Abstract
Purpose
Distal radius fractures (DRF) often serve as the initial indication of bone mineral disease. This study aims to determine the extent to which the risk of subsequent fragility fractures can be reduced by initiating anti-osteoporotic therapy after initial presentation of a DRF.
Methods
This study utilized TriNetX, an online database with de-identified patient data from 79 US healthcare organizations. Females above the age of 50 were categorized based on receiving initial anti-osteoporotic treatment within a year of the DRF. Group characteristics, antiosteoporosis medications, and bone density evaluations were analyzed. After propensity matching, the risk of subsequent DRF, hip and vertebral fractures, as well as incidence of additional DEXA scans from 2004 to 2024 was explored.
Results
The Medication (M) group (n = 6709) had a mean age of 69, the No Medication (NM) group (n = 181,065) had a mean age of 65 at the index incidence. Baseline differences included higher rates of bone density disorders, inflammatory polyarthropathies, spondylopathies, metabolic disorders, obesity, malnutrition, and neoplasm in the M group. Notably, 43% of the M group had a prior DEXA scan compared to 8.7% of the NM group. The most prescribed anti-osteoporotic medication in the M group was Alendronate (49%). After propensity matching (n = 6627), the M group had 32% more DEXA scans and were 25% less likely to have a subsequent DRF fracture compared to the NM group. There was no difference between groups in combined intertrochanteric and femoral neck fractures. Vertebral compression fractures demonstrated a small but statistically significant increase in the M group, with an absolute risk difference of 0.8% (number needed to treat = 125) and an effect size (Cohen’s h = 0.079), suggesting limited clinical relevance.
Conclusions
Timely assessment and medical intervention can prevent future DRF. However, post-DRF bone density evaluations remain infrequent. This study highlights the hand surgeon’s role in identifying osteoporosis. Level of Evidence: Level III Cohort Study.
Introduction
Osteoporosis and other bone density pathologies pose a significant risk for sustaining life-altering fragility fractures, particularly hip and vertebral fractures. Distal radius fractures (DRFs) are often among the first injuries associated with osteoporosis, accounting for 18% of fractures in patients over 65 years old. 1 In patients with bone density pathology, a DRF can precede a devastating hip fracture by approximately 15 years. 2 This underscores the importance of identifying and managing bone pathology following a DRF as a key opportunity for fracture prevention.
The risk of subsequent fragility fractures, particularly hip fractures, is notably high, raising concerns about the lack of post-injury investigations. 3 A significant decrease in hip fractures was seen from 2002 to 2012, followed by a slight increase after 2012, coinciding with declines in osteoporosis treatment initiation following fragility fractures. 4 These trends were thought to be secondary to Medicare’s decreased reimbursement of DEXAs scans which significantly decreased the rate of bone density screening. The change in screening rates was estimated to result in 11,000 additional hip fractures. 4 Additionally, there has also been a decrease in the use of anti-osteoporotic therapy following all fragility fractures. 4 The consequences of severe fragility fractures, such as vertebral or hip fractures, on individual morbidity and the economic burden on individuals and the healthcare system are substantial, with billions spent annually in the United States. The compounding effect of subsequent fractures is evident, with vertebral fractures being among the most associated osteoporotic fractures, carrying up to a fivefold higher risk of a second vertebral fracture after the first event. This underscores the necessity of preventive therapy.5,6 As DRF often serves as the initial indication of bone mineral disease, it presents a glaring opportunity to prevent a future cascade of events. 7
A standardized evaluation followed by medical therapy could be the intervention that prevents these future events.4,8-10 However, the proportion of patients who receive treatment following a DRF remains low, despite evidence that those who receive therapy have a reduced risk of subsequent fractures. Patients who do receive bone density evaluations are more likely to receive treatment. 11 Furthermore, a significant percentage of orthopedic surgeons report rarely discussing osteoporosis management after a DRF, citing time constraints and the perception that it falls outside their scope of practice.12,13
Few studies have examined how much initiating anti-osteoporotic therapy following a DRF directly reduces future fracture risk. This study aims to address that gap by determining the extent to which initiating anti-osteoporosis therapy following a DRF reduces the risk of subsequent fragility fractures. Previous studies on anti-osteoporotic therapy have often been limited by small sample sizes, short follow-up periods, or less rigorous methodology. While this study builds on this work by using a large, multicenter real-world dataset from 79 U.S. healthcare organizations (TriNetX), applying propensity score matching, and evaluating outcomes over a 20-year period. By focusing on DRFs as a sentinel event, this study not only underscores a critical opportunity for early intervention but also demonstrates the long-term real-world consequences of initiating, or failing to initiate, appropriate therapy. The findings highlight existing treatment gaps and their measurable impact on future fragility fracture risk.
Methods
This study was conducted using TriNetX, an online database that contains de-identified patient information, and followed the STROBE Guidelines when preparing the manuscript. Using this platform, female patients above the age of 50 from 79 healthcare organizations who had reported an incidence of a distal radius fracture were identified from 2004 to 2024. The population was categorized into those who subsequently received treatment for osteoporosis within 6 months following their fracture (the medication group, M) and those who did not (the no medication group, NM). International Classification of Diseases (ICD), Tenth Revision (10), Clinical Modification (CM) diagnosis codes (UMLS:ICD10CM.509 A, UMLS:ICD10CM.S52.5, UMLS:ICD10CM.S52.90XA, UMLS:ICD10CM.S52.133 A) were used to identify distal radius fractures. Anatomical Therapeutic Chemical Classification System codes (NLM:ATC, NLM:RXNORM:77655, NLM:RXNORM:1311287, NLM:RXNORM:32915, NLM:RXNORM:73056, UMLS:CPT:82308, NLM:RXNORM:1921069, NLM:ATC, NLM:RXNORM:993449, NLM:RXNORM:46041, NLM:ATC, NLM:RXNORM:115264, NLM:RXNORM:2123126, UMLS:HCPCS, NLM:RXNORM:1921069) were used to identify anti-osteoporotic medication use.
To ensure that the cohort represented “new” DRF cases, we restricted the analysis to patients with no prior documented DRFs, and no prior prescriptions for anti-osteoporosis medications. This filtering process ensures that the study cohort represents patients who had no prior bone health treatments or fractures at the time of their DRF, confirming that the medications initiated after the DRF were truly new prescriptions. Patients with prior DRFs were excluded.
Characteristics of the groups included the types of anti-osteoporosis medication used and their rates, as well as comorbidities associated with bone density pathology. The groups were also examined to determine the rate of DEXA scan evaluations before and after the DRF incident. After collecting baseline characteristics, propensity score matching was performed on 14 characteristics: Demographics: Current Age, Age at Index, Female; Diagnosis: Disorders of bone density and structure, Spondylopathies, Other osteopathies, Inflammatory polyarthropathies, Metabolic disorders, Overweight, obesity and other hyperalimentation, Malnutrition, Neoplasms, Fracture of thoracic vertebra, Intertrochanteric fracture of femur; Procedure: Dual-energy X-ray absorptiometry (DEXA), or bone density study. Notably, propensity score matching accounted for pre-fracture DEXA scans, ensuring comparability between groups before the DRF event. The reported differences in DEXA scans between cohorts reflect post-fracture evaluations. Following propensity matching, risk of subsequent fragility fractures was assessed beginning one year after the index of injury and over a 20-year follow-up period. The subsequent injuries examined included distal radius, hip/femur, and thoracic compression fractures that occurred after the initial DRF. Patients with prior vertebral or femur fractures before the DRF were excluded.
A second query was generated to clarify if the increase in DEXA studies after a DRF prompted more medication use. The same ICD codes for DRF were used. The groups were based on those who received their first DEXA scan after the DRF incidence, and those who did not. Outcomes of anti-osteoporotic medication use after the index of incidence were then assessed.
Statistics
De-identified patient demographics and baseline characteristics were calculated using t-test to compare cohorts using the TrinetX statistical software, reporting the mean values and standard deviations. Other outcomes were analyzed using measure of association statistical analysis after propensity matching. Results were reported using the count of patients with outcomes in both the M and NM cohorts, providing a risk fraction. The risk ratio and risk difference were derived from the risk fractions, with results presented with ninety-five percent confidence intervals and P-values. A formal power analysis was not conducted, as this was a retrospective cohort study utilizing a fixed sample size determined by all eligible patients meeting the inclusion criteria within the TriNetX database. The study aimed to examine real-world associations within a large, representative population rather than test a pre-specified hypothesis requiring minimum sample size thresholds. The primary outcomes measured included the incidence of subsequent fragility fractures (distal radius, hip/femur, and thoracic compression fractures), the rate of DEXA scan evaluations following the index DRF, and the use of anti-osteoporotic medication after DEXA scan completion.
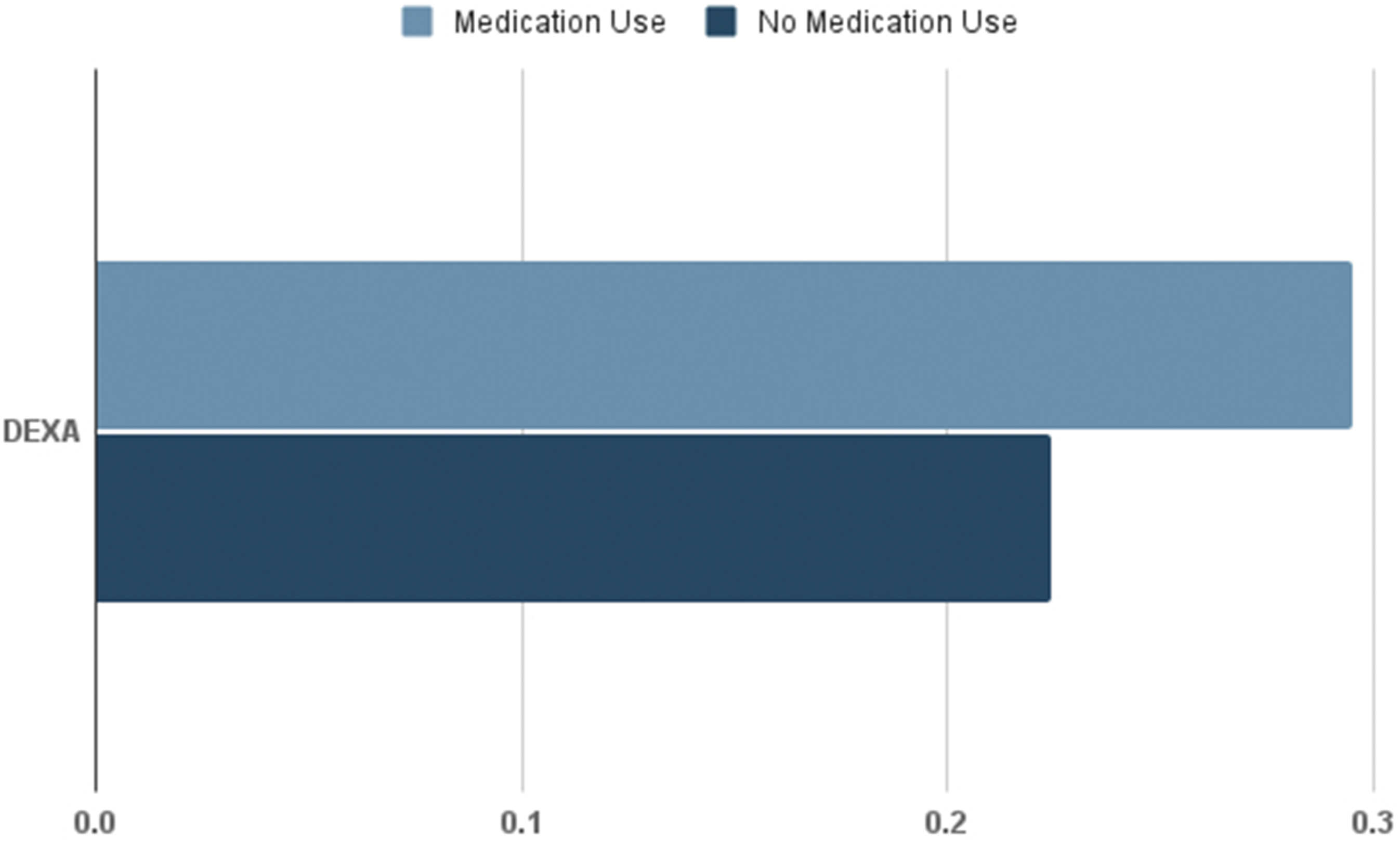
Results
Group Characteristics

Prescribed antiosteoporosis therapy use rates also showed a much higher use of resorptive medications compared with anabolic use (Figure 1). After propensity matching each group (n = 6627), the risk analysis revealed the medication group had a 25 percent decreased risk of subsequent DRF (Risk Difference = −0.052, 95% CI [-0.067, −0.038], Cohen’s h = 0.124. Risk Ratio = 0.799, 95% CI [0.751, 0.850]) and a higher rate of bone density evaluations (Risk Difference = 0.072, 95% CI [0.057, 0.087] Cohen’s h = 0.164. Risk Ratio = 1.321, 95% CI [1.246, 1.400], P < 0.0001) (Figures 2 and 3). There was no difference between groups found in combined intertrochanteric and femur neck fractures (Risk Difference = 0.002, 95% CI [-0.002, 0.006], P = 0.257. Risk Ratio = 1.188, 95% CI [0.882, 1.600]), Cohen’s h = 0.020. Thoracic compression fracture had a small increase in fracture risk (Risk Difference = 0.008, 95% CI [ 0.003, 0.012, P = 0.01]. Risk Ratio = 1.494, 95% CI [1.163, 1.919]), Cohen’s h = 0.056. The second query revealed that patients were five times more likely to use medication after undergoing an initial DEXA study, as shown in Table 2. Anti-osteoporotic Therapy Use Among the Medication Group Results of Subsequent Distal Radius Fractures Between Groups Results of Subsequent DEXA Scans Between Groups Likelihood of Medication Use With a DEXA Study Verses Without a DEXA Study


Discussion
The group using anti-osteoporotic therapy (M) after a DRF had a higher overall risk for fragility fractures due to their increased rate of comorbidities compared to the group not using therapy. Despite this risk, those who initiated anti-osteoporosis therapy after a DRF were 25% less likely to experience a subsequent DRF. This aligns with findings from Bawa et al, who demonstrated that anti-osteoporotic therapy after a fragility fracture significantly reduces the risk of future fractures. 14 Additionally, The M group had 32% more subsequent DEXA scans compared to the NM group, raising the question of whether the NM group missed follow-up evaluations, potentially leading to undetected cases and preventable fragility fractures. To explore if the increased DEXA scans were related to higher medication use or simply due to follow-up protocols, a separate query was made categorizing patients based on whether they had their first DEXA scan after the DRF incident. Those who received follow-up bone density evaluations after a DRF were about five times more likely to start on osteoporosis medications.
The increased number of DEXA scans in the M group could be attributed to their higher-risk characteristics, prompting clinicians to be more vigilant regarding their risk of osteoporosis and, therefore more likely to evaluate and manage them. Despite having a sicker profile, they had better outcomes with a decreased rate of subsequent DRF. This highlights that the incidence of low-energy DRF in women over the age of 50 should be considered a sentinel warning for potential future fragility fractures, regardless of the patient’s medical profile. Yet, many healthier women with a clear risk of decreased bone density due to the DRF event are not being evaluated or initiated on therapy. One randomized prospective trial found that 78.7% of patients were never screened following a DRF. 8 The root cause may stem from the lack of follow-up evaluations, leading to these missed cases. As osteoporosis treated early can have a favorable prognosis, it would seem intuitive that clinicians should aim for the earliest identification and initiation of treatment. While DEXA scans are relatively simple and inexpensive studies, they are currently underutilized. To address this issue, it’s necessary to identify and overcome obstacles that contribute to the problem. Barriers from the patient’s perspective include transportation limitations, fatigue from excessive appointments, mobility restrictions, financial limitations, and denial of pathology. 13 Physicians report a lack of time and comfort with the topic, stating bone density disorders are out of the scope of their practice. 15 While metabolic bone specialists may be most equipped to manage complex bone pathology, hand surgeons of any subspecialty may be the first point of contact with a presentation of a DRF, initiating evaluation, or a referral to a specialist could drastically improve future outcomes.
Study characteristics within the M group revealed a notably higher utilization rate of Alendronate, a nitrogen-containing bisphosphonate, compared to other medications. Alendronate has demonstrated efficacy in reducing vertebral fracture risk by about half and diminishes the risk of other nonvertebral fractures by approximately 30%. 16 Being orally administered and cost-effective in comparison to alternative therapies, it is frequently chosen for both prophylactic and therapeutic purposes.17,18 While bisphosphonates like alendronate are the most used anti-osteoporotic therapy, osteoanabolic agents have a greater anabolic effect, and are now the recommended first-line therapy for osteoporosis. 19 In the lumbar spine, Teriparatide increases bone mineral density by 7%, Abaloparatide by 11.2%, and Romosozumab by 13.3%.20-22 Additionally, there is greater bone-forming potential if initiated on an osteoanabolic medication before a resorptive medication, further supporting the choice to make them first-line agents. At the very least, when encountering a DRF from a mechanical fall, physicians should not hesitate to initiate evaluation, or referral to a specialist or fracture liaison service. 23 This proactive approach is crucial, considering potential barriers to care and the heightened risk of insufficient investigation following a DRF.
This study does have limitations. These include the inability to analyze individual DEXA scan results for more in-depth analysis and relying solely on statistical trends to conclude due to database constraints. Since the database cannot examine each case to elucidate the reasons behind the trends, we can only observe the trends and must further investigate using other tools. Although, the analysis used ICD codes specifically identifying DRF that fragility fractures, such as Colle’s fractures, there are limitations to know the exact mechanism to each injury, as a detailed case-by-case analysis is impossible using the TrinetX database. Additionally, variation in ICD coding across institutions and the absence of sensitivity analyses to assess underreporting or misclassification may affect the accuracy of event rates and risk estimates. When assessing the difference in the rate of femur neck and intertrochanteric fractures, no significant findings were uncovered, possibly due to the low reported incidence overall (n = 93 from the M group and n = 79 from the NM group). It was also not possible to distinguish high-energy from low-energy fractures simply utilizing ICD codes. Thoracic compression fractures were reported as mildly elevated in the M group (n = 148 from the M group and n = 101 from the NM group). The validity of these results was called into question due to the potential of incidental findings on imaging and underreported cases. 24 Additionally, medication adherence could not be monitored within the database, potentially impacting the observed treatment effects. An overall higher-risk patient profile in the M group, having equivocal results of a fragility fracture of their “healthier” counterparts, is still an optimistic result. A detailed case-by-case review would be a more appropriate way to gain a more accurate understanding of the incidence rate and provide a comprehensive understanding of severity and fracture healing outcomes. Although propensity score matching was used to balance baseline characteristics, patients with risk factors such as metabolic disorders and neoplasms were not excluded, which may introduce residual confounding given their association with osteoporosis risk.
This study did not include a formal power analysis, as the sample size was determined by available data within the TriNetX network. While the large sample enhances generalizability and statistical precision, the absence of a predefined power calculation may limit certain interpretations of statistical significance or nonsignificance, particularly for rare outcomes.
Future plans involve examining vertebral fractures and detailed DEXA results within our institution to provide a deeper understanding of the results obtained in this study. Additionally, our next steps include launching a fracture liaison service and evaluating improvements in osteoporosis fragility fracture intervention.
In 2007, the estimated burden of disease for fragility fractures in the US was 17 billion dollars, 19% of which were due to wrist fractures. 25 A 25% decrease in DRF as demonstrated in this study has the potential for saving healthcare costs up to 136 million dollars. If more proactive bone density evaluations occurred, this could result in a reduction of fragility fractures. A study by Harness et al. demonstrated that a structured osteoporosis management program led to a significant decrease in distal radius fracture risk, further supporting the need for comprehensive post-fracture interventions. 26 The current study reflects real-world patient data from 79 healthcare organizations. It provides an opportunity to analyze clinically relevant data to inform treatment decisions thereby contributing to improved outcomes for patients with osteoporosis. Hand surgeons, as frontline healthcare providers, play a crucial role in recognizing and addressing osteoporosis in their patients presenting with DRF.11-13,15 This frontline role, shared by many general orthopaedic surgeons as well as orthopaedic trauma surgeons, presents a key opportunity to initiate bone health evaluation and prevent secondary fractures.
Conclusion
Patients who initiated anti-osteoporotic therapy after a DRF had a higher overall risk profile but were 25% less likely to experience a subsequent DRF compared to those not treated. Despite more comorbidities, the treatment group had better outcomes, highlighting the importance of recognizing a low-energy DRF as a sentinel event for future fractures. Alendronate was the most frequently used medication, but newer osteoanabolic agents offer superior bone-forming effects and are now considered first-line therapy. Regardless of specialty, clinicians encountering DRF patients should initiate evaluation or referral to optimize long-term outcomes.
Footnotes
Acknowledgments
We would like to acknowledge Avnish Katoch for his technical support and guidance in navigating and utilizing the TriNetX online database platform, which was essential to the completion of this study.
Ethical Statement
Author Contributions
Chenée Armando, MD, Role: The first author led the study design, conducted the data analysis using TriNetX, and was the primary author of the manuscript. She was responsible for the conception and execution of the study and took the lead in drafting and revising the manuscript. Edward Fox, MD, Role: The middle author served as a metabolic bone disease specialist and advisor. He contributed to refining the study methods and provided expert review and revisions of the full manuscript, ensuring clinical accuracy and relevance. Kenneth Taylor, MD, Role: The last author, an orthopaedic hand surgeon, contributed to the study concept and methods and provided senior guidance throughout the project. He reviewed and approved the final manuscript, contributed substantively to its direction, and made final decisions regarding submission and revisions.
Funding
The authors received no financial support for the research, authorship, or publication of this article.
Declaration of Conflicting Interests
The authors declare no potential conflicts of interest with respect to the research, authorship, or publication of this article.
Data Availability Statement
The data that support the findings of this study were obtained from the TriNetX research network. Access to the data is subject to licensing agreements and restrictions; therefore, the raw data are not publicly available. However, aggregated results and analytic methods are available from the corresponding author upon reasonable request and with permission from TriNetX.
