Abstract
A siddha polyherbal preparation consisting of 5 medicinal plants, namely, Asparagus racemosus, Emblica officinalis, Salacia oblonga, Syzygium aromaticum, and Tinospora cordifolia, in equal ratio, was formulated to examine the molecular mechanism by which it exhibits antidiabetic effects in the liver of high-fat and fructose-induced type 2 diabetic rats. The polyherbal preparation treated type 2 diabetic rats showed an increase in insulin receptor, Akt, and glucose transporter2 mRNA levels compared with diabetic rats. Insulin receptor, insulin receptor substrate-2, Akt, phosphorylated Akt substrate of 160kDaThreonine642, α-Actinin-4, β-arrestin-2, and glucose transporter2 proteins were also markedly decreased in diabetic rats, whereas the polyherbal preparation treatment significantly improved the expression of these proteins more than that of metformin-treated diabetic rats. The expression pattern of insulin signaling molecules analyzed in the present study signifies the therapeutic efficacy of the siddha polyherbal preparation.
Type 2 diabetes is now a pandemic and shows no signs of abatement. 1 Chronic fuel surfeit is the primary pathogenic event that drives the development of type 2 diabetes in genetically and epigenetically susceptible people. 2,3 In modern societies, both increased consumption of unhealthy high-calorie diets and sedentary behavior have been recorded, which form the basis for the occurrence of type 2 diabetes. 4 It has long been regarded as a chronic progressive condition, which can be ameliorated but not cured. The burden of this disorder is enormous, owing to its rapidly increasing global prevalence, the devastating damage it can do to many organs of the body, as well as direct and indirect costs. 1 Increased endogenous glucose production, predominantly of hepatic origin, is a major determinant of fasting hyperglycemia in type 2 diabetes. Lack of suppression of glucose production after having food contributes to fed-state hyperglycemia. 3 The mechanisms underlying this fed-state hyperglycemia are complex, involving increased supply of gluconeogenic substrates from peripheral tissues, an effect of raised concentration of nonesterified fatty acids to activate hepatic gluconeogenesis, and hepatic response to raised concentration of glucagon. 3,5
The liver facilitates sustaining normal blood glucose concentration in the fasting and postprandial states, whereas a decreased level of insulin leads to glycogenolysis and an increase in hepatic glucose production. 6 Abnormalities of triglyceride storage and lipolysis in insulin-sensitive tissues, such as liver, are the early manifestation of conditions characterized by insulin resistance and are detectable earlier than fasting hyperglycemia. 7 The 3 major tissues largely responsible for clearing glucose from the blood in healthy individuals, namely, liver, muscle, and adipose tissue, all become insulin resistant in type 2 diabetes. It has been demonstrated that targeted inactivation of the insulin receptor gene, specifically in the liver, leads to diabetes-like symptoms in mice and suggests that insulin has a direct role in regulating liver metabolism. 8 In another study, development of insulin resistance in the liver involves selective inactivation of the capacity of insulin to block hepatic glucose production and the ability of the hormone to stimulate fatty acid synthesis is retained. 9 These studies emphasize the significance of insulin receptor signaling in cells.
Gluconeogenesis is a life-sustaining process for maintaining blood glucose levels within the physiologic range and providing the solitary fuel source for the brain, testes, and erythrocytes during starvation. 10 It occurs mainly in the liver in a metabolic pathway that is tightly regulated by insulin. When hepatic insulin signaling goes awry, gluconeogenesis becomes uncontrolled, resulting in excessive glucose production and causing fasting hyperglycemia in diabetes. 10 While it has a variety of functions, the liver plays a distinctive role in managing carbohydrate metabolism by retaining glucose concentrations in an optimum range.
In countries such as India and China, use of herbal medicines is a very common practice from ancient time, and herbal medicines are considered to be much safer and less expensive therapeutic strategies for the treatment of various diseases. 11 A proper scientific investigation of traditional herbal remedies, in particular, can provide valuable leads for the development of alternative drugs and strategies for the management of diabetes. Isolation of pure compounds and evaluation of its pharmacological properties leading to drug discovery is a long, tedious, time-consuming, and an expensive process. Natural products are rich source of ligands for nuclear receptors and are promising therapeutic agents in clinical practice. In particular, plants provide an abundant source of biologically active molecules that have played critical roles in pharmacology. 12,13
Currently available synthetic antidiabetic agents produce serious side effects, such as hypoglycemic coma and hepatorenal disturbances. 14 –19 While high saturated fat and fructose containing Western diet is thought to contribute to an increased lifetime risk of certain diseases, such as cancer and diabetes, plant-based diets offer protective effects. 20 –23 Increasing side effects caused by chemotherapeutic agents and drugs have induced attention toward the use of natural products–based drugs to minimize hazards caused by synthetic drugs. In fact, phytotherapy has been widely used because of the low cost and the easy availability of medicinal plants.
The present study was designed to evaluate the efficiency and the molecular mechanisms underlying the antidiabetic effects of a polyherbal preparation (500 mg/kg body weight) in the liver of high-fat and fructose-induced type 2 diabetic rats. 24,25 Metformin is a potent antihyperglycemic agent widely used in the management of type 2 diabetes, whose main actions are the suppression of gluconeogenesis and the enhancement of glucose uptake and insulin sensitivity, making the body to use glucose in an effective way. 26 Therefore, in the present study, we compare the antidiabetic efficacy of metformin with that of a polyherbal preparation.
Materials and Methods
Chemicals and Supplies
All chemicals and reagents used in the present study were of molecular and analytical grade and were purchased from Sigma Chemical Company, St Louis, MO, USA; Amersham Biosciences, Buckinghamshire, UK; and Sisco Research Laboratories, Mumbai, India. 14 C-glucose and 14 C-2-deoxyglucose were purchased from the Board of Radiation and Isotope Technology, Mumbai, India. Total RNA isolation reagent from Invitrogen (Grand Island, NY), reverse transcriptase enzyme from New England Biolabs (Ipswich, MA), and polymerase chain reaction (PCR) mastermix from Promega (Madison, WI) were used for the present study. The primers for β-Actin, insulin receptor, glucose transporter2 (GLUT2), Akt, and the β-Actin monoclonal antibody were purchased from Sigma Chemical Company. Polyclonal insulin receptor β-subunit, Insulin receptor substrate-2 (IRS-2), p-AS160Threonine642, Akt1/2/3 and p-AktSerine473, β-arrestin-2, α-Actinin-4, and glucose transporter2 antibodies were purchased from Santa Cruz Biotechnology Inc (Santa Cruz, CA). Akt substrate of 160 kDa (AS160) monoclonal antibody was purchased from Cell Signaling Technology Inc (Danvers, MA).
Induction of Type 2 Diabetes
Adult male rats were made diabetic (type 2) by a single intraperitoneal injection of streptozotocin (35 mg/kg body weight), after feeding the animals with high-fat diet 24 containing cholesterol (2 g), cholic acid (1 g), coconut oil (30 mL), along with standard rat feed (100 g) and high-fructose feeding through drinking water (25%) for 30 days. The low dose of streptozotocin was given to generate a slight trauma to beta cells of pancreas to mimic the chronic hypoinsulinemic insulin resistant condition. Animals were maintained as per the National Guidelines and Protocols approved by the Institutional Animal Ethical Committee (IAEC No. 03/019/2009, dated April 1, 2009).
Experimental Design
Rats were divided into the following groups:
Group 1: Control.
Group 2: Rats were made diabetic (type 2) by a single intraperitoneal injection of streptozotocin (35 mg/kg body weight) after 30 days of high-fat diet and high-fructose feeding through drinking water (25%).
Group 3: Diabetic (type 2) rats were treated with the polyherbal preparation (500 mg/kg body weight) twice a day (8.00
Group 4: Diabetic (type 2) rats were treated with metformin (50 mg/kg bodyweight/day) for 30 days after 5 days of streptozotocin treatment.
Group 5: Control rats were treated with the polyherbal preparation (500 mg/kg body weight) twice a day (8.00
Isolation of Total RNA
Total RNA was isolated from control and experimental samples using total RNA isolation reagent from Invitrogen. Briefly, 100 mg fresh tissue was homogenized with 1 mL total RNA isolation reagent and the homogenate was transferred immediately to a microfuge tube and kept at 4°C for 5 minutes to permit the complete dissociation of nucleoprotein complexes. Then, 0.2 mL of chloroform was added, vortexed vigorously for 15 seconds, and placed on ice at 4°C for 10 minutes. The homogenates were centrifuged at 12 000g for 15 minutes at 4°C. The aqueous phase was carefully transferred to a fresh microfuge tube and an equal volume of isopropanol was added and stored for 10 minutes at 4°C. Samples were centrifuged at 12 000g for 10 minutes. The supernatant was removed and the RNA pellet was washed twice with 75% ethanol by vortexing and subsequent centrifugation for 5 minutes at 7500g at 4°C. RNA pellets were mixed with 50 μL of autoclaved Milli-Q water. The concentration and purity of RNA were determined spectrophotometrically at A260/280 nm. A ratio of absorbance of >1.8 was considered as good-quality RNA. 27 The purity of RNA obtained was 1.85. The yield of RNA is expressed in microgram.
Reverse Transcriptase Polymerase Chain Reaction (RT-PCR)
Total RNA was used for the synthesis of complementary DNA (cDNA). The following specific oligonucleotide primers were used for the generation of complementary DNAs. Rat insulin receptor (IR)
28
(amplicon size—224 bp) Sense primer: 5′-GCC ATC CCG AAA GCG AAG ATC-3′ Anti-sense primer: 5′-TCT GGG GAG TCC TGA TTG CAT-3′ Rat Akt (amplicon size—146 bp) Sense primer: 5′-GGA AGC CTT CAG TTT GGA TCC CAA-3′ Anti-sense primer: 5′-AGT GGA AAT CCA GTT CCG AGC TTG-3′ Rat glucose transporter-2
29
(amplicon size—238 bp) Sense primer: 5′-CTC GGG CCT TAC GTG TTC TTC CTT-3′ Anti-sense primer: 5′-TGG TTC CCT TCT GGT CTG TTC CTG-3′ Rat β-actin (amplicon size—96 bp) Sense primer: 5′-AAG TCC CTC ACC CTC CCA AAA-3′ Anti-sense primer: 5′-AAG CAA TGC TGT CAC CTT CCC-3′
For the first strand cDNA synthesis, 2 μg of RNA template was added with master mix containing 10 μL of RT-PCR buffer, 2.0 μL of deoxynucleotide triphosphates mix, 2.0 μL of RT-PCR enzyme, appropriate volume of 0.6 μM primer (forward and reverse each), and made up to 50 μL with RNase-free water. RT-PCR was performed using the thermal cycler (Eppendorf) programmed as reverse transcriptase reaction at 50°C for 30 minutes, initial PCR activation at 95°C for 15 minutes, denaturation at 94°C for 1.5 minutes, annealing at 58°C for 1.5 minutes, and extension at 72°C for 3 minutes. Thirty-five cycles were performed, with final extension at 72°C for 10 minutes. Finally, the reaction mixture containing PCR products were separated by 2% agarose gel electrophoresis along with 100 bp marker DNA. Gels were densitometrically (Bio-Rad, Hercules, CA) scanned and the cDNAs were normalized to that of the house keeping gene or internal control (β-actin), which was co-amplified along with the cDNA of interest.
Western Blot Analysis
Protein isolation
For the present study, 2 different fractions of proteins (cytosolic and plasma membrane) were isolated by a previously published method. 30 About 100 mg of tissue was homogenized in 1.5 mL of buffer-A containing 10 mM/L NaHCO3 (pH 7.0), 250 mM/L sucrose, 5 mM/L NaN3, protease inhibitor cocktail (Sigma Chemical Company), and 100 μM/L phenylmethylsulfonyl fluoride using a homogenizer (Proscientific, Oxford, CT). The resulting homogenate was centrifuged at 1300g for 10 minutes. The supernatant was centrifuged at 190 000g for 1 hour. The resultant supernatant was saved and sampled as a cytosolic fraction for protein analysis. The pellet was resuspended in buffer-A and applied on discontinuous sucrose gradients (25%, 32%, and 35% w/w) and centrifuged at 150 000g for 16 hours. Plasma membrane at 25% to 32% interface was recovered, diluted with sucrose-free buffer-A, and centrifuged at 190 000g for 1 hour. Plasma membrane fraction (pellet) was resuspended in buffer-A and kept at −80°C until used for protein expression analysis. Protein concentration was estimated as per the method of Lowry et al 31 using bovine serum albumin as a standard. The insulin receptor protein level was evaluated in plasma membrane fraction. Other insulin signaling molecules were seen in cytosolic fraction. Glucose transporter2 level was assessed both in cytosolic and plasma membrane fractions.
Separation of proteins
Proteins were separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis as described by Laemmli. 32 Briefly, each sample (50 μg) was subjected to heat denaturation at 96°C for 5 minutes with Laemmli buffer (1.25 mL of 0.5 M Tris–HCl [pH 6.8], 2.5 mL glycerol, and 2 mL of 10% [w/v] sodium dodecyl sulfate were added to 0.2 mL of 0.5% [w/v] bromophenol blue and the total volume was brought to 9.5 mL with 3.55 mL deionized water and stored at room temperature; 50 μL of β-mercaptoethanol was added to 950 μL of Laemmli buffer prior to use). Proteins were resolved by sodium dodecyl sulfate–polyacrylamide gel electrophoresis on 10% polyacrylamide gels and then transferred to polyvinylidene fluoride membrane (Amersham Biosciences, Buckinghamshire, UK). The membrane was blocked with 5% blocking buffer (Amersham Biosciences) in Tris-buffered saline and Tween 20 for 1 hour at room temperature followed by incubation with primary antibody to insulin receptor, Akt, p-AktSerine473, Akt substrate of 160kDa, phosphorylated-Akt substrate of 160kDaThreonine642, β-arrestin-2, α−Actinin-4, Sterol regulatory element binding protein-1c (SREBP-1c), and glucose transporter2 at a dilution of 1:1000. The membrane was subjected to repeated washing 3 times with Tris-buffered saline and Tween-20 and then incubated for 1 hour in horseradish peroxidase–conjugated mouse/rabbit secondary antibody at the dilution of 1:7500 in Tris-buffered saline and Tween-20 solution. The membrane was again subjected to repeated washing 3 times with Tris-buffered saline and Tris-buffered saline - Tween-20. Protein bands were visualized in Chemidoc instrument (Bio-Rad) using enhanced chemiluminescence reagents (Thermo Scientific). The detected bands were quantified by Quantity One software (Bio-Rad). Later, the membranes were incubated in stripping buffer (5 mL, containing 62.5 mM Tris–HCl [pH 6.7], 1 g sodium dodecyl sulfate, and 0.34 mL β-mercaptoethanol) at 55°C for 25 minutes. Following this, the membranes were reprobed using β-actin antibody (1:5000). In the present study, rat β-actin was used as the loading control.
Insulin Receptor Assay
Insulin receptors were quantified by the method of Torlinska et al. 33 Plasma membrane was isolated by the method of Dombrowski et al. 30 The final pellet of the plasma membrane was resuspended in 0.04 M Tris–HCl buffer (pH 7.4) containing 0.1% bovine serum albumin. All procedures mentioned above were carried out at 4°C. Protein concentration was estimated 31 using bovine serum albumin as a standard. Membrane preparation (100 μg protein) was incubated at 4°C for 16 hours with increasing concentration of 125I-labeled porcine insulin in a final volume of 0.5 mL of 0.04 M Tris buffer (pH 7.4) containing 0.1% bovine serum albumin. Nonspecific binding was determined in the presence of excess (1000-fold) unlabeled insulin. Bound and free fractions of insulin were separated by centrifugation at 20 000×g for 10 minutes and then the radioactivity of the pellets was determined using a gamma counter. Total number of receptors as a binding capacity was determined by the Scatchard analysis. 34 The receptor concentration is expressed as fmol/mg protein.
14 C-2-Deoxyglucose Uptake
Glucose uptake in control and experimental liver samples were estimated by the standard method using 14 C-2-deoxyglucose. 35 Briefly, 10 mg of tissue was incubated in 5 mL of Dulbecco’s modified Eagle medium with 5 mmol/L glucose for 30 minutes under the condition of 95% air, 5% carbon dioxide. After this, the tissue was washed and incubated for 30 minutes in glucose-free medium for 30 minutes with or without 100 nmol/L of insulin. Glucose uptake was initiated by the addition of 2 μL of 0.05 μCi 14 C-2-deoxyglucose. At the end of 10 minutes of incubation, tissue was removed, rapidly rinsed in isotope-free medium, homogenized in 2 mL of 5% trichloroacetic acid, and placed in scintillation vials containing 5 mL of scintillation fluid and counted in a beta counter. Results are expressed as counts per minute of 14 C-2-deoxy- glucose/10 mg tissue.
14 C-Glucose Oxidation
After perfusion, the liver was dissected out and used for the assay of 14 C-glucose oxidation as described earlier. 36 Briefly, 10 mg of tissue was weighed and placed in a 2-mL ampoule containing 180 μL Dulbecco’s modified Eagle medium (pH 7.4), 10 U penicillin in 10 μL Dulbecco’s modified Eagle medium, and 0.5 μCi 14 C-glucose. Then the ampoule was tightly closed with rubber cork containing carbon dioxide trap and incubated at 37°C. The carbon dioxide traps were replaced every 2 hours and 0.1 mL of 1 N sulfuric acid was added to the ampoule on the removal of the second trap to halt further metabolism. The ampoule was again closed for 1 hour before the third and final trap was removed and all the carbon dioxide traps were placed in the scintillation vials containing 10 mL of scintillation fluid and were counted with a beta counter. Results are expressed as counts per minute of 14 CO2 released/10 mg tissue.
Statistical Analysis
The data were subjected to statistical analysis using one way analysis of variance and Duncan multiple range test to assess the significance of individual variations between the control and treatment groups using a computer-based software (SPSS 7.5 for Windows Student Version; SPSS, Chicago, IL). In the Duncan test, the significance was considered at the level of P < .05.
Results
Effect of Polyherbal Preparation on Insulin Receptor mRNA and Protein Levels and Insulin Receptor Binding in the Liver of Type 2 Diabetic Rats
Insulin receptor mRNA levels were diminished in type 2 diabetic rats (Figure 1). Polyherbal preparation and metformin treatment augmented the insulin receptor mRNA levels significantly when compared with diabetic rats. The polyherbal preparation showed a significant improvement in insulin receptor protein in cytosol (Figure 2), which was found to be decreased in the diabetic group. Figure 3 depicts the effect of the polyherbal preparation on insulin receptor concentration in the plasma membrane of liver. Polyherbal preparation and metformin treatment significantly improved the insulin receptor concentration in diabetic rats. Administration of the polyherbal preparation to normal rats produced no significant change in these parameters.
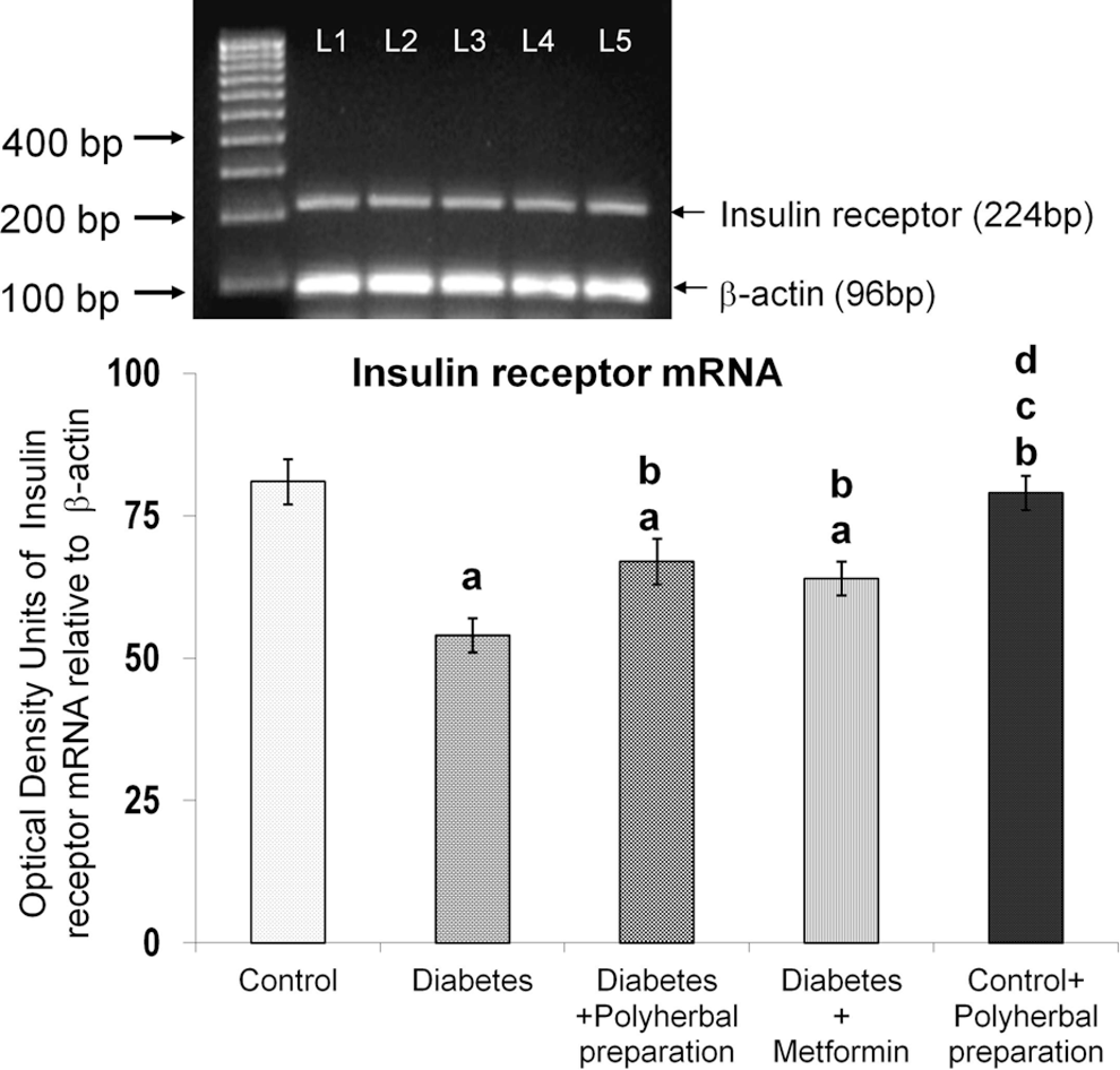
Effect of polyherbal preparation on insulin receptor mRNA level in the liver of type 2 diabetic adult male rats.

Effect of polyherbal preparation on insulin receptor protein level in the liver of type 2 diabetic adult male rats.
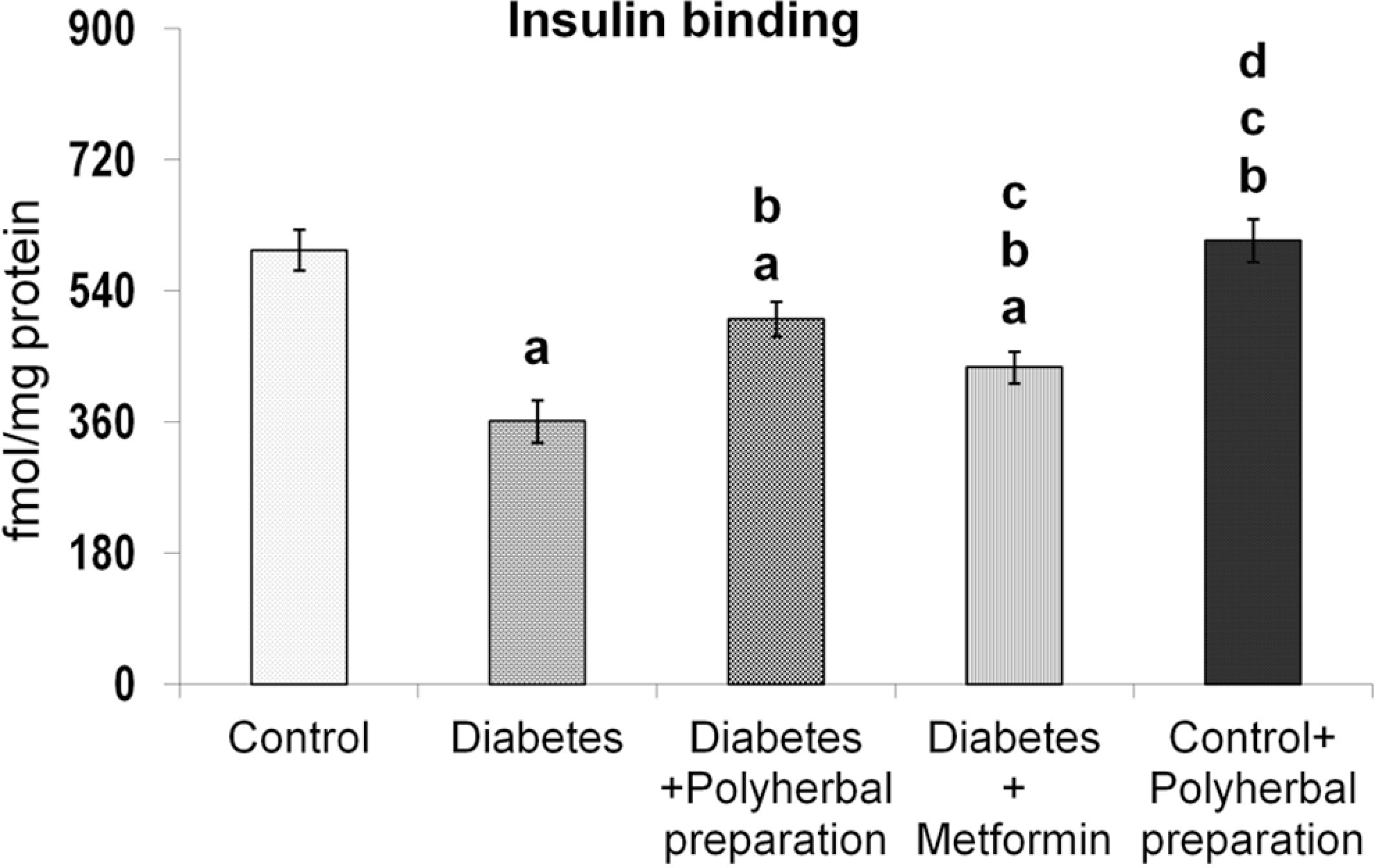
Effect of polyherbal preparation on insulin binding in the liver of type 2 diabetic adult male rats.
Effect of Polyherbal Preparation on Insulin Receptor Substrate-2 Protein Level in the Liver of Type 2 Diabetic Rats
The decreased insulin receptor substrate-2 protein observed in diabetic rats was significantly improved after polyherbal preparation treatment, which was better than metformin treatment (Figure 4). Treatment with the polyherbal preparation to control rats induced no significant change.
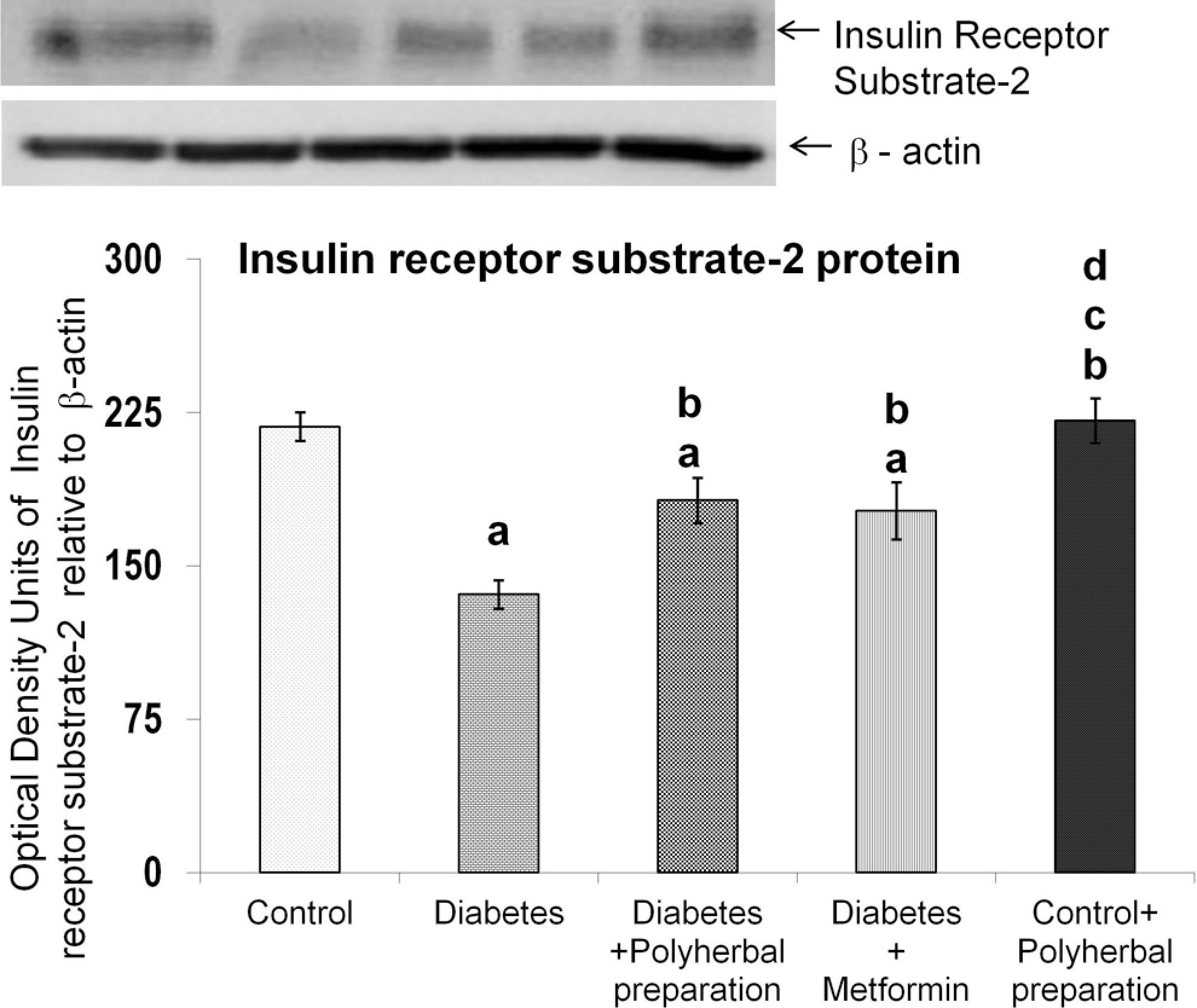
Effect of polyherbal preparation on insulin receptor substrate-2 protein level in the liver of type 2 diabetic adult male rats.
Effect of Polyherbal Preparation on Akt mRNA and Protein Level in the Liver of Type 2 Diabetic Rats
In diabetic rats, a significant decrease was observed in Akt mRNA levels, but the polyherbal preparation treatment significantly restored it to near normal level (Figure 5). Akt protein was found to be decreased in the diabetic group, but the polyherbal preparation treatment improved it, better than metformin treatment (Figure 6). The response to phosphorylated-Aktserine473 protein level is similar to that of Akt protein, attesting the fact that the polyherbal preparation treatment enhanced the Akt phosphorylation, which was slightly more than that of metformin treatment (Figure 7).
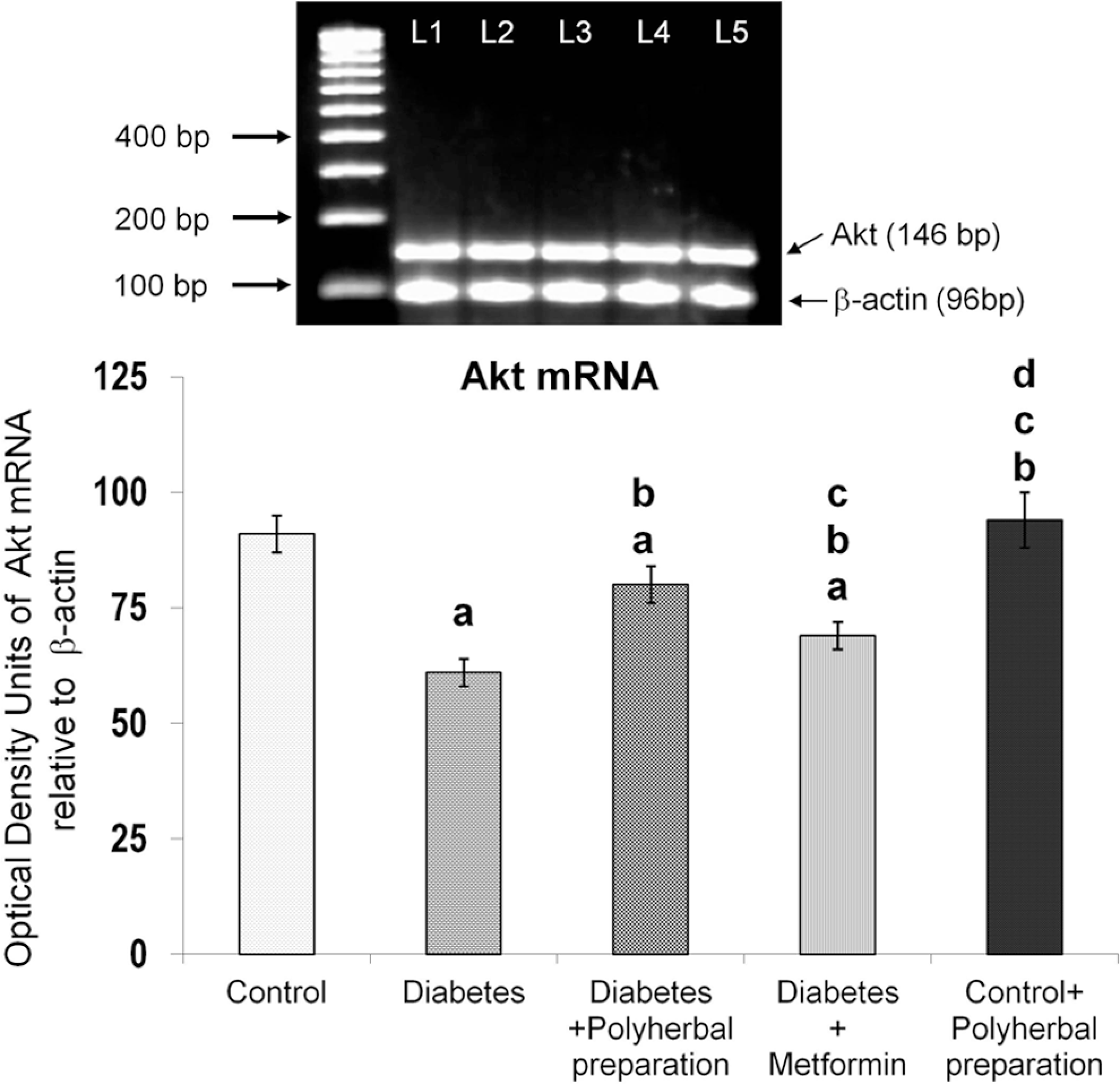
Effect of polyherbal preparation on Akt mRNA level in the liver of type 2 diabetic adult male rats.
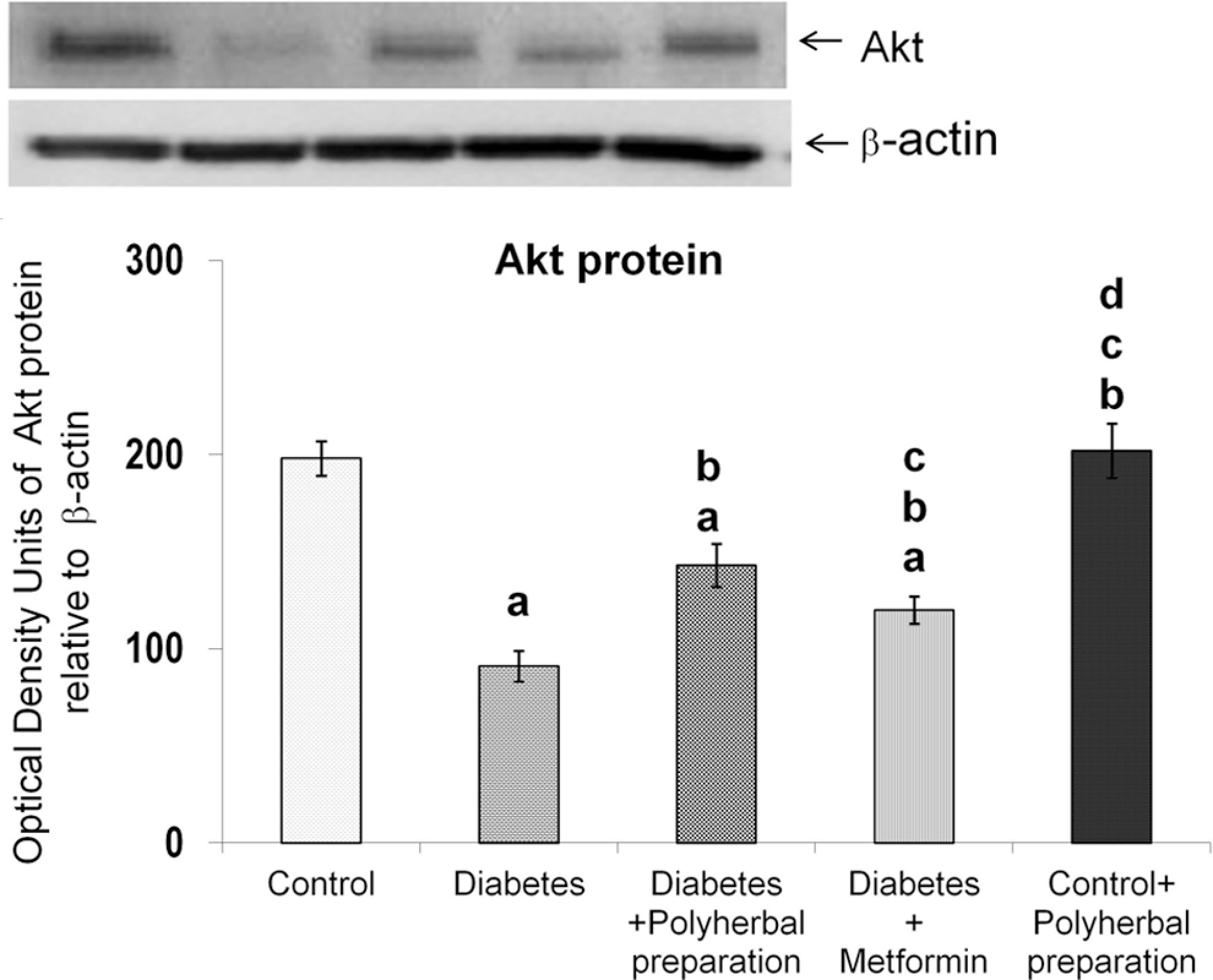
Effect of polyherbal preparation on Akt protein level in the liver of type 2 diabetic adult male rats.
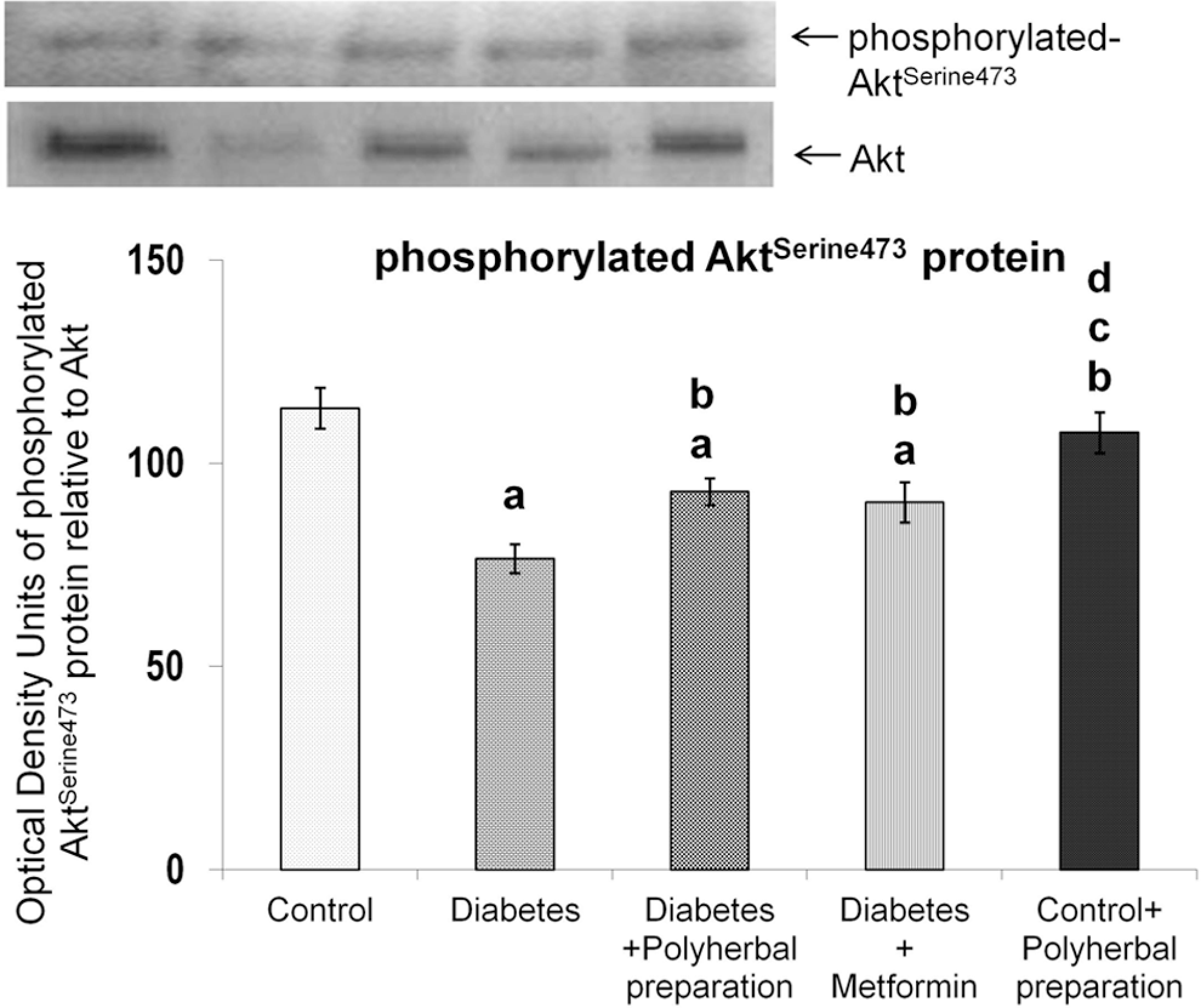
Effect of polyherbal preparation on phosphorylated-AktSerine473 protein level in the liver of type 2 diabetic adult male rats.
Effect of Polyherbal Preparation on Akt Substrate 160 kDa (AS160) Protein Level in the Liver of Type 2 Diabetic Rats
Figure 8 depicts the level of Akt substrate 160 kDa protein, the substrate of Akt. Diabetic rats showed a marked increase in Akt substrate 160 kDa, whereas polyherbal preparation and metformin treatment significantly restored it to near normal level. Polyherbal and metformin treatment significantly increased the phosphorylated Akt substrate 160kDaThreonine642 (Figure 9) level than type 2 diabetic rats. AS160 phosphorylation was slightly improved in metformin-treated rats, whereas the polyherbal preparation treatment showed an additional enhancement in Akt substrate 160 kDa phosphorylation.

Effect of polyherbal preparation on Akt substrate of 160 kDa protein level in the liver of type 2 diabetic adult male rats.

Effect of polyherbal preparation on phosphorylated-Akt substrate of 160kDaThreonine642 protein level in the liver of type 2 diabetic adult male rats.
Effect of Polyherbal Preparation on α-Actinin-4 Protein Level in the Liver of Type 2 Diabetic Rats
Type 2 diabetic rats showed a significant decrease in α-Actinin-4 protein level compared with control. However, a significant enhancement was observed in polyherbal preparation–treated and metformin-treated diabetic rats (Figure 10). Treatment with polyherbal preparation to control rats induced no significant change.

Effect of polyherbal preparation on α-Actinin-4 protein level in the liver of type 2 diabetic adult male rats.
Effect of Polyherbal Preparation on Phosphatase and Tensin Homolog Protein Level in the Liver of Type 2 Diabetic Rats
Phosphatase and tensin homolog protein was markedly increased in diabetic rats. However, significant decrease was observed in both polyherbal preparation–treated and metformin-treated diabetic rats (Figure 11). Polyherbal treatment to control rats had no influence on phosphatase and tensin homolog protein concentration. Administration of the polyherbal preparation to normal rats produced no significant change.

Effect of polyherbal preparation on phosphatase and tensin homolog protein level in the liver of type 2 diabetic adult male rats.
Effect of Polyherbal Preparation on β-arrestin-2 Protein Level in the Liver of Type 2 Diabetic Rats
β-arrestin-2 protein level was significantly decreased in type 2 diabetic rats compared with control (Figure 12). Polyherbal preparation treatment to type 2 diabetic rats reverted the β-arrestin-2 protein to near normal level than metformin treatment, whereas the polyherbal treatment to control rats did not induce any change.

Effect of polyherbal preparation on β-arrestin-2 protein level in the liver of type 2 diabetic adult male rats.
Effect of Polyherbal Preparation on SREBP-1c Protein Level in the Liver of Type 2 Diabetic Rats
In diabetic rats, a significant increase in Steroid Regulatory Element Binding Protein-1c (SREBP-1c) was recorded (Figure 13), whereas the polyherbal preparation treatment markedly decreased the expression of SREBP-1c than diabetic rats and such an effect was more than in metformin treatment.

Effect of polyherbal preparation on sterol regulatory element binding protein-1c protein level in the liver of type 2 diabetic adult male rats.
Effect of Polyherbal Preparation on Glucose Transporter2 mRNA and Protein Levels in the Liver of Type 2 Diabetic Rats
Figure 14 shows the expression levels of glucose transporter2 mRNA & protein in the cytosol (Figure 15), and plasma membrane (Figure 16) fractions of liver. Glucose transporter2 mRNA level was decreased in diabetic group and a significant improvement was seen in all the treatment groups. Decreased glucose transporter2 protein in the cytosolic fraction (Figure 15) of diabetic rats was improved by polyherbal preparation treatment. Plasma membrane glucose transporter2 protein level (Figure 16) showed a steep increase in diabetic rats but the polyherbal preparation treatment maintained the protein at near normal level.

Effect of polyherbal preparation on Glucose transporter-2 mRNA level in the liver of type 2 diabetic adult male rats.

Effect of polyherbal preparation on Glucose transporter-2 protein level (cytosol) in the liver of type 2 diabetic adult male rats.

Effect of polyherbal preparation on Glucose transporter-2 protein level (plasma membrane) in the liver of type 2 diabetic adult male rats.
Effect of Polyherbal Preparation on 14 C-2-Deoxy Glucose Uptake in the Liver of Type 2 Diabetic Rats
Type 2 diabetic rats showed a significant decrease in 14C-2-deoxy- glucose uptake (Figure 17). Improved glucose uptake was achieved in polyherbal preparation–treated and metformin-treated rats. Administration of polyherbal preparation to normal rats produced no significant change in 14C-2-deoxyglucose uptake.

Effect of polyherbal preparation on 14C-2-deoxyglucose uptake in the liver of type 2 diabetic adult male rats.
Effect of Polyherbal Preparation on 14 C-Glucose Oxidation in the Liver of Type 2 Diabetic Rats
There was a significant reduction of 14 C-glucose oxidation in type 2 diabetic rats (Figure 18). The polyherbal preparation treatment caused a significant rise in 14C-glucose oxidation level, more than that of metformin treatment. Administration of the polyherbal preparation to normal rats produced no significant change in 14C-glucose oxidation.

Effect of polyherbal preparation on 14C-glucose oxidation in the liver of type 2 diabetic adult male rats.
Discussion
Glucose homeostasis is vital for the balanced use of energy sources, such as carbohydrates, amino acids, and fatty acids by the body. In addition, high circulating glucose concentrations are unfavorable because of the chemical reactivity of glucose with amino-groups in proteins. Insulin plays a critical role in maintaining glucose homeostasis. High portal insulin concentrations rapidly trigger stimulation of glycogen synthesis, lipogenesis, inhibition of very low density lipoprotein secretion, and suppression of gluconeogenesis and glycogenolysis. 37 These processes are regulated by phosphorylation and dephosphorylation of varieties of proteins involved in the metabolic pathways, expression of several genes, and mRNA stability. 38,39 In the liver, insulin signaling is a crucial pathway for the regulation of glycogen synthesis, glycogenolysis, gluconeogenesis, and lipogenesis. 40 Increased hepatic glucose production will enhance insulin release, causing the suppression of hepatic insulin receptor substrate-2 and insulin-mediated inhibition of gluconeogenesis. High-fat diet has been known to increase free fatty acid level in the circulation and the same can be attributed to significant reduction in insulin receptor mRNA and protein expression in the liver. 41 Insulin-stimulated tyrosine kinase activity per insulin receptor was reported to be significantly lowered in high-fat-fed animals, accompanied by diminished autophosphorylation of the β-subunit of the receptor and lower proportion of tyrosine-phosphorylated receptors. 42 Increased lipid peroxidation and free radical generation recorded in our earlier study 24 might have caused severe plasma membrane disruption, thus leading to decreased insulin receptor concentration. In addition, it has been reported that free fatty acid inhibits insulin receptor (IR) gene expression and contributes to a reduced amount of insulin receptor protein in insulin target cells. 43
Insulin receptor substrates-1 and -2 play crucial and complementary roles exhibiting differential regulation in hepatic insulin signaling. Insulin receptor substrate-1 is regulated at the protein level and is reported to be more closely linked to glucose homeostasis such as suppression of gluconeogenic genes, whereas insulin receptor substrate-2 controls lipid metabolism. 44 Studies on insulin receptor substrate and signaling in ob/ob mouse, a model of the insulin resistance of obesity and non-insulin-dependent diabetes mellitus, have reported that the levels of expression of both insulin receptor substrates-1 and 2 were decreased approximately 50% in muscle, whereas in liver, the decrease was significantly greater for insulin receptor substrate-2 (72%) than for insulin receptor substrate-1 (29%). 45 Significant improved levels of insulin receptor substrate-2 observed in the present study might be attributed to the therapeutic effects of the polyherbal preparation.
Insulin also promotes hepatic triglyceride synthesis via increasing the transcription factor steroid regulatory element-binding protein-1c, 46 and this lipogenic effect of insulin also appears to be mediated via the phosphoinositide-3-kinase pathway. Steroid regulatory element-binding protein-1c directly binds to the promoter of insulin receptor substrate-2 and suppresses its expression, leading to impaired insulin sensitivity. 47 Glycogen synthesis, a marker of insulin sensitivity, was suppressed by steroid regulatory element-binding protein-1c, whereas fatty acid synthesis was activated. Thus, increased level of steroid regulatory element-binding protein-1c could be responsible for lipotoxicity in the liver by causing both insulin resistance and fatty liver. 48 The decreased insulin receptor substrate-2 and increased steroid regulatory element-binding protein-1c are well correlated in type 2 diabetic rats. 9,46 Presence of numerous polyphenols 49 and flavonoids-like anthacyanidins 50 in the polyherbal preparation might have contributed to the decreased expression of steroid regulatory element-binding protein-1c in the polyherbal preparation–treated type 2 diabetic rats.
Akt has a crucial role in glucose metabolism. Of the 3 Akt isoforms, Akt-2 represents the predominant isoform in insulin responsive tissues and is most strongly associated with regulation of glucose homeostasis. 51 Glycogen synthase kinase-3β, an important Akt substrate, is inactivated by phosphorylation, thereby leading to increased glycogen accumulation. 52 Akt phosphorylation and inhibition of glycogen synthase kinase-3β also promotes sterol regulatory element binding proteins, which induces transcription of genes involved in cholesterol and fatty acid biosynthesis. 53 Similarly, Akt phosphorylation of phosphofructokinase stimulates glycolysis. 54 Furthermore, Akt activation enhances the association of hexokinase isoforms in the mitochondria, where they more readily phosphorylate glucose. 55 Glucose-6-phosphate can be stored by conversion to glycogen or catabolized to produce cellular energy through glycolysis; Akt signaling is known to regulate both these processes. 53 Moreover, through regulation of the Forkhead box protein O1 (a transcription factor that plays important roles in regulation of gluconeogenesis and glycogenolysis by insulin signaling), Akt inhibits hepatic glucose production. In hepatocytes, Akt inhibits gluconeogenesis and fatty acid oxidation through direct phosphorylation of peroxisome proliferator-activated receptor-gamma coactivator-1 alpha, a co-activator of Forkhead box protein O1. 56 Thus, Akt regulates many facets of glucose and lipid metabolism. Syzygium aromaticum regulates phosphoenolpyruvate carboxykinase and glucose-6-phosphatase gene expression by affecting the expression of peroxisome proliferator-activated receptor-gamma coactivator-1 alpha, which is an important co-activator for both of these genes. 57,58 Both Syzygium aromaticum extract and insulin downregulate the gene encoding peroxisome proliferator-activated receptor-gamma coactivator-1 alpha, suggesting the mechanism for downregulation of the phosphoenolpyruvate carboxykinase and glucose-6-phosphatase genes. 59 All these reports have suggested the efficacy of polyherbal preparation on lowering the hepatic glucose production and thereby improving glucose homeostasis in liver.
Akt substrate 160 kDa is phosphorylated at multiple sites by a variety of agonists that stimulate different protein kinases in intact cells, 60 –63 indicating its complex regulation. In the present study, polyherbal preparation treatment has significantly increased the Akt substrate 160 kDa protein level in liver than diabetic and metformin treatment. Further studies are required in this regard, because the role of Akt substrate 160 kDa has been correlated with glucose transporter4 translocation in skeletal muscle and not glucose transporter2 in the liver. Circumstantial evidences from this study can tend to hypothesize that increased Akt substrate 160 kDa can have a role in improvement of glucose homeostasis.
Experimental studies have documented that β-arrestin-2 and to a lesser extent β-arrestin-1 are downregulated in the liver of insulin-resistant and type 2 diabetic mouse models. 64 β-arrestin-2, Akt, and Src form a complex in vivo and interaction of Akt and Src with insulin receptor depends on the expression level of β-arrestin-2 (essential for proper insulin signaling and its action). 64 Additionally, it is also shown that both β-arrestin-1 and -2 are downregulated in the liver of type 2 diabetic humans, 65 which has been correlated with the findings in type 2 diabetic rats. Cytoskeletal protein α-Actinin-4, present in cellular architecture, 66 plays a major role in the translocation dynamics of glucose transporter4, but its role in liver is not clearly explained, but the polyherbal preparation treatment significantly enhances its expression.
Phosphatase and tensin homolog is a protein phosphatase involved in signal transduction and tumor suppression. 67 It is capable of dephosphorylating phosphatidylinositol (3,4,5)-trisphosphate, 68 and cells in which phosphatase and tensin homolog protein activity has been inhibited have elevated phosphatidylinositol-(3,4,5)-trisphosphate concentrations and higher levels of Akt phosphorylation. 69 Inhibition of phosphatase and tensin homolog protein expression dramatically reduced insulin concentrations and improved the performance of db/db mice during insulin tolerance tests and increased Akt phosphorylation in the liver in response to insulin. 70 These results suggest that phosphatase and tensin homolog protein plays a significant role in regulating glucose metabolism in vivo by negatively regulating insulin signaling. 70 In the present study, high-fat and fructose-induced diabetic state was associated with increased expression of phosphatase and tensin homolog protein but the polyherbal treatment significantly reduced the same, suggesting the antidiabetic property of flavonoids present in the polyherbal preparation. 71 –73
Glucose transporter2 (facilitated glucose transporter), also known as solute carrier family 2 member 2, a transmembrane carrier protein, is known to transport glucose across the hepatic plasma membrane in a bidirectional manner. 74 it is a glucose-sensitive gene in liver cells, 75 together with the genes encoding L-type pyruvate kinase, and S14, fatty acid synthase. 76 it is concomitantly regulated by glucose and by a lipogenic factor; the control of glucose transporter2 transcription thus represents a possible step on the way to glucolipotoxicity. 77 –79
Fruit, vegetables, and red wine provide the main sources of dietary polyphenols. 80 Quercetin, a naturally occurring flavonoid in Emblica officinalis, strongly inhibits apical glucose transporter2-mediated sugar uptake by diminishing its overexpression in Caco-2E cells. 81 When ingested with glucose, quercetin diminishes hyperglycemic excursions by 40% in diabetic Zucker fa/fa rats that express apical and basolateral glucose transporter2. 82 Insulin has been shown to trigger glucose transporter2 internalization, thereby slowing sugar uptake in the intestine during digestion, 83 and these reports supports our earlier finding that the polyherbal preparation favors improved glucose tolerance during oral glucose tolerance test. 24
Flavonoids are one of the numerous and widespread group of phenolics found in vegetables and fruits. 84 Some of them, due to their phenolic structure, are known to be involved in the healing process of free radical–mediated diseases including diabetes. 85 The basic pathological mechanisms in type 2 diabetes are insulin resistance leading to pancreatic stress and consequent pancreatic β-cell failure in susceptible individuals. To date, while glucose-lowering medications are successful in reducing blood glucose levels in the short term, they have not been able to prevent the progressive natural history of type 2 diabetes, as demonstrated by the continual loss of β-cell function over time. 86 Thus, polyphenols and flavonoid-rich polyherbal preparation prevents the type 2 diabetic rats from the detrimental effects of high-fat and high-fructose diet.
Recent findings have shown that dietary fructose facilitates metabolic derangement and induces oxidative damage. 87 –91 Also, numerous studies suggest that increased fructose consumption can be an important contributor to the metabolic syndrome. 92 –96 In addition, a high-fructose diet leads to a well-characterized metabolic syndrome, typically resulting in hyperinsulinemia, insulin resistance, hypertension, hypertriacylglycerolemia, dyslipidemia, and a decline in the level of high-density lipoproteins. 97,98 Also, animals fed high-fructose diet have been shown to exhibit altered lipid metabolism due to hepatic stress as a result of the burden of fructose metabolism. 88 Multiple mechanisms of berberine actions have been proposed such as increase in glycolysis 99 and insulin receptor expression level 100 and activation of 5′-adenosine monophosphate-activated protein kinase, 101,102 a pathway activated by metformin as well. 103 In view of these findings, it is suggested that the improvement in insulin resistance in polyherbal preparation–treated animals can be attributed to the presence of berberine in the formulation. 104
Polyphenols of green tea extracts have been shown to increase the genes (glucose transporter-1, -2, -4, insulin receptor substrate-1 and -2, Akt, phosphoinositide-3-kinase, glycogensynthase kinase-3β) involved in insulin signaling, which leads to enhancement of glucose uptake activity. 105 Enhanced glucose uptake and oxidation observed in the present study might be attributed to the presence of polyphenols 106 in the polyherbal preparation.
Conclusion
The present study clearly reveals that the chosen polyherbal preparation improves glucose homeostasis by decreasing the insulin-resistant condition in the liver of type 2 diabetic male rats. The expression pattern of insulin signaling molecules analyzed supports the efficacy and therapeutic values of the siddha polyherbal preparation. Hence, it is concluded that supplementation of the polyherbal preparation, along with regulated dietary habits, can offer an effective, holistic approach for the management of type 2 diabetes.
Footnotes
Acknowledgements
We thank Ms L. Sheerin Banu for correcting the manuscript for proper English usage.
Author Contributions
VB conceptualized the study, collected the data, developed the figures, analyzed the data, and wrote the first draft of the article. JS, SS, and CM developed the figures and helped in statistical analysis. KB conceptualized the study, critically analyzed and discussed the data, and corrected and reviewed the article.
Declaration of Conflicting Interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The financial assistance from DST-PURSE (Grant Letter No. RPO/PURSE/2009/4063, dated December 2, 2009, Department of Science & Technology, Government of India) and UGC-SAP-DRS (Sanction Letter No. F-3/58/2009/(SAPIII), dated September 17, 2010, University Grants Commission, New Delhi, India) are gratefully acknowledged.
Ethical Approval
Ethical clearance was obtained from the institutional animal ethical committee (IAEC No. 03/019/2009, dated April 1, 2009).
