Abstract
Hepatotoxicity associated with isoniazid and rifampicin is one of the major impediments in antituberculosis therapy. The present study explored the prophylactic and therapeutic efficacies of Spirulina maxima in isoniazid and rifampicin induced hepatic damage in a rat model. Hepatic damage induced in Wistar rats by isoniazid and rifampicin resulted in significant alterations in biomarkers of liver function, namely, bilirubin, aspartate transaminase, alanine transaminase, alkaline phosphatase, and oxidative stress markers such as superoxide dismutase, catalase, glutathione, and thiobarbituric acid reactive substances. Co-administration of Spirulina maxima along with antituberculosis drugs protected liver from hepatotoxicity due to isoniazid and rifampicin. Administration of Spirulina maxima consecutively for 2 weeks to hepatodamaged animals resulted in restoration of hepatic function as evident from normalization of serum markers of liver function. Thus, the present study revealed remarkable prophylactic and therapeutic potential of Spirulina maxima. Co-administration of Spirulina maxima and antituberculosis drugs is advantageous as it provides extra nutritional benefit.
Introduction
Tuberculosis is a common problem in India and worldwide, especially after the recent increase in incidence of acquired immunodeficiency syndrome, and it is the leading cause for adult deaths in the tropical world. 1 Isoniazid and rifampicin are the first-line antituberculosis drugs and are associated with hepatotoxicity. 2 Isoniazid is directly or indirectly metabolized to acetyl hydrazine and hydrazine by N-acetyltransferase and amidohyrolase and these isoniazid metabolites have been implicated as the causative hepatotoxins. 3 Oxidative activation of these metabolites in liver by cytochrome P-450 monooxygenases generates electrophilic intermediates and free radicals that cause damage to the hepatic tissue. 4 Cytochrome P-450 2E1 is responsible for the biotransformation of therapeutic agents and endogenous compounds such as fatty acids and ketone bodies. Cytochrome P-450 2E1–mediated metabolism of drugs may be responsible for generation of reactive oxygen species, 5 with resultant lipid peroxidation and DNA strand breaks. 6 Rifampicin, an antibiotic used routinely for tuberculosis chemotherapy, is documented to be a potent hepatotoxicant. 7 Incidence of hepatotoxicity increases significantly when it is combined with rifampicin, 8 and higher risk of hepatotoxicity has been reported in Indian tuberculosis patients than in their Western counterparts. 9 Several studies have shown that rifampicin causes oxidative injury of liver, its membrane, and organelles, leading to lipid peroxidation and depletion of glutathione and the free radical scavenging enzymes. 10 Since long-term use of these drugs causes liver damage, supplementation with hepatoprotective drugs is highly desired to protect from hepatic damage caused by tuberculosis drugs.
Spirulina maxima, a filamentous alga, is a cyanobacterium belonging to the family Oscillatoriaceae, which blooms well in alkaline water, and contains a unique blend of nutrients that no single source can provide. 11 The alga is characterized by high protein content (60% to 70%) and a wide spectrum of nutrients that include B-complex vitamins, minerals, γ-linolenic acid and the super antioxidants, β-carotene, vitamin E, trace elements, 12 and other uncharacterized bioactive compounds. Because of its apparent ability to stimulate the immune system, Spirulina has several therapeutic functions such as antidiabetic, anti-inflammatory, antiviral, and anticancer activities. In vitro and in vivo studies also suggest that Spirulina helps in protection against harmful allergic reactions. Silymarin, obtained from Silybum marianum, is one of the most popular herbal remedies for liver disease. 12 The hepatoprotective function of silymarin is attributed to antioxidant effects of its flavanolignan and of other polyphenolic constituents. 13 –15 The present study explored the prophylactic and therapeutic potentials of aqueous extracts of Spirulina maxima in the hepatic damage generated by isoniazid and rifampicin.
Materials and Methods
Chemicals and Drugs
Isoniazid and rifampicin, were purchased from Lupin Research Laboratories (Mumbai, India). Silymarin was purchased from Micro Labs Limited (Bangalore, India). Spirulina maxima, originally obtained from Indian Agricultural Research Institute, New Delhi, is being regularly grown in our laboratory. All other analytical laboratory chemicals, kits, and reagents were purchased from Crest Biosystems (Goa, India), Sigma (St Louis, MO), and SRL Chemicals (Mumbai, India).
Culturing of Spirulina maxima
Routine culture of Spirulina maxima in a photobioreactor is being done in the laboratory since the past 4 years. The cultures were grown in Zarrouk’s medium (1966) at 25°C at a proven flounce rate of 50 μmol m−2 s−1 at the surface of vessel. Cells were harvested, and dried biomass was used to treat animals.
Experimental Design
Experiments were performed on male Wistar rats (with mean body weight of 135 ± 15 g) obtained from the animal house facility of Defense Research and Development Establishment, Gwalior, India. Prior to experiments, rats were acclimatized for 7 days to laboratory conditions, that is, temperature of 25 ± 2°C with regular alternate cycles of 12 hours light and darkness. Rats were maintained on standard pellet diet (Amrit Feeds, Pranav Agro, New Delhi, India) and water ad libitum throughout the experiment. The experiment was carried out for 30 days. Thirty animals were randomly divided into 5 groups of 6 rats each and kept on different regimens as given below:
Group I: Normal control animals maintained on normal diet.
Group II: Animals fed with isoniazid (50 mg/kg, orally) + rifampicin (75 mg/kg, orally) consecutively for 2 weeks and left untreated.
Group III: Animals cofed with isoniazid (50 mg/kg, orally) + rifampicin (75 mg/kg, orally) and Spirulina maxima (500 mg/kg, orally) for 4 weeks.
Group IV: Animals fed with isoniazid + rifampicin consecutively for 2 weeks (as in group II) followed by administration of Spirulina maxima (500 mg/kg, orally) for 2 consecutive weeks.
Group V: Animals fed with isoniazid + rifampicin consecutively for 2 weeks (as in group II) followed by administration of Silymarin (100 mg/kg, orally) for 2 consecutive weeks.
Doses of Spirulina maxima (500 mg/kg, orally), isoniazid (50 mg/kg, orally), and rifampicin (75 mg/kg, orally) were selected on the basis of previous studies. 16 –19 Isoniazid and rifampicin (50 and 75 mg/kg body weight) were prepared separately in sterile distilled water. Spirulina maxima and Silymarin were prepared in sterile distilled water. At the end of the experiment (30 days), animals were sacrificed by cervical dislocation under light ether anesthesia 48 hours after the last dosing. Blood was collected in heparinized vials. Liver was excised and homogenate was made. Variations in specific biochemical and molecular parameters were assessed in blood and tissue samples of all groups of animals. The study protocol was approved by the Institutional animal ethics committee.
Biochemical Investigations
Plasma was assayed for serum glutamate pyruvate transaminase (alanine transaminase) and serum glutamate oxaloacetate transaminase (aspartate transaminase) by the method of Reitman and Frankel, 20 alkaline phosphatase by the method of Kind and King, 21 and bilirubin by the method of Jendrassik and Grof. 22 All estimations were carried out by using standard kits purchased from Crest Biosynthesis, a Division of Coral Clinical Systems, Goa, India.
Superoxide dismutase was assayed by the method of Winterbourn et al, 23 which is based on the formation of NADH-phenazine methosulfate–nitroblue tetrazolium formazan complex. Catalase assay was based on reaction of dichromate in acetic acid with H2O2. 24 Thiobarbituric acid reactive substances were estimated by the method of Ohkawa et al 25 and reduced glutathione as described by Ellman. 26 The biomarkers of oxidative stress were determined in blood as well as liver tissue. DNA damage was measured by Comet assay as described by Singh et al 27 and protein was estimated by the method of Lowry et al. 28
Statistical Analysis
Data are expressed as mean ± SEM. Data comparisons were carried out using one-way analysis of variance using GraphPad Instat version 3.05 (Tukey Kramer multiple comparison).
Results
Effect of Spirulina maxima Administration on Isoniazid and Rifampicin Induced Hepatotoxicity
Significant elevations in the plasma activities of alanine transaminase, aspartate transaminase, alkaline phosphatase, and bilirubin were recorded in animals exposed to isoniazid and rifampicin. Administration of Spirulina maxima to isoniazid and rifampicin exposed animals restored almost all enzyme markers of hepatic function, namely, aspartate transaminase, alanine transaminase, and alkaline phosphatase, to near normal levels. Co-administration of Spirulina maxima with isoniazid and rifampicin, however, offered a significant protection (P < .001) from hepatic damage (Table 1).
Plasma Markers of Liver Function in Wistar Rats Exposed to INH + RIF and Spirulina maxima. a

Abbreviations: ALT, alanine transaminase; AST, aspartate transaminase; ALP, alkaline phosphatase; INH, isoniazid; RIF, rifampicin.
a Values given are mean ± SE.
b P < .05 versus normal control.
c P < .01 versus INH + RIF exposed.
d P < .001 versus INH + RIF exposed.
Effect of S. maxima on Drug Induced Oxidative Stress
Exposure to isoniazid and rifampicin induced severe oxidative stress in experimental animals. Significant reduction in blood levels of glutathione was effectively reversed by Spirulina maxima. The superoxide dismutase activity was decreased moderately when exposed to isoniazid and rifampicin, and treatment with Spirulina maxima almost restored the superoxide dismutase activity to normal level. Similarly, the catalase activity was decreased in isoniazid and rifampicin exposed rats significantly; however, treatment with Spirulina maxima or Silymarin showed restoration of catalase activity. A significant elevation in thiobarbituric acid reactive substances was recorded in rats exposed to isoniazid and rifampicin and Spirulina maxima treatment normalized the levels of thiobarbituric acid reactive substances. Even co-administration of Spirulina maxima with isoniazid + rifampicin resulted in significant elevation in glutathione levels but the elevation was moderate in case of other antioxidant markers, that is, superoxide dismutase, catalase, and thiobarbituric acid reactive substance levels (Table 2).
Effect of Spirulina maxima on Blood Biomarkers of Oxidative Stress in INH + RIF Exposed Animals.a
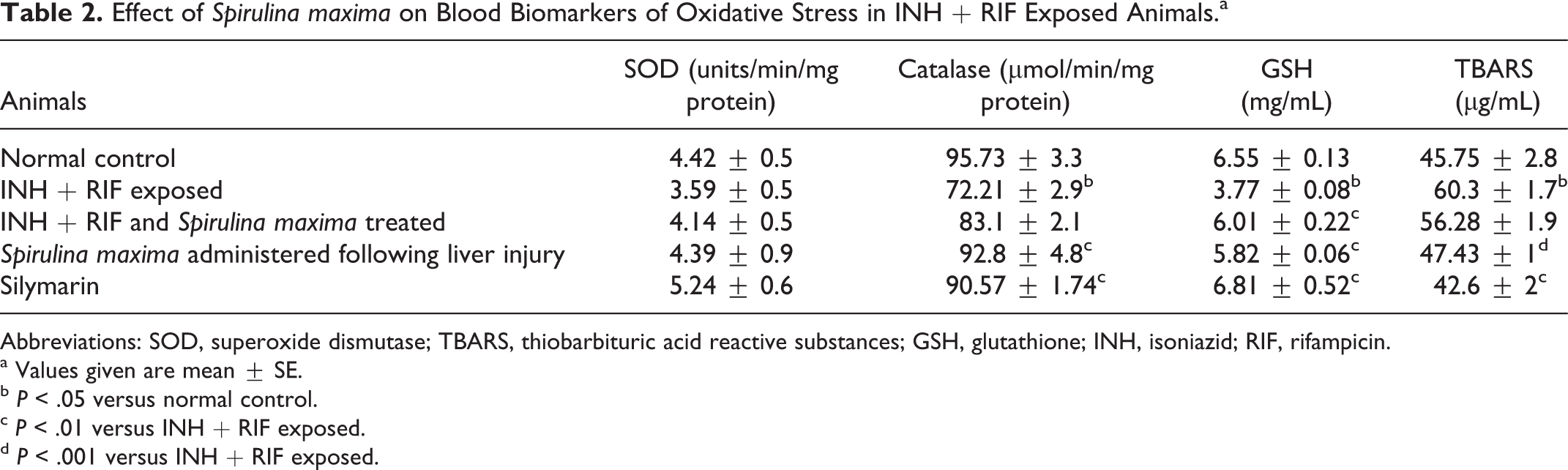
Abbreviations: SOD, superoxide dismutase; TBARS, thiobarbituric acid reactive substances; GSH, glutathione; INH, isoniazid; RIF, rifampicin.
a Values given are mean ± SE.
b P < .05 versus normal control.
c P < .01 versus INH + RIF exposed.
d P < .001 versus INH + RIF exposed.
Liver tissue showed moderate reductions in antioxidant enzymes; superoxide dismutase and catalase and thiobarbituric acid reactive substances following exposure to isoniazid and rifampicin, but the difference was not statistically significant. However, treatment with Spirulina maxima or concomitant administration of Spirulina maxima with anti-tuberculosis drugs resulted in restoration of these biomarkers (Table 3).
Effect of Spirulina maxima on Liver Tissue Antioxidant Markers of INH + RIF exposed Animals.

Abbreviations: SOD, superoxide dismutase; TBARS, thiobarbituric acid reactive substances; GSH, glutathione; INH, isoniazid; RIF, rifampicin.
Values given are mean ± SE. Differences between values with matching symbol notations within each column are not statistically significant at 5% level of probability.
DNA Damage
DNA damage had taken place in isoniazid and rifampicin treated animals as evident from tail formation in Comet assay. Administration of Spirulina maxima either with anti-tuberculosis drugs or after inducing hepatic damage reverted the DNA damage induced by these drugs (Figure 1).

Analysis of DNA damage in blood cells through comet assay.
Discussion
Tuberculosis is a reemerging infectious disease that was declared a global public health problem by the World Health Organization in 1993. 29 The anti-tuberculosis regimen comprises isoniazid, rifampicin, ethambutol, and pyrazinamide daily for 2 months, followed by isoniazid and rifampicin daily for the next 4 months. 30 Protective agents of plant origin with anti-peroxidative and anti-oxidant properties play an important role in protecting the liver against drug-induced toxicity. Spirulina is gaining more attention from medical scientists as a neutraceutical and source of potential pharmaceuticals as well. The antioxidant properties of Spirulina maxima have attracted the attention of researchers recently. 12 The present study investigated the in vivo effects of Spirulina maxima on antituberculosis drug induced hepatotoxicity in Wistar rats.
Isoniazid and rifampicin administration for 14 days generated hepatotoxicity in Wistar rats. Significant alterations in plasma hepatic markers, viz, bilirubin, alanine transaminase, aspartate transaminase, and alkaline phosphatase, had taken place in isoniazid and rifampicin exposed animals, indicating severe hepatic damage induced by these antituberculosis drugs. The hydrazine produced directly from isoniazid or indirectly from acetyl hydrazine is reported to play a key role in liver damage. 31 Rifampicin is a potent inducer of cytochrome P-450 systems, which is responsible for generation of toxic metabolites of drugs and their covalent binding to hepatic macromolecules. 32 Significant reductions in superoxide dismutase, catalase, and glutathione in blood as well as liver tissue with simultaneous increase in thiobarbituric acid reactive substance level in isoniazid + rifampicin exposed group suggest that isoniazid- and rifampicin-induced hepatotoxicity is mediated through oxidative stress.
Treatment with Spirulina maxima resulted in near normalization of liver function and it could be attributed to phycocyanin, β-carotene found in Spirulina maxima. Rats treated with silymarin also showed reduction in plasma alanine transaminase, aspartate transaminase, alkaline phosphatase, and bilirubin. It is known that superoxide dismutase, catalase, and glutathione peroxidase constitute a mutually supportive team of antioxidant enzymes that provide a defense system against reactive oxygen species. 33 Activity of catalase was found to be increased in blood and liver tissue of Spirulina maxima treated rats. Glutathione, which is a potent nonenzymatic antioxidant, was increased in Spirulina maxima treated group of animals. Lipid peroxidation is the process by which lipids (poly unsaturated fatty acids) are oxidized when they react directly with molecular oxygen. 34,35 The insertion of an oxygen molecule is catalyzed by free radicals (nonenzymatic lipid peroxidation) or enzymes (enzymatic lipid peroxidation). 36 The Spirulina maxima treatment resulted in lowering of thiobarbituric acid reactive substance levels in both blood and liver tissue.
Antituberculosis drugs caused increase in cytotoxicity as evident from reduced comet tail length in isoniazid and rifampicin treated animals. Restoration of DNA damage (Figure 1) was clearly evident following Spirulina maxima or silymarin treatment. Concomitant administration of Spirulina maxima with isoniazid and rifampicin protected the animals from the cellular DNA damage suggesting the cytoprotective role of Spirulina maxima. Carotenoids, phenolics, and tocopherols from Spirulina maxima are associated with antioxidant activity. The alcoholic extract of Spirulina maxima inhibited lipid peroxidation more significantly (65%) than the chemical antioxidant like α-tocopherol (35%), BHA (45%), and β-carotene (48%). 37 The water extract of Spirulina maxima was also shown to have more antioxidant effect (76%) than gallic acid (54%) and chlorogenic acid (56%). Phycocyanin also inhibited liver microsomal lipid peroxidation. 38 When antioxidant effects of 2 fractions of a hot water extract of Spirulina maxima using 3 systems that generate superoxide, lipid, and hydroxyl radicals were studied, it was found that both fractions showed significant capacity to scavenge hydroxyl radicals. 39 Earlier studies by our group revealed significant antioxidant functions of Spirulina maxima in experimental diabetes. 40 The study revealed that concomitant administration of Spirulina maxima with antituberculosis therapy offers significant protection from the drug-induced liver damage. The interaction of nutrition and infections is well established. Malnutrition is frequently observed in patients with pulmonary tuberculosis. Several studies reported that patients with active pulmonary tuberculosis are under or malnourished. 41,42 It is further reported that age as well as nutritional status significantly influences the isoniazid acetylator status as well as drug levels in tuberculosis subjects. 30 Hence, nutritional support is needed for undernourished tuberculosis subjects. Spirulina being rich in protein and other nutrients, the present study offers a novel therapeutic approach that addresses both nutritional and hepatoprotection requirements in tuberculosis subjects. Studies directly on tuberculosis subjects, however, would further substantiate its utility as an antidote in isoniazid–rifampicin therapies.
Footnotes
Acknowledgment
The authors thank the Department of Science & Technology, India, for providing infrastructural facility under the FIST program.
Author Contributions
SKJ was associated with the estimation of biochemical parameters. AK and SKJ were associated with induction of hepatotoxicity, collection of blood samples from animals, and so on. NS and SKJ were associated with the comet assay; AZ and GT were associated with the cultivation and maintenance of Spirulina cultures, harvesting, and so forth. PSB and GBKSP were associated with planning the study design and the overall supervision of the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The study was supported in part by research grants from AYUSH, New Delhi, India, and the University Grants Commission, New Delhi, India, and DST-FIST, New Delhi, India.
Ethical Approval
Approval was obtained from the institutional animal ethics committee of Jiwaji University, Gwalior, India.
