days. Serum samples were collected and the time intervals between symptom onset and serum sample collection were documented. The date of disease onset was defined as the day that symptoms were first noticed. The admission dates of patients were from 9 February to 9 March 2020. The durations from symptom onset to hospital admission and to hospital discharge were recorded. Clinical outcomes were followed up until 4 April 2020.
Statistical analysis
Non-normally distributed continuous variables were presented as medians (interquartile ranges [IQR]). Differences between non-normally distributed continuous variables were assessed using the Mann–Whitney U test. Categorical variables were presented as counts (%). Differences between categorical variables were assessed using the χ2 or Fisher’s exact tests. A two-sided value of p < 0.05 was considered statistically significant. All statistical analyses were conducted using the IBM SPSS version 26 (IBM, Armonk, NY, USA) and GraphPad Prism version 8.2.0 (GraphPad Inc., San Diego, CA, USA).
Results
Demographic and clinical features of RT-PCR positive and negative COVID-19 patients admitted to Leishenshan Hospital
A total of 125 COVID-19 patients (87 RT-PCR positive and 38 RT-PCR negative) were enrolled in the study. We found no significant difference in the median age of RT-PCR positive and negative cases (58.0 years vs. 63.0 years, Table 1). Most patients were between 50 and 69 years of age (Figure 1a). More than half of patients had underlying comorbidities (Table 2) including hypertension (54, 43%), diabetes (20, 16%), chronic kidney disease (14, 11%), cardiovascular disease (12, 10%), and cerebrovascular disease (9, 7%). There were no significant differences in comorbidities between RT-PCR positive and negative cases except for chronic kidney disease (16% vs. 0%, p = 0.005). The symptom onset dates were distributed from 20 January to 10 February 2020 in both groups of patients (Figure 1b). The median duration from symptom onset to admission to Leishenshan Hospital was 27.0 days (IQR 15.0–33.0 days).
Demographics and clinical outcomes of 125 COVID-19 patients admitted to Leishenshan Hospital.

COVID-19, coronavirus disease 2019; IQR, interquartile range; p values indicate differences between RT-PCR positive and negative cases.

Distribution of coronavirus disease 2019 (COVID-19) patient age (a) and date of symptom onset (b). Cases are stratified into reverse transcriptase polymerase chain reaction (RT-PCR) positive and RT-PCR negative subgroups.
Comorbidities of 125 COVID-19 patients.
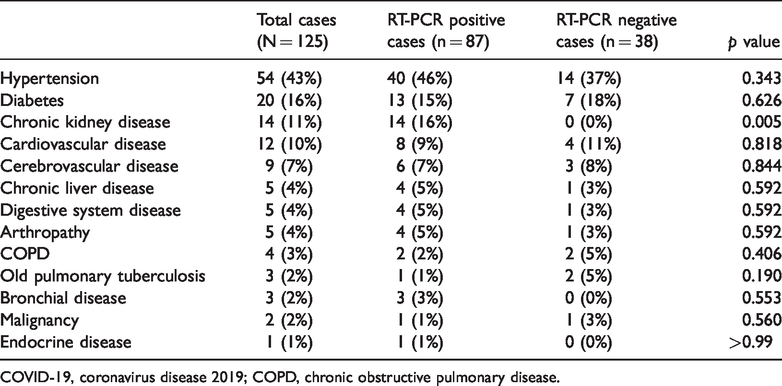
COVID-19, coronavirus disease 2019; COPD, chronic obstructive pulmonary disease.
Common symptoms on admission were fever (111, 89%), dry cough (111, 89%), shortness of breath (92, 74%), respiratory distress (90, 72%), fatigue (86, 69%), myalgia (63, 50%), and anorexia (58, 46%). Less common symptoms included dizziness (8, 6%), rhinorrhea (4, 3%), chest pain (4, 3%) and headache (4, 3%) (Table 3). Compared with RT-PCR negative patients, RT-PCR positive patients were more likely to present with shortness of breath (79% vs. 61%, p = 0.028) and myalgia (57% vs. 34%, p = 0.017).
Clinical symptoms of 125 COVID-19 patients.
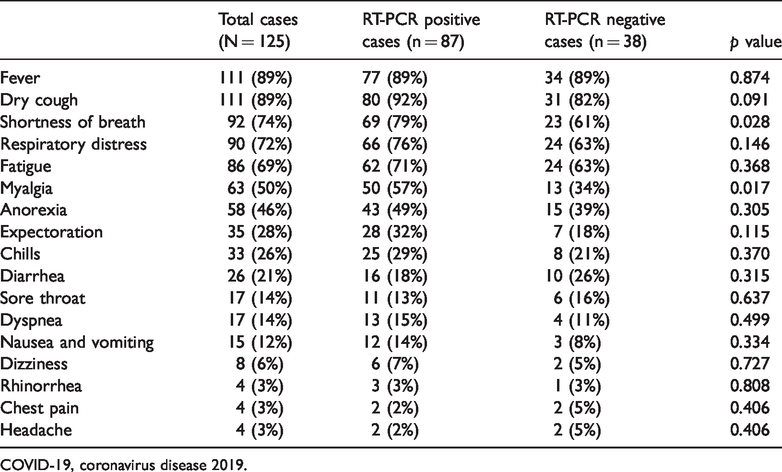
COVID-19, coronavirus disease 2019.
The frequencies of complications, including respiratory failure, arrhythmia, acute respiratory distress syndrome and shock, were not significantly different between the two groups of patients (Table 4). All patients admitted to Leishenshan Hospital were treated in isolation. Common treatments administered in both groups were antiviral therapy (119, 95%), oxygen inhalation (116, 93%) and antibiotic therapy (83, 66%). Non-invasive ventilation was required more often in RT-PCR negative cases (2% vs. 16%, p = 0.007), and no patients received extracorporeal membrane oxygenation as rescue therapy. For RT-PCR positive patients who were also diagnosed with chronic kidney disease, kidney replacement therapy was required. Our data suggested that there were no significant differences in most demographic and clinical features, complications and treatments between RT-PCR positive and negative COVID-19 patients.
Complications and treatments of 125 COVID-19 patients.

COVID-19, coronavirus disease 2019; ARDS, acute respiratory distress syndrome; CKRT, continuous kidney replacement therapy; NIV, noninvasive ventilation; IMV, invasive mechanical ventilation; ECMO, extracorporeal membrane oxygenation.
Serological features and dynamics of SARS-CoV-2-specific IgM/IgG
Most patients (115 of 125) were seropositive by colloidal gold assay. The time intervals between symptom onset and sample collection ranged from 3 to 70 days. The timing of serum collection and serological tests was not exactly the same in each patient. The time intervals between serological tests for RT-PCR positive cases and negative cases are shown in Table 5. There was no difference in the duration between symptom onset and testing between the two patient groups. Overall, 82 of 87 RT-PCR positive patients were positive for serological tests and 33 of 38 RT-PCR negative patients were positive for serological tests (Table 5). Thus, at least 87% of RT-PCR negative cases were missed by nucleic acid testing.
Seropositivity rates of RT-PCR positive and negative COVID-19 patients.

COVID-19, coronavirus disease 2019.
We further analyzed the dynamics of IgM/IgG at different time points post-symptom onset (Figure 2a, b). The exact number of patients tested at each time interval is shown in Supplementary Table 1 (corresponding to Figure 2a). Because serological tests were performed on patients at different time points, IgM and IgG results differed. In general, IgM is easier to detect during the early stages of infection soon after symptom onset, while IgG can be detected at later times. In Figure 2a, “Patients tested for SARS-CoV-2 antibodies” refers to the total number of patients undergoing SARS-CoV-2 antibody testing during different time intervals. “Patients with positive IgM or IgG” refers to the number of patients with positive SARS-CoV-2 IgM and IgG, respectively, among patients undergoing serological testing. IgM was detectable as early as 6 days after symptom onset and was undetectable after 60 days. In contrast, IgG could be detected only 10 days after symptom onset and rose gradually, reaching a peak after 60 days (Figure 2b). Therefore, serological tests performed during the appropriate time window of disease progression may be helpful for diagnosis of COVID-19, especially in patients with negative RT-PCR tests. Companion serological tests may help to diagnose SARS-CoV-2 infected patients and evaluate stages of infection more accurately.
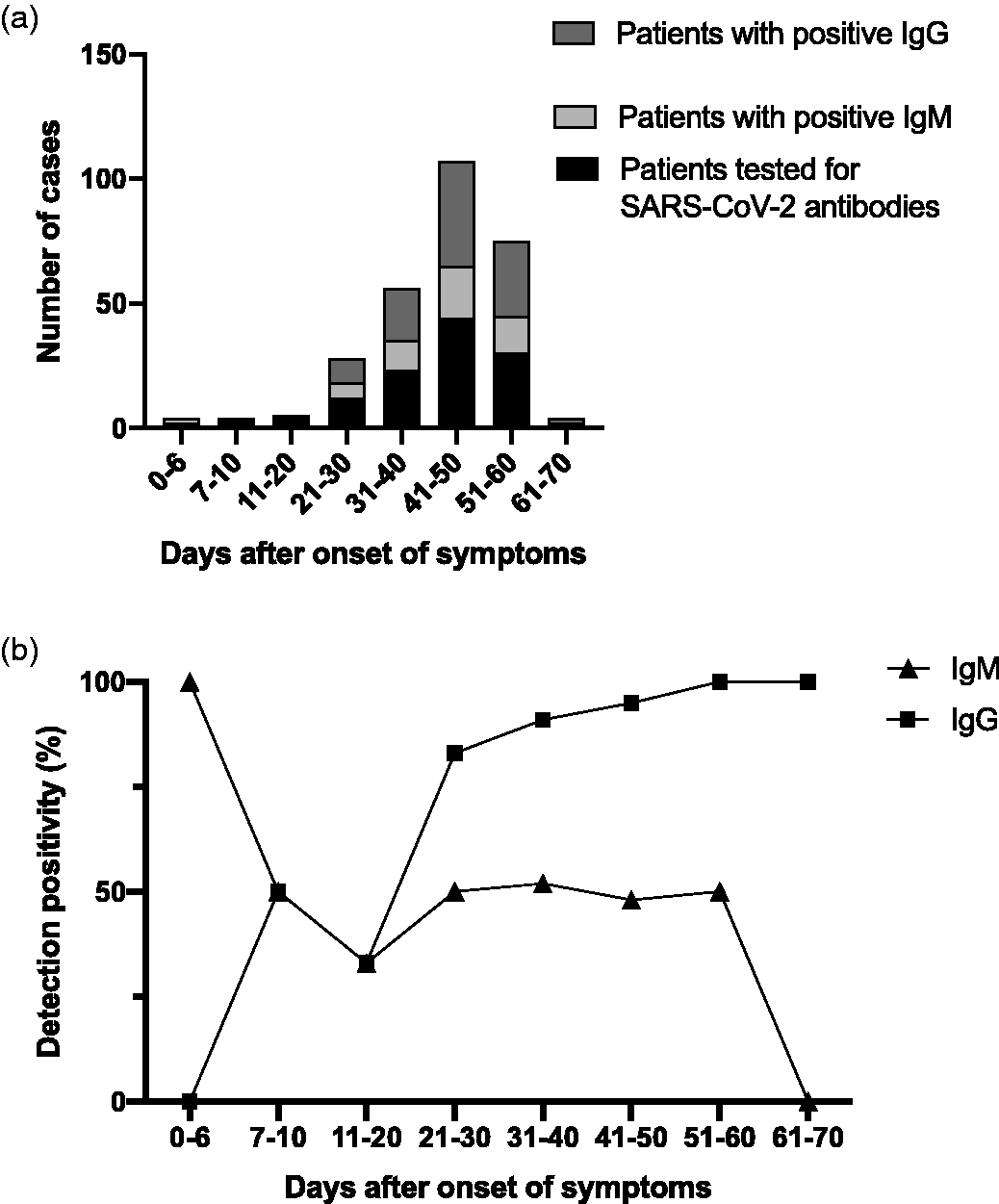
Summary of serological test results of coronavirus disease 2019 (COVID-19) patients. (a) Numbers of seropositive cases at different time intervals. (b) IgM/IgG dynamics at different time intervals.
Discussion
COVID-19 can cause serious, sometimes fatal, pneumonia19–21 and asymptomatic COVID-19 patients may also be capable of transmission. 22 Thus, timely and accurate diagnosis of COVID-19 is crucial. RT-PCR amplification of viral nucleic acid has been widely recognized as the gold standard for diagnosis of COVID-19 and can effectively confirm early SARS-CoV-2 infection. 15 However, some studies have reported that assessment of antibody responses induced in COVID-19 patients using serological tests is a useful complement for diagnosis of patients with negative RT-PCR results.18,23–25 In this study, we conducted a comprehensive analysis of the demographic, clinical and serological features of 125 COVID-19 patients transferred to Leishenshan Hospital (87 RT-PCR positive cases and 38 RT-PCR negative cases with clinical manifestations of COVID-19). The clinical value of serological tests for diagnosis of COVID-19 patients with negative RT-PCR tests was demonstrated by our results.
The symptom onset dates of the patients studied here were distributed from 20 January to 10 February 2020 and the admission dates were distributed from 9 February to 9 March 2020. By 4 April 2020, 84 of 125 patients had been discharged and another 41 patients were still undergoing treatment. Our data showed no significant differences between the majority of demographic features, clinical symptoms, complications and treatments of RT-PCR positive and negative COVID-19 patients.
SARS-CoV-2 IgM/IgG was detected in 82 (94%) and 33 (87%) RT-PCR positive and negative COVID-19 patients, respectively. This result demonstrates the clinical value of serological tests for diagnosis of COVID-19, especially in patients with negative RT-PCR tests. We found that SARS-CoV-2 specific IgM appeared 3 days after symptom onset and was easily detected during the first week. After this time, IgM decreased sharply. IgG could be detected 10 days after symptom onset and rose gradually, reaching a peak 60 days after symptom onset. During this period, IgG can be detected to determine whether SARS-CoV-2 infected has occurred. Our results suggest that the appropriate time window for IgM serological tests is from 3 days to 1 week after symptom onset; during this time IgM can be used as a diagnostic indicator of acute or recent infection. By contrast, the appropriate time window for IgG serological tests is from 10 days to 2 months after symptom onset; IgG can be detected during the later stages of symptoms or the recovery period and reflects past infection. Therefore, serological tests performed during appropriate time windows of the disease course could be considered as valuable auxiliary methods. It is important to note that serological tests are effective methods for patients with clinical symptoms typical of COVID-19 or an epidemiological contact history but negative nucleic acid tests. However, serological tests are not suitable for screening the general population in areas with low infection rates.
Our study had several limitations. First, only 125 COVID-19 cases at Leishenshan Hospital were included. There are several reasons for this relatively small sample size. In response to the COVID-19 outbreak, Leishenshan Hospital was a designated mobile hospital and mainly admitted COVID-19 patients with severe clinical symptoms transferred from other hospitals in Wuhan. Thus, the total number of patients admitted to Leishenshan Hospital was relatively small. To maintain consistency of serological testing and to ensure the rigor of the analysis, we analyzed a cohort of patients tested with the same colloidal gold kits; these two kits have been approved by the National Medical Products Administration of China. Second, the numbers of RT-PCR positive and negative cases were dissimilar (87 and 38, respectively). The proportion of patients who cannot be diagnosed by RT-PCR is relatively small, which means that the sizes of the two groups were quite different. It would be optimal to include as many patients as possible in other cities in China, and even in other countries to obtain a more comprehensive understanding of SARS-CoV-2. Third, because of the severe epidemic and need for urgent treatment, the timing of nucleic acid and serological tests was not consistent in all patients. However, we found no difference in the timing of sampling or testing between the two patient groups (RT-PCR positive and negative). Currently, RT-PCR and serological tests are the most commonly used methods for diagnosing COVID-19 patients. In our study, all patients were tested using two validated and approved kits. We only analyzed the dynamics of IgM/IgG from 2 to 70 days after symptom onset. Finally, detailed patient information including clinical outcomes was unavailable at the date of analysis.
Conclusion
Serological tests performed during the appropriate time window of disease progression are useful auxiliary tools for diagnosis of COVID-19, especially in patients with negative RT-PCR tests. Timely and accurate diagnosis requires an RT-PCR combined with typical clinical symptoms and serological tests.
Footnotes
Acknowledgements
We are grateful to the medical team of Shanghai General Hospital working in Leishenshan Hospital, Wuhan, China.
Authors’ contributions
Drs. Weidong Li and Jun Liu had full access to all study data and take responsibility for the integrity of the data and the accuracy of the analysis. Ms. Caiqin Li, Dr. Qi Su and Dr. Jun Liu contributed equally to this work.
Concept and design of the work: Jun Liu, Weidong Li
Acquisition, analysis, or interpretation of data: Jun Liu, Qi Su, Caiqin Li
Drafting the article: Caiqin Li
Critical revision of the article for important intellectual content: Weidong Li, Jun Liu, Lei Chen
Statistical analysis: Caiqin Li, Qi Su, Yuting Li
Administrative, technical, or material support: Xiaoli Tian, Qi Su, Jun Liu
Approval of the version to be submitted: Weidong Li, Jun Liu
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by the Shanghai Education Commission Research and Innovation Program (grant number 2019-01-07-00-02-E00037) and “Eastern Scholar” (Shanghai Municipal Education Commission).
