Abstract
The clinical efficacy of purple passion fruit peel extract (a flavonoid-rich dietary supplement) in reducing cardiovascular risk factors in adult type 2 diabetic subjects was investigated in a randomized, double-blind, placebo-controlled trial. Forty-one subjects were randomly assigned to receive a daily dose of purple passion fruit (220 mg) or a matched placebo for 16 weeks. Body mass index, blood pressure, fasting and postprandial blood glucose, glycated hemoglobin, and lipid profile were determined at baseline and at monthly intervals. A significant reduction in systolic blood pressure and fasting blood glucose was observed following administration of purple passion fruit (P < .05). Purple passion fruit was well tolerated, and no adverse events were reported. These data suggest that purple passion fruit supplementation for 16 weeks in type 2 diabetics results in a significant reduction in systolic blood pressure and fasting blood glucose, indicating that purple passion fruit is safe and well tolerated by diabetics.
Keywords
Introduction
Prevalence of type 2 diabetes is increasing worldwide because of increase in life expectancy, prevalence of obesity, and sedentary lifestyle. 1,2 Numerous epidemiological studies have demonstrated that type 2 diabetes increases the risk of cardiovascular morbidity and mortality. 3,4 Prolonged exposure to hyperglycemic environment and changes in key metabolites and biomarkers damage blood vessels, which leads to atherosclerosis and hypertension, further leading to cardiovascular complications. The concomitant presence of hyperglycemia, dyslipidemia, and hypertension increases the risk of cardiovascular disease and requires aggressive management of all cardiovascular risk factors. 5 Treatment of hypertension and dyslipidemia in diabetic patients substantially decreases cardiovascular and microvascular complications. 6 However, it is difficult to achieve this goal with the currently available drugs without incurring undesirable side effects.
A promising and cost-effective alternative strategy is to test natural products and dietary supplements that reduce the cardiovascular disease risk factor in diabetes patients. Plant extracts that contain high antioxidants have been shown to be effective in reducing blood glucose levels in type 2 diabetes. 7,8 The purple passion fruit peel (Passiflora edulis Sims f edulis) extracts have been used widely in folk medicine in South America to treat anxiety, insomnia, bronchitis, and asthma. 9 The purple passion fruit peel extract contains 3 major components: cyanidin-3-O-glucoside, quercetin-3-O-glucoside, and edulilic acid. 10 In a rat model of human essential hypertension with increased expression and activity of inducible nitric oxide, purple passion fruit peel supplementation significantly reduced the blood pressure by decreasing nitric oxide. 10 Purple passion fruit peel treatment of moderate to severe hypertensive patients also markedly reduced both systolic and diastolic blood pressure. However, no data are available with respect to the clinical usefulness of purple passion fruit peel extract in improving cardiovascular risk factors in patients with type 2 diabetes, especially with concurrent pharmaceutical antihypertensive drug therapy. The fact that antioxidants have beneficial effects on hypertension 11 and that purple passion fruit peel extract contains high levels of antioxidants, 11 and exhibits antihypertensive effects, 10,11 prompted us to investigate the clinical efficacy of purple passion fruit peel extract in altering the cardiovascular disease risk factors in diabetic patients.
Experimental Section
Study Subjects
A total of 49 eligible type 2 diabetic subjects of both genders in the age range of 30 to 70 years, who are maintaining their fasting blood glucose levels >140 mg/dL and systolic/diastolic blood pressure >140/90 mm Hg, were enrolled for the study. Forty-one subjects completed the study. The clinical trial was conducted as per International Conference on Harmonisation Good Clinical Practice guidelines and approved by the Alerts Ethics Committee, Mumbai, India. The study was conducted at Gnanaganga Ayurvedic Trust Hospital and Research Center, Tirupati, Andhra Pradesh, India. Written informed consent was obtained from the participants. All the subjects were identified by a unique number, and no personal information was used to identify the study subjects.
Components of Purple Passion Fruit Peel Extract
The purple passion fruit peel extract was purified as described earlier 8 –10 by Kemin Foods, and the same extract was used in an earlier clinical trial. 8 Kemin Foods performed tests to show the purity and similarity to previous purple passion fruit peel extracts 8,10 and found microbial contamination and debris to be at levels acceptable for human use. The passion fruit was obtained by Kemin Foods from growers in Florida, United States of America, and the extract was prepared by Kemin Foods in Des Moines, Iowa. The extract is 0.6% of the wet weight of the purple fruit skin, and a person receiving a maximum 200 mg/day is eating the equivalent of 3.33 passion fruit peel. Purple passion fruit peel extract contains a mixture of several key phytochemicals as indicated by several constituent high-performance liquid chromatography peaks. Initial high-performance liquid chromatography analysis identified the following major constituents in the purple passion fruit peel extract: cyanidin-3-O-glucoside, quercetin-3-O-glucoside and edulilic acid, a novel cyclic acid glucoside found only in Passiflora edulis. Minor ingredients at less than 1% include catechin, epicatechin, kampferol-3-O-glucoside, kampferol, luteolin-8-C-neohesperoside, luteolin-8-C-digitoxoside, protocatechuic acid, quercetin, and prunasin. 9,10 The compounds identified thus far represent about 20% of the product by weight, while other constituents remain undefined. 10
Study Procedure
Each subject’s personal history of current and previous medicines used, disease symptoms, type of diet, family history of disease, onset of disease if already under medication, and current life styles were recorded. The study participant’s demography, physical activity, smoking and usage of alcohol, besides the physical assessment including height, weight, and circumferences of waist and hip, were recorded. 12
Experimental Design
Study subjects were randomly assigned to receive either purple passion fruit pill (220 mg) or matched placebo for 16 weeks in a double-blind trial. The placebo and purple passion fruit peel extract capsules were prepared and kindly provided by Kemin Health LLC (Des Moines, IA) as described previously. 8,10 Subjects were requested to take the pill after food. Subjects were asked to complete a daily diary—recording the time they took the pill and other concomitant medications or alternative treatments. The subjects were asked to maintain normal diet and activity throughout the study. Subjects were also asked to complete a 3-day meals record at the beginning and at the end of the treatment phase. No significant changes in diet were noted between the placebo and purple passion fruit peel groups (data not shown).
Blood samples were collected at the beginning and after 4, 8, 12, and 16 weeks of purple passion fruit peel supplementation to determine the lipid profile, fasting and postprandial blood glucose, glycated hemoglobin, and safety parameters such as liver and kidney function tests. Blood samples taken at monthly intervals were used to determine the fasting and postprandial blood glucose concentrations. Biochemical and hematological safety parameters such as hematocrit, liver function tests, and kidney function tests were performed at the beginning and at the end of the treatment phase. Eight subjects withdrew from the study before 2 months due to personal reasons unrelated to the study and hence their data were not included in analyzing the data. The remaining subjects completed the study (19 subjects, purple passion fruit treated group; 21 subjects, placebo group).
Biochemical Estimations
Clinical chemistry was performed with diagnostic assay kits as per manufacturer’s instructions. Glucose was determined by GOD-POD method, and total cholesterol, low-density lipoprotein cholesterol, and high-density lipoprotein cholesterol were determined by the oxidase peroxidase method. Triglycerides were determined by the glycerol phosphate oxidase dehydrogenase method, and glycated hemoglobin was determined using the ion exchange method. 13 –16
Statistical Analysis
Analysis was performed according to the intention to treat principle, using a standard deviation of 0.7, to detect the effect with 80% power. Thus, all randomized patients who received at least 1 dose of study medication and who had both a baseline measurement and at least 1 postbaseline measurement were analyzed. The data are expressed as mean ± standard error of the mean. Statistical analyses were performed with SPSS version 16.0 (SPSS Institute, Chicago, IL). Chi-square test with Yates correction was used for noncontinuous variables. Student’s t test was used to determine the statistical significance of the continuous variables. Comparable nonparametric test (Mann–Whitney U test) was substituted when tests for normality and equal variance failed. A value of P < .05 was used as a criterion for statistical significance. Linear growth rate was calculated for blood glucose and blood pressure by taking the average values every 4 weeks.
Results
Table 1 shows the demographic data of the diabetic subjects. The study population was 49% male. Fifty-nine percent of the study population had a family history of diabetes. Activity pattern indicates that 20% of the subjects are sedentary, 29% come under mild activity, and 51% come under moderate activity (Table 1). The majority of the subjects who participated in the trial belong to socioeconomic status 4. (In India, socioeconomic status is divided into 5 grades. Grade 1 is highest class and grade 5 is lowest class.) In this study, 37% of the subjects had diabetes for <4 years, 46% of the study subjects had the disease for 5 to 9 years, and 17% of the subjects had diabetes for >10 years. Subjects who participated in the current trial were using metformin/glibenclamide to control the glucose levels and atenol to control high blood pressure. The study was initiated in September 2010 and completed in October 2011. The subjects were divided into 2 groups and were asked to take 1 pill of either placebo or purple passion fruit every day. The mean age of the purple passion fruit treated group is 52 ± 9.7 years and that of the placebo group 51 ± 12 years (Table 1). The mean body mass index of the purple passion fruit treated group and placebo remained unchanged after 16 weeks. A significant reduction in systolic blood pressure was observed in the purple passion fruit treated group (day 0, 133.8 ± 2.9; vs week 16, 126.1 ± 2.5; % change = 7.7 ± 3.8 mm Hg; P < .05), when compared with the placebo group (day 0, 139.2 ± 2.3; vs week 16, 132.4 ± 2.7; % change = 6.8 ± 3.6 mm Hg; ns). Diastolic blood pressure did not show any significant change in both purple passion fruit treated and placebo treated groups (Table 2). As shown in Table 3, fasting blood glucose levels in the purple passion fruit treated group significantly declined (22.6 ± 10.8 mg/dL; P = .04) after 16 weeks. Similarly, the postprandial blood glucose levels decreased (21.8 ± 21.9 mg/dL; P = .33) on purple passion fruit administration. However, in the placebo-treated group, an increase in the levels of fasting (22.6 ± 21.5 mg/dL, ns) and postprandial blood glucose (49.0 ± 19.4 mg/dL; P < .05) levels was observed (Table 3).
Demographic Profile of the Study Subjects.

Abbreviation: SES, socioeconomic status.
a Socioeconomic status is divided into 5 grades: grade 1 is the highest class and grade 5 is the lowest class.
Blood Pressure Levels in Study Subjects After Placebo or PFP Supplementation.a

Abbreviation: PFP, passion fruit peel.
a Data are presented as mean ± standard error of the mean.
Changes in Blood Glucose Levels in Placebo and PFP Treated Subjects.a
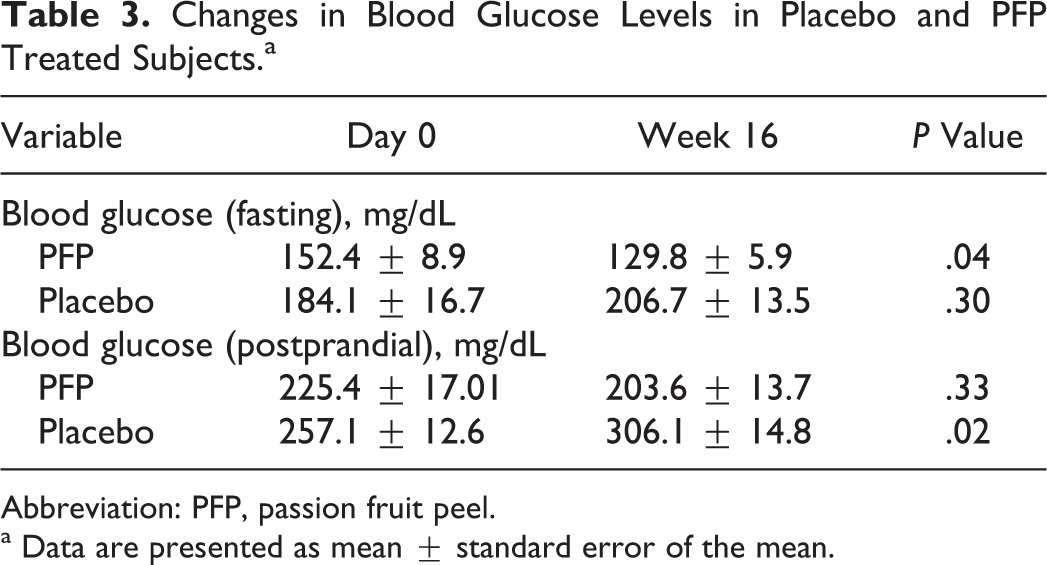
Abbreviation: PFP, passion fruit peel.
a Data are presented as mean ± standard error of the mean.
Mean levels for glycated hemoglobin and lipid profile are shown in Table 4. Purple passion fruit administration did not reduce glycated hemoglobin (statistically insignificant). Total cholesterol, high-density lipoprotein cholesterol, low-density lipoprotein cholesterol, and triglycerides did not change significantly during the study period. In the placebo group, glycated hemoglobin, total cholesterol, low-density lipoprotein cholesterol, and triglycerides tend to increase and high-density lipoprotein cholesterol level tends to decrease at the end of the study when compared to the values at the initiation of the study. These changes were not significant.
Changes in Glycated Hemoglobin and Lipid Profile in Placebo and PFP Treated Subjects.a

Abbreviation: PFP, passion fruit peel.
a Data are presented as mean ± standard error of the mean.
Changes in blood pressure and blood glucose levels at monthly intervals (growth rate) are shown in Figures 1 and 2, respectively. In purple passion fruit administered group, both fasting and postprandial blood glucose show a decreasing trend (fasting = −3.73, postprandial = −3.01) compared with the placebo group, which showed an increasing trend (fasting = 2.33, postprandial = 2.59). The growth rate for blood pressure levels in purple passion fruit treated group shows a negative trend in both systolic blood pressure (−1.61) and diastolic blood pressure (−0.50). But we observed a positive trend (an increase) in the placebo group (systolic blood pressure= 0.98, diastolic blood pressure = 0.55). All the subjects were monitored throughout the study period and adverse events or serious adverse events were recorded. There were no adverse events reported during the trial period. Purple passion fruit supplementation did not cause any significant changes in safety parameters (hematological, liver function, and kidney function tests). There were no toxicities associated with purple passion fruit administration, and the administered dose (220 mg/day) was well tolerated.
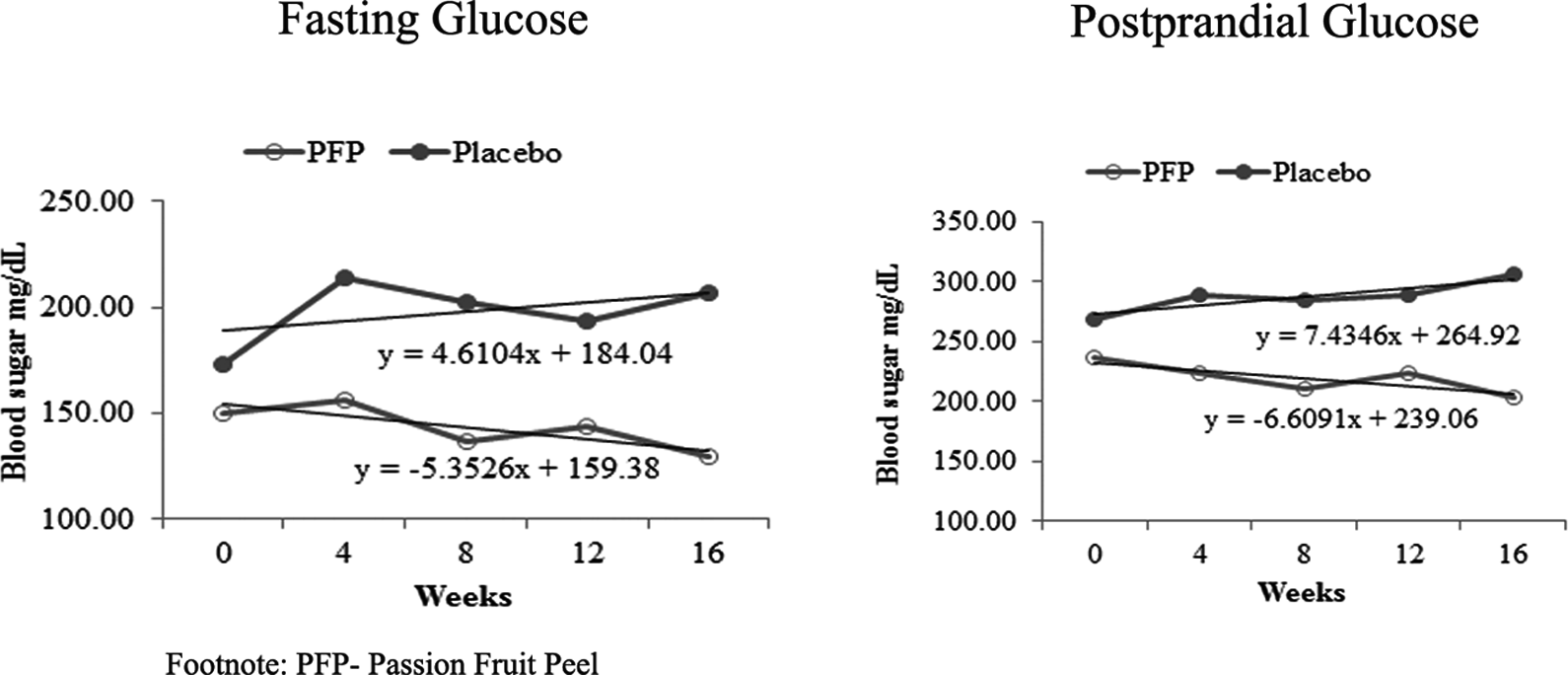
Changes in the levels of fasting and postprandial blood glucose levels in type 2 diabetes patients administered with passion fruit peel (PFP) extract or placebo for 16 weeks.

Changes in the levels of systolic and diastolic blood pressure in type 2 diabetes patients administered with passion fruit peel (PFP) extract or placebo for 16 weeks.
Discussion
Dietary supplements with a complex array of naturally occurring bioactive nonnutrients confer significant long-term health benefits. Passion fruit peel extract is a novel dietary supplement containing bioactive nonnutritive molecules with health promoting activities. 8 –10,17
The principal findings of the current study include a decrease in the systolic blood pressure in purple passion fruit treated subjects while it increased in the placebo treated group. Thus, even in the presence of antihypertensive medicines, purple passion fruit had an additional positive effect in reducing systolic blood pressure. Chronic administration of purple passion fruit (1 capsule containing 220 mg/day) for 16 weeks did not result in any adverse events, and hence purple passion fruit can be considered as safe for diabetic subjects. In previous in vitro studies, 10 the purple passion fruit peel extract did not show any inhibitory effect on cell growth but reduced DNA damage. In addition, in in vitro toxicity studies, passion fruit peel extract did not show any hepatotoxicity. 10
A significant reduction in fasting blood glucose and systolic blood pressure was observed with purple passion fruit administration. The negative linear growth rates for blood glucose and blood pressure supports the beneficial effects of purple passion fruit in diabetes subjects. Oral administration of passion fruit juice to streptozotocin-induced diabetic rats for 30 days resulted in a significant decrease in blood glucose, total cholesterol, triglycerides, and low-density lipoprotein cholesterol levels and an increase in high-density lipoprotein cholesterol levels. 18 The results presented in the current study also indicate a significant decrease in blood glucose levels in human subjects. But we did not observe significant changes in cholesterol and triglycerides.
The present study shows that purple passion fruit supplementation helps maintain control of blood pressure in individuals with type 2 diabetes. In light of the increased use and interest in dietary supplements, it is essential to obtain a better understanding of their potential positive and negative health effects and the underlying mechanisms. The present study was undertaken to investigate the efficacy and safety of the purple passion fruit peel extract. Hypertension affects nearly 1 in 3 American adults and as many as 1 billion people worldwide. The prevalence of hypertension increases with advancing age, with more than half of the people in the age group of 60 to 69 years suffering from hypertension. 19 Hypertension is an established risk factor for myocardial infarction, congestive heart failure, stroke, end-stage renal disease, and sudden death. Improvements in identifying and treating hypertension have contributed to a major reduction in the incidence of cardiovascular disease and stroke. 6 A growing body of evidence suggests the oxidative/nutritive stress in the arterial wall as an important causative agent in the pathogenesis of various forms of hypertension. 20
The antihypertensive effect of the purple passion fruit peel extract was first investigated in a spontaneously hypertensive rat model, a well-recognized rodent model of human essential hypertension. 20 There is much experimental evidence that the pathophysiology of spontaneously hypertensive rats closely mirrors that of human patients with essential hypertension. Similar defects in membrane transport of sodium, increased sympathetic activity, and hypercoagulability have been identified in spontaneously hypertensive rats and patients with essential hypertension. In addition, long-term complications of hypertension such as left ventricular hypertrophy and stroke have been shown to be common in both spontaneously hypertensive rats and humans with essential hypertension. 10 The antihypertensive effect of purple passion fruit peel extract can be explained, at least in part, by the lowering of the elevated nitric oxide level in the hypertensive animals.
Oxidative stress plays an important role in diabetes, and supplementation with herbal extracts containing high levels of antioxidants have been shown to offer beneficial effects. 20,21 The principal compounds in purple passion fruit peel extract include edulilic acid, a novel cyclic acid glucoside, quercetin 3-O-glucoside, and cyanidin 3-O-glucoside. Several compounds in the purple passion fruit peel extract have been identified (Figure 3), and their potential role in modifying hypertension has been described. 10 Several epidemiological studies have shown a significant inverse association between dietary flavonoids and long-term mortality from coronary heart disease. 22 In vivo biological actions of quercetin, one of the flavonoids found in purple passion fruit peel extract, can support the observed antihypertensive effect of purple passion fruit peel extract. Quercetin has been shown to lower arterial blood pressure, attenuate cardiac hypertrophy, and decrease aortic medial thickening in different animal models of cardiovascular disease. 20,23 The quercetin 3-O-glucoside content in the purple passion fruit peel extract was estimated to be about 2.8% by weight of the total material. Therefore, it is possible that quercetin might be a significant contributor to the observed antihypertensive effect. Taken together, the results of the current study, the results of rat hypertension study, 18 and previous study in hypertensive persons 10 suggest that the purple passion fruit peel extract affects hypertension in part by modulating nitric oxide production. This could be probably because of the downregulation of inducible nitric oxide synthase expression by quercetin, luteolin, cyanidin 3-O-glucoside, or other compounds, or scavenging of nitric oxide (as a radical itself) by quercetin 24 with assistance from other flavonoids. 21 Modulated nitric oxide production and free oxygen species scavenging by flavonoids in purple passion fruit peel extract can decrease peroxynitrite anion generation. This results in the inhibition of the peroxynitrite anion’s effects on the endogenous antioxidant system, which in turn can modify vascular tone and peripheral vessel resistance, thus lowering blood pressure. 18,25,26 In spontaneously hypertensive rats, purple passion fruit lowered nitric oxide production, 10 an important indicator of its mechanisms of action. Another plant extract, pycnogenol, made from pine tree bark and partially purified to enrich for polyphenols, reduced blood pressure in type 2 diabetes patients treated with antihypertensive drugs 27 by lowering nitric oxide. Purple passion fruit lowered nitric oxide in spontaneously hypertensive rats. 10 Aged mice treated chemically to induce heart failure and given pycnogenol lowered blood pressure by changing nitric oxide production and prevented structural cardiac remodeling by excessive metallomatrix proteinases synthesis, thus reducing high blood pressure. 28 This study 28 suggests how purple passion fruit’s polyphenols might reduce blood pressure by modulating lysyl oxidase and metallomatrix proteinases and their changes in cardiac structure, fibrosis, and thus function.
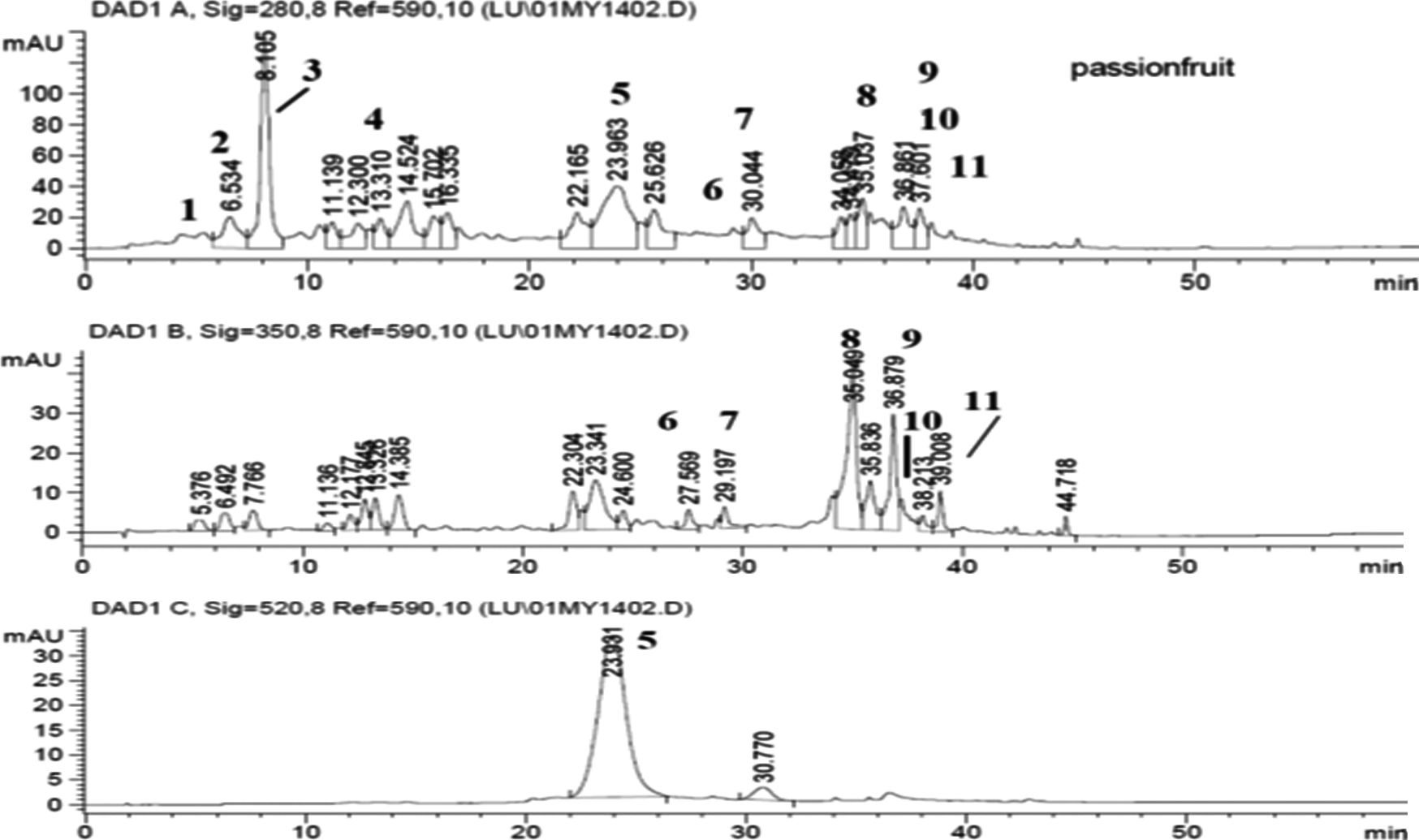
Key phytochemicals identified in passion fruit peel extract.
There are few potential limitations to this study. The first limitation of our study concerns the relatively moderate sample size. Another limitation was that we treated diabetics with blood pressure lowered by standard pharmaceutical drugs. Finally, the purple passion fruit dose (220 mg) used in this study was less than that previously used in a hypertension trial 10 of “nondiabetics.” But this study was done in the absence of any pharmaceutical drug therapy. It was effective in treating asthma and osteoarthritis. 8,17 The results are also supported by the efficacy of purple passion fruit components in rat models of diabetes. 18 It is possible that higher doses of purple passion fruit (more than 220 mg/day) for prolonged periods of time (more than 16 weeks) would be more likely to have much more beneficial effects in diabetic subjects. The dose of 220 mg of the extract given to the diabetic subjects may be insufficient to make significant changes in the HbA1c. Also, since the duration of the study is 12 weeks, we observed a significant decrease in fasting blood glucose levels. But the postprandial blood glucose levels did not change significantly. Future studies with higher dose of passion fruit peel extract (>220 mg) for longer periods (>3-6 months) can alter HbA1C levels. In addition, a larger sample size could have further strengthened our findings about the beneficial effect of purple passion fruit in subjects with type 2 diabetes. The change in HbA1C levels (6.7%, after PFP treatment) vs (7.1%, placebo) did not reach statistical significance, which could be due to the limited number of subjects (n=20) in the current study. If similar changes are observed in large clinical trials before and after PFP treatment, the result would achieve statistical significance indicating the potential of PFP in lowering the HbA1C. In spite of the above limitations, we observed a significant decrease in systolic blood pressure and also a decrease in fasting blood glucose, indicating that the high levels of antioxidants in the purple passion fruit peel extract alter cellular targets that affect blood pressure and blood glucose in the diabetic subjects.
Conclusion
The results of the present study support the antihypertensive potential and safe use of the purple passion fruit peel extract in diabetes subjects with high blood pressure. Because the purple passion fruit peel extract did not show any clinical toxicity, it is possible to consider purple passion fruit as a supplement to antihypertensive medications, such as angiotensin-converting enzyme inhibitors. As many patients may not be able to afford the expenses of currently available medications, a natural product with low or no toxicity is an attractive candidate that offers additional benefits to diabetic subjects. In summary, our data support the hypothesis that purple passion fruit supplement favors a reduction in cardiovascular disease risk in individuals with type 2 diabetes.
Footnotes
Acknowledgments
Support of Southwest Scientific Editing and Consulting, LLC, a grant from US Government Stimulus funds to SWSEC in 2010 and 2011, and a grant from Venture LLC are appreciated. The purple passion fruit pills were kindly provided and made by Kemin Foods, Des Moines, IA, USA.
Author Contributions
Dr Watson is one of the key investigators of the study design. He is also responsible for procuring the purple passion fruit from Kemin Foods. Also, he has been actively involved in designing the clinical trial and in the interpretation of the results. Dr C. Damodar Reddy monitored the studies in India and spent months there on this study. Dr S. Dattatreya Rao is the principal investigator of study. Dr K. Kodandarami Reddy: investigator selection, regulatory approvals, statistical analysis, manuscript writing. I. Naga Raju: data management, statistical analysis, manuscript writing, and GCP/regulatory compliance. C. Krishna Kumari: labeling, coding, and accountability of investigational product.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Support from Southwest Scientific Editing and Consulting, LLC came from US Government Stimulus funds to SWSEC in 2010 and 2011, and a grant from Venture LLC which are appreciated. The purple passion fruit pills were kindly provided and made by Kemin Foods, Des Moines, IA, USA.
Funding
Dr Watson received research funding from US Government Stimulus funds to Southwest Scientific Editing and Consulting, LLC (SWSEC) in 2010 and 2011, and a grant from Venture LLC which is developing product related to the research described in this paper. In addition, the author serves as president of SWSEC and receives no compensation for the clinical research services. He may potentially benefit from the research results. The terms of this arrangement have been reviewed and approved by Dr Watson in accordance with its conflict of interest policies.
Ethical Approval
The clinical trial was approved by the Alerts Ethics Committee, Snehal Hospital, Bombay (Protocol No. SLS/CT-01-10; May 27, 2010). Informed consent was prepared in accordance with guidelines laid down by ICH-GCP, Declaration of Helsinki, the World Health Organization, the Indian Council of Medical Research, Central Drugs Standard Control Organization, and the applicable laws and regulation. Written informed consent was obtained from the participants. All the subjects were identified by a unique number and no personal information was used to identify the study subjects.
