Abstract
Across scientific disciplines, 3-D organoid culture systems offer platforms to integrate basic research findings with clinical care. The National Eye Institute mounted a $1.1 million 3-D Retina Organoid Challenge. Organoids developed through the Challenge will be valuable resources for drug screening, disease modeling, and precision and regenerative medicine.
Due to advances in stem cell research and tissue engineering, the research community is poised to create 3-D retina organoid culture models that faithfully recapitulate the in vivo biology of human tissue. Retina organoids have the potential to further knowledge of embryonic eye development and disease modeling and help identify mechanisms and signals involved in retinal degeneration and repair, and have practical applications for drug testing and supplying cells for transplantation.
3-D retina organoids (also referred to as mini-retinas, optic cups, and optic vesicles) are generated by manipulating cells outside of traditional two-dimensional culture systems to result in multiple cell types that organize along three dimensions into microphysiological systems. 1 Many of these efforts, which use advanced techniques in 3-D tissue culture, microfluidics, and bioprinting, are informed by principles developed from organs-on-chips, biomimetics, biomechanics, and synthetic biology, and are conducted at the interface of vision research and various disciplines including developmental biology, bioengineering, and regenerative medicine.
The National Eye Institute (NEI) has mounted a $1.1 million 3-D Retina Organoid Challenge (3-D ROC; https://nei.nih.gov/3droc), to provide leadership and incentives to accelerate paradigm-shifting advances in 3-D retina organoid generation, distribution, and use, with specific focus on disease modeling and high-content screening. Acting now to marshal resources and coordinate efforts across these disciplines will pay high dividends in the future of vision research.
The potential of eye organoids
Eye development has long been studied in lower vertebrate models, which have provided valuable insights into the requisite signaling and guidance cues necessary to form a structurally and functionally healthy eye. These model organisms have served the development research community well, as they are genetically tractable, and the in vivo tools and techniques to study them have been optimized over the years.
Human pluripotent stem cells provide a new platform to investigate human development, patient-specific drug effects, gene repair strategies, and disease mechanisms in vitro. However, stem cell-based retina modeling does not inherently incorporate the 3-D tissue architecture or interactions between multiple cell types. To address these shortcomings, vision biologists explored growing pluripotent stem cells using sophisticated 3-D techniques to control eye organogenesis and recapitulate development or genetic diseases affecting the eye. 3-D organoid platforms lend themselves to improved ex vivo investigations of the retina because they have the potential to mimic its structure, organization, and function. Critical discoveries and innovations have contributed to protocols for generating retina organoids and used them to learn about the mechanisms underlying select mutation-induced diseases (Table 1).
Select publications that demonstrate advances in retina organoid generation protocols, use of retina organoids to understand disease mechanisms, and other findings relevant to creating in vitro retina model systems.

Despite these significant developments in a short time, limitations remain in growing these tissues. For example, most published protocols require manual dissection, which is time intensive and introduces variability. Current protocols also favor differentiation of a subset of neurons due to limitations in oxygen and nutrient diffusion through the tissue layers. Furthermore, widespread adoption is hindered because human retina organoids require extended time to mature and significant resources to maintain.
NEI Ideation Challenge competition to advance 3-D retina organoid research
To address these limitations and build upon this exciting technology, the NEI created the 3-D ROC competition. The prize competition is timely, as techniques to grow and maintain organoids from a variety of tissue types are becoming more robust. For example, miniaturized 3-D printed culture products can increase throughput and reproducibility while reducing costs. Organoid culture methods, combined with engineering techniques such as microfluidic platforms like those developed under the National Center for Advancing Translational Sciences (NCATS) Tissue Chip programs (https://ncats.nih.gov/tissuechip), offer promising improvements for modeling tissue functions and drug testing. 18
The NEI 3-D ROC aims to capitalize on these cross-disciplinary advances to improve retina organoid technology such that all major neuron classes in the tissue reach appropriate states of maturity and differentiation. Protocols developed as part of the challenge should also increase the throughput with which organoids can be grown and manipulated, and enable standardization and broad dissemination. The overarching vision of the NEI challenge is that retina organoid systems will add great value to drug validation, disease modeling, and other therapeutic applications.
An initiative to produce organoids satisfying specific structural and functional requirements is well suited to a prize challenge. Unlike grant mechanisms, which fund multiyear research applications based on preliminary data, applicants’ experience and working environment, prize competitions reward creative solutions and outcomes, and attract challenge solvers with diverse experience. Solvers retain the intellectual property, and the incentive to commercialize their inventions drives innovation beyond the prize purse. Congress encouraged NEI to use a challenge to accelerate therapy development for retinal disease. NEI held a planning meeting in April 2016 to determine the state of the science and technical needs, and to help design realistic targets for retina organoids, bringing together experts in gut and liver organoid biology, tissue engineering, and retinal developmental biology. Also participating in the planning meeting was US Congressman Pete Sessions, a long-time advocate for translational retinal research and for promoting prize competitions in science.
NEI designed the NEI 3-D ROC prize competition as a two-part series that began with an “ideation” challenge and led to “reduction to practice.” NEI launched the prize ideation competition in May 2017 as the first part of the 3-D ROC. This phase asked the research community to form interdisciplinary teams and propose groundbreaking ideas to generate 3-D retina organoids that would address the shortcomings of current protocols. The protocols were required to generate robust organoids for use in screening drugs or modeling retinal degenerative diseases. Thirteen submissions were received by the August 1, 2017 deadline and were reviewed by both technical expert and federal review panels. Erin Lavik, ScD, and her team at the University of Maryland, Baltimore County, were awarded $90,000 for their proposal to screen-print retina organoids using retina neurons derived from adult neuronal progenitors. Five teams were recognized with honorable mention (team leads listed): Rebecca Carrier, PhD, Northeastern University; David Gamm, MD, PhD, University of Wisconsin; Wei Liu, PhD, Albert Einstein College of Medicine; Daniel Pelaez, PhD, University of Miami; and Katja Schenke-Layland, PhD, Fraunhofer Institute for Interfacial Engineering and Biotechnology.
The submitted protocols incorporated the use of bioprinting, scaffolds, hydrogels, microfluidics, and the addition of vasculature to build organoids more faithful to normal human retina tissue. In the “reduction to practice” or implementation phase, these and other techniques will be tested for the ability to improve the derivation of retina organoids.
3-D ROC 2020: The next NEI Challenge competition to realize the promise of retina organoids
In February 2018, NEI launched the 3-D ROC: Reduction to Practice (3-D ROC 2020) as a $1 million follow-on to the ideation challenge. This prize competition asks participants to implement protocols to generate 3-D retina organoids and submit publication-quality data showing their functionality and utility in modeling diseases or screening drugs. In August 2018, NEI will recognize interim progress by awarding up to $100,000 each to as many as six teams whose models best address the scientific criteria outlined in the challenge announcement. The final challenge submissions will be accepted in March 2020, and submitting teams will have the opportunity to win up to $400,000.
Companies including Genentech, a member of the Roche group, and XCell Science, with expertise in areas such as retinal diseases, stem cells, tissue modeling, engineering, bioprinting, cell culture, drug screening, and regenerative medicine, have joined the NEI challenge and are offering resources to the challenge participants. Support includes in-kind consulting, access to optimization and validation platforms for drug screening, discounts on products and reagents, and other incentives. NEI continues to build relationships with businesses and organizations interested in supporting challenge participants during 3-D ROC II (https://nei.nih.gov/content/3-d-roc-sponsors).
At the end of the competition, NEI hopes that this challenge will have developed retina organoids as platforms for modeling retinal degenerative diseases in ways that closely mimic the in vivo disease pathology and to screen therapeutics. While the research community will provide robust and reproducible retina organoids, companies involved will offer the expertise and infrastructure necessary to move the tissue products toward commercialization. With functional human stem-cell derived retinal tissue built with standardized protocols, robust tools will be available to a broader group of vision researchers to move into translational and clinically relevant work.
Organoids as tools for translational research
3-D tissue culture and organoid technology has been at the forefront of interest of several scientific organizations, societies, and funding agencies. Initiatives and funding opportunities in tissue engineering, biomimetics, and microphysiological systems are being supported by the National Institutes of Health and other federal agencies including NASA, Department of Energy, the National Science Foundation, the Department of Defense, and the Office of Science and Technology Policy. Organoid biology is frequently featured in research conferences for a variety of disciplines including developmental biology, cell biology, tissue engineering, disease modeling, regenerative medicine, and cancer.
Interest in organoid biology stems in part from the promise they hold for applications in translational research because of their ability to closely recapitulate the structure and function of human organs. This is already being exploited in cancer research, where projects like the Stand Up to Cancer Dutch Cancer Society Translational Cancer Research Dream Team (http://www.standuptocancer.org/progress_reports/dream_team_progress_reports_tumor_organoids), the Broad Cancer Cell Line Factory (https://portals.broadinstitute.org/cellfactory#home), and the National Cancer Institute Human Cancer Models Initiative (https://ocg.cancer.gov/programs/HCMI) are building biobanks of patient-derived tumor organoids. Once established, the organoid repositories will provide many researchers with access to models of a variety of tumor types and associated data, increasing knowledge about the diseases and eventually serving as tools for screening tumor-specific or patient-specific therapies. Human forebrain organoids have been used to elicit the mechanisms underlying developmental defects caused by Zika virus and as a tool to screen and identify therapeutic compounds that may mitigate Zika-induced microcephaly. 19 In the Netherlands, Hans Clevers, his team at Hubrecht Organoid Technology, and their colleagues at Utrecht University have piloted the use of organoids derived from patients to inform clinical decisions. 1 They have employed rectal organoids grown from tissue of cystic fibrosis patients to screen drugs and subsequently prescribe successful treatments based on the results. 20,21 The group recently announced an upcoming clinical trial in collaboration with Dutch insurance companies to validate their screening platform (http://hub4organoids.eu/hub-and-dutch-health-insurance-companies-to-validate-use-of-organoids-for-cystic-fybrosis/).
The future of stem cell-derived retina organoids: Translational research and regenerative medicine
As the vision field continues to optimize retina organoids, goals set forth by the NEI 3-D Retina Organoid Challenge will move retina organoids toward use for translational and clinical applications.
In the UK, the National Centre for the Replacement Refinement & Reduction of Animals in Research (NC3Rs), together with Merck, Hoffmann La Roche, and Novartis, developed the “Retinal 3D” CRACK IT Challenge to establish a 3-D retinal cell model predictive of human physiology for early assessment of the efficacy and toxicity of novel ophthalmology drugs (http://www.crackit.org.uk/challenge-23-retinal-3d). A £1,000,000 contract was awarded to a project team led by Professor Lyle Armstrong at Newcells Biotech Ltd. to create 3-D retina organoids from human induced pluripotent stem cells (iPSCs) and validate the model as a tool for drug screening.
NEI is currently working with NC3Rs to ensure that the NEI challenge complements their recent “Retinal 3D” CRACK IT Challenge. Although the end goals and the paths to achieve the two goals are different, these two challenges ultimately strive to provide the research community with a wide set of tools to support better understanding of human retinal biology in health and disease. Technologies and techniques that emerge from these two challenges can be combined and leveraged to understand normal retina development and degeneration, model human diseases, test efficacy of drug therapies, and screen for retinal toxicity induced by therapeutics for systemic conditions. Combining drug screening and disease modeling applications has potential for precision medicine applications. In the future, using induced pluripotent stem cells to grow patient-specific retina organoids may allow physicians to tailor treatments for complex genetic diseases.
Another promising use for retina organoids is as tissue sources for cell transplantation to treat retinal degenerative diseases. Investigators are starting to use stem-cell derived 3-D retina organoids as sources for isolating photoreceptor precursors and transplanting as cell suspensions into animal models. 22 –24 Retina organoids offer a source of complex tissue that can be transplanted as a 3-D tissue sheet that integrates with and replaces diseased retinal cell layers. Masayo Takahashi and colleagues in Japan have demonstrated the feasibility of this approach in both murine and non-human primate models of retinal degeneration. 25,26 A separate NEI program, the Audacious Goals Initiative (AGI), is currently soliciting proposals to develop translation-enabling animal models of human retinal degenerative disease that can be used as proof-of-principle for regenerative medicine. The NEI Audacious Goals Initiative is committed to understanding the biology of regeneration and reconnection and overcoming the challenges of cell therapies with regard to photoreceptors and retinal ganglion cells (https://nei.nih.gov/audacious). Other collaborative programs under the AGI are developing non-invasive imaging techniques and identifying regeneration-promoting factors. Knowledge gained from AGI, along with platforms developed under the 3-D retina organoid challenge, will support the translation of cell replacement therapies to treat retinal degenerative diseases.
Administrative efforts are underway to standardize and accelerate the regenerative medicine field. The National Academies Forum on Regenerative Medicine is convening experts to define and overcome hurdles in the regenerative medicine field, such as appropriate cell sourcing, manufacturing, and logistics and delivery (http://nationalacademies.org/hmd/Activities/Research/RegenerativeMedicine.aspx). Under the Production Assistance for Cellular Therapies (PACT), the National Heart, Lung, and Blood Institute (NHLBI) has funded a coordinating center and five processing facilities to produce and test novel cell therapies. The California Institute of Regenerative Medicine is already supporting multiple cell therapy trials, including trials for retinal degenerative diseases. Interest and expertise from groups and organizations that can pull information across many fields to set standards and move therapies to trials will be critical for making regenerative medicine treatments safe and widely available to patients (Figure 1).
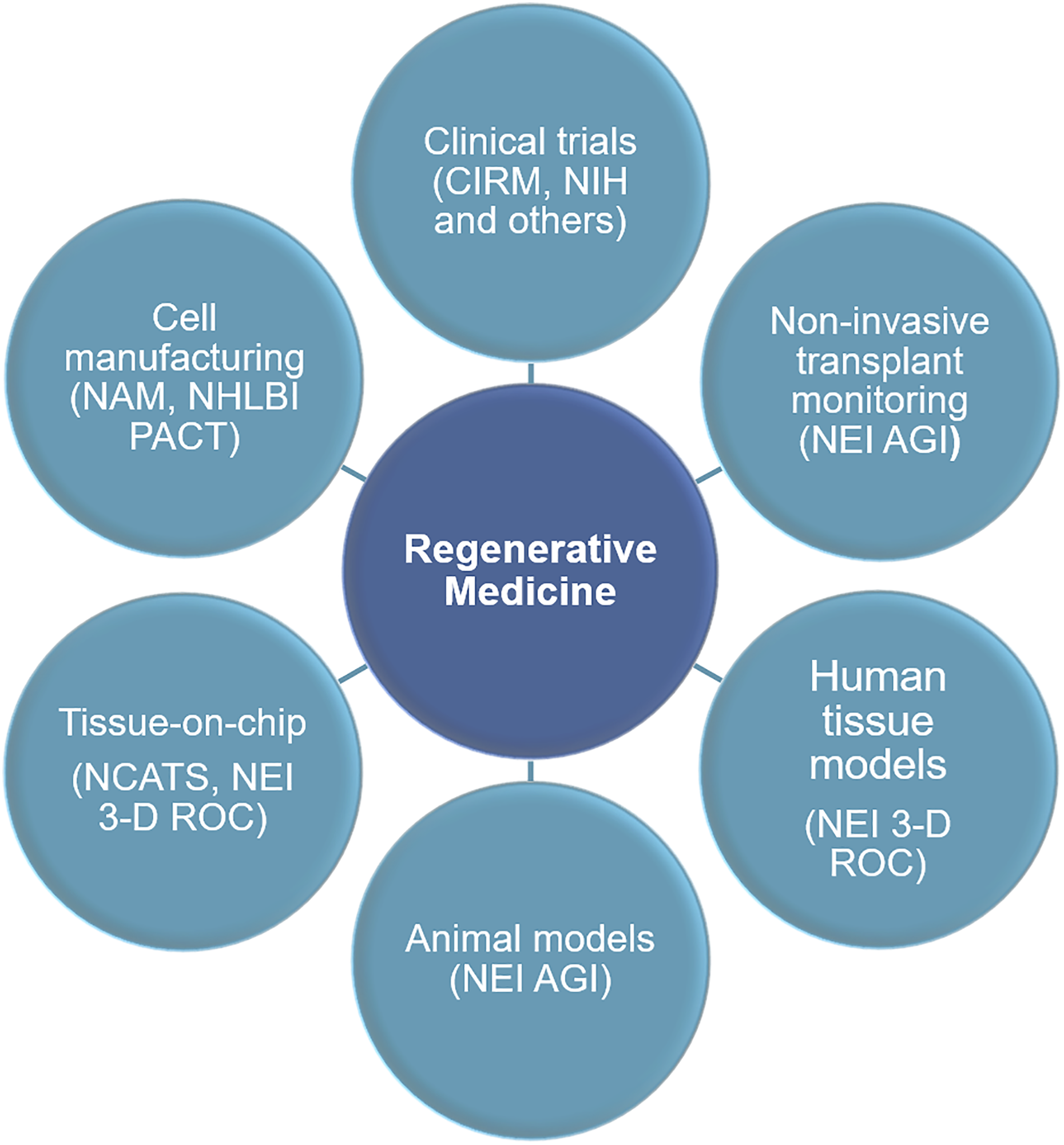
Basic research programs will continue to support technology generation, discovery, and translation in the regenerative medicine space. Scientific advances coming out of programs like the NEI Audacious Goals Initiative, the NEI 3-D Retina Organoid Challenge, and NCATS Tissue Chip programs will lay the foundation for translation of cell therapies into clinical trials such as those supported by the California Institute of Regenerative Medicine (CIRM) and the National Institutes of Health (NIH).
As the regenerative medicine community continues to define standards for bioproduct and cell manufacturing, the vision and broader research community has mobilized to advance and standardize retina organoid production. NEI encourages these fields to collaborate to develop retina organoids to understand retinal disease, improve drugs, and use as a tissue source for transplantation. With the 3-D Retina Organoid Challenge, NEI will accelerate the production of organoids to build a knowledge base and lay the groundwork for using these tissue products to provide better treatment options for patients with retinal degeneration through future uses in precision medicine and transplantation.
Footnotes
Author contributions
S Becker, JN Mazerik, and PA Sieving wrote this manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
