Abstract
Hawthorn extract has been used for heart failure and may decrease cardiac cell injury and improve cardiac function. One proposed mechanism for hawthorn action is vasodilation. We hypothesized that hawthorn extract would increase coronary blood flow in isolated perfused rat hearts. Coronary flow was measured in nonworking perfused rat hearts (Langendorff, constant pressure) using a flow probe; data were collected electronically in real time. Hawthorn extract showed an early (30-120 seconds) vasodilation, followed by a later (3-5 minutes) decrease in coronary flow. Maximum vasodilation occurred with 240 μg/mL hawthorn extract. Hawthorn’s pattern of activity was unlike that of several known vasoactive drugs. Both nitric oxide synthase inhibitors and indomethacin abolished early vasodilation, but they had no effect on the late phase decrease in flow. We suggest that a hawthorn-induced increase in nitric oxide generation leads to an increase in prostacyclin production, thus causing early phase vasodilation.
Introduction
In European countries today, hawthorn extract is the primary alternative therapy prescribed for cardiovascular diseases including angina, hypertension, arrhythmias, and symptomatic heart failure (including ischemia). 1 Clinical studies have shown that hawthorn extract increases heart contractility and myocardial oxygen utilization while decreasing heart rate.2–4 Animal studies have shown that hawthorn decreases infarct size in rats whose left anterior descending coronary artery was occluded for 240 minutes followed by 15 minutes of reperfusion. 4 On the basis of these studies, the German Commission E approved hawthorn extract for use in cardiovascular disease, especially New York Heart Association Class I and II heart failure. 5 This is usually administered orally as the unpurified (crude) extract of the leaves/flowers or berries rather than as individual components. 6
Hawthorn extract has been shown to increase cardiac contractility and myocardial oxygen utilization in patients diagnosed with New York Heart Association Class III heart failure. 6 The positive inotropy may be related to an increase in intracellular cAMP. The net effect of protein kinase A activation in heart is an improvement of excitation–contraction coupling and an increase in heart rate and force of contraction. 7 Although acutely beneficial, the slow inward calcium current can induce arrhythmias and increase the energetic demands of the myocardium. 8 , 9
Hawthorn extract caused relaxation of vascular smooth muscle in at least 2 studies. Anselm et al 10 used porcine coronary arteries, which they cut into rings and bathed with aerated buffer. When hawthorn extract was applied to either rat aortic rings 11 or porcine coronary arteries, 10 , 11 the extract caused significant relaxation of the smooth muscle. Both groups felt that the effect was mediated by nitric oxide.
One study suggested that hawthorn extract increases coronary flow. 12 This group used several components of hawthorn extract (rather than the crude extract itself) to dose-dependently increase coronary flow in guinea pig hearts. They saw not only increased coronary flow but also a small positive inotropic effect (possibly elevated cyclic AMP) and a large increase in cardiac relaxation velocity (possibly reduced calcium traffic or elevated cyclic GMP).
Other studies, however, suggest that hawthorn has no significant vasodilatory effects on coronary vasculature. One such study, performed by Nasa et al 13 used the Langendorff technique with Krebs–Henseleit buffer and hawthorn extract dissolved in water. The hearts were made ischemic by removing afterload pressure from the heart for an extended period of time. The hearts were then allowed to reperfuse for 20 minutes with hawthorn extract dissolved in water added to the perfusate. While it was found that hawthorn extract contains cardioprotective qualities, it was determined that hawthorn did not contain vasodilatory qualities.
The interaction of hawthorn extract and nitric oxide (one endothelium-derived vasodilatory compound) has been investigated in several recent studies. Varying levels of hypotensive activity were observed in rats and humans after administration of hawthorn extract.14–16 Two studies showed relaxation of phenylephrine-precontracted artery rings when hawthorn extract was added, demonstrating that hawthorn is effective in relaxing smooth muscle. Both groups abolished the effect by denuding the rings of endothelium and concluded that nitric oxide was involved. 11 , 14
The first goal of this study was to determine the most effective dose of hawthorn extract (WS 1442) to increase coronary flow in the isolated rat heart. The second goal was to explain the physiological mechanism(s) by which hawthorn achieves vasodilation. In this study, we used a crude hawthorn extract (WS 1442), standardized such that oligomeric proanthocyanindins comprise 18.75% of the total powdered extract, by weight, to cause vasodilation in adult rat hearts. We found that the hawthorn extract causes vasodilation in the intact heart system, possibly via nitric oxide–induced prostacyclin synthesis.
Methods
Use of Animals
Male Sprague-Dawley rats (Hilltop Laboratory Animals, Scottdale, PA) weighing between 250 and 300 g were used in this study. Animals were housed in a fully staffed AAALAC-approved facility at A. T. Still University, Kirksville College of Osteopathic Medicine, under environmentally controlled conditions (temperature = 21 ± 1°C, 12-hour light/dark cycle, standard rat chow and water provided ad libitum). The standards of care were in accordance with the National Research Council’s Guide of the Care and Use of Laboratory Animals. 17
Perfusion of Hearts
Rats were heparinized (500 IU, ip) 20 minutes prior to administration of anesthesia (sodium pentobarbital, 105-140 mg/kg, ip). Hearts were excised from the rat and securely attached via the aorta to a fluid-filled 9-gauge cannula, then moved to the apparatus. The attached hearts were perfused for 1 to 2 minutes with nominally oxygenated basal salts buffer while being trimmed of excess tissue. The buffer consisted of either Krebs–Henseleit buffer (a bicarbonate-buffered crystalloid, equilibrated with 95% O2/5% CO2; composition of Krebs–Henseleit, in mM: NaCl 118.0, KCl 4.7, NaH2PO4 1.2, NaHCO3 25.0, CaCl2 3.0, ethylenediaminetetraacetic acid 0.5, MgCl2 1.2, HEPES 5.0, glucose 11.0, pH 7.4). 18
Once moved, the hearts were perfused with a constant pressure Langendorff system (70 mm Hg), using a reservoir of fluid located above the heart. 18 Coronary flow was measured by a flow probe inserted in line ahead of the heart (Carolina Medical Equipment, King, NC). Output from the probe was relayed to a digitizer (DataQ Instruments, Akron, OH 19 ) and read/stored on a laptop computer. In this model, the hearts were not provided with any preload.
Preparation of Hawthorn Extract
The hawthorn extract used in this study was pharmaceutical-grade extract, the kind gift of Willmar Schwabe GmbH (Karlsruhe, Germany). This extract is a dried tincture (80% aqueous ethanol) prepared from the leaves and flowers of the white hawthorn (Crataegus monogyna, also called Crataegus oxycantha in older literature), standardized to 18.92% oligomeric proanthocyanidins. We developed 2 experimental protocols, based on our handling of the powdered extract for use with the hearts. In protocol 1, the extract was dissolved in dimethyl sulfoxide and added to perfusion buffer in that form. In protocol 2, the extract was dissolved to the extent possible in warm water, centrifuged to remove any insoluble material, and the aqueous supernatant added to the perfusion buffer.
Protocol 1: Hawthorn Extract in Dimethyl Sulfoxide
In this protocol, hawthorn extract was dissolved in dimethyl sulfoxide to a concentration that allowed the desired concentration to be achieved with the addition of 0.6 mL of dimethyl sulfoxide solution (ie, [dimethyl sulfoxide] was constant at 0.6 mL/120 mL perfusate, or 0.5%).
Hearts were cannulated and perfused as described above. After a washout and stabilization period of 5 minutes, drug additions (including hawthorn extract) in the protocol were made and allowed to recirculate; the addition of a drug to the system marked time zero (t = 0 minutes) for that experiment. Data were collected for 5 minutes after each drug addition (t = 5 minutes). At the conclusion of data collection for the first drug (drug “A”), a second drug (drug “B”) was added to the system in some experiments. The addition of drug “B” would constitute time zero for that drug (t = 5 minutes overall) and data would be collected for an additional 5 minutes (t = 10 minutes overall). Drugs were added according to the schedule shown pictorially in Figure 1. When all trials were completed for each heart, it was removed from the apparatus and weighed.

Protocol diagram. Rectangular bar shows the basic protocol, below which are times (minutes) at which each step begins and ends. Note that all times are expressed relative to the addition of drug 1. C = cannulation; washout = 5 minutes stabilization period post-cannulation. Experimental series are shown below the bar, as well as the number of hearts at each condition (n). Abbreviations: BSA, bovine serum albumin; DMSO, dimethyl sulfoxide; IBMX, isobutylmethylxanthine; KH, Krebs–Henseleit buffer (described in Methods); L-NAME, L-NG-nitroarginine methyl ester hydrochloride; PDE, cyclic nucleotide phosphodiesterase.
Protocol 2: Hawthorn Extract in Water
In this protocol, hawthorn extract was suspended in water to a concentration that allowed the desired concentration to be achieved with the addition of 0.6 mL of aqueous solution. Handling the extract in this way produced an opaque suspension, which was cleared by centrifugation at 13 000 × g for 1 minute. The supernatant was removed and added in toto to the perfusate. Concentrations were expressed as if all of the extract had gone into solution. The pellet was held and used in subsequent experiments.
In experiments using the pellet as a drug, the pellet was reconstituted in 0.6 mL warm dimethyl sulfoxide. The resulting solution was added to the perfusate as if it was hawthorn extract.
The hearts were allowed a 30-minute stabilization period. At this time hawthorn extract was added to the reservoir and allowed to circulate. This was designated time T = 0. In studies using L-NG-nitroarginine methyl ester hydrochloride, the heart was allowed a 25-minute stabilization period. At the end of the stabilization period, L-NG-nitroarginine methyl ester hydrochloride was administered. This was designated time T = −5. Hawthorn extract (60 μg/mL) was administered 5 minutes following the injection of L-NG-nitroarginine methyl ester hydrochloride (T = 0) and allowed to circulate for 30 minutes. After the hour-long study, each heart was removed from the cannula, filleted, blotted dry, and weighed. A timeline for each experiment is illustrated in Figure 2.

Protocol diagram. Rectangular bar shows the basic protocol, below which are times (minutes) at which each step begins and ends. As in protocol 1, times are expressed relative to the addition of the first drug. Abbreviation: L-NAME = L-NG-nitroarginine methyl ester hydrochloride.
Data Collection and Handling
Digitization rate varied between 15 and 60 samples per second in individual experiments. Data points were digitally meaned using a time-moving average technique. One second’s samples (approximately 10) on both sides of an individual time point were averaged to determine the mean value at that time point. The time point was advanced by 1 sample and the process was repeated. This had the effect of producing a single “mean” flow from the pulsatile coronary flow traces.
The mean flow was used to calculate coronary flows relative to the time the drug was added. These were all normalized to heart weight. Baseline flow was defined as flow at the time drug was added to the apparatus (by definition, 0 minutes). Early phase is defined as flow 30 to 120 seconds after the addition of a drug. Due to the distance the drug had to travel in the apparatus, no effects would be seen before 30 seconds had elapsed, and all observable effects of the early phase were recorded by 120 seconds. Late phase (also known as 5-minute flow) is defined as flow 5 minutes after the addition of a drug.
Hawthorn Extract Metabolism or Extraction by the Heart
Hawthorn extract was added to Krebs–Henseleit buffer in the apparatus with a heart in place, as indicated above. Samples (1.5 mL) were collected before and after passage through the coronary circulation, and at other predetermined times throughout the experiment. Each sample was assayed qualitatively and quantitatively for hawthorn extract by gradient high-performance liquid chromatography essentially as described by Kirakosyan et al. 20 A 200-μL portion of each sample was applied to a reversed-phase (C18) column, then eluted with a gradient system consisting of 0.1% aqueous triflouroacetic acid (solvent A) and acetonitrile (solvent B) at 1.0 mL/min. The column was initially equilibrated with 95% solvent A and 5% solvent B. immediately following sample injection, solvent B was increased to 45% over 40 minutes, followed by a 5-minute period at 45% solvent B. Flow was constant at 1.0 mL/min. Detection wavelength was 280 nm.
This assay produced an integrator tracing with 63 nominally individual peaks representing the individual components of the hawthorn extract. Each peak was analyzed for individual area (μV-s) and percentage of total peak area. Pre- and post-heart samples were compared for changes in the relative areas of individual peaks. (Specifically, post-heart samples were examined for missing or added peaks relative to pre-heart samples.) As per agreement with Willmar Schwabe GmbH (the hawthorn extract supplier), no attempt was made to identify individual peaks.
Statistics
Differences between individual experiments and experimental series were examined using analysis of variance followed by Student’s t test, as performed by SigmaPlot and Microsoft Excel. The t test was used to confirm differences between (a) baseline and early flow and (b) baseline and late flow. Flows were defined as significantly different at α = .05. Data are represented as an average compared to the baseline (t = 0) and shown with standard deviation.
Reagents
Salts, bovine serum albumin, and drugs were purchased from either Sigma-Aldrich Chemical Company (St Louis, MO) or MP Biochemical Company (Solon, OH) and were the highest quality commercially available (reagent grade or better). Hawthorn extract was the kind gift of Willmar Schwabe GmbH (Karlsruhe, Germany).
Results
Protocol 1: Hawthorn Extract in Dimethyl Sulfoxide
Four doses of hawthorn extract (final concentrations = 80, 160, 240, 320 μg/mL) were dissolved in 0.6 mL dimethyl sulfoxide (final concentration in perfusion = 0.5% v/v), and added separately to the crystalloid medium (Krebs–Henseleit) perfusing the hearts. Coronary flow was evaluated as described in Methods. The flow rate pattern from a typical heart treated with hawthorn extract is shown in Figure 3. Both the early phase increase in flow and the late phase (5 minutes) decrease in flow are clearly visible.

Sample tracing with hawthorn addition. Coronary flow tracing (mL/min/g) of a single heart versus time is shown. Baseline flow, early phase flow, and late phase (5 minutes) flow are labeled.
Dose and Time of Exposure
Dimethyl sulfoxide alone did not change weight-normalized flow from baseline after the time of introduction into the circulating media. Flow rates did not differ from the zero time point at either the early phase or the 5-minute recording (Figure 4, open bars).
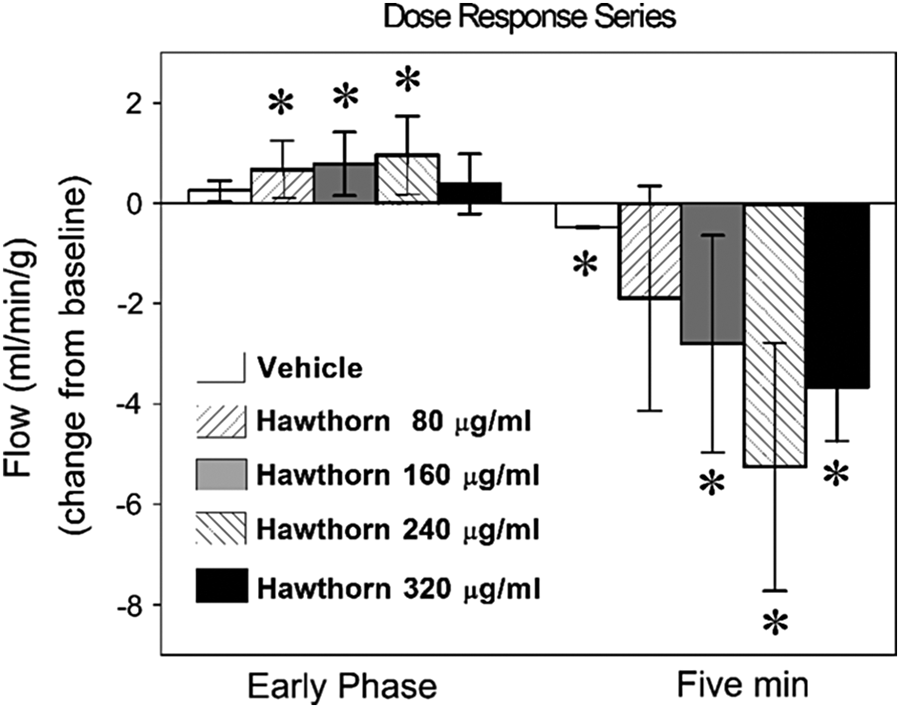
Coronary flow (hawthorn dose response series). Coronary flows (mL/min/g) at 4 doses of hawthorn (80, 160, 240, and 320 μg/mL) are shown with vehicle control (0.5% dimethyl sulfoxide). Numbers are expressed as absolute change ± SD from baseline (baseline = 0).
Hawthorn extract dissolved in dimethyl sulfoxide was added to the crystalloid medium perfusing the hearts, using the described doses. The medium containing the drug was recirculated for 5 minutes after addition. Hawthorn generated 2 distinct time-dependent effects on flow. First, there was a hawthorn-induced increase (P < .05) in coronary flow 30 to 120 seconds after addition of the drug to the system; this occurred as soon as the drug reached the heart. We have termed this early rise “early phase.” This activity reached a maximum at 240 μg/mL of hawthorn extract (Figure 4, striped bars). Second, flow decreased (P < .05) after the measured peak and continued to fall during the following 5 minutes (the “late phase”; Figure 4). This late-phase reduction in flow was not related to the dose of extract, nor did it appear in other hearts that were exposed to drugs with known mechanisms. Flows recorded longer than 5 minutes after the drug was introduced into the system did not show recovery (data not shown). Flow continued to decline to almost zero and an arrhythmia could be seen on the recording device. Hawthorn extract at 240 μg/mL was most effective at increasing flow in the early phase and was thus used for subsequent experiments.
Published reports have made no reference to the late-phase reduction in flow that we observed using hawthorn extract. Therefore, we attempted to evaluate this phenomenon and tried several approaches to diminish its severity.
Single-Pass Exposure Versus Recirculation
Since hawthorn extract is a complex chemical mixture, it could contain both vasodilatory and vasoconstrictive components. If the heart extracted or metabolized a vasodilatory component but left a vasoconstrictive component in the circulation, a phenomenon something like the observed late-phase reduction in flow might result. We therefore designed a series of single-pass (ie, non-recirculating) experiments. In these experiments, each heart was allowed to equilibrate with recirculated oxygenated Krebs–Henseleit media. At 5 minutes, hawthorn extract was introduced at a concentration of 240 μg/mL and recirculation discontinued. This set of experiments did not show a significant increase in coronary flow. More important, a significant late-phase reduction in flow (P < .05) was still present at the 5-minute flow recording (data not shown).
In addition, we collected perfusate from the apparatus before passage through the heart and at several time points after passage through the heart during recirculating experiments (7 total of these latter samples). These samples were evaluated by reversed-phase high-performance liquid chromatography and compared to each other. The object was to document either the disappearance of a component or the appearance of extra peaks, which might represent metabolites of an extract component.
Sample chromatograms from these experiments are shown in Figure 5 (before passage through the heart [panel A], after 2 minutes of hawthorn exposure [early phase, panel B], or after 5 minutes of hawthorn exposure [late phase, panel C]). No added or missing peaks were noted in any chromatograms, nor were individual peak areas different between chromatograms. These results indicate that individual components observed with this chromatographic system are neither metabolized nor extracted by the heart. As per agreement with our supplier, no attempt was made to further evaluate the extract components.

Examination of putative hawthorn metabolism/extraction by intact heart. Hawthorn (240 μg/mL) was added to Krebs–Henseleit buffer in the apparatus and assayed for the extract by high-performance liquid chromatography. Sample chromatograms of the extract diluted in the apparatus before passage through the heart (panel A), at the maximum of early phase flow stimulation (about 2 minutes; panel B), and at 5 minutes after extract addition (panel C) are shown. Numbers above each peak are retention times (minutes), which fluctuate ±5% due to variations in room temperature and solvent composition.
Comparison of Hawthorn to Known Metabolic Inhibitors or Stimulators
To evaluate possible mechanism(s) for hawthorn extract’s vasodilatory (or putatively vasoconstrictive) effects, flow was examined using several known vasoactive substances and metabolic inhibitors. These agents were used alone or in conjunction with hawthorn at the 240 μg/mL level. Figure 1 is valuable for tracking the experimental protocols used in this section.
Phosphodiesterase Inhibition: 3-Isobutyl-1-methyxanthine ± Forskolin
A set of hearts were cannulated and allowed to equilibrate according to protocol. At 5 minutes, 1 mM 3-isobutyl-1-methyxanthine was added and allowed to circulate for 5 minutes. 3-Isobutyl-1-methyxanthine is a nonspecific inhibitor of cyclic nucleotide phosphodiesterases. Some research groups believe that hawthorn relaxes smooth muscle by acting as a phosphodiesterase inhibitor, which will increase cellular levels of cyclic AMP. The early phase showed a decrease in flow with 3-isobutyl-1-methyxanthine (P < .05), followed by a return to baseline at 5 minutes (Figure 6, open bars).

Coronary flow (phosphodiesterase series). Coronary flow (mL/min/g) is shown using 3-isobutyl-1-methylxanthine (IBMX, 1 mM; white bars) and forskolin (10 μM; right-slanted hatching) in single-drug protocols, and 1 mM IBMX followed by 10 μM forskolin in a 2-drug protocol (black bars). See Figure 2 for a schematic. Numbers are expressed as absolute change ± SD from baseline (baseline = 0). Hawthorn alone (240 μg/mL; left-slanted hatching) is shown for comparison purposes.
A second set of hearts was cannulated and allowed to equilibrate. In this set, 10 μM forskolin was added instead of 3-isobutyl-1-methyxanthine. (Forskolin activates adenylyl cyclase, increasing cyclic AMP.) Forskolin caused an early phase decrease in flow (P < .05), followed by a return to baseline at 5 minutes (P < .05; Figure 6, light gray bars).
A third set of hearts was perfused according to the 2-drug protocol. After cannulating the hearts and allowing them to equilibrate for 5 minutes, 1 mM 3-isobutyl-1-methyxanthine was added to the perfusate and allowed to circulate for an additional 5-minute incubation (10-minute elapsed time). Forskolin (10 μM) was then added to the perfusate and allowed to circulate for 5 minutes. This set of experiments was designed to elevate cyclic AMP while not allowing it to degrade. When compared to baseline, early phase flow (between 30 and 120 seconds) was increased significantly (P < .05) and returned to baseline at 5 minutes (Figure 6, dark gray bars).
Nitric Oxide Inhibition: L-NG-nitroarginine methyl ester Hydrochloride ± Hawthorn Extract
To test if endothelial derived nitric oxide generation was a possible mechanism by which hawthorn increases coronary flow, L-NG-nitroarginine methyl ester hydrochloride (100 μM, an isoform-insensitive inhibitor of nitric oxide synthase) was added at 5 minutes and allowed to circulate. L-NG-nitroarginine methyl ester hydrochloride caused no significant change in flow at the early phase (30-120 seconds) nor at the 5-minute recording (Figure 7, open bars).

Coronary flow (nitric oxide series). Coronary flow (mL/min/g) is shown using L-NG-nitroarginine methyl ester hydrochloride (L-NAME; 100 μM; white bars) and hawthorn (240 μg/mL; left-slanted hatching) in single-drug protocols, and 100 μM L-NAME followed by 240 μg/mL hawthorn in a 2-drug protocol (black bars). See Figure 2 for a schematic. Numbers are expressed as absolute change ± SD from baseline (baseline = 0).
Another set of hearts was perfused according to the 2-drug protocol. L-NG-nitroarginine methyl ester hydrochloride (100 μM) was added and allowed to circulate for 5 minutes as described above. After this incubation period (10-minute elapsed time), hawthorn extract (240 μg/mL) was added and allowed to circulate for an additional 5 minutes (Figure 7, grey bars). L-NG-nitroarginine methyl ester hydrochloride prevented hawthorn from increasing coronary flow. Early phase flow (30-120 seconds) was not different from the baseline recording. At the 5-minute reading, flow was still dramatically decreased (P < .05).
We questioned whether the hearts were limited in their ability to create nitric oxide as a result of a shortage of arginine. Krebs–Henseleit buffer is normally not supplemented with amino acids. Therefore, 3 hearts were perfused using Krebs–Henseleit supplemented with 1% essential and 1% nonessential amino acids. Hawthorn extract (240 μg/mL) was administered at 5 minutes and allowed to circulate for 5 minutes. Flow recordings from these hearts produced both significant increases (P < .05) in the early phase and a significant decrease (P < .05) in flow at the 5-minute recording. Hence, amino acid supplementation did not affect the temporary increase in flow or alleviate the decrease in flow at 5 minutes (data not shown).
Cyclooxygenase Inhibition: Indomethacin ± Hawthorn Extract
To rule out vasoconstriction by locally produced thromboxane as a cause of the late-phase reduction in flow, 100 μM indomethacin was added to hearts. Indomethacin alone caused a significant increase in flow (similar to hawthorn), as well as a significant decrease in flow at 5 minutes (P < .05; Figure 8, open bars). While these changes were significant, the pattern did not closely resemble that of hawthorn extract.
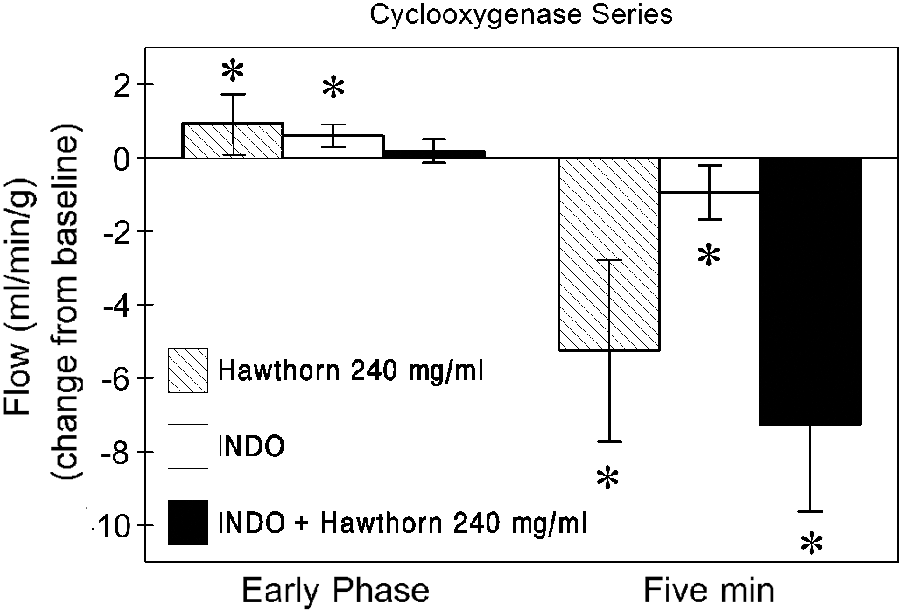
Coronary flow (cyclooxygenase series). Coronary flow (mL/min/g) is shown using indomethacin (INDO, 100 μM; white bars) and hawthorn (240 μg/mL; left-slanted hatching) in single-drug protocols and 100 μM indomethacin followed by 240 μg/mL hawthorn in a 2-drug protocol (black bars). See Figure 2 for a schematic. Numbers are expressed as absolute change ± SD from baseline (baseline = 0).
A second set of hearts were perfused with indomethacin for 5 minutes, after which a bolus of hawthorn extract (240 μg/mL) was added and allowed to circulate for 5 minutes. No significant change in flow was observed (Figure 8). However, the 5-minute exposure to hawthorn still produced a late-phase decrease in flow (P < .05) that had been seen in hearts exposed to hawthorn alone.
Protocol 2: Hawthorn Extract in Water
Hawthorn extract was added to the perfused heart in aqueous suspension, as described in Methods. At each dosage, hawthorn extract caused an increase in coronary flow, all of which were significant except for the 120 μg/mL dose (Figure 9).

Dose-dependent increase in coronary flow by hawthorn extract. Flow above baseline levels after addition of the hawthorn extract (20, 40, 60, 80, 100, and 120 μg/mL) is shown as mL/min/g wet weight of heart. * = significantly different from baseline flow in each heart (P < .05).
Each concentration of hawthorn extract increased coronary flow past baseline until the maximum increase in flow was reached using 80 μg/mL (Figure 9). Concentrations of the extract beyond 80 μg/mL resulted in a decline in the observed vasodilation. This resulted in a biphasic dose response relationship, peaking at 80 μg/mL hawthorn extract. Since hearts exposed to 80 μg/mL hawthorn showed a more profound decrease in flow than that seen with 60 μg/mL of hawthorn extract, subsequent experiments were conducted with 60 μg/mL hawthorn extract. Increase in flow was significantly different between dosages 20 and 80 μg/mL and 80 and 120 μg/mL. There was no difference between other dosages (ANOVA).
Hearts exposed to higher levels of hawthorn extract (especially 100 and 120 μg/mL) responded with a biphasic response to the drug itself. (A comparison is shown in Figure 10A [60 μg/mL] and B [100 μg/mL]). Concentrations of hawthorn extract higher than 80 μg/mL caused vasodilation followed by a decrease in flow that fell below baseline.

Coronary flow in perfused heart with hawthorn extract. Heart was perfused according to the protocol described in Methods. Flow is expressed as mL/min/g wet weight. (A) An amount of hawthorn extract appropriate to a final concentration of 60 μg/mL was added at the arrow. Note that flow never falls below baseline flow. This is a typical response to concentrations of hawthorn extract between 20 and 80 μg/mL. (B) An amount of hawthorn extract appropriate to a final concentration of 100 μg/mL was added at the arrow. Note the late-phase reduction in flow past baseline flow (T = 0). This is a typical response to 100 and 120 μg/mL hawthorn extract (n = 5).
Studies With L-NG-Nitroarginine Methyl Ester Hydrochloride
When L-NG-nitroarginine methyl ester hydrochloride (100 μM) was added to the perfusate 5 minutes before the addition of hawthorn extract, we observed an increase in flow of 0.97 mL/min/g. However when L-NG-nitroarginine methyl ester hydrochloride was added to the perfusate without hawthorn extract, a slight vasoconstriction was observed (Figure 11, top, and Figure 12). L-NG-nitroarginine methyl ester hydrochloride reduced the vasodilatory effect produced by hawthorn extract (60 μg/mL; 4.34 mL/min/g increase in flow) an average of 11% (Figure 11, bottom). L-NG-nitroarginine methyl ester hydrochloride did not completely abolish the vasodilatory effect of hawthorn extract. Figure 11 (bottom) shows the average flow tracing when L-NG-nitroarginine methyl ester hydrochloride preceded hawthorn extract.

Effect of L-NG-nitroarginine methyl ester hydrochloride (L-NAME) on hawthorn-induced vasodilation in rat heart. Flow tracings of coronary flow after addition of L-NAME (T = −5) with and without hawthorn extract (60 μg/mL, T = 0) are shown in the top and bottom panels, respectively. Baseline is defined as the flow at the point when the drug is added (T = 0).

Effect of L-NG-nitroarginine methyl ester hydrochloride (L-NAME) on hawthorn-induced vasodilation in rat heart. Summary of results shown in Figure 11.
Pellet Studies
The pellets from the 60 and 100 μg/mL hawthorn extract/water solution were retained and reconstituted in dimethyl sulfoxide (0.6 mL). This solution was added to the buffer, and the perfusion performed otherwise using our established protocol. Addition of the pellet did not cause a significant increase or decrease in flow past baseline (Figure 13).

Lack of late-phase reduction in flow after addition of hawthorn pellet to the perfusate. Flow tracings of coronary arteries after addition of hawthorn pellets corresponding to 60 and 100 μg/mL, reconstituted in dimethyl sulfoxide. No significant increase or decrease in flow is seen.
Discussion
The goal of this project was to determine (a) the most effective dose of hawthorn extract to increase coronary flow in the isolated rat heart and (b) the physiological mechanism by which hawthorn increases coronary flow.
Doses of hawthorn extract for this study were chosen based on available literature. Popping Pöpping et al 21 studied hawthorn’s effect on the contraction amplitude of cardiac myocytes. They used the extract in the same range as this study (30-180 μg/mL), but a different preparation of the extract (LI 132). Schüssler et al 22 used individual flavonoids found in hawthorn at concentrations from 0.1 to 500 μM (again, the same range as this study) to investigate cardiac effects of these compounds. Long et al 23 used 30 μg/mL hawthorn extract to investigate the extract as an anti-arrhythmic. These levels of hawthorn extract were proven to generate effects, which is what we wished to study with our isolated organ preparation.
The hawthorn concentrations used in both this study and the literature cited above are pharmacologic doses. Exactly what serum concentration of individual hawthorn components (or, indeed, any polyphenol) constitutes a “physiologic” level is unclear. (We use the term physiologic level here to mean the concentration of these compounds one might expect to see in body fluids after a given oral dose.) The bioavailability of the general class of polyphenols 24 as well as more specific subclasses of proanthocyanidins 25 , 26 and flavonoids 27 has been reviewed. There is general agreement that polyphenols (flavonoids, proanthocyanidins, and related compounds) do not cross the intestinal wall well (3% to 10% of dose) and experience quantitatively large first-pass metabolism by the liver. There is much less agreement as to the levels of these compounds one might expect to see in body fluids after a given oral dose. There is also little agreement as to the changes that gastric secretions or intestinal microflora exert on the composition of an ingested oral dose of the extract. Thus, we elected to use the simpler and proven method of a pharmacologic dose of extract as purchased.
We found in this study that hawthorn extract used in this way is an effective coronary vasodilator. In our first protocol (extract in dimethyl sulfoxide), hawthorn extract caused a transient increase in coronary flow followed by a later decrease in flow. In our second protocol (extract in water), hawthorn extract caused a dose-dependent increase in coronary flow up to 80 μg/mL. Higher concentrations of extract were not as effective and showed a small late-phase reduction in flow.
Hawthorn-induced vasodilation in coronary arteries is consistent with 3 previous studies. Schussler et al 12 demonstrated that luteolin-7-glycoside and theophylline (both are components of hawthorn extract) elevate coronary flow by 50% in isolated guinea pig hearts. In another study, Kim et al 11 showed that the proanthocyanidins in hawthorn extract were vasorelaxants in rat aortic rings. Finally, Anselm et al 10 showed vasodilatory activity of hawthorn extract in pig coronary arteries. In this study, 240 μg/mL of dimethyl sulfoxide–dissolved hawthorn extract was the most effective dose for increasing coronary flow, while 80 μg/mL hawthorn extract was most effective using water as a vehicle.
The Late-Phase Reduction in Flow
As mentioned above, hawthorn extract had a biphasic influence on coronary flow. The initial response was vasodilation. Coronary flow increased once the extract reached the heart (30-120 seconds after addition to the circulating medium), but rapidly declined below baseline. This effect was especially striking in protocol 1 (dimethyl sulfoxide vehicle, short preincubation), but could still be observed with the higher doses of hawthorn extract in protocol 2 (water vehicle, longer preincubation).
In protocol 1, this biphasic response of the hearts to hawthorn was remarkably consistent. Coronary flow increased by approximately 5% between 30 and 75 seconds after the addition of the drug to the apparatus, followed by a brisk decline. In the initial phase, flow increased at a steady rate. In the later phase, flow declined at approximately the same rate dropping significantly below baseline flow. This trend continued until data collection was terminated. Diminishing flow was often accompanied by arrhythmias. We believe that this is the first description of this biphasic flow phenomenon following the addition of hawthorn extract to isolated hearts.
We tried to eliminate the late-phase reduction in flow using several different drugs and protocols (vide infra). The first protocol consisted of passing the hawthorn-containing medium through the heart only once (single pass), as opposed to the recirculating method, which was our standard protocol. The crude extract of most plant products are made up of many components (at least 63 are present in hawthorn extract), any of which could be vasodilatory or vasoconstrictive. If one or more of the vasodilatory components of hawthorn was metabolized or altered by the heart, the remaining vasoconstrictive compounds could cause severe reduction in flow. We used 240 μg/mL hawthorn extract in the single-pass protocol to test this possibility. We observed no difference in flow pattern with this protocol; the initial peak in flow was present as well as the later reduction.
We also collected fractions of the perfusate at various points in the recirculating protocol and analyzed the hawthorn content by high-performance liquid chromatography. No differences in the number or quantity of individual components were observed when prepass samples were compared to postpass samples. These results, when taken together with the lack of differences observed in the single-pass protocol, leads us to believe that individual components of the hawthorn extract are not being eliminated or altered by the heart. The late-phase flow reduction therefore must result from some other process.
Since we saw a pellet in the aqueous hawthorn that was not present in dimethyl sulfoxide–reconstituted hawthorn, we developed the notion that certain compounds found in the dimethyl sulfoxide–reconstituted extract were water insoluble. In this scenario, the late-phase reduction in flow was a mechanical phenomenon. Water-insoluble portions of the extract would fall out of solution when the dimethyl sulfoxide–reconstituted extract was added to the perfusate, occluding the coronary vasculature and blocking flow. We tested this idea by reconstituting the pellets from the hawthorn/water preparation with dimethyl sulfoxide and adding them to the perfusate. If the late-phase reduction in flow was purely a mechanical phenomenon, these “pellet preparations” should have caused a reduction in flow with no accompanying vasodilation. Instead, the added pellets had no real effect; neither an increase nor a decrease in flow was observed (60 and 100 μg/mL). We were forced to conclude that the late-phase reduction in flow is based somehow in physiology, not in mechanics.
One possible explanation for the late-phase decrease in flow (and, indeed, for the reduction in flow observed using higher concentrations of hawthorn [100 and 120 μg/mL] in protocol 2) is that there are competing mechanisms (vasodilatory and vasoconstrictive) occurring in the coronary vasculature. It is conceivable that vasodilatory mechanisms alone are initiated with lower concentrations of hawthorn, while vasoconstrictive mechanisms require a much higher concentration of the extract. This theory would explain why the higher concentrations of hawthorn do not cause as significant a vasodilation and cause a greater decrease in flow than the lower concentrations of hawthorn extract. Further studies are necessary to examine this phenomenon.
Mechanism of Vasodilation
Phosphodiesterase Series
Our initial efforts to uncover a mechanism by which hawthorn extract either increases flow (initial phase vasodilation) or decreases flow (late phase vasoconstriction) were predicated on the prevailing suggestion that hawthorn extract inhibits cyclic nucleotide phosphodiesterase. 6 Several groups have suggested, however, that cyclic nucleotide phosphodiesterase inhibition is a less likely mechanism than some other possible mechanisms. 11 , 14 , 15 The ability of hawthorn extract to inhibit cyclic phosphodiesterase had clearly not been tested sufficiently prior to our study.
3-Isobutyl-1-methylxanthine, a nonspecific inhibitor of cyclic nucleotide phosphodiesterases, was used to elevate cyclic AMP in the vascular tissue to determine if this would mimic the response evoked by hawthorn extract. Flow was found to sharply decrease at the time 3-isobutyl-1-methylxanthine containing medium came into contact with the heart tissue. This early phase decrease was followed by a return to baseline flow. This was almost the exact opposite of the pattern evoked by the administration of hawthorn extract.
One must be careful in interpreting data from cyclic nucleotide phosphodiesterase inhibitor studies. Often, real elevations in cyclic nucleotides are not achieved due to lack of stimulation of the cyclase enzymes that form these molecules. Forskolin activates adenylate cyclase. We used this drug alone and in combination with 3-isobutyl-1-methylxanthine to confirm our results. In these experiments, nothing resembling the vasodilatory peak or 5-minute decline of flow due to hawthorn was observed. Thus, cyclic AMP is not a physiologically significant vasodilator in our model, and the hawthorn-induced increase in early phase flow is unlikely to be due to inhibition of cyclic nucleotide phosphodiesterase.
Nitric Oxide Series
It has been suggested that hawthorn relaxes smooth muscle by activating nitric oxide synthase. 14 Results from a recent study suggest that hawthorn-induced vasodilation is related to an unknown endothelium-derived factor related to nitric oxide. 11 We used L-NG-nitroarginine methyl ester hydrochloride (an inhibitor of nitric oxide synthase) to determine if nitric oxide plays a role in hawthorn’s vasodilatory effect. Baseline flow was unchanged by L-NG-nitroarginine methyl ester hydrochloride alone, indicating that nitric oxide is not critical to maintaining baseline coronary flow. When L-NG-nitroarginine methyl ester hydrochloride preceded hawthorn, hawthorn no longer caused an increase in early phase coronary flow. This suggests that hawthorn-induced vasodilation is the result of nitric oxide production. The flow reduction seen at 5 minutes still occurred after L-NG-nitroarginine methyl ester hydrochloride, indicating that it is not related to the early phase flow increase.
Krebs–Henseleit medium is not normally supplemented with amino acids. If, in fact, hawthorn vasodilates by activating nitric oxide synthase, it is conceivable that the late-phase reduction in flow is caused by a shortage of the NOS substrate arginine. However, hearts perfused with amino acid–supplemented Krebs–Henseleit medium showed the same flow pattern as those perfused with unsupplemented Krebs–Henseleit. This suggests that the heart is not depleted of arginine or other amino acids during our protocol.
Cyclooxygenase Series
Release of prostacyclin causes vasodilation in coronary arteries; thromboxane A2 causes vasoconstriction. 28 We considered the possibility that the early phase vasodilation was secondary to prostacyclin production, while the late-phase reduction in flow was a result of thromboxane A2 release. To test this possibility, hearts were perfused with 100 μM indomethacin (a nonspecific cyclooxygenase inhibitor). Hearts treated with indomethacin alone showed a significant early phase increase in flow (similar to hawthorn); however, there was no late-phase decrease in flow at the 5-minute recording. When hearts were perfused with indomethacin followed by 240 μg/mL hawthorn extract (our 2-drug protocol), the initial (early phase) increase in flow was not observed. However, the reduced late-phase flow was still evident. This suggests that prostacyclin synthesis contributes to the early phase vasodilation. In contrast, thromboxane A2 synthesis seems unimportant to the late-phase reduction in flow.
Recent studies suggest that cyclooxygenase and nitric oxide synthase reciprocally regulate each other. 29 These authors suggested that nitric oxide production stimulates the cyclooxygenase enzyme, producing more prostacyclin and E-series prostaglandins. It is conceivable that hawthorn extract, by increasing nitric oxide production, actually stimulates cyclooxygenase to preferentially produce more prostacyclin as part of the effect. L-NG-nitroarginine methyl ester hydrochloride, of course, would still block this activity by reducing nitric oxide production. In this model, indomethacin (by blocking cyclooxygenase) should also eliminate the early phase hawthorn induced flow increase. This, in fact, can be observed in Figure 8 (grey bar).
Hawthorn Extract and Nitric Oxide
We further examined the participation of nitric oxide in the hawthorn-induced vasodilatory process using protocol 2. We introduced the optimum dosage of hawthorn extract (60 μg/mL) into the heart, which produced the expected level of vasodilation (70% increase, Figure 11). When L-NG-nitroarginine methyl ester hydrochloride (an inhibitor of nitric oxide synthase, and thus nitric oxide production) preceded hawthorn addition, there was no significant increase in flow in response to the hawthorn (Figure 11B). Thus, protocol 2 confirms that aqueous hawthorn appears to act at least in part via the nitric oxide pathway.
Summary
In summary, hawthorn extract had a biphasic influence on coronary flow. The initial response was vasodilation. In the early phase, coronary flow increased by approximately 5% between 30 and 75 seconds after the addition of the drug to the apparatus. In the later phase, flow declined at approximately the same rate dropping significantly below baseline flow. We believe that this is the first description of this biphasic flow phenomenon following the addition of hawthorn extract to isolated hearts. We suggest that the mechanism by which hawthorn increases coronary flow is not phosphodiesterase inhibition as previously thought but most likely involves nitric oxide and prostacyclin.
Footnotes
Acknowledgments
We thank Wilmar Schwabe Pharmaceuticals, Karlsruhe, Germany, for their generous gift of hawthorn extract (WS 1442). We are grateful to A. T. Still University’s Graduate Program and the KCOM Warner-Fermaturo Fund for their funding and sponsorship.
Author Contributions
Kenneth P. Dood was mainly responsible for the experiments, data, and data analysis outlined in Protocol 1. Aaron D. Frey was mainly responsible for the experiments, data, and data analysis outlined in Protocol 2. Timothy P. Geisbuhler is the laboratory director and mentored the other two authors as graduate students. Dr Geisbuhler also combined the theses of Mr Dood and Mr Frey into a single article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this project came from the ATSU/KCOM Biomedical Sciences Research Fund and the ATSU/KCOM Warner-Fermaturo Fund.
Ethical Approval
This project was approved by the KCOM Animal Care Committee (the KCOM Internal Animal Care and Use Committee), Protocol 153.
