Abstract
Few homeopathic complexes seemed to produce significant effects in osteoarthritis; still, individualized homeopathy remained untested. We evaluated the feasibility of conducting an efficacy trial of individualized homeopathy in osteoarthritis. A prospective, parallel-arm, double-blind, randomized, placebo-controlled pilot study was conducted from January to October 2014 involving 60 patients (homeopathy, n = 30; placebo, n = 30) who were suffering from acute painful episodes of knee osteoarthritis and visiting the outpatient clinic of Mahesh Bhattacharyya Homeopathic Medical College and Hospital, West Bengal, India. Statistically significant reduction was achieved in 3 visual analog scales (measuring pain, stiffness, and loss of function) and Osteoarthritis Research Society International scores in both groups over 2 weeks (P < .05); however, group differences were not significant (P > .05). Overall, homeopathy did not appear to be superior to placebo; still, further rigorous evaluation in this design involving a larger sample size seems feasible in future. Trial registration: Clinical Trials Registry, India (CTRI/2014/05/004589).
To improve the management of hip or knee osteoarthritis, a multidisciplinary guideline-based stepped-care strategy has been developed with recommendations regarding the appropriate nonsurgical treatment modalities and optimal sequence for care. 1 In spite of that, conventional medical treatment of osteoarthritis successfully relieves pain only, alongside producing adverse gastrointestinal and cardiovascular effects, especially with long-term use. 2 Hence, many patients use complementary and alternative medicine, including homeopathy, to prevent, control, and manage the pain of rheumatologic conditions. 3,4 These rheumatologic problems are among the most common disease conditions encountered by complementary and alternative medicine practitioners. 5 However, scientific research has so far not provided evidences solid enough to support the effectiveness of complementary and alternative medicine as treatment options for rheumatologic conditions, including osteoarthritis, and has remained ambiguous. 6 Reviews have remained contradictory in conclusions. 7–13 Few low-potency homeopathic complexes in the randomized controlled trials seemed to have significant effects in osteoarthritis, 14,15 but the potential of individualized homeopathy remained untested so far. Hence, based on small to moderate effect sizes for the wide range of symptomatic treatments, conventional medicine in a personalized approach remains the mainstay of treatment. 16,17 Further high-quality studies are warranted to elucidate the efficacy and side effects of complementary and alternative medicine methods. 2
Keeping in view the necessity and the large number of patients with rheumatologic conditions, especially osteoarthritis, being reported in the outpatient clinic of Mahesh Bhattacharyya Homeopathic Medical College and Hospital, 18–20 Howrah, West Bengal, India, a pilot randomized controlled trial was undertaken to evaluate the feasibility of a larger efficacy study of individualized homeopathy in knee osteoarthritis in near future.
Materials and Methods
The study was prospective, parallel arm, double blind, randomized, and placebo controlled, and it was conducted from January to October 2014. Patients were recruited from the outpatient clinic of Mahesh Bhattacharyya Homeopathic Medical College and Hospital. A specific protocol conforming to the declaration of Helsinki and good clinical practice in India was designed for the study. The institutional ethics committee approved the study protocol. Before participation in the study, a patient information sheet written in local vernacular Bengali was provided to all patients, and written consent was taken from each.
On account of absence of any randomized controlled study of individualized homeopathy in osteoarthritis, a formal sample size calculation was not possible. We aimed to achieve a target sample of 60 within the stipulated time frame.
As per specified eligibility criteria, patients underwent preliminary and detailed screening (Table 1). Following recruitment, selection of single individualized medicine was based on the presenting symptom totality, repertorization and consultation with materia medica, and individualized dose on the judgment of susceptibility of the patients. Overall decision making was influenced by consensus among 2 physicians (M.K. and S.G.). Subsequent prescriptions were generated according to Kent’s observations and second prescription 21 and the Patient Response Assessment Tool after Homeopathic Treatment. 22
Eligibility Criteria for the Study.

Intervention/comparator was allocated per randomization chart generated by using the StatTrek random number generator. Equal numbers of patients were randomized to code 1 or code 2 (either of verum/homeopathy or control/placebo) of which the treating physician and the patients were kept unaware (blinded/masked) all through the study. Confidentiality of random-number generation and code allocation was maintained strictly by S.S., who was not allowed to influence the study in any ways. The randomization chart was available only with the pharmacist, who was responsible for dispensing of placebo/medicine to the patients according to the chart. Unblinding or breaking of the randomization codes was done after the study had been completed and the database was frozen (Figure 1).
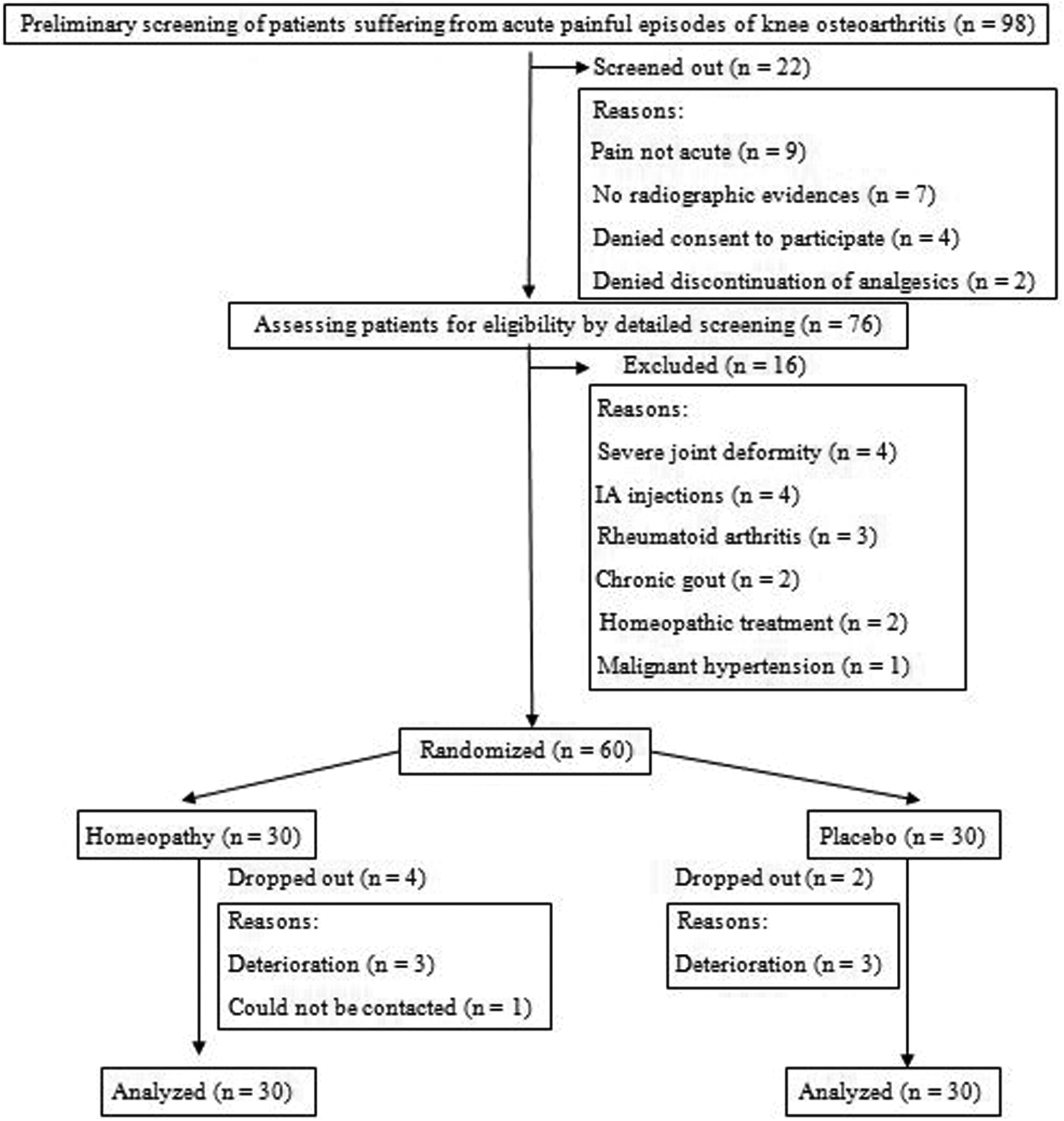
Study flow diagram. Abbreviation: IA, intra-articular.
The outcome measures were chosen per the Cochrane recommendation. 23 Primary outcome measures were visual analog scales (VASs; 0-100 mm) for pain, stiffness, and limitation of physical function, measured at baseline and after weeks 1 and 2, as well as the Osteoarthritis Research Society International osteoarthritis intermittent/constant pain measure at baseline and after second week. Throughout the study, adverse events or serious adverse events, lack of efficiency, and deaths were planned to be reported. Study end point was lowering of global assessment pain (VAS) by 5.1 to 13.3 mm (minimal clinically important difference 24 ; “responders”). Safety end point was any adverse event during the study.
Symptoms based on the homeopathic medicines prescribed and, in the subsequent visits, the changes in these symptoms were mentioned clearly. Homeopathic medicines, dilution method, potency, pharmaceutical form, and dose were also recorded. All medicines were procured from a GMP-certified firm: SBL Pvt Ltd. Each dose was directed to be taken orally on clean tongue, consisted of 4 cane sugar globules of size 30, and moistened with a single drop of indicated medicine prepared and preserved with 88% v/v ethanol. Repetition was done depending on the intensity of the complaints. The patient might report on follow-up days as mentioned above and by telephone as per need.
Reporting adhered to the “reporting data on homeopathic treatments” guidelines 25 and criteria for reporting individualization in homeopathy. 26
Statistical analyses were performed using computational websites. The intention-to-treat population (n = 60) was analyzed in the end. Missing values were computed by last value carried forward. Outcome values at different points of time were compared with paired t test, followed by post hoc repeated-measure analysis of variance. Due to increased likelihood of type I (alpha) error on account of multiple testing, Bonferroni-Holm correction was made to set the significance level at P < .01, 2-tailed. Group differences were tested with independent t test, keeping significance level set at P < .05, 2-tailed.
Results
A total of 98 patients were preliminarily screened, of which 22 (22.4%) were screened out; 76 (77.6%) underwent detailed screening per the inclusion and exclusion criteria (Table 1); 16 (21.1%) were excluded, and 60 (78.9%) were enrolled out. Six patients (10%) dropped out, and 54 (90%) completed the study. Intention-to-treat population (n = 60; 100%) was analyzed in the end. When Kolmogorov-Smirnov test was carried out, no significant departure from normality was found (Figure 1).
Baseline demographics, dropout rates, and frequency of placebo prescriptions were comparable between the groups (P > .05, 2-tailed). Outcomes were also comparable (P > .05, 2-tailed), except stiffness VAS (t = 2.089, P = .041, 2-tailed, independent t test; Table 2).
Comparing Baseline Demographics, Outcomes, Dropouts and Prescriptions Between Groups.a

Abbreviation: OARSI, Osteoarthritis Research Society International.
aContinuous values presented as mean ± SD (all others as No.). Data were compared between groups with Pearson chi-square and independent t tests for categorical and continuous variables, respectively. Fisher exact P values were taken if any cell value became <5 in 2 × 2 contingency tables.
b P < .05, 2-tailed, considered as statistically significant.
Following homeopathic intervention, over 2 weeks, reduction of pain VASs (–15.1; 95% CI, –45.3, 15.1; P < .0001, 2-tailed, paired t test), stiffness VASs (–17.2; 95% CI, –62.5, 28.1; P = .0004), loss of function VASs (–13.4; 95% CI, –47.9, 21.1; P = .0003), and Osteoarthritis Research Society International scores (–3.2; 95% CI, –11.0, 4.6; P < .0001) were significant after adjustment of baseline differences (Table 3). Post hoc repeated-measure analysis of variance also revealed significance of lowering of the VAS outcomes: pain (F 2,58 = 15.719; P < .01), stiffness (F 2,58 = 10.567; P < .01), and loss of function (F 2,58 = 10.601; P < .01).
Changes in Outcome Measures Over Time in the 2 Groups.

Abbreviation: OARSI, Osteoarthritis Research Society International.
a P < .01, 2-tailed compared with the baseline.
The placebo group, over 2 weeks, showed significant reduction in pain VAS (–10.8; 95% CI, –36.7, 15.1; P = .0001), loss of function VAS (–8.5; 95% CI, –34.2, 17.2; P = .001), and Osteoarthritis Research Society International score (–3.5; 95% CI, –9.2, 2.2; P < .0001); however, a decrease in stiffness VAS could not achieve significance (–3.5; 95% CI, –23.9, 16.9; P = .079; Table 3). Still, post hoc repeated-measure analysis of variance revealed significance of lowering of all the outcomes: pain VAS (F 2,58 = 11.248; P < .01), stiffness VAS (F 2,58 = 1.618; P < .01), and loss of function VAS (F 2,58 = 8.188; P < .01).
Although significant reductions were achieved in all the outcomes across the 2 groups, group differences were not significant (P > .05, 2-tailed) in any occasion. Among the 54 complete case records, 22 (84.6%) were responders in the homeopathy group and 20 (71.4%) in the placebo group (P = .332, 2-tailed, Fisher exact test), as defined by minimal clinically important difference.
Between the homeopathy and placebo groups, Bryonia alba (23.2% and 22.4%), Rhus toxicodendron (14.3% and 20.7%), Calcarea carbonica (8.9% and 3.4%), Arnica montana (7.1% and 3.4%), and Natrum muriaticum (5.4% and 5.2%) were the most frequently prescribed medicines, and the frequencies were comparable between groups as well (P > .05, 2-tailed; Table 4).
Five Most Frequently Prescribed Medicines, No. (%).

aPearson chi-square test (Yates corrected) or Fisher test. P < .05, 2-tailed considered significant.
Homeopathic therapy was found to be safe throughout the study period as no deaths or (serious) adverse events were reported across the 2 groups.
Discussion
While acute painful episodes of knee osteoarthritis were being treated, statistically significant reductions of pain, stiffness, and loss of function VAS scores and Osteoarthritis Research Society International scores were elicited in both homeopathy and placebo groups; however, group differences were nonsignificant in every occasion. In conclusion, homeopathy did not appear to be superior to placebo; still, further rigorous evaluation in this design involving a larger sample size seems feasible in future.
The was a short-term, parallel-arm, double-blind, randomized controlled pilot study with a limited number of patients. One serious limitation may be that we depended only on patient-assessed subjective outcomes. Another limitation was that the stiffness VAS scores varied significantly at baseline between the 2 groups, even after running an analysis of covariance adjusting for covariates (age, sex, height, and weight; F 1,57 = 4.32; P = .042). Still, double blindness and randomization were the strengths to keep the intervention exactly the same in the contrasted groups and to ensure unbiased assessment of the effects. When prognostic factors of the illness other than the intervention under study are insufficiently known, random allocation to the contrasted treatments is useful to ensure comparable prognoses. However, 10% of dropouts from a small recruited sample makes interpretation of the study results tricky and less likely to be generalizable.
Serious piloting prior to the conducting of any large multicentric randomized controlled trial is always a prerequisite. 27 This pilot study inevitably raised the feasibility issues. Further randomized controlled trials seem feasible in future, but the high rate of dropouts should be a matter of concern that should be reflected in the sample size calculation by keeping provision for at least 20% dropouts. The follow-up duration may be increased to 2 to 3 months, as in a few occasions, “chronic”/“polychrest” remedies were prescribed due to indistinct “characteristic symptoms” indicating a single medicine or following inadequate results obtained after prescribing on “acute totality.” Recruitment and follow-up took only 10 months in the trial site, indicating ease and less time consumption for successful accomplishment of future randomized controlled trials. Fifty millesimal potencies may be thought of to encounter the disease condition under question. The cost incurred in the project seemed to be minimal. To validate the findings further, study of biochemical markers (eg, matrix metalloproteinase) and calcium pyrophosphate dihydrate crystals in joint fluid; analysis of messenger RNA expression of proinflammatory cytokines, interleukins, and catabolic enzymes; and ultrasonographic and magnetic resonance imaging studies detecting synovial fluid and bone changes may be taken into consideration.
Finally, it can be concluded that although evidences seemed to be inadequate in support of efficacy of individualized homeopathy over placebo in acute painful episodes of knee osteoarthritis, larger randomized controlled trials in a multicenter design seem quite feasible in near future, especially in the context that no work concerning the subject matter has been undertaken to date.
Footnotes
Acknowledgments
The authors acknowledge Prof (Dr) Amitava Biswas, Principal, Mahesh Bhattacharyya Homeopathic Medical College and Hospital, for allowing us to carry out the study successfully in his institution. The authors also remain grateful to the patients for their participation in the study.
Author Contributions
MK, SS: concept, design, literature search, data interpretation, statistical analysis, preparation of the article. SG: clinical study, data acquisition. All the authors edited, reviewed, and approved the final article.
Author Notes
List of abbreviated names of the all authors for bibliographic citation when the manuscript is published: Koley M, Saha S, Ghosh S.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Approval was obtained from the Institutional Ethics Committee of Mahesh Bhattacharyya Homeopathic Medical College & Hospital prior to the initiation of the study (No. 47/Mahesh Bhattacharyya Homeopathic Medical College and Hospital/CH/PRIN/ADM; date, 12.02.2013). The study is registered with the Clinical Trials Registry, India (CTRI/2014/05/004589).
