Abstract
Supplementation with vitamin D has become increasingly popular over the past decade, and numerous organizations have developed recommendations for the appropriate intake of vitamin D. Vitamin D supplements come in a variety of formulations and strengths and vary in their directions for use. This study was designed to compare vitamin D label dosing information with the recommendations in North American guidelines. A systematic search was conducted to identify 62 single-ingredient vitamin D products of which 1000 IU was the most common strength. Assessment of North American guidelines found recommended vitamin D dosing to range from 400 to 1000 IU daily, depending on age. Twenty-four (39%) of the products recommended a maximum dose within the range of 400 to 1000 IU daily. Thirty-eight (61%) and 19 (31%) products recommended maximum doses more than 1000 IU daily and 2000 IU daily, respectively. Labeled dosing recommendations of commercially available vitamin D supplements are largely inconsistent with North American recommendations.
Keywords
The popularity of vitamin D supplementation has grown substantially over the past decade. In fact, according to the Nutrition Business Journal, consumers purchased approximately $430 million worth of vitamin D supplements in 2009 compared with $40 million in 2001. 1,2 Furthermore, one survey identified vitamin D as the fastest growing dietary supplement on the market, with use increasing by 52% from 2008 to 2010. Fifty-six percent of survey respondents reported using vitamin D, making it the third most popular dietary supplement behind fish oil and multivitamins. 3 The ascent of vitamin D in the supplement world is not surprising. Practitioner awareness of and testing for vitamin D deficiency has recently increased. Additionally, new research suggests that increased vitamin D intake can be beneficial in a variety of disease states. Media reports and consumer health information Web sites have made this information available to the public. 4
Physiologically, vitamin D regulates calcium absorption in the intestine, providing adequate serum calcium levels necessary for bone building and repair. 5 Thus, it is not surprising that vitamin D plays an important role in bone health. The use of vitamin D supplementation in preventing and treating rickets in children and osteomalacia in adults is well established, and clinical evidence suggests its efficacy in the prevention and treatment of osteoporosis as well as fall prevention. 6 Aside from its function on bone health, vitamin D is also believed to modulate cellular and genomic processes. Because of this, researchers have studied the effects of vitamin D supplementation on a variety of disease states ranging from depression and diabetes to multiple sclerosis and cancer. 5
A number of organizations in North America have developed guidelines for recommended vitamin D intake. In 2010, the Institute of Medicine updated its Dietary Reference Intakes for vitamin D. The Dietary Reference Intakes were developed based on a thorough review of evidence related to vitamin D and human health. Accordingly, the recommended dietary allowance for individuals 1 to 70 years of age is 600 IU/day. The recommended dietary allowance for individuals older than 70 years of age is 800 IU/day. The United States Institute of Medicine also recommends an adequate intake of 400 IU/day for infants 0 to 12 months of age. 4 The National Osteoporosis Foundation recommends vitamin D intake of 400 to 800 IU/day for individuals under 50 years of age and 800 to 1000 IU/day for those age 50 and older. 7 Osteoporosis Canada, on the other hand, recommends supplementation of 400 to 1000 IU/day in healthy adults at low risk of vitamin D deficiency. Subsequently, Canadian adults over 50 years of age are considered to have moderate risk of vitamin D deficiency and should supplement with 800 to 1000 IU/day. Osteoporosis Canada’s guidelines also note optimal supplementation may require more than 1000 IU/day, with doses up to 2000 IU/day being safe. 8 The North American Menopause Society recommends 700 to 800 IU/day for women at risk of deficiency due to inadequate exposure to sunlight. 9 The National Comprehensive Cancer Network bases its vitamin D intake recommendations for bone health in cancer on the Institute of Medicine’s report. 10 Conversely, the Canadian Cancer Society recommends supplementation of 1000 IU/day during the fall and winter for all adults living in Canada. Furthermore, Canadian adults who are older, have dark skin, do not go outside often, and/or wear clothing covering most of their skin should consider supplementation of 1000 IU/day year round. 11 An American Academy of Pediatrics report on the prevention of rickets and vitamin D deficiency recommends all infants, children, and adolescents receive at least 400 IU/day of vitamin D. 12 Table 1 outlines the national guideline recommendations for vitamin D intake.
North American Guideline Recommendations for Vitamin D Dosing.

The recent rise in consumer supplementation with vitamin D has led to an increase in available vitamin D products on the market. We wonder, however, how closely the dosing information on these over-the-counter supplements follows the guidelines. Therefore, this study was designed to compare vitamin D label dosing information with the recommendations in national guidelines.
Methods
A systematic search was conducted using Google search and Google shopping from January 18 through February 9, 2012. The search term used was vitamin D. Commercially available single-ingredient vitamin D products were identified and selected for further review. Product information was collected for each product identified, including brand and manufacturer if different from brand; vitamin D form, strength, or concentration; dosing directions on label; and USP verification status.
North American national guidelines related to vitamin D dosing were identified based on the knowledge of the investigators and mention in review literature discussing vitamin D. The recommended dosing of vitamin D from these guidelines was tabulated. Dosing recommendations from commercially available vitamin D products were then compared against dosing recommendations of the various guidelines.
Results
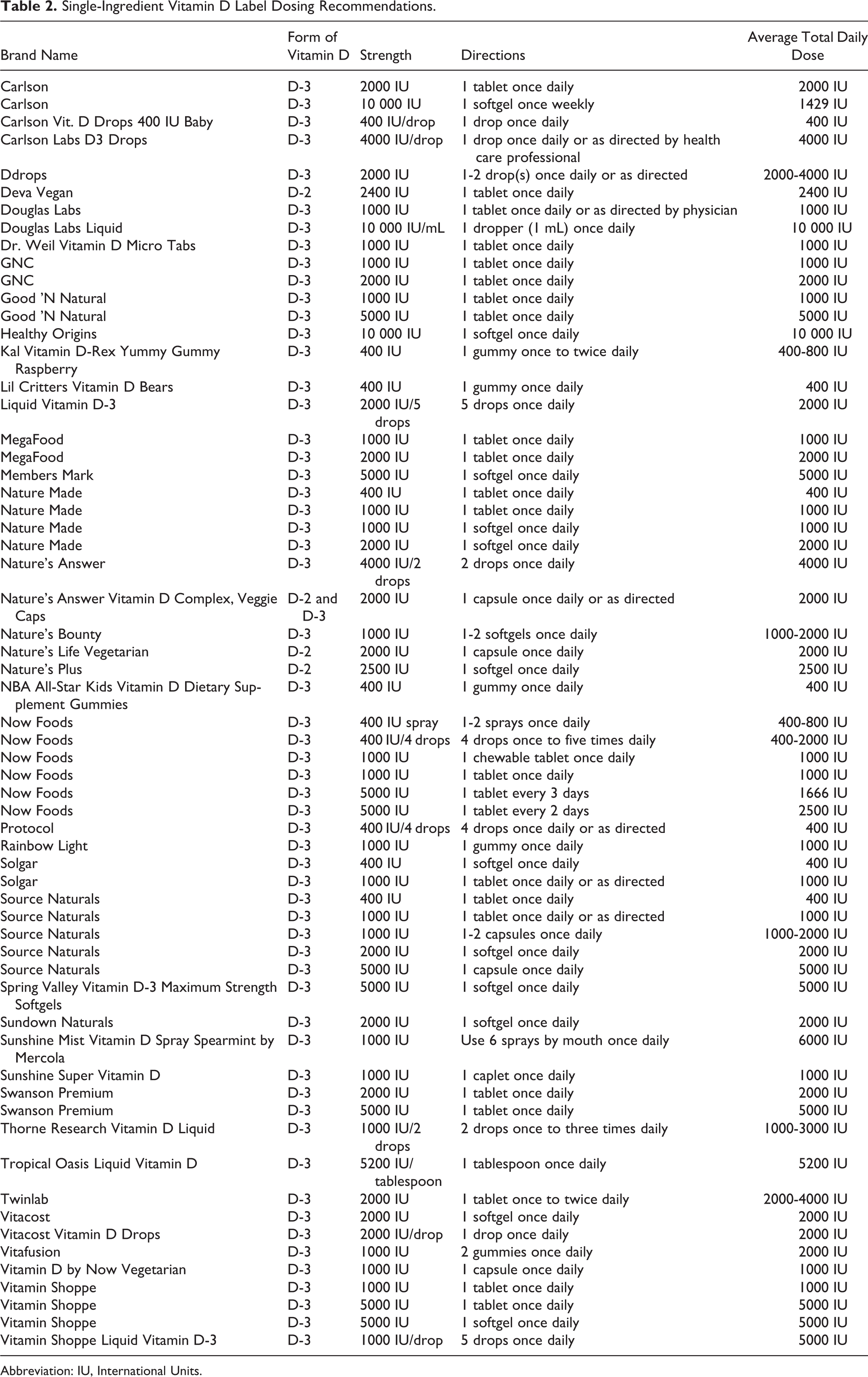
The systematic search identified 62 single-ingredient vitamin D products (Table 2). Of these, 58 contained vitamin D3 (cholecalciferol), 3 contained vitamin D2 (ergocalciferol), and 1 contained both vitamins D3 and D2.
Single-Ingredient Vitamin D Label Dosing Recommendations.
Abbreviation: IU, International Units.
The most common strength of vitamin D products was 1000 IU, followed by 2000 IU. The most common daily dose recommended by labeling of vitamin D products was 1000 IU, followed by 2000 IU. Recommended daily dosing ranged from 400 to 10 000 IU. The labeling on 51 of the products identified recommended a set daily dose (eg, 1 tablet daily or as directed), whereas the labeling on 8 products allowed for a range of dosing options (eg, 2 drops 1 to 3 times daily). The labeling on 3 identified vitamin D products recommend an extended dosing frequency, allowing for every other day, every 3 days, and weekly dosing, respectively.
North American guidelines that recommend vitamin D dosing range from 400 IU daily to 1000 IU daily, depending on age. Canadian guidelines recommend the highest vitamin D dosing. For adults 50 years of age and older, the National Osteoporosis Foundation and Osteoporosis Canada recommend 800 to 1000 IU daily. 7,8 The Canadian Cancer Society recommends 1000 IU daily for all adults at risk for vitamin D deficiency. 11
Of the vitamin D products identified, 24 (39%) recommended a maximum dose within the range of 400 to 1000 IU daily. There were 38 (61%) products with a recommended maximum dose more than 1000 IU daily. There were 19 (31%) products with a maximum recommended dose more than 2000 IU daily.
Discussion
Vitamin D insufficiency in North America is more common than most health professionals realize. Some estimates suggest that it could be as high as 80%. 13 Vitamin D status is most commonly measured using 25-hydroxyvitamin D blood levels. Vitamin D insufficiency is typically considered to be levels <20 ng/mL. However, some argue that levels <30 ng/mL should be considered insufficient. Most laboratories consider 20 to 100 ng/mL to be within the normal range. 14,15
The most appropriate vitamin D dose to prevent insufficiency, maintain appropriate levels, and prevent fractures is highly debated. The Institute of Medicine suggests that 600 to 800 IU daily is the minimum amount needed to prevent osteomalacia. 4 However, higher doses are probably needed to prevent fractures. A recent meta-analysis suggests that doses of about 800 to 2000 IU daily reduce hip fracture in people 65 years of age or older. 16 Still, even with vitamin D dosing of 1600 IU daily, as much as 20% of patients will still have levels less than 30 ng/mL. 17
Our data show that the labeled dosing recommendations of commercially available vitamin D supplements are largely inconsistent with recommendations from national guidelines in North America. The majority of the products recommend dosing that is substantially higher than what is commonly recommended by guidelines. However, given the controversy regarding the most appropriate dose and the data suggesting that dosing up to 2000 IU daily may be needed for fracture prevention, the higher recommended dosing is not necessarily unreasonable.
Toxicity from vitamin D is uncommon. The biggest concern related to vitamin D overdose is hypercalcemia, but this adverse effect typically does not occur until 25-hydroxyvitamin D levels exceed 150 ng/mL. This level typically would not be achieved until vitamin D doses of 10 000 IU daily for an extended period was used. 13
Health care providers should be cognizant that labeled dosing on vitamin D supplements may not be consistent with what is recommended by treatment guidelines. Therefore, it is particularly important for health care providers to give a specific dose to be taken when recommending or prescribing a vitamin D supplement rather than to have patients follow the labeled dosing recommendations provided by supplement manufacturers.
Footnotes
Author Contributions
PJG, DJH, and AMA completed the first draft of the article. All authors were involved in reviewing, revising, and development of the final article. PJG designed and conceptualized the project. DJH, AMA, and PJG collected and analyzed the study data.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not required as no human subjects or animals were involved in the analysis.
