Abstract
Introduction
Osteoarthritis (OA) is a prevalent chronic joint disease affecting approximately 7% of the global population, with increasing significance in aging populations. Sarcopenia and obesity, two interconnected age-associated conditions, have potential pathogenic roles in OA development, yet their precise interactions remain incompletely understood.
Material and Methods
A cross-sectional analysis was conducted using National Health and Nutrition Examination Survey (NHANES) data from 1999-2006, focusing on 2309 participants aged 65 years and older. Sarcopenia and obesity were assessed via dual-energy X-ray absorptiometry-derived body composition measures, and OA status was determined through self-reported physician diagnoses. Multiple logistic regression models were employed to investigate the correlations between sarcopenia, obesity status, and the prevalence of OA.
Results
Sarcopenia was positively associated with OA, with an adjusted OR of 1.38 (95% CI: 1.07-1.79). Obesity demonstrated a consistent positive correlation with OA (adjusted OR: 1.31, 95% CI: 1.03-1.65). Subgroup analyses revealed nuanced associations, particularly among specific demographic groups such as non-Hispanic Black individuals and those with comorbidities like hypertension and diabetes.
Conclusions
The study reveals complex interrelationships between sarcopenia, obesity, and OA prevalence in older adults. The findings underscore the importance of body composition in joint health and suggest the need for targeted interventions considering demographic and clinical variations.
Introduction
Osteoarthritis (OA), a pervasive chronic joint disease affecting approximately 7% of the global population, represents a complex systemic condition characterized by intricate molecular and biomechanical interactions within joint tissues.1,2 As global demographics shift toward increasingly aging populations, OA has emerged as a critical public health challenge with profound implications for individual well-being and healthcare infrastructure. 3 The escalating prevalence of OA transcends traditional age-related mechanisms, becoming intricately linked with contemporary lifestyle patterns characterized by physical inactivity and metabolic dysregulation. 4
Sarcopenia and obesity, two interconnected age-associated conditions, have garnered substantial research attention for their potential pathogenic roles in OA development.5,6 The pathophysiological interface between these conditions is multifaceted: diminished muscle mass compromises joint biomechanical stability, while adipose tissue generates a pro-inflammatory microenvironment that potentially accelerates cartilage degradation.7,8 These interconnected processes suggest a dynamic, bidirectional relationship between body composition and joint health, with complex mechanisms of disease progression.
The relationship between sarcopenia and OA is complex and multifaceted, characterized by shared biomechanical and lifestyle vulnerabilities, with lower limb muscle atrophy emerging as a potential prognostic marker for disease progression. 9 Despite extensive investigations, the scientific literature presents conflicting and inconclusive evidence regarding the precise interactions between these conditions. 10 Similarly, the intricate association between obesity and OA remains enigmatic, shaped by a complex interplay of biomechanical loading, metabolic dysregulation, and inflammatory processes.11,12 Traditional anthropometric measures have proven insufficient for comprehensively elucidating these nuanced relationships. 13 Recognizing these critical knowledge gaps, our study aimed to comprehensively investigate the correlations of sarcopenia and obesity status with OA prevalence in older adult populations. By employing advanced body composition assessment techniques, including dual-energy X-ray absorptiometry (DXA), we sought to provide a more refined understanding of the associations between these interconnected age-related conditions.
Methods
Study Design and Population
We conducted a comprehensive cross-sectional analysis using data from the National Health and Nutrition Examination Survey (NHANES), which is a biennial population-based study that systematically collects comprehensive health and nutritional data from a representative sample of the United States population. Our research focused on 4 consecutive survey cycles spanning 1999-2006, with a specific focus on adults aged 65 years and older. The NHANES 1999-2006 cycles were utilized because they provided the most recent comprehensive DXA-derived body composition data for older adults (≥65 years). Subsequent NHANES cycles discontinued whole-body DXA scans in this population, making these cycles uniquely suited for examining sarcopenia-obesity-OA correlations in older adults.
The initial study population comprised 5538 older adults aged ≥65 years. After excluding 2214 individuals with non-OA arthritis diagnoses or undefined arthritis types, 993 individuals with missing data on appendicular lean mass (ALM), total fat percentage (TFP), or body mass index (BMI), 16 individuals with unclear medical histories of hypertension, diabetes, or cancer, and 6 individuals with undefined physical activity status, the final analytical sample consisted of 2309 subjects (Figure 1). The study protocol received approval from the National Center for Health Statistics Research Ethics Review Board, and all participants provided informed written consent. This study followed the STROBE guidelines for reporting observational research.
14
Flowchart of Participant Selection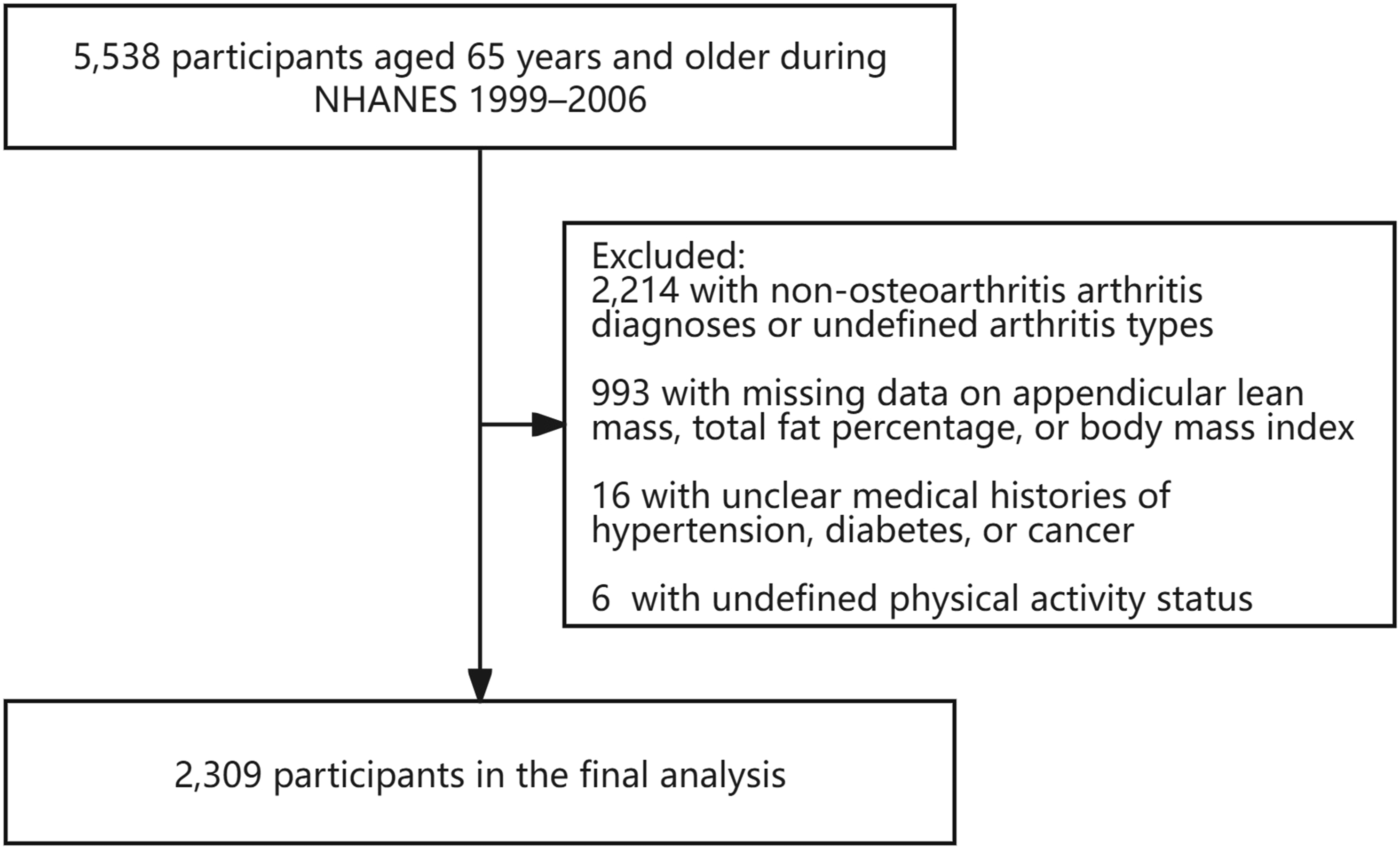
Defines of Sarcopenia and Obesity Status
In our study, sarcopenia was defined according to the guidelines established by the Foundation for the National Institutes of Health. It was characterized by an appendicular lean mass index (ALMI), calculated as ALM adjusted for BMI, with thresholds set at <0.789 for men and <0.512 for women. 15 Obesity was defined by TFP, with criteria of ≥40% for women and ≥30% for men. 16 Both ALM and TFP were measured using whole-body DXA scans conducted with a Hologic QDR-4500A fanbeam densitometer (Hologic, Inc., Bedford, Massachusetts), performed by certified radiology technologists to ensure accuracy and reliability in the data collection process.
Define of OA Status
OA status was determined through self-reported physician diagnoses. Participants were asked, “Has a doctor or other health professional ever told you that you had arthritis?” Those who responded affirmatively were subsequently queried, “What type of arthritis was it?” Based on their responses to these 2 questions, participants were classified as either having OA or non-OA.
Covariates
Covariates in our analysis included age, sex, race, educational level, marital status, physical activity, and medical history. Race was categorized as non-Hispanic White, non-Hispanic Black, Mexican American, and other. Educational level was classified into 3 groups: less than high school, high school, and more than high school. Marital status was categorized as married/living with partner, widowed/divorced/separated, never married, and unrecorded. Physical activity levels were classified as sedentary, low, moderate, and high. Participants’ medical histories regarding hypertension, diabetes, and cancer were obtained through a structured questionnaire.
Statistical Analyses
Descriptive statistics were presented as means ± standard deviations for continuous variables and as percentages for categorical variables. Comparative analyses utilized the Kruskal–Wallis test for continuous variables and the χ2 test for categorical variables. To investigate the correlations of sarcopenia and obesity status with the prevalence of OA, we conducted multiple logistic regression models: Model 1, no covariates were adjusted; Model 2, age, sex and race were adjusted; Model 3, all covariates were adjusted. Additionally, subgroup analyses were conducted, stratified by sex, race, and medical history of hypertension, diabetes, and cancer.
Statistical analyses were performed using R software (version 3.4.3) and Empower Stats software (X&Y Solutions, Inc., Boston, MA). A significance level of P < 0.05 was established to determine statistical significance (2-sided).
Results
The Characteristics of Participants According to Arthritis Status

Association Between of Sarcopenia, Obesity and Prevalence of Osteoarthritis
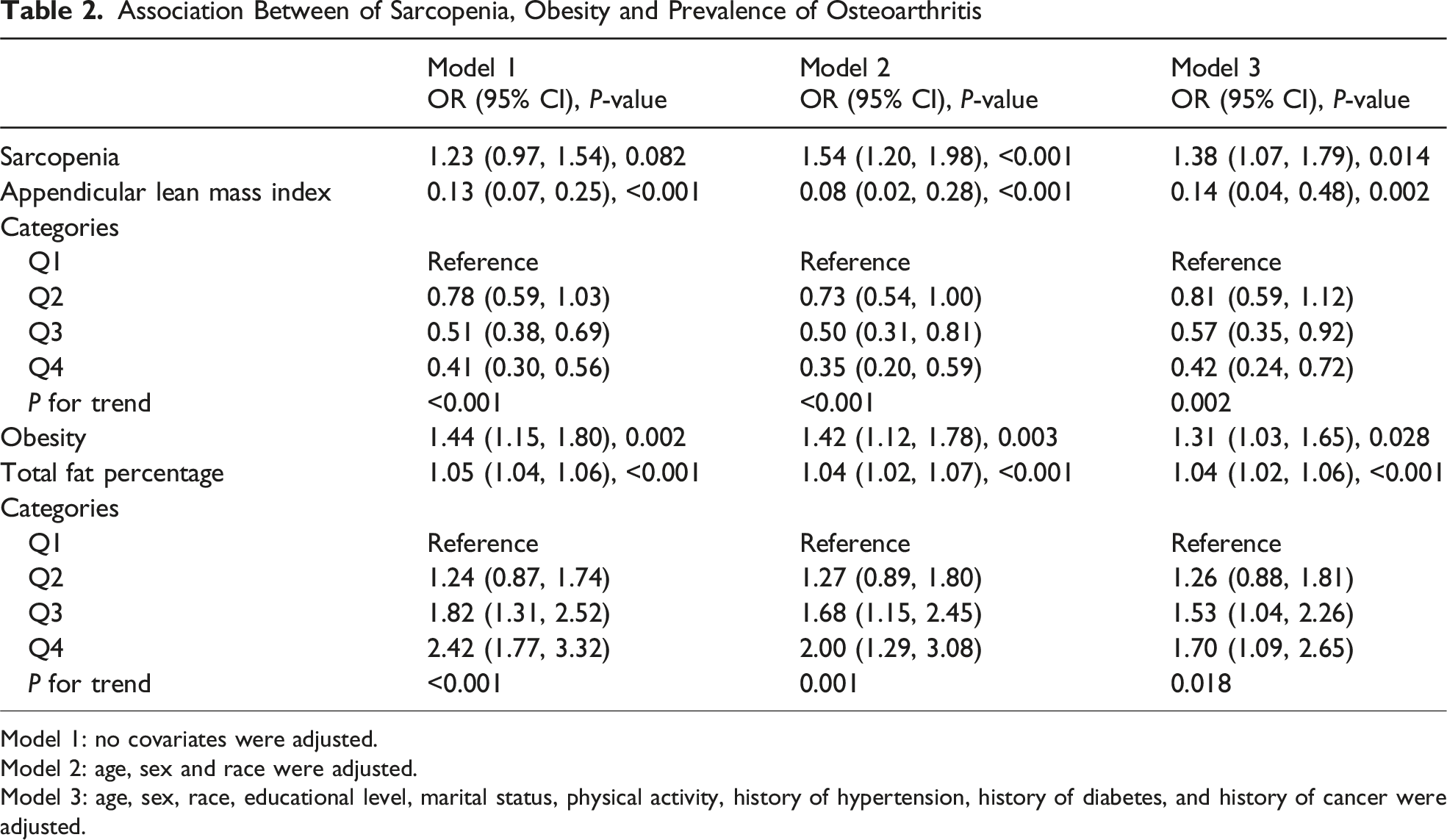
Model 1: no covariates were adjusted.
Model 2: age, sex and race were adjusted.
Model 3: age, sex, race, educational level, marital status, physical activity, history of hypertension, history of diabetes, and history of cancer were adjusted.
Association Between of Sarcopenia Status and Prevalence of Osteoarthritis

Age, sex, race, educational level, marital status, physical activity, history of hypertension, history of diabetes, and history of cancer were adjusted. In the subgroup analysis, the model is not adjusted for the stratification variable itself.
Association Between of Obesity Status and Prevalence of Osteoarthritis
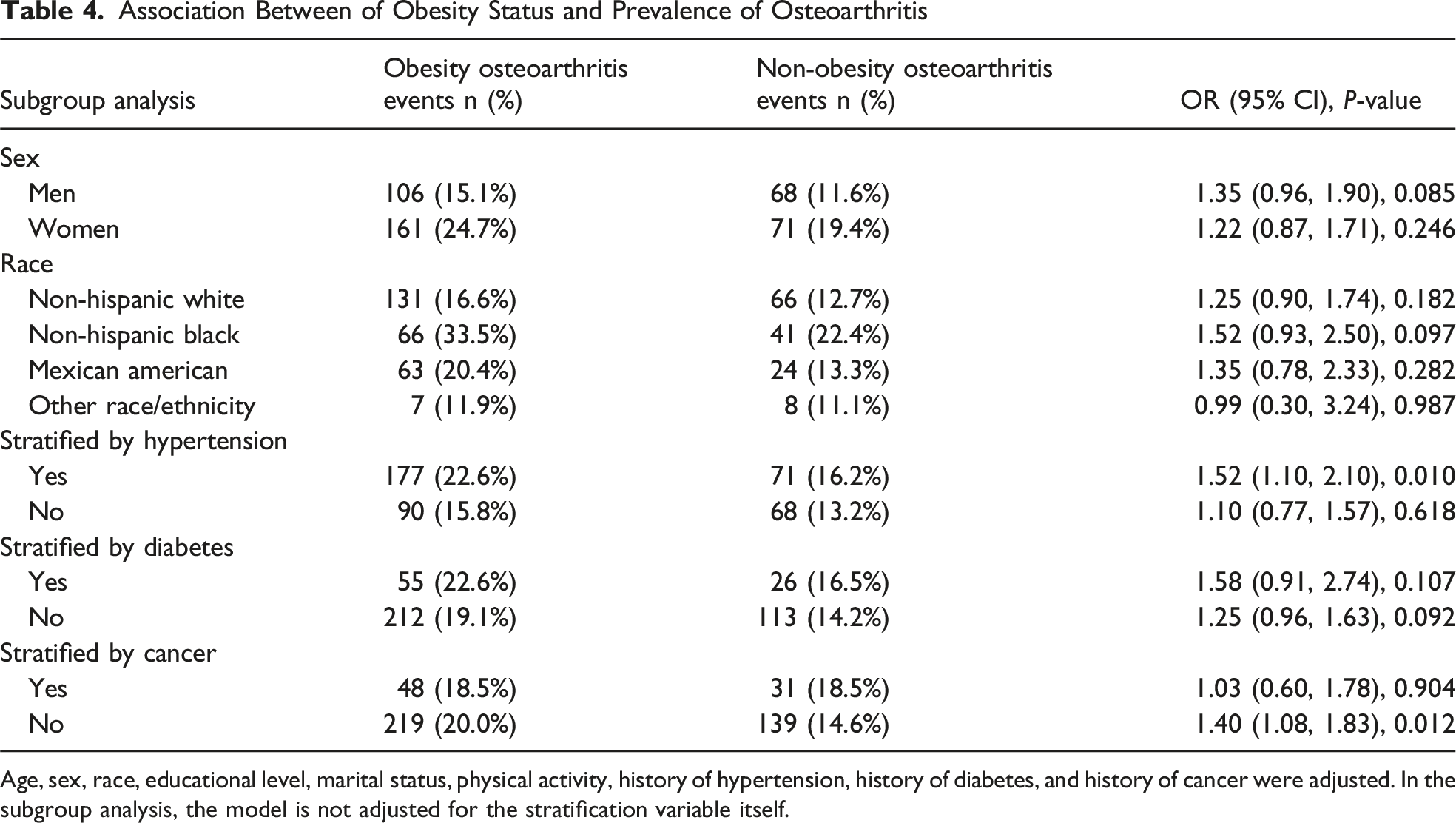
Age, sex, race, educational level, marital status, physical activity, history of hypertension, history of diabetes, and history of cancer were adjusted. In the subgroup analysis, the model is not adjusted for the stratification variable itself.
Discussion
Our findings reveal significant associations between sarcopenia, obesity, and OA prevalence in older adults, illuminating the intricate interactions among these conditions across diverse demographic and clinical groups.
The existing literature has preliminarily indicated the potential convergence of sarcopenia and OA in geriatric populations. A meta-analysis of 7495 participants revealed a significantly higher prevalence of sarcopenia in knee OA patients (45.2% vs 31.2%), with pooled OR demonstrating more than twice the likelihood of sarcopenia compared to control groups. 10 However, the Multicenter Osteoarthritis Study found that sarcopenia alone was not significantly associated with the risk of knee OA. 17 A recent longitudinal study of 6212 older Chinese adults found that sarcopenia significantly increased the risk of new-onset knee OA (OR: 1.91, 95% CI: 1.15-3.18), with particularly notable associations in females and potential implications for low muscle mass as a key contributing factor. 18 Our results indicated that older men with sarcopenia exhibited a markedly higher OR for OA, while this association was not significant in older women, suggesting potential gender-specific differences. Furthermore, the pronounced correlation between sarcopenia and OA among non-Hispanic Black individuals underscores the importance of considering racial disparities in OA risk.
Sarcopenia and OA exhibit a bidirectional relationship, with sarcopenia potentially exacerbating OA symptoms through muscle dysfunction and increased fall risk, 19 while OA simultaneously promotes sarcopenia by limiting mobility and physical activity. 20 The mechanisms linking sarcopenia and OA are complex and multifaceted, potentially related to inflammaging, 21 myokines and osteokines, 22 and obesity as a mediator.23,24 Emerging biomarkers, such as amino acids, may enhance early detection of sarcopenia and OA through non-invasive diagnostic approaches.25,26 Sarcopenia and OA are linked to inflammaging, a chronic low-grade inflammatory state associated with aging, where the inflammatory microenvironment in OA promotes the secretion of pro-inflammatory cytokines that exacerbate muscle atrophy in sarcopenia and cartilage degradation in OA. 21 Irisin, a key myokine with significant interactions between muscle and bone tissues, demonstrates how reduced levels in sarcopenia can compromise lower limb strength and joint load-bearing capacity, potentially accelerating OA progression through disrupted myokines and osteokines signaling. 22 Furthermore, obesity serves as a mediator in the relationship between sarcopenia and OA. Sarcopenic obesity demonstrates a more pronounced association with OA than non-sarcopenic obesity, suggesting that excess adiposity mediates the relationship between sarcopenia and joint pathology through enhanced inflammatory stress and biomechanical dysfunction.23,24
Obesity emerges as a significant risk factor for OA, particularly knee OA. A meta-analysis of 14 prospective studies demonstrated that obesity increases knee OA risk, with overweight and obese individuals showing 2.45- and 4.55-fold increased risks, respectively. 27 Additionally, the Framingham Study demonstrated that obesity at the onset of the study was associated with an increased risk of developing knee OA later in life. 28 Moreover, the Framingham Study revealed that weight loss over a decade significantly mitigates OA risk in women, suggesting obesity management as a pivotal prevention strategy. 29 Our study used TFP measured by DXA to define obesity, and found that obesity was consistently linked to increased OA prevalence, particularly in non-Hispanic Black participants and in high-risk populations with specific comorbidities such as hypertension and diabetes. Further research is needed to fully understand the pathogenetic features of obesity phenotypes and their specific impacts on OA development.
The association between obesity and OA is driven by a complex interplay of biomechanical loading, 30 metabolic factors,31,32 and obesity-induced sarcopenia effects. 33 Obesity escalates mechanical joint stress through increased load on weight-bearing joints and altered biomechanics, driving cartilage degradation and substantially elevating OA risk. 30 Adipose tissue, particularly infrapatellar fat, secretes pro-inflammatory mediators like leptin and resistin that directly promote joint degradation, while obesity-associated metabolic syndrome amplifies OA risk through chronic inflammation, oxidative stress, and systemic metabolic dysregulation.31,32 Obesity-induced sarcopenia, particularly affecting the quadriceps, compromises knee joint stabilization by impairing force absorption and distribution, consequently escalating mechanical stress on articular surfaces. 33
This study leveraged a comprehensive, population-based dataset and employed advanced body composition measurements using DXA, offering more precise assessments of lean mass and fat percentage compared to traditional anthropometric methods. However, the study also has several important limitations. First, the cross-sectional nature of the research precludes establishing definitive causal relationships between sarcopenia, obesity, and OA, limiting our ability to determine the precise temporal sequence of these conditions. Second, the reliance on self-reported physician diagnoses for OA introduces potential recall bias and may not capture the full spectrum of OA severity or early-stage manifestations. Third, while the study included various demographic stratifications, the sample sizes for some subgroups were relatively small, which may impact the generalizability of specific subgroup findings. Fourth, the study was limited to older adults in the United States, which may restrict the direct applicability of results to populations with different genetic backgrounds, lifestyle patterns, or healthcare contexts. Fifth, while our NHANES-derived sample size provided sufficient statistical power for primary analyses, the lack of a formal power analysis during study design may limit the interpretability of subgroup findings, particularly in smaller strata.
Conclusion
Our findings indicate that both sarcopenia and obesity significantly correlate with increased OA prevalence, particularly among specific demographic groups such as non-Hispanic Black individuals and those with comorbidities like hypertension and diabetes. These findings underscore the complex interplay between body composition and joint pathology, warranting further investigation into the molecular mechanisms underlying these epidemiological trends and potential targeted interventions.
Footnotes
Acknowledgements
The authors appreciate the time and effort given by participants during the data collection phase of the NHANES project.
Ethical Approval
The ethics review board of the National Center for Health Statistics approved all NHANES protocols.
Consent to Participate
Written informed consents were obtained from all participants (Protocol Number: Protocol #98-12 for 1999-2004 cycles; and Protocol #2005-06 for 2005-2006 cycle).
Author Contributions
YQW and GFG contributed to data collection, analysis and writing of the manuscript. ZXZ contributed to study design, analysis, writing and editing of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by the National Natural Science Foundation of China (82374493).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
