Abstract
The purpose of this study was to investigate the effects of electroacupuncture at Zusanli acupoint on the enteric neuropathy in diabetic rats. Sprague–Dawley rats were divided into different groups depending on the total electroacupuncture span and frequency. The expression of nitric oxide synthase (nNOS), choline acetyltransferase (CHAT), protein gene product 9.5 (PGP9.5), and doublecortin was significantly decreased in the diabetic group compared with the control group. Long-term electroacupuncture at Zusanli with either high frequency or low frequency could increase the expression levels of nNOS, CHAT, PGP9.5, and doublecortin, and the increase was greater in the high-frequency group. But no obvious changes were seen in the short-term electroacupuncture groups. These results suggest that electroacupuncture at Zusanli can restore the deficiency of enteric neurons in diabetes partly but a comparative long duration of stimuli (6 weeks) is required. The increase of doublecortin may be involved in this positive process.
Introduction
Recently, it has been demonstrated that gastric empting is delayed in 30% to 50% of patients with long-standing type 1 and type 2 diabetes. 1 The current pharmacological treatment of diabetic gastroparesis remains unsatisfactory and oftentimes disappointing for both the patient and the clinician. 2
As a traditional Chinese medicine treatment, acupuncture has been practiced in China for several millennia. It is also a very important therapeutic method in the complementary medicine. Electroacupuncture is a modification of this technique. It stimulates acupoints with electrical current and appears to have more consistently reproducible results than manual manipulations. Some observations promise that electroacupuncture may help diabetic patients by accelerating gastric emptying and reducing symptom severity. 3 –5 Zusanli is the most commonly used acupoint in treating gastrointestinal diseases. In animal models, electroacupuncture at Zusanli has been reported to have ameliorating effects on gastric motility, such as acceleration of delayed gastric emptying, restoration of impaired gastric accommodation in vagotomized dogs, and relaxation of the gastric fundus in rats. 6 However, the underlying mechanism of the promotility effect of electroacupuncture at Zusanli is still unknown. Iwa et al 7 demonstrated the stimulatory effect of electroacupuncture at Zusanli on stress-induced delayed gastric emptying mediated via cholinergic pathways. Electroacupuncture at Zusanli is able to restore the rectal distension-induced impaired gastric slow waves and motor activities, possibly by enhancing vagal activity and is partially mediated via the opioid pathway. 8 Recently, Yin et al 6 found that electroacupuncture at Zusanli improves gastric dysrhythmia, delayed gastric emptying and intestinal transit, and impaired accommodation in streptozotocin-induced diabetic rats, and the improvement seems to be mainly mediated via the vagal pathway. But there was no report about the effects of electroacupuncture at Zusanli on enteric neuropathy in the diabetes.
Over the past years, several studies on the role of enteric nervous system and its neurotransmitters in the pathogenesis of altered gastrointestinal functions in diabetes have gained great significance. As a result of adaptive responses to different types of stimuli/conditions, enteric neurons are able to change their structural, functional, or chemical phenotype in order to maintain homeostasis of gut functions. 9 They can also increase in numbers in response to some physiological or pathological condition. 10,11 Some researchers have proved that electroacupuncture stimulation at Zusanli can induce cell proliferation and differentiation in central nervous system and improve hippocampus-related neuropathologies. 12,13 Our previous research also shows that long-pulse gastric electrical stimulation could induce regeneration of myenteric plexus synaptic vesicles in diabetic rats. 14 We speculated that, for the enteric nervous system, which histologically and to some extent functionally, resembles the central nervous system, electroacupuncture stimulation at Zusanli can also rescue the damages to the enteric neurons in diabetes.
In our current research, we first investigated the alteration of nitric oxide synthase (nNOS) neuron, cholinergic neuron (choline acetyltransferase [CHAT] stained), total neuron (protein gene product 9.5 [PGP9.5] stained), and newly generated neuron (doublecortin stained) in the stomach of diabetic rats, so as to explore the mechanism of the promotility effect of electroacupuncture at Zusanli for diabetic gastroparesis and supply theoretical foundation for the use of electroacupuncture for diabetic patients clinically.
Materials and Methods
Animals
Adult Sprague–Dawley male rats weighing approximately 250 to 300 g were used for the experiment. Rats were obtained from the Tongji Medical College of Huazhong University of Science and Technology. All animals were housed at a controlled temperature (20°C to 25°C) and were maintained in light controlled rooms with free access to water and standard solid food. All experiments are subjected to and approved by the Animal Care and Use Committee of the university.
Experimental Protocol
Animals were divided into 8 groups (n = 8): the control group, the diabetic group, the sham stimulation with long-term group, the low-frequency stimulation with long-term group, the high-frequency stimulation with long-term group, the sham stimulation with short-term group, the low-frequency stimulation with short-term group, and the high-frequency stimulation with short-term group (Figure 1).

Schematic representation of electroacupuncture study protocol. After the streptozotocin injection, the diabetes rats were divided into 6 groups depending on the total electroacupuncture span and frequency: CHS, CLS, CSS, AHS, ALS, and ASS
Diabetes was induced by a single intraperitoneal injection of streptozotocin in 0.05 mol/L citrate buffer, pH 7.4, at a dose of 60 mL/kg. The controls were injected with citrate buffer at the same dose as diabetic groups. The blood glucose level was measured 1 week after injection by cutting off the tip of the tail. Diabetes was considered if the random blood glucose is higher than 16.7 mmol/L. Blood glucose was also checked 3 weeks later and before the rats were killed. The animals that did not meet this criterion were excluded from the study. All the long-term stimulation groups were given electroacupuncture stimulation for 6 weeks (30 min/d) through the whole time course, and the short-term ones were given electroacupuncture for 30 min/day only at the last week (sixth week). The frequency of stimulation was 100 Hz in the high-frequency groups and 20 Hz in the low-frequency groups, and the amplitude of stimulation was 1 mA. Stainless acupuncture needles of 0.3 mm in diameter were bilaterally inserted into the locus of Zusanli (5 mm lateral and distal to the anterior tubercle of the tibia). For the real stimulation groups, the needles were attached to an electrical stimulator (G6805-2A; Shanghai Huayi Medical Instrument Factory, Shanghai, China), and needles of the same diameter were left in place without electrical stimulation for the sham stimulation groups.
After the 6-week intervention, all the rats were killed and the gastric
Materials
In the current study, we used the following materials: primary antibodies to nNOS (Abcam, Cambridge, UK), CHAT (Chemicon International, Temecula, CA), PGP9.5 (Abcam, Cambridge, UK), doublecortin (Abcam, Cambridge, UK), and mouse anti-rat actin (Boster, Wuhan, China). Secondary antibodies for Western blot include horseradish peroxidase (HRP)-linked goat anti-rabbit IgG, HRP-linked goat anti-mouse IgG, and HRP-linked mouse anti-goat IgG (Boster, Wuhan, China); anti-mouse Histostain-plus Kit, anti-rabbit Histostain-plus Kit and anti-goat Histostain-plus Kit (MAIXIN, Fuzhou, China).
Other reagents were as follows: streptozotocin (Alexis Biochemicals, San Diego, CA), TRIzol Reagent (Invitrogen, Carlsbad, CA), Oligo (dT) (Promega, Madison, WI), Rnase inhibitor (BioStar International, Toronto, Canada), dNTPmix (Promega), MMLV (Promega), NC membrane (Millipore, Bedford, MA), Fast SYBR Green PCR Master Mix (Applied Biosystems, Foster City, CA), and enhanced chemiluminescence agent (Thermo Scientific, Waltham, MA).
Immunohistochemical Analysis
The gastric specimens were fixed by immersion in 4% paraformaldehyde for 24 hours, and then processed for paraffin embedding in vacuum and cut at a thickness of 4 μm. Sections were deparaffinized in xylene and hydrated in a graded solution of ethanol. After blocking endogenous peroxidase activity in water containing 3% hydrogen peroxide for 10 minutes and microwave (750 W) for 3 minutes. Nonspecific binding was blocked by treatment with blocking reagent serum for 30 minutes. The primary antibodies for nNOS (1:600), CHAT (1:100), PGP9.5 (1:200), and doublecortin (1:100) were applied to the sections, and each specimen was incubated in a moist chamber overnight at 4°C. The slides were then washed in phosphate-buffered saline (PBS) and incubated for 30 minutes with the specific biotinylated secondary antibodies (goat anti-rabbit IgG, mouse anti-goat IgG, or goat anti-mouse IgG). Later, sections were again washed in PBS and incubated with HRP-linked streptavidin for 30 minutes. After 3 washes in PBS, the localization of target protein was visualized by incubating the sections for 5 to 10 minutes in freshly prepared 3,3-diaminobenzidine solution. For the observation, Olympus FV500 optical microscope (Olympus, Tokyo, Japan) was used.
mRNA Expression Analysis
mRNA expression levels were measured by real-time quantitative reverse-transcription PCR. Total RNA was isolated using the TRIzol reagent, and the purity and concentration of total RNA were evaluated by absorbance readings (A260/A230 and A260/A280 values) using a spectrophotometer. Two milligrams RNA was reverse transcribed into cDNA according to the manufacturer’s instructions. The cDNA was subsequently diluted in nuclease free water and stored at −20°C. The specific primers used are listed in Table 1. All PCR reactions were performed in a 20 μL final volume, comprising 10 μL SYBR green/enzyme reaction mix, 50 pmol/μL of each sense and antisense primer, and 1 μL of diluted cDNA. The samples were first heated to 95°C for 10 minutes. This was followed by a step at 95°C for 15 seconds and 60°C for 1 minute. This loop was completed a total of 40 times, and a melt curve was also inserted. During the annealing temperature, fluorescent signals were collected and cycle threshold (CT) values were extracted. We calculated ▵▵CT values using housekeeping gene GAPDH as control and including a no-template control using water instead of cDNA. The 2−▵▵CT method as described by Livak and Schmittgen 15 was used to quantify the relative change in gene expression.
List of Designed primer Sets for Real-Time Quantitative Polymerase Chain Reaction (PCR)
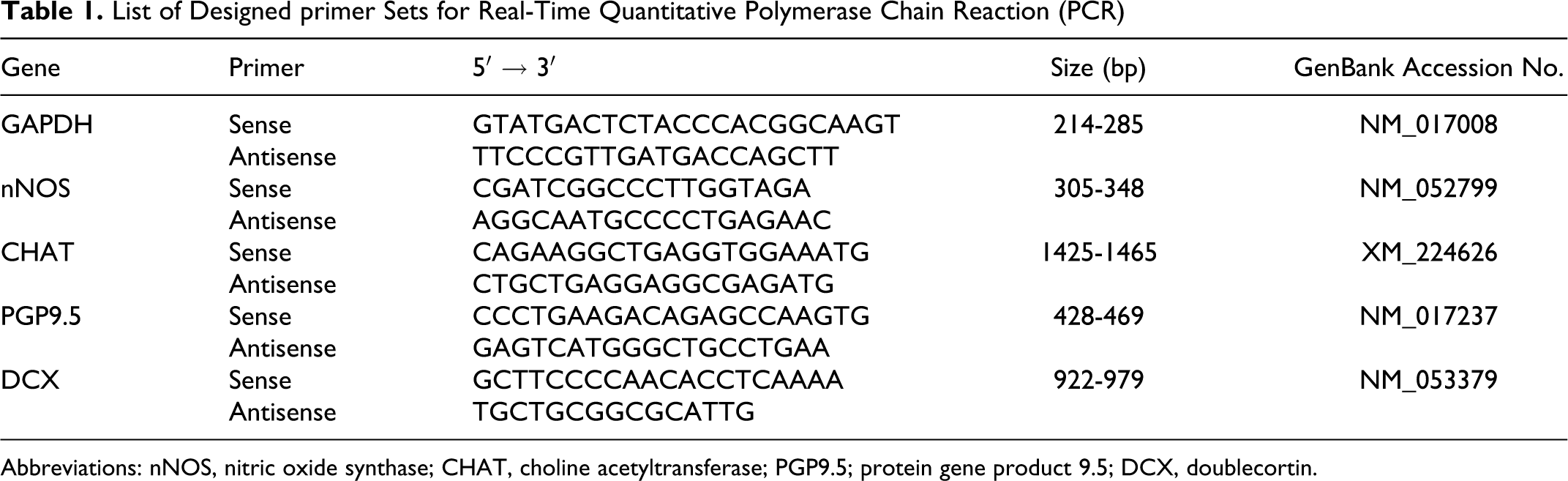
Abbreviations: nNOS, nitric oxide synthase; CHAT, choline acetyltransferase; PGP9.5; protein gene product 9.5; DCX, doublecortin.
Western Blot Analysis
Frozen samples were thawed and homogenized in extraction buffer for 1 hour. The resulting homogenates were centrifuged at 12000
Statistical Analysis
The results are expressed as mean ± standard error of the mean. Differences were compared statistically by independent
Results
Histology
Figure 2 shows the immunohistochemical staining of nNOS, CHAT, PGP9.5, and doublecortin. The brown immunoreactive nNOS, CHAT, PGP9.5, and doublecortin fibers and positive ganglial neuronal bodies were easily found in the antrum specimen of the control group. After 6 weeks of diabetes, these 4 kinds of immunoreactive products were decreased. Compared with the age-matched diabetic groups with sham stimulation, the immunopositive area of nNOS, CHAT, PGP9.5, and doublecortin were increased both in the high-frequency stimulation with long-term group and in the low-frequency stimulation with long-term group, but no obvious changes were found between the sham stimulation and short-term group and the high-frequency stimulation with short-term group, or the low-frequency stimulation with short-term group.
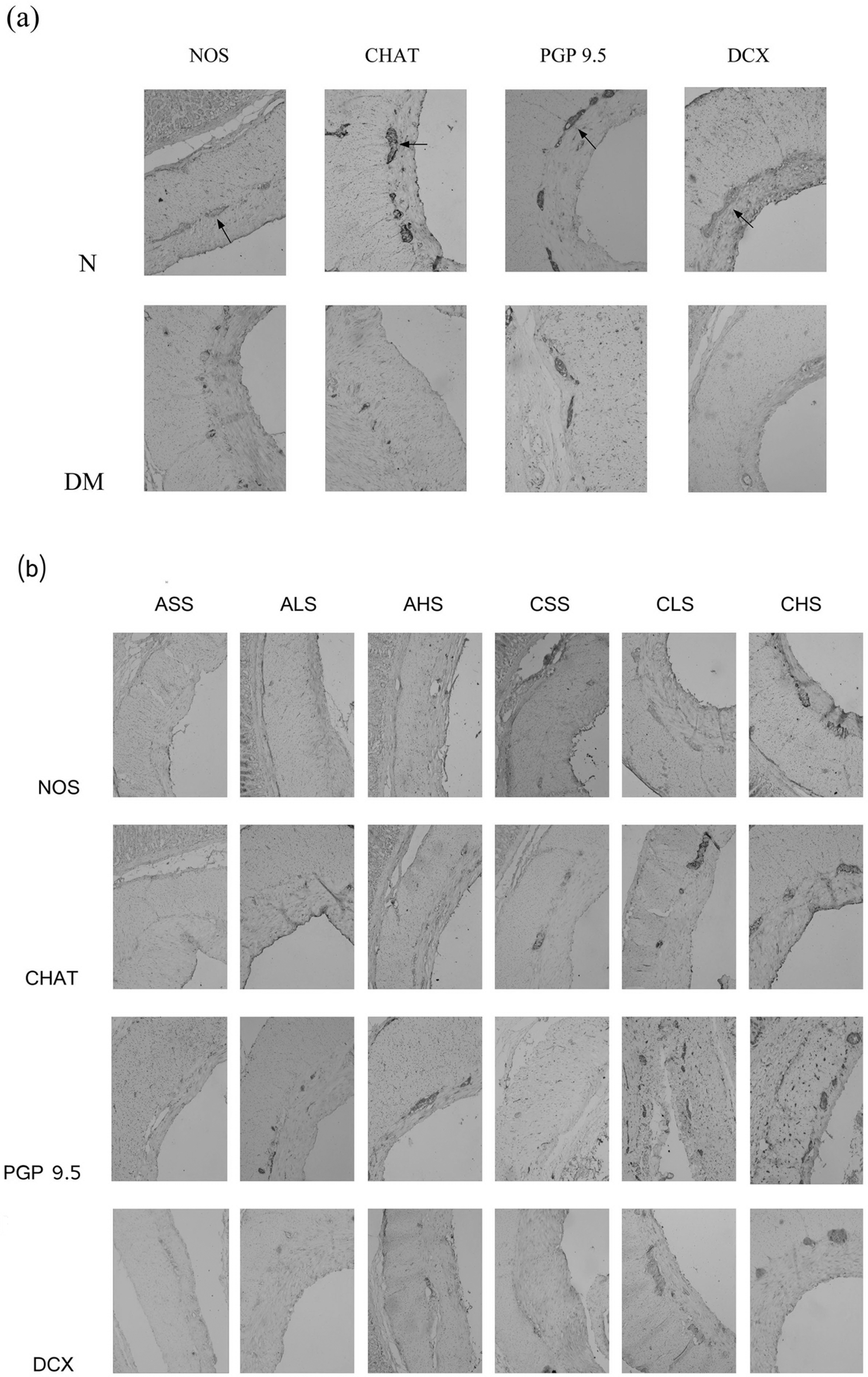
Immunohistochemical staining of nNOS, CHAT, PGP9.5, and DCX
mRNA Analysis by Real-Time Polymerase Chain Reaction
The mRNA expression of nNOS, CHAT, PGP9.5, and doublecortin are shown in Figure 3. Significant decreases in the mRNA expression of nNOS (

mRNA values of nNOS, CHAT, PGP9.5, and DCX
Protein Expression by Western Blot
The protein expression of nNOS, CHAT, PGP9.5, and doublecortin are shown in Figure 4. Significant decreases in the protein expression of nNOS (

The protein expression of nNOS, CHAT, PGP9.5, and DCX
Discussion
Acceleration of gastric emptying with electroacupuncture at Zusanli has been reported in both animals and humans, 4,5,16,17 but the mechanism is still unclear. Several studies show that enteric nervous system plays an important role in the pathogenesis of altered gastrointestinal functions in diabetes. In this study, we focus on the effect of electroacupuncture at Zusanli on the enteric neuronal changes in the stomach.
In the current study, we found enteric neuronal loss in the stomach of diabetic rats at the sixth week, including inhibitory neurons (nNOS stained), excitatory neurons (CHAT stained), and the total neurons (PGP9.5 as a pan-neuronal marker). Several studies have demonstrated loss of nNOS neurons in diabetes in the stomach. 18 –20 But for the changes of CHAT neurons in diabetes, there are opposite results in other regions of gastrointestinal tract. The possible reason may be that in different regions of gastrointestinal tract, nerves containing the same neurotransmitter are differently affected. The loss of the total neurons is consistent with our earlier research. 14
Several research studies about the central nervous system have proved that electroacupuncture stimulation can promote neural plasticity and induce cell proliferation and differentiation. 12,13,21 Enteric nervous plasticity in the enteric nervous system is also possible after the electroacupuncture stimulation. Our current study showed for the first time that electroacupuncture at Zusanli rescued the enteric neuron loss in the diabetic stomach, not only inhibitory neurons (nNOS stained), which can produce the NO, a major noradrenergic, norchinergic inhibitory neurotransmitter that mediates the smooth muscle relaxation in the gastrointestinal tract, but also excitatory neurons (CHAT stained) in the stomach. It seems that the electroacupuncture does not only restore one specific subpopulation of the enteric neurons to elicit one-side function. It can also balance the subpopulations of the enteric neurons to have an overall improvement in the enteric nervous system recovery.
The increase of the positive neurons in this study may come from 2 sources: the recovery of the injured neurons and neurogenesis. The occurrence of cell birth and neurogenesis has been demonstrated in the central nervous system. Although several studies found that stem cells in the adult enteric nervous system can also give rise to neurons in vitro, few studies prove neurogenesis in vivo. Recently, Liu et al 10 first demonstrated that the progenitor cells in vivo can give rise to neurons in the adult enteric nervous system by the activation of 5-HT4 receptors. Doublecortin is regarded as a developmentally regulated protein, expressed in immature neurons of both central nervous system and peripheral nervous system 22 and is considered as a transient marker of newly generated neurons in the central nervous system. Here we describe for the first time the changes of the doublecortin in the diabetic stomach. We found that the expression of doublecortin was decreased after 6 weeks of diabetes. It suggests that the diabetic condition may affect the neurogenesis in the stomach. Furthermore, we demonstrated that electroacupuncture at Zusanli can rescue the loss of doublecortin partly, and it can possibly be the mechanism of the restoring effect of electroacupuncture at Zusanli on enteric neurons. Electroacupuncture at Zusanli may promote the new neurons to be generated so as to increase the enteric neurons in quantity. In the central nervous system, parallel research studies also reported that neurogenesis can be induced by electroacupuncture. 12,13 However, the mechanism of the restoring effect of the electroacupuncture at Zusanli for the enteric neurons is still unclear, and more studies are needed to unmask it.
Research studies about the analgesia indicated that the effect of electroacupuncture is frequency dependent, with low-frequency (2 Hz) electroacupuncture increasing the release of an array of opioid peptides to interact with receptors in the central nervous system and high frequency (100 Hz) stimulation increasing the release of dynorphin to the receptor in the spinal cord.
23
For the gastrointestinal system, several studies reported the regulative effects of
Our result also showed that only long-term electroacupuncture can rescue the damages of the enteric nervous system, which implied that electroacupuncture did not affect enteric neurons in a quick way, but other researchers found quick regulative effect of gastric motility and myoelectrical activity after
In conclusion, electroacupuncture at Zusanli can restore the loss of enteric neurons in diabetes partly, but requires a long duration of stimuli, and the increase of doublecortin is likely involved in this positive process. The positive effect of electroacupuncture over the enteric system can contribute to the promotility effect of electroacupuncture. However, the precise mechanism of the promoting effect of electroacupuncture on enteric nervous system is still unclear. More studies are needed to unmask this mechanism and supply new therapeutic strategy in the treatment of enteric neuropathy in diabetes.
Footnotes
Author Contributions
The work presented here was carried out through collaboration between all authors. S. Liu defined the research theme. S. Liu and F. Du designed methods and experiments, M. Hu and F. Du carried out the laboratory experiments, M. Hu analyzed the data, interpreted the results, and wrote the article. All authors have contributed to, seen, and approved the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by grant from the National Natural Science Foundation of China (Project No. 30670775) and project of the National Key Technologies R&D Program in the 11th Five Plan period (2007BAI04B01).
Ethical Approval
All animals in our study were treated strictly in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and the work was approved by the Ethics Committee of Laboratory Animals.
