Abstract
Objectives:
Chronic rhinosinusitis with nasal polyposis (CRSwNP) is a pathological condition which leads to high healthcare-related costs and low quality of life for patients. The introduction of new biological therapies (monoclonal antibodies, MAbs) in CRSwNP patients has allowed new therapeutic options for non-responders to conventional therapies and Dupilumab represents the first approved biological agent. The aim of this paper is to provide a practical clinical multidisciplinary protocol which might help clinicians involved in this field to monitor the clinical outcomes.
Methods:
Our centre of Rhinology and Rhino-Allergology has developed a dedicated collection form to observe the outcomes of patients treated with Dupilumab. Our research started from the indications given in EPOS 2020 as main reference. We then implemented these references in an electronic database trying to apply Evidence Based Medicine (EBM) in current clinical practice.
Results:
The result consists of three parts: an anamnestic collection data, a clinician reported outcome and two patient reported outcomes (PROMs) questionnaire, the visual analogue scale (VAS) and the Sino-Nasal Outcome Test-22 (SNOT-22). The tables we propose should provide an adequate correlation with the patients’ adherence to therapy and their treatment outcomes. Such periodical evaluation (after a month, after 3 months, after 6 months and at one year) should quickly allow to monitor if the patient is correctly assuming the therapy and the eventual objective improvements.
Conclusions:
We propose a practical monitoring protocol formulated to analyse both objective and subjective aspects of patients with severe uncontrolled CRSwNP treated with MAbs, thus helping to define in future a better comparison between the clinical results of different institutes.
Keywords
Introduction
Chronic rhinosinusitis with nasal polyposis (CRSwNP) is a heterogeneous pathological condition due to an inflammatory disorder characterized by multiple phenotypes which leads to high healthcare-related costs and low quality of life of patients [1,2].
CRSwNP is defined as the presence of symptoms of rhinosinusitis (nasal obstruction, anterior/posterior nasal drip, facial pain/pressure, reduction in or loss of smell) for at least 3 months associated with finding of bilateral nasal polyps at the exam with nasal endoscope and/or with reported sinus disease on a CT scan [3,4].
Chronic rhinosinusitis presents an elevated prevalence in the world population: this condition affects about 11% of adults in Europe and about 12% of adults in the United States [5]. In China, the prevalence is about 8% [6] and in Brazil the 5.5% of population is affected by this condition [7].
Recently, several biological agents (monoclonal antibodies, MAbs) have dramatically changed the therapeutic approach for the treatment of chronic eosinophilic diseases such as severe asthma and atopic dermatitis. These molecules, acting on type 2 inflammation, are also demonstrating efficacy on severe uncontrolled CRSwNP and research in this regard is increasing.
Dupilumab represents the first biological agent approved in 2019 by the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) to treat adults with CRSwNP, in addition to topic treatment with nasal steroids, in those cases which are uncontrolled with oral steroids and/or surgery [8,9].
Aim of our paper is to develop a multidisciplinary protocol which might help clinicians involved in this field to monitor the clinical outcomes of patients treated with Dupilumab. The main goal of this protocol is to provide the ENT specialists, facing this new clinical challenge, with a combined clinical evaluation, together with patient reported outcomes (PROMs).
Materials and Methods
In order to comply with these goals, our centre of Rhinology and Rhino-Allergology of ENT Department has developed a dedicated collection form.
Our research started from the indications given in EPOS 2020 as main reference. We then implemented these references in an electronic database trying to apply Evidence Based Medicine (EBM) in current clinical practice. The result consists of three parts: an anamnestic collection data, a clinician reported outcome and two PROM questionnaire namely a visual analogue scale (VAS) and the Sino-Nasal Outcome Test-22 (SNOT-22) [10,11].
Results
In detail, the first part includes, in addition to the patient's personal data (sex, age, job); clinical assessment (comorbidities, drugs, allergies); alcohol and tobacco consumption; asthma; GORD; atopic dermatitis; Immune diseases; aspirin sensitivity; Lund and Mackay radiologic score [12]. Additionally, we included an anamnestic collection about CRSwNP (year of diagnosis; drugs taken and time; type, time and number of any surgical procedures).
The second part, describes the presence and the extent of nasal polyposis detected objectively trough nasal endoscopy. The staging systems used to quantify the severity of the disease at the endoscopic assessment is the Nasal Polyp Score proposed by Meltzer. It consists of a 0 to 4 polyp grading system (0: no polyps, 1: polyps limited to the middle meatus, 2: multiple polyps occupying the middle meatus, 3: polyps extending beyond middle meatus, 4: polyps completely obstructing the nasal cavity) [13].
The subjective evaluation is done through the VAS and the SNOT-22. VAS evaluates the intensity of specific symptoms, measured on a horizontal 10-cm line. Investigated symptoms are nasal obstruction, rhinorrhoea, local pain, hypo / anosmia, and sadness. A mean score for each symptom valuated may be obtained using the average value of the scores [14].
The second subjective evaluation is performed through the SNOT-22, so called because it is constituted by 22 CRS-related items for the patient scored from 0 to 5 (total score range 0-110 and higher scores mean worse symptoms), which evaluates the severity of complaints that patients have been adverting due to the disease. SNOT-22 items can be divided into 2 categories: first part of questionary regarding physical symptoms (items 1-12) which considers nasal, ear, and facial symptoms, and second part of questionnaire regarding quality of life and health of patients (items 13-22) which studies psychological issues and sleep functions [15].
The items are formulated based on the clinical diagnostic criteria of rhinosinusitis, as defined by the international consensus statement on allergy and rhinology: rhinosinusitis 2021 (ICAR-RS-2021) [16].
The first section is a single time pre-administration questionnaire. The other two parts (one objective and one subjective) should be completed before the start of biological therapy, and subsequently at 1 month, at 3 months, at 6 months and at 12 months.
Our proposal of practical clinical questionnaire for monitoring the effects of Mabs on treatment of CRSwNP patients is showed in Table 1 (anamnestic patient's data), and in Table 2 (CRSwNP monitoring scheduling).
Anamnestic Patient's Data.

CRSwNP Monitoring Scheduling.

Modified from Reference 13
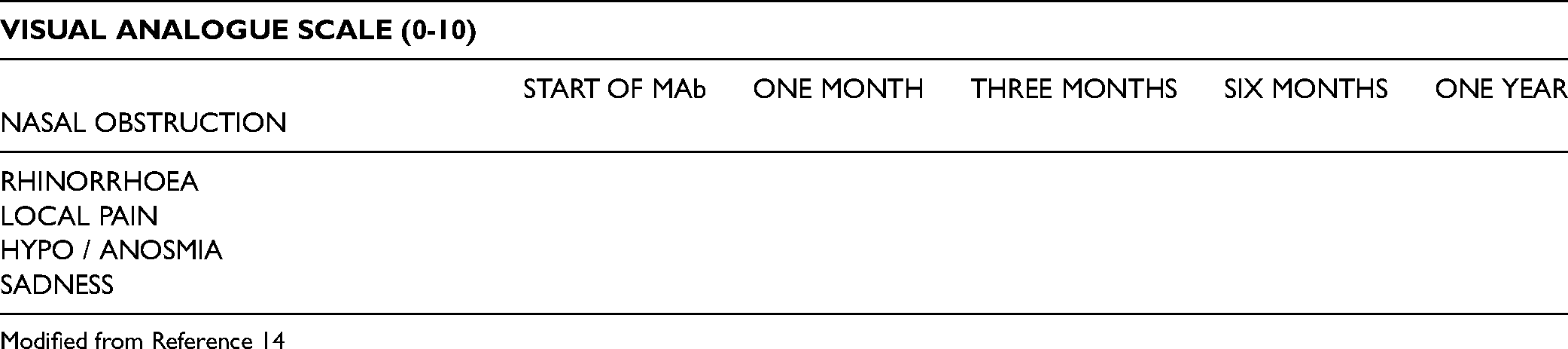
Modified from Reference 14

2. Please mark the most important items affecting your health (maximum of 5 items) _______↑.
Modified from Reference 15.
Discussion
CRS has been traditionally classified into CRS without nasal polyps (CRSsNP) and CRS with nasal polyps (CRSwNP). The two forms are different not only for macroscopic and clinical aspects but also for underlying biological mechanisms: the first one is correlated with type 1 inflammatory response and neutrophilia, and it is more common in the Asian population. The second one is correlated with type 2 inflammatory response and hyper-eosinophilia and mainly involves the Caucasian population [17].
The initiation of type 2 inflammatory response begins with antigen/allergen uptake by dendritic cells that elaborate the antigens and influence the T-cell response through the secretion of cytokines including IL4 which is responsible to convert Th0 cells into Th2 lymphocytes. The Th2 differentiation represents a specific inflammatory mechanism shared with atopic dermatitis and severe asthma. Further studies have shown that interleukins IL5, IL13 and IL 33 are also involved in the pathogenesis of CRSwNP [18].
Moreover, recent position papers, give increasing importance to the endotype of CRS as responsible for the underlying molecular and cellular inflammatory mechanisms associated with the disease. In CRSwNP the dominant endotype is the Type 2 that is characterized by the involvement of type 2 cytokines (IL-4, IL-13 and IL-5) and high levels of circulating IgE and/or local IgE [19,20].
The prevalence of CRSwNP in Europe is estimated to be between 1% to 5% [21].
In literature it has been reported the high association between CRS and asthma, with a prevalence of asthma around 25% in patients with CRS compared to 5% in the general population. CRS is also associated with other diseases as gastro-oesophageal reflux disease (GORD), chronic obstructive pulmonary disease (COPD) or nonsteroidal antiinflammatory drug (NSAID)-exacerbated respiratory disease (NERD). Other conditions negatively correlated with CRS are smoking, occupational exposure or air-pollution [4].
Furthermore, CRSwNP is typically associated with anatomical alterations of the sinonasal tract and high resistance to medical therapy [17].
CRSwNP still remains a social and clinical challenge not only for the ENT specialist but also for the scientific community due to several reasons: the elevated prevalence, which is higher than that of asthma and COPD in the general population [22]; the frequent relapses of the disease; the high direct social costs (therapies, and hospitalizations) and indirect social costs (absences from work).
The therapeutic approaches of CRSwNP have focused primarily on symptom relief over the years as reported in literature. Medical treatment mainly consists of nasal saline irrigations with steroid nasal sprays for maintenance therapy and short course of oral steroids [23].
Moreover, it is important to consider the role of antibiotics to treat any acute exacerbations.
Recent guidelines also describe the role of optional treatments as anti-leukotrienes, macrolide therapy and aspirin desensitization for those patients with aspirin-exacerbated respiratory disease [4].
Any co-management of asthma further complicates medical management by increasing the dosage and administration of steroids, in this case preferably oral, increasing the risk of adverse side effects and the potential for antibiotic prophylaxis and bacterial resistance [24].
When the medical therapy is inadequate for symptoms control, functional endoscopic sinus surgery (FESS) is indicated to remove mucosal inflammatory disease, reducing nasal obstruction, and facilitating the passage of topical therapies [4].
However, despite the evolution of surgical techniques and appropriate post-operative medical therapy, there are recurrence rates, about 30% at 5 years, which make further surgical treatments necessary [25].
The introduction of new biological therapies in CRSwNP patients has suggested new therapeutic options for non-responders to conventional therapies.
In the most recent literature, there are very few randomized studies on the therapeutic effect of Monoclonal antibodies on the CRSwNP population (less than ten reports) with only one study including also CRSsNP patients [26].
An interesting Cochrane systematic review highlighted the efficacy of biological treatment, in addition to regular administration of topical nasal steroids, in reducing local extension of the disease as measured on CT scan and improving the related quality of life. On the other hand, the evidence was poor/moderate regarding the general quality of life, the size of polyps assessed with the nasal polyp score and the risk of rhinopharyngitis [27].
The action mechanisms of these molecules are different: Omalizumab is an anti-IgE therapeutic antibody; Mepolizumab, Benralizumab and Reslizumab are selective IL-5 inhibitors; Dupilumab inhibits IL-4 and IL-13 cytokine-induced responses [28].
Dupilumab is a fully human monoclonal antibody that stops the signal of IL-4 and IL-13 targeting to their shared receptor component, the α-chain subunit of IL-4 receptors (IL-4Rα). This subunit is common to IL-4 (IL-4Rα / ɣc) and IL-13 (IL-4Rα / IL-13Rα) receptors and the molecular mechanism is inhibiting IL-4 and IL-13 signal, two essential cytokines in type 2 inflammation in CRSwNP [29].
The most recent EPOS guidelines delineate the indications for MAbs in patients with CRSwNP: biologics are indicated in that patients with bilateral nasal polyps, already treated with endoscopic sinus surgery or not fitting for surgical treatment and who show three of the following features: evidence of type 2 inflammation (tissue eosinophils ≥10/HPF or blood eosinophils ≥250 u/L OR total IgE ≥100 u/mL), need for at least two courses of systemic steroids or continuous use of systemic corticosteroids [≥2 courses per year OR long term (>3 months) low dose steroids OR contraindication to systemic steroids], significantly impaired quality of life (SNOT-22≥40), anosmic on smell test and/or a diagnosis of comorbid asthma needing regular inhaled steroids [4].
However, this potential for treatment has brought with it numerous, still unanswered questions among clinicians.
The non-univocal indications, the duration of administration, the timing of beginning of treatment, the period to be considered to achieve a therapeutic result, are just some of the questions that the international scientific community will have to answer in the literature of coming years.
The need of these answers is not only a challenge, but it represents a duty to allow uniformity of data collection of single institutes authorized to administer Mabs.
The tables we propose should provide an adequate correlation with the patients’ adherence to therapy (as the patient self-administers Dupilumab injections, after the first session performed by the otolaryngologist) and the relative treatment outcome.
Such periodical evaluation (after a month, after 3 months, after 6 months and at a year) should quickly allow to monitor if the patient is correctly assuming the therapy and the eventual objective improvements.
Since we have started such protocol only a few months ago, mature clinical data are not still available therefore we are aware that this is a point of weakness.
Conclusion
There are many open questions about the methods of use of Mabs, their symptomatic clinical efficacy, and the possibility of identifying more responsive subgroups of the population by type of pathology, entity, type of disorders. There are currently no universally accepted guidelines to evaluate the individual objective-subjective benefit, thus making difficult the comparison between the clinical results of different institutes. The result our effort was to provide a practical monitoring protocol formulated to analyse both objective and subjective aspects of patients with severe uncontrolled CRSwNP treated with Mabs, thus helping to define in future a better comparison between the clinical results of different institutes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Authors’ Note
Dr Stefano Millarelli was involved with the concept of the manuscript.
Dr Antonella Loperfido and Dr Fulvio Mammarella were involved with drafting the manuscript.
Dr Cristina Giorgione, Dr Alessandra Celebrini and Dr Massimiliano Del Ninno were involved with critically reviewing the manuscript.
Prof. Gianluca Bellocchi gave final approval to the manuscript.
All authors read and approved the final manuscript.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
