Abstract
Objectives
To determine the efficacy of zinc sulfate supplementation in managing dysmenorrhoea.
Methods
In total, 103 high school students were randomised into an experimental arm (52 students) and a control arm (51 students) and received 40-mg zinc sulfate or placebo, respectively, over three cycles. Primary outcome measures were the mean Visual Analogue Scale score, which measured pain over three cycles, and the frequency of nausea and vomiting. Secondary outcomes were the use of additional analgesics and the frequency of allergic reactions.
Results
Fifty participants were analysed in each group. Mean pain scores were not significantly different between the groups before administering zinc sulfate therapy. Following the intervention, the mean pain scores for the treatment (2.80 ± 2.28) and placebo (3.48 ± 2.85) groups were not significantly different in the first cycle; however, scores in the treatment group were significantly better in the second (2.56 ± 1.97 vs 3.80 ± 2.77) and third (1.95 ± 1.72 vs 3.95 ± 2.82) cycles. No significant differences were observed between the groups in the nausea and vomiting incidence and the requirement for additional analgesics.
Conclusions
Zinc sulfate reduces dysmenorrhoea severity with minimal or no adverse effects, especially with more than one cycle of usage.
Trial Registration Number: PACTR202105843292338. The trial is publicly available and was registered at www.pactr.org on 25 May 2021.
Introduction
During the early days of their menses, healthy young women typically experience lower abdominal pain of varying severity that can affect daily activities and productivity and—at the population level—can potentially lead to a loss of revenues globally. Primary dysmenorrhoea is defined as recurrent crampy pain in the lower abdomen at the onset of menstruation in the absence of identifiable pelvic disease. 1 Dysmenorrhoea commences in adolescence after the establishment of ovulatory cycles, and the absence of pelvic disease distinguishes primary from secondary dysmenorrhoea. 2 Primary dysmenorrhoea is common in adolescent females and remains a leading cause of pelvic pain in adolescents.1–5
The prevalence of primary dysmenorrhoea is highly variable and ranges from 20% to 90% globally.6–9 A study of Japanese women of reproductive age showed a prevalence rate of 15.8%. 10 A multicentre study in an urban Swedish population revealed that up to 72% of adolescents had primary dysmenorrhoea. 11 Proctor et al. acknowledged the difficulty in estimating the prevalence of primary dysmenorrhoea, citing estimates ranging from 45% to 95%. 12 A prevalence rate of 71.8% has been reported in Nigeria. 13
Primary dysmenorrhea occurs as a result of the increased production and secretion of prostaglandins (PGF2ᾳ and PGE2) in the uterus during endometrial sloughing at the initiation of menstruation. These prostaglandins cause myometrial contraction, vasoconstriction, ischemia and stimulation of pain fibres leading to pelvic pain.1,12 Women who have severe forms of dysmenorrhoea have higher levels of prostaglandins in menstrual fluid than women with less severe dysmenorrhoea; prostaglandin levels are highest during the first two days of menstruation. 12 Inhibition of prostaglandin synthesis has been shown to decrease the severity of dysmenorrhoea. 14
Primary dysmenorrhoea is treated with nonsteroidal anti-inflammatory drugs (NSAIDs) as the first-line option.2,3,15 One particular NSAID has not been proven to be superior to another; the choice of NSAID is based on cost, effectiveness, tolerability and convenience. 2 Medication is usually initiated a day or two before the onset of menstrual flow and then continued for up to 2 to 3 days.2,3,15 Given that NSAIDs are administered on a long-term basis for the management of primary dysmenorrhoea, counselling patients on possible NSAID adverse effects including gastrointestinal intolerance, headaches and drowsiness is important.
Zinc supplementation in the management of primary dysmenorrhoea has evolved as a useful intervention strategy as consumers seek useful adjuncts to NSAIDs or effective and more inexpensive alternatives to NSAIDs with considerably fewer long-term side effects.14,16–19 Zinc is shown to decrease and regulate levels of the cyclooxygenase enzyme, increase microcirculation to avert ischemia and inactivate free oxygen radicals by increasing levels of the dismutase enzyme and downregulating inflammatory cytokines.20,21 Therefore, zinc supplementation may reduce the need for chronic NSAID use and the associated side effects in the management of primary dysmenorrhoea.
Primary dysmenorrhoea has considerable medical, social and economic costs, and identifying a well-tolerated and effective treatment with considerably fewer side effects than NSAIDs is critical. This double-blind, randomized, placebo-controlled study evaluated the efficacy of zinc sulfate supplementation in the treatment of primary dysmenorrhoea in female senior secondary students in Alor, South East Nigeria.
Methods
Study setting
This study was conducted at Alor Girls Secondary School, Alor, Idemili South Local Government, Anambra State, Nigeria from 1 June 2021 to 1 October 2021.
Study population
The study population comprised consenting senior secondary students aged 13 to 21 years with primary dysmenorrhoea.
Inclusion criteria
The study included young women between the ages of 13 and 21 years who had regular menses (every 28 ± 7 days), similar diets, a history of dysmenorrhea for at least 1 day per month and the ability to score pain using a 10-cm Visual Analogue Scale (VAS).
Exclusion criteria
The study excluded students who had other gynaecological diseases, secondary dysmenorrhoea, a history of chronic or systemic disease and a history of adverse reactions to zinc. Further excluded were students who used hormonal medications or contraceptives and those who refused to participate in the study or whose parents declined to provide consent.
Participant recruitment
Participants were selected from a pilot study that identified individuals with primary dysmenorrhoea.
Intervention
The experimental arm consisted of 52 students who received 40 mg of zinc sulfate and the control arm consisted of 51 students who received placebo alone over three cycles.
Study design
This was a double-blind, randomized, placebo-controlled clinical study. Recruited participants were randomised into two arms—the zinc sulfate supplementation and placebo arms—using a random number table. The randomisation sequence was computer-generated using randomly permuted blocks (blocks of 4 with a 1:1 allocation ratio) from software available online (http://www.randomization.com). Randomisation was performed by a third party who was not involved in the study. Information regarding the study medications was written on a piece of paper and placed in sequentially numbered sealed opaque envelopes. Specifically, each envelope contained a folded slip of paper displaying zinc sulfate—which placed the patient into the treatment group (Group A; 40-mg tablet of zinc sulfate)—or placebo—which placed the patient in the placebo group (Group B, receiving lactose-containing placebo). The envelopes were stored and opened by an independent third party. Participant allocation was not changed after the envelopes were unsealed. The medications were prepared by Chi Pharmaceutical Company (Lagos, Nigeria) and were indistinguishable from zinc sulfate tablets in shape, size and colour, ensuring the blinding of the participants. The study was approved by the ethics committee of Nnamdi Azikiwe University Teaching Hospital Nnewi, Nigeria on 28 November 2020 (NAUTH/CS/66/VOL.13/VER/173/2018/109).
Sample size determination
A minimum sample size of 92 participants (46 per arm) was required in a study by Zekavat et al. 18 that compared the effects of zinc sulfate and placebo on pain severity and duration in adolescent girls with primary dysmenorrhoea. The study reported mean pain severity scores of 6.58 and 4.23 in the control and intervention groups, respectively, and had 90% power, a 95% confidence interval, a standard deviation of 1.7 and a 40% dropout rate. The sample size had 97% power to detect a 25% difference in the rate of nausea and vomiting between the intervention and control arms.
Study procedure
All of the participants were informed about the study and participants provided written informed consent. Consent was obtained from parents at parent–teacher association meetings for participants younger than 18 years of age. After participants were randomised into the two groups, those in Group A received a 40-mg tablet of zinc sulfate and those in Group B received the inactive placebo containing lactose. All of the participants took the medications for 3 days from the day of commencement of menstruation to the third day of menstrual bleeding. To record compliance with the trial drugs, the participants were contacted daily by telephone throughout the duration of menstruation. Where possible, the participants were encouraged to record any adverse events in a document that was reviewed at each follow-up visit, and the participants were explicitly queried about these events in each interview. Where possible, drug compliance was verified during the follow-up visit by checking the used drug packets. The participants took the medication at approximately 8:00 in the morning. Medications were administered by hostel matrons and form teachers who had been appropriately trained in the procedure. Menstrual pain perception was recorded using the VAS at the end of each school day (i.e., at approximately 17:00 hours) after the participant had taken the medication. The VAS was explained to the participants by the research assistant before the study. The participants were asked to mark a line to indicate pain intensity, with 0 indicating no pain and 10—the highest score on the VAS—indicating the worst possible pain, which was labelled “the worst pain imaginable” or “pain as bad as it can be”. The pictorial and easy-to-use VAS was self-administered by the participants. VAS scores between 1 and 4 indicated mild menstrual pain; moderate and severe pain was indicated by VAS scores of 5 to 10. Recordings were collected at the end of every menstrual cycle and the results were collated for analysis at the end of three menstrual cycles. To avoid bias, the outcome was assessed by the researcher, who was blinded to the medications received; participants were also blinded. All adverse effects including nausea and vomiting, pruritus, allergy or other abnormal findings were recorded.
The primary outcomes were the mean VAS score of the three menstrual cycles and the proportion of participants with nausea and vomiting. The secondary outcomes were the use of additional analgesics and the frequency of allergic reactions.
Data analysis was conducted using SPSS version 23 (IBM Corp., Armonk, NY, USA). Depending on the normality of distribution, the independent t-test or the Mann–Whitney U-test was used to compare the mean (± standard deviation) or median (± interquartile range) of continuous variables between the groups The Shapiro–Wilk test was used to test for normally or non-normally distributed continuous variables. Data were presented in tables and charts, and continuous data were presented as mean and standard deviation or median (± interquartile range). Categorical variables were analysed using chi-square tests where appropriate, whereas continuous data, if parametric, were analysed using t-test. The mean pain score was evaluated for each cycle using the paired t-test. A p-value ≤ 0.05 was considered statistically significant. The reporting of this study adhered to the CONSORT guidelines. 22
Results
Of the 251 participants who were assessed for eligibility, 40 declined to participate in the study, 80 did not meet the inclusion criteria and 28 were excluded for other reasons. Ultimately, 103 participants were randomised into the zinc sulfate (n = 52) and placebo (n = 51) groups. Three of the participants (n = 2 in the zinc sulfate group; n = 1 in the placebo group) were ultimately not analysed because they discontinued the intervention. A diagram describing participant flow through the study is shown in Figure 1.

CONSORT flow chart.
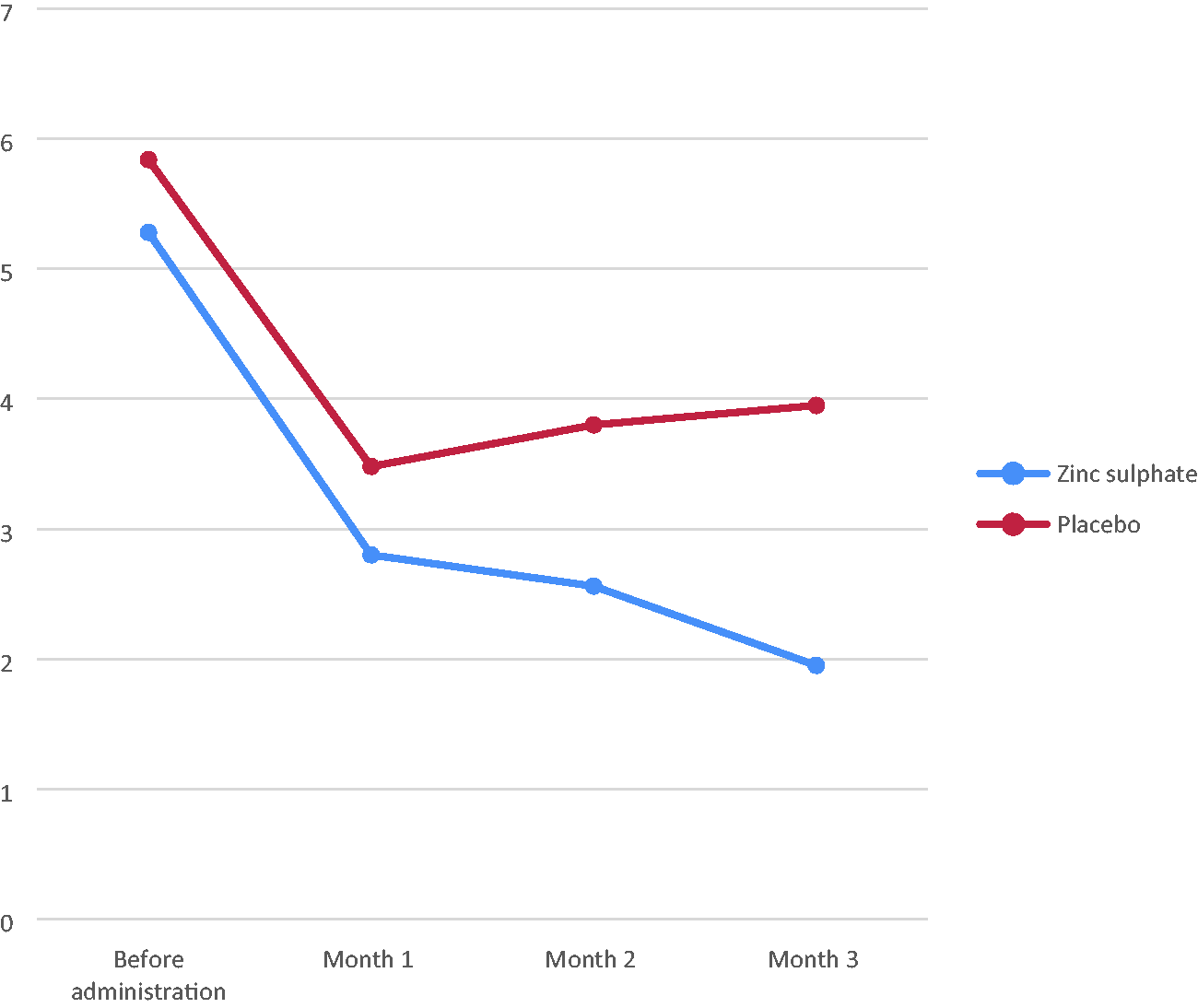
Line graph showing the distribution of mean pain scores in the zinc sulfate and placebo groups.
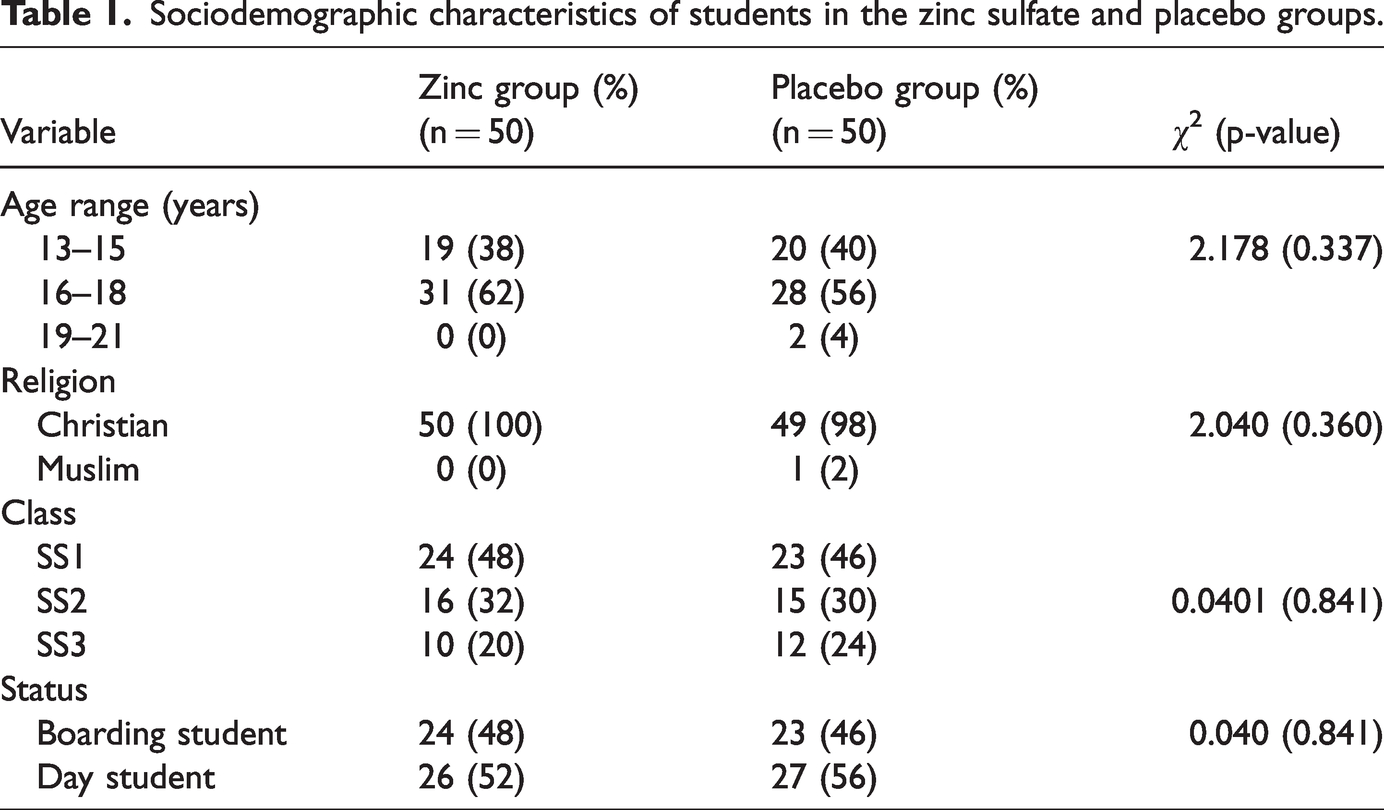
Table 1 summarises the socio-demographic characteristics of both groups. No significant differences were observed in the baseline sociodemographic and clinical characteristics between the groups. Table 2 shows the baseline (i.e., pre-intervention) therapies of the students in the zinc sulfate and placebo groups. Behavioural therapy was a popular modality in the management of primary dysmenorrhoea, with 48% and 44% adopting this therapeutic option before study interventions in the zinc sulfate and placebo groups, respectively. Postural modifications, such as lying down on one side of the body, were adopted before study interventions by 100% and 77.27% of participants who used behavioural therapy in the zinc sulfate and placebo groups, respectively. Avoiding sugary foods was a pre-intervention behavioural strategy in 54.17% and 91.30% of the study population that practised behavioural therapy in the zinc sulfate and placebo groups, respectively, and breathing exercises were used pre-intervention in 4.17% and 13.64% of those who used behavioural therapy in the treatment and placebo groups, respectively.
Sociodemographic characteristics of students in the zinc sulfate and placebo groups.
Baseline therapies of students in the zinc sulfate and placebo groups.

# Some participants chose more than one option.
Participants’ opinion about whether a safe drug could improve their academic performance is shown in Table 3. Overall, 40% of all participants believed that a new drug with an insignificant side effect profile could improve their academic performance. Most of the participants—44% and 62% in the zinc sulfate and placebo groups, respectively—were undecided, whereas 20% and 24% in the treatment and placebo groups, respectively, asserted that they would not experience an improvement in their academic performance regardless of the availability of an effective drug therapy with a favourable side-effect profile.
Participant opinion about the role of a safe drug in the improvement of academic performance.

As shown in Table 4, the baseline (i.e., pre-administration) mean pain scores were 5.28 ± 0.95 and 5.24 ± 1.51 for the treatment and placebo groups, respectively (p = 0.874). Following the intervention, the mean pain scores for the zinc sulfate (2.80 ± 2.28) and placebo (3.48 ± 2.85) groups were not significantly different in the first cycle (p = 0.205) but were significantly different in the second (2.56 ± 1.97 vs 3.80 ± 2.77; p = 0.014) and third (1.95 ± 1.72 vs 3.95 ± 2.82; p = 0.001) cycles of intervention.
Mean pain severity scores before and after intervention in the zinc sulfate and placebo groups.

STD: standard deviation.
Table 5 illustrates the frequency of adverse effects and the need for additional therapy among study participants following the intervention. The combined incidence of nausea and vomiting was not statistically different between the groups (2% vs 6%; p = 0.307). Allergic reactions were not reported in either group. Although more participants in the placebo group than in the treatment group required the use of additional analgesics, the difference was not significantly different (p = 0.645).
Adverse effects and need for additional therapy among study participants following intervention.

Tables 6 and 7 show the pre-intervention and post-intervention effects, respectively, of menstrual pain on the academic activities of senior secondary school students. Pre-intervention, 36% and 44% of students in the zinc sulfate and placebo groups, respectively, agreed that menstrual pain affected their academic activities (p = 0.932). However, post-intervention, 12% and 44% of participants in the zinc sulfate and placebo groups, respectively, agreed that menstrual pain affected their academic activities (p < 0.001).
Pre-intervention effect of menstrual pain on the academic performance of senior secondary school students.

Post-intervention effect of menstrual pain on the academic performance of senior secondary school students.

Discussion
The mean age of the study population was 15.89 ± 1.54 years, comparable to the findings of Kashefi et al. (15.2 ± 1.7 years) and Zekavat et al. (17 ± 4.3 years) in their studies of Iranian high school students.16,18 As expected, these findings differ from those of studies conducted on university students or medical staff. Sangestani et al. 23 observed a mean age of 21.5 ± 2.5 years in 2015, whereas Teimoori et al. in Iran 2016 and Farrah et al. in Bali in 2017 reported similar mean ages of 21.4 ± 2.2 years and 22.95 ± 1.33 years, respectively, in university students.14,19 The higher age range for university students is explained by the higher age of university students compared with high school students.
Zinc sulfate for primary dysmenorrhoea was shown to be effective and superior to placebo. Before the administration of zinc sulfate therapy, the mean pain score for the study group was 5.28 ± 0.95. Following zinc sulfate therapy, the mean pain score dropped to 2.80 ± 2.28, 2.56 ± 1.97 and 1.95 ± 1.72 in the first, second and last cycles, respectively. This shows effective and sustained reduction in pain from primary dysmenorrhea in the zinc group. The mean VAS score in the control group was 3.48 ± 2.85 in the first month, increasing to 3.80 ± 2.77 in the second month and 3.95 ± 2.82 in the third month. The reduction in the mean pain scores in the first month was not sustained and was not concurrent with the time period in which participants received placebo. Prior to intervention in both groups, no significant difference was observed in the mean pain severity score between the two groups (p > 0.05). However, following intervention, the zinc group had significantly lower mean pain severity scores in the second (p < 0.01) and third (p < 0.01) months compared with the placebo group. This result is similar to Zekavat et al.’s finding of no significant difference in pain severity between the zinc and placebo groups in the first month (p = 0.497). Furthermore, pain severity and duration in the zinc group were significantly lower than in the placebo group in the second and third months (P < 0.001). Similarly, Farrah et al. reported that the mean pain score decreased from 4.92 ± 1.80 before zinc supplementation to 2.7 ± 2.03 over three cycles, a statistically significant reduction. 19 Teimoori et al. further observed a statistically significant reduction in the pain score from 5.3 ± 1.8 to 1.2 ± 1.9 (p < 0.001) with the administration of zinc and mefenamic acid following one cycle of therapy compared with mefenamic acid and placebo alone, which was associated with a mean pain score reduction from 5.8 ± 2.1 before therapy to 2.9 ± 1.7 (p < 0.001) after one cycle of therapy. The difference in pain levels before and after treatment were 4.1 ± 2.8 and 2.9 ± 1.7(p > 0.050) in the intervention and control groups, respectively. 14 Additionally, although more participants in the placebo group required additional analgesics, the use of this therapy class was not significantly different versus the treatment group (p = 0.45). In a 2015 study of zinc intervention versus placebo over one cycle, Sangestani et al. in Iran showed that zinc supplementation reduced the severity of dysmenorrhoea and menstrual bleeding compared with placebo (p < 0.001). 23 However, unlike the previously cited studies, this was a single-cycle study.
Allergic reactions were not reported, and the combined incidence of nausea and vomiting (2% vs 6%; p = 0.307) was not statistically different across the treatment and placebo groups. The absence of undue side effects is an important advantage of zinc sulfate therapy for dysmenorrhoea, making this therapy safe and readily acceptable by patients for the treatment of the condition—especially in resource-poor settings. Most of the works cited in our literature review did not report side effects with zinc sulfate therapy.21,23–25
The impact of primary dysmenorrhoea on academic activities was also evaluated. Overall, 40% of participants agreed—and 10% strongly agreed—that primary dysmenorrhoea adversely affects their academic performance. This finding was similar to that of Orhan et al.’s study of 471 university students in Turkey which established the negative influence of primary dysmenorrhoea on academic performance. 24 Despite assessing university or medical students, most of the studies we cite did not evaluate the impact of primary dysmenorrhoea on participants’ academic activities. Evaluating the effect of dysmenorrhoea on academic performance is challenging because assessing exam results based solely on the effects of dysmenorrhoea cannot sufficiently rule out other confounders such as individual difficulty with various subjects and the timing of tests/examinations vis-à-vis the onset of menses. Furthermore, the degree of improvement or decline in results that can be considered statistically significant is difficult to determine. However, given that most of the studies reviewed concluded that dysmenorrhoea affects the quality of daily living of which academic activities are an important part, we can extrapolate that dysmenorrhoea affects the academic activities of students by affecting their quality of life. The extent of this effect remains unknown and is difficult to appropriately quantify.
Incidental findings from this study suggest that behavioural therapies are important non-pharmacological approaches in the management of primary dysmenorrhoea. Behavioural therapies are readily available and focus on psychological and physical coping mechanisms rather than drug usage for pain management. Behavioural therapy was a popular modality in the management of primary dysmenorrhoea, whereas herbal therapy was the least employed in both groups. Postural modification such as lying down on one side of the body was the most common behavioural therapy among the zinc group, whereas avoiding sugary food was the most common measure in the placebo group. This result may be related with our finding that most of the study population believed that behavioural therapies provided some symptomatic improvement, with significant percentages in each group experiencing a moderate improvement in symptoms. Overall, 54.17% and 81.82% of the zinc sulfate and placebo groups, respectively, experienced mild improvement with behavioural therapies. In their analysis of findings from five trials, Proctor et al. in 2007 concluded that some evidence supports that behavioural interventions are effective in dysmenorrhoea; however, the authors asserted that results should be interpreted with caution given small trial sizes, poor methodology and inconsistencies in data reporting. 25
The clinical implications of our findings have widespread ramifications for adolescent gynaecological health in Nigeria and the West African sub-region. Adolescent gynaecology tends to be neglected in the national discourse and is not typically allocated funding in the national and state budgets despite the pivotal and growing adolescent segment of the population. Zinc sulfate reduces the incidence and severity of primary dysmenorrhoea—especially after more than one cycle of usage—with negligible side effects. Furthermore, the 3-day regimen used in this study is significantly less expensive than a three-day course of NSAIDs. Finally, the negative impact of dysmenorrhea on academic activities, which may be alleviated with zinc sulfate therapy, is a neglected and understudied phenomenon.
A study strength is the double-blind randomised placebo-controlled study design, which reduces bias from participants and outcome assessors. Furthermore, our study adds to the literature on the role of zinc sulfate in primary dysmenorrhea, an area in which studies are limited. However, a study limitation is the inability of researchers to directly observe the participants taking the medications given the peculiarities of the study population (most of the participants were adolescents) and the considerable associated financial outlay. Additionally, pelvic ultrasound evaluation was not performed to rule out other organic causes of dysmenorrhea. Finally, in our power analysis, we could not use a mixed model for within-subject effects with a time-point variable. However, within a subjects design, one participant provides multiple data points that can correlate with one another because they are associated with the same participant, thus allowing the measurement of systematic item-level variability within participants and participant-level variability within the study groups. Therefore, we acknowledge the error in planning the statistical analysis, given that the mixed model analysis was only conducted for the mean severity score at baseline and 1, 2 and 3 months post-intervention, limiting the strength of the evidence.
Conclusion
Zinc sulfate reduces the incidence and severity of primary dysmenorrhoea—especially after more than one cycle of usage—with minimal or no adverse effects. The academic performance of participants who received zinc sulfate appears to have improved compared with that of participants who received placebo.
Research Data
Research Data for Efficacy of zinc supplementation for the treatment of dysmenorrhoea: a double-blind randomised controlled trial
Research Data for Efficacy of zinc supplementation for the treatment of dysmenorrhoea: a double-blind randomised controlled trial by Hillary I. Obiagwu, George U. Eleje, Nworah J.A. Obiechina, Betrand O. Nwosu, Gerald O. Udigwe, Joseph I. Ikechebelu, Joseph O Ugboaja, Chukwuemeka C. Okoro, Ifeanyi O. Okonkwo, Ayodele O. Okwuosa, Charlotte B. Oguejiofor, Chisom G. Chigbo, Chukwudi A. Ogabido, Chidinma Charity Okafor and Chigozie G. Okafor in Journal of International Medical Research
Footnotes
Acknowledgements
The authors are grateful to all of the students who participated in the study, Alor Girls Secondary School and the staff involved in the study.
Author contributions
Hillary I. Obiagwu was involved in conceptualisation/design, manuscript writing & revision and data collection. George U. Eleje and Nworah J.A Obiechina were involved in supervision, manuscript writing and revision. Joseph I. Ikechebelu, Bertrand O. Nwosu, Gerald O. Udigwe, Chigozie G. Okafor, Joseph O. Ugboaja, Chukwuemeka C. Okoro, Ifeanyi O. Okonkwo, Ayodele O. Okwuosa, Charlotte B. Oguejiofor, Chisom G. Chigbo, Chukwudi A. Ogabido and Chidinma C. Okafor were involved in manuscript writing, revision, data collection and analysis. All of the authors contributed to the final revision and approved the manuscript for publication.
Data availability statement
Research data are available upon request from the authors.
Declaration of conflicting interest
All of the authors declare that they have no conflicts of interest. None of the authors received financial support from Chi Pharmaceutical Company (Lagos, Nigeria) or any other company whose product was mentioned in this study. The views and opinions expressed in this study are those of the authors and do not necessarily reflect those of any company whose product was discussed or mentioned in this study.
Funding
The authors declare that no external funding was received for this study. The principal researcher bore the cost of the drugs.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
