Abstract
Introduction
For older adults, maintaining muscle strength and balance is crucial to preserve an upright posture and independently manage their basic activities of daily living (ADL). This study aimed to examine whether muscle strength and balance mediate the relationship between frailty syndrome (FS) and osteoporosis in a large sample of community-dwelling older adults.
Material and Methods
This cross-sectional study is part of the second phase (2016-2017) of the Amirkola Health and Ageing Project (AHAP), a cohort study conducted on all elderly aged 60 and over in Amirkola, Northern Iran, since 2011. Data from 2018 older adults were collected by a trained person using bone mineral density (BMD), frailty index, activities of daily living (ADL), instrumental activities of daily living (IADL), handgrip strength (HGS), quadriceps muscle strength (QMS), Berg Balance Scale (BBS), and Timed Up and Go test (TUG test) and analyzed using analysis of variance, chi-square, and path analysis tests.
Results
The mean indices of femoral neck BMD and lumbar spine BMD, HGS, QMS, BBS, ADL, and IADL were lower in the frail older adults than in the pre-frail and non-frail older adults. In addition, the mean TUG test level was higher in the frail older adults than in the non-frail and pre-frail older adults. The results of the present study have indicated that frailty is significantly related to osteoporosis, and that balance and muscle strength can predict osteoporosis; these variables play a mediating role in the relationship between frailty and osteoporosis.
Conclusion
From the results of the present study, it can be concluded that frailty may increase the odds of osteoporosis. The results of the current study have indicated that balance (BBS and TUG test) and muscle strength (HGS and QMS) are associated with osteoporosis and these variables play a mediating role in the relationship between frailty and osteoporosis.
Highlights
• Osteoporosis is the most common metabolic bone disease and a public health problem, especially in the older adults, worldwide and in Iran. • Based on the findings of the present study, frailty syndrome was associated with osteoporosis in the older adults in Iran. • Muscle balance and strength mediate the relationship between frailty syndrome and osteoporosis in community-dwelling individuals aged ≥60 years.
Introduction
Osteoporosis is the most common metabolic bone disease and is considered a public health problem worldwide. 1 The worldwide prevalence of osteoporosis and osteopenia is 19.7% (95%CI, 18.0%-21.4%) and 40.4% (95%CI, 36.9%-43.8%), respectively, and varies widely by country (from 4.1% in the Netherlands to 52.0% in Turkey) and continent (from 8.0% in Oceania to 26.9% in Africa). 2 In Iran, a total of 1006 (41.5%) older adults were diagnosed with osteoporosis. The age-standardized prevalence was 62.7 (95% CI: 60.0-65.4) in females and 24.6 (95% CI: 21.9-27.3) in males, using the reference value derived from 20-29-year-old females. 3
Osteoporosis may not be diagnosed until the older adults have already suffered multiple fragility fractures. On the other hand, the results of the studies represent that after the diagnosis, the treatment of osteoporosis in the older adults is usually not carried out and adherence to diet therapy is usually poor. Therefore, it is necessary for health care providers and researchers to recognize the potential predictors of osteoporosis in order to reduce the incidence of osteoporosis so that evidence-based interventions can be implemented.4,5
Osteoporosis is known to be a multifactorial disease, so some diseases such as hyperthyroidism, gonadal dysfunction, rheumatoid arthritis, Cushing’s syndrome, and long-term use of certain medications such as cortisone may predict its occurrence. Other factors such as inactivity are also involved in the development of osteoporosis.6,7 Today, in addition to the above factors, frailty syndrome (FS) in the older adults has attracted the attention of researchers as one of the predictors of osteoporosis.8,9 FS is associated with body compositional changes, sarcopenia, and osteoporosis and overlaps with pathogenic pathways associated with loss of lean muscle mass and skeletal deterioration. 10
The results of various studies suggest that people with severe FS have an increased likelihood of common and accidental fractures due to osteoporosis.11–13 On the other hand, FS increases the susceptibility of the older adults to adverse health outcomes such as falls, fractures, hospitalization, and disability due to a decrease in the individual’s ability to compensate for disruptions in homeostasis and minor stressors. 8,14
In addition to FS, researchers have suggested that another factor predicting osteoporosis is muscle balance and strength. 15 However, epidemiological findings on the relationship between muscle strength and osteoporosis or fracture risk are contradictory.16–18
For example, in the study by Song et al 19 (2022), no causal relationship was found between muscle strength and bone mineral density (BMD). On the other hand, balance and muscle strength are also considered predictive variables for FS 14,20 and consideration of this reciprocal relationship may lead to more effective interventions. The inconsistency of results in various studies and the fact that in the past, most studies on the factors influencing osteoporosis in the older adults have examined simple linear relationships between variables make the need for studies explaining the relationship between multiple factors and osteoporosis in terms of complex models more evident than ever. 21
On the other hand, the researcher did not find any study that investigated the mediating role of muscle strength and balance in the relationship between FS and osteoporosis. Research on osteoporosis risk factors such as FS helps us to understand the importance of frailty management in reducing the occurrence of osteoporosis. 22 In addition, investigating the causal relationship between osteoporosis with muscle balance and strength is an important public health issue and this relationship has not been fully elucidated 19 ; therefore, this study aimed to examine whether muscle strength and balance mediate the relationship between FS and osteoporosis in a large sample of community-dwelling older adults.
Hypotheses
• There is a significant relationship between FS and femoral neck BMD (FN-BMD) and lumbar spine BMD (LS-BMD in older adults. • There is a significant relationship between FS and muscle strength (handgrip strength (HGS) and quadriceps muscle strength (QMS)) in older adults. • There is a significant relationship between FS and balance (Berg Balance Scale (BBS) and Timed Up and Go test (TUG test) in older adults. • Muscle Strength (HGS and QMS) mediates the relationship between FS and Osteoporosis (FN-BMD and LS-BMD) in older adults. • Balance (BBS and TUG test) mediates the relationship between FS and Osteoporosis (FN-BMD and LS-BMD) in older adults. • The structural equation explaining the mediating role of muscle strength (HGS and QMS) and Balance (BBS and TUG test) in the relationship between FS and Osteoporosis (FN-BMD and LS-BMD) has a good fit.
Conceptual Model
Based on the results of various studies, the conceptual model of this study was designed15,20,23,24 (Figure 1). The conceptual model.
Materials and Methods
This cross-sectional study is part of the second phase (2016-2017) of the Amirkola Health and Ageing Project (AHAP), a cohort study conducted on all elderly aged 60 and over in Amirkola, Northern Iran, since 2011. 25
Inclusion criteria included individuals who were ≥60 years of age, lived in the city of Amirkola, did not use mobility aids for walking, had no uncorrected hearing or vision defects, had no limbs amputated due to disease, had no transient balance problems on the day of testing, and were not wheelchair dependent. Older adults who suffered from conditions such as stroke, Parkinson’s disease, peripheral neuropathy, de novo scoliosis, etc., and were unable to cooperate with the research team for balance tests or other assessments were excluded from the study. Exclusion criteria were deficiencies in the collected data, lack of ability to answer questions (due to severe speech impairment or hearing loss), and cognitive impairment. Cognitive impairments were assessed by Mini-Mental State Examination (cutoff score = 21). 26 The researchers invited all the older adults to participate in the study by calling them and visiting them at home while giving them the necessary information about the project.
In this study, considering a frailty prevalence rate of 34.75% in Iran, a Type I error rate of 5%, a statistical power of 80%, and an absolute error of 2.97%, the sample size was estimated to be 2018 individuals based on the following formula.
Authors followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Guidelines when preparing the manuscript. The STROBE checklist is available as Supplemental Material 1. Data were collected by trained individuals using the following instruments:
Bone Mineral Density (BMD)
Osteoporosis was determined by measuring BMD, which in turn was measured by dual energy X-ray absorptiometry (DXA) scanning by a Hologic Horizon-WI densitometer with an instrument accuracy of <1% and a coefficient of variation (%CV) of 0.25%. Femoral neck BMD (FN-BMD) and lumbar spine BMD (LS-BMD) were measured. BMD is the amount of bone mass (areal density, g/cm2) expressed by the T-score. According to World Health Organization, BMD is normal when the T-score is greater than −1. However, osteopenia is said to occur when the T-score is between −1 and −2.5, and osteoporosis when the T-score is less than −2.5 (in this condition, bone density is 2.5 SD < mean for a 30-year-old male or female). 27
Frailty Index
To identify the older adults with FS, the indicators defined by Fried et al 28 were used in the current study. It has a solid foundation of biological causative theory28,29 and has been applied to multiple epidemiological studies where it is predictive of adverse clinical outcomes, including mortality.30–33 These 5 indicators were 1-shrinking (unintentional weight loss of 4.5 kg or more in the last year), 2-weakness (low grip strength), 3-exhaustion (self-reported), 4-slowness (slow walking speed) and 5-low physical activity. 28 This instrument was completed through interviews with older adults. In the present study, frailty and pre-frailty were defined as ≥3 indicators and 1-2 indicators out of 5 indicators, respectively. 34 The validity and reliability of this instrument were confirmed by the study of Dent et al 35 (2016).
Muscle Strength
All research phases were conducted on both categories of participants (first on the dominant group and then on the non-dominant group). At the beginning of the study, the dominant group was identified by asking participants which hand they used for writing. To measure handgrip strength (HGS), the Korean-made DIGI Hand Dynamometer, which measures muscle strength of the upper body and arms in kilograms was used. The term ‘maximum HGS’ referred to the highest force exerted by participants on the dynamometer handle, which was recorded as the maximum HGS. To standardize the position of participants’ hands for evaluating HGS, the recommended position by the American Society of Hand Therapists was used. In this position, the participant sat on a chair without armrests at an appropriate height, with the shoulder of the assessed limb in adduction (without any rotation), the elbow bent at a 90-degree angle, the forearm in a neutral (mid-position), and the wrist in a neutral position. The participant was then asked to hold the dynamometer vertically aligned with the forearm and exert maximum force within a pain-free range.36–39 This assessment was repeated three times for each participant with a 60-second interval between attempts. The highest recorded value from both the right and left hands was used as the grip strength in this study. Subsequently, the maximum grip strength of the participant was recorded.
The Manual Muscle Testing (MMT) clinical grading system was used to measure quadriceps muscle strength (QMS). 40 The elderly patient was asked to sit on the edge of the examination table with the hip and knee joints bent at 90°. A strap was used to stabilize the thighs to prevent any unwanted movements. The dynamometer force gauge was positioned on the anterior part of the shin, just above the medial malleolus. An inflexible strap was used to secure the force gauge and the fixed base of the table, which was positioned behind the elderly patient’s foot. The participant was instructed to push their foot against the strap as much as possible. QMS (leg strength) was calculated based on the maximum force exerted by the participant on the spring gauge in kg and for the left and right legs separately. The data from these measurements were sorted from the lowest to the highest value. The lower third was considered as low muscle strength and the rest as normal muscle strength. 41
Balance
Two instruments were used to test balance, the Berg Balance Scale (BBS) and the Timed Up and Go test (TUG test):
1. Stand up from the chair, 2. Walk to the line on the floor at your normal pace, 3. Turn around, 4. Walk back to the chair at your normal pace, and 5. Sit down again. The execution time of the test was measured with a timer. In this study, the total time to perform this test was considered. This test was repeated three times in the interval when the person did not feel tired, and the average of the three tests was recorded as the record for each subject. The cut-off point for this test was ≥12 s for community-dwelling older adults. More than 12 s indicated that the older adults were at high risk for falls. 47 A tape measure was used to measure the distance.
Other Covariates
Baseline characteristics include age, sex, marital status, positive self-report of diabetes mellitus
Statistical Analysis
In the present study, the path analysis approach using AMOS24 software was applied to examine the research models. The assumptions of univariate normality were evaluated based on skewness and elongation values, and multivariate normality was evaluated using Merdia’s standardized elongation coefficient and critical ratio. The results showed that the univariate normal distribution of the research variables was confirmed since the values of skewness and elongation of the variables were in the range of ±2. In the present study, since the Merdia coefficient was 4.621 and the critical ratio was 2.598 less than 5, the normality assumption was confirmed for several variables. In addition, chi-square tests, analysis of variance, and Pearson’s correlation coefficient were performed based on SPSS 26.
Ethical Approval
The present study was conducted after approval by the Ethics Committee of Babol University of Medical Sciences with code IR. MUBABOL.HRI.REC.1397.019. All methods were carried out in accordance with the relevant guidelines and regulations. Participants were informed about the aims of the study and all signed the informed consent form.
Results
Demographic Characteristics of the Older Adults Living in Amirkola City and Their Comparison Based on Frailty.
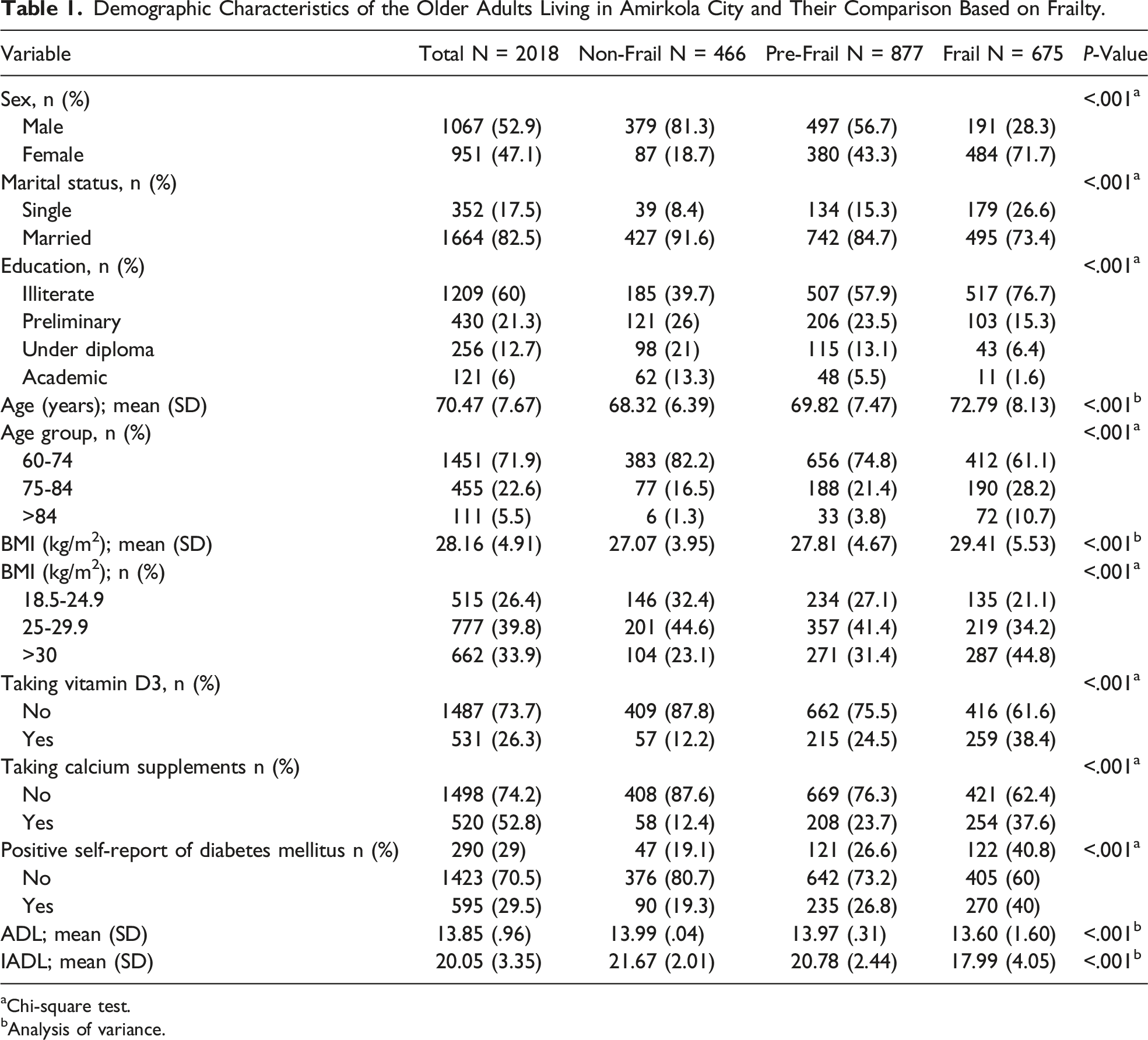
aChi-square test.
bAnalysis of variance.
Mean and Standard Deviation of Muscle Strength and Balance Variables and Their Comparison Based on Frailty.
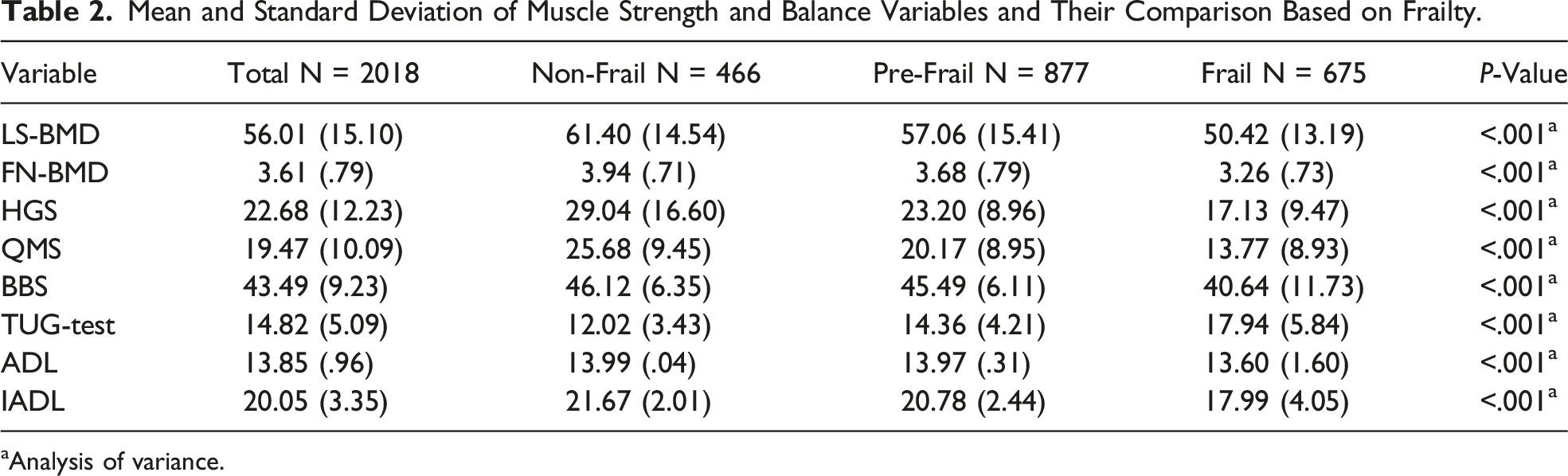
aAnalysis of variance.
In the present study, the structural equation modeling method was used to investigate the role of the mediator variable. Because the independent variable had three levels, three separate models (Model 1: Frail = 1; Non-Frail = 0, Model 2: Pre-Frail = 1; Non-Frail = 0, and Model 3: Frail = 1; Pre-Frail = 0) were assessed. In the ongoing study, background and clinical variables that indicated a significant relationship with dependent variables (main outcomes) were included as covariates. To identify the covariate variables, tetrachoric correlation coefficient tests were first used for two-level demographic variables, multidisciplinary correlation coefficients for more than two-level demographic variables, and Pearson correlation for quantitative variables. Results illustrated that gender and IADL variables were included as covariates in the model (Model 1).
Model 1
The Goodness-Of-Fit Indices of the Three Proposed Models.

Abbreviations: Model1: Frail vs non-Frail, Model2: Pre-Frail vs non-Frail, Model3: Frail vs Pre-Frail; RMSEA: Root Mean Square Error of Approximation; PCFI: Parsimonious Comparative Fit Index; PNFI: Parsimonious Normed Fit Index; GFI: Goodness-of-Fit Index; IFI: Incremental Fit Index; CFI: Comparative Fit Index; SRMR: Standardized Root Mean Residual; AIC: Akaike Information Criterion.
Note: Fit indices: PNFI, PCFI (>0.5), CFI, IFI, GFI (>0.9), RMSEA, SRMR (<0.08), CMIN/DF (<3 good, <5 acceptable).

Model 1 with standardized estimates.
Standard Coefficients of the Direct Paths of Model 1.

Note: IADL: Instrumental activities of daily living; TUG-test: Timed Up and Go Test; BBS: Berg Balance Scale; QMS: Quadriceps Muscle Strength; HGS: Handgrip Strength; FN-BMD: Femoral Neck Bone Mineral Density
Standard Coefficients of the Indirect Paths of Model 1.
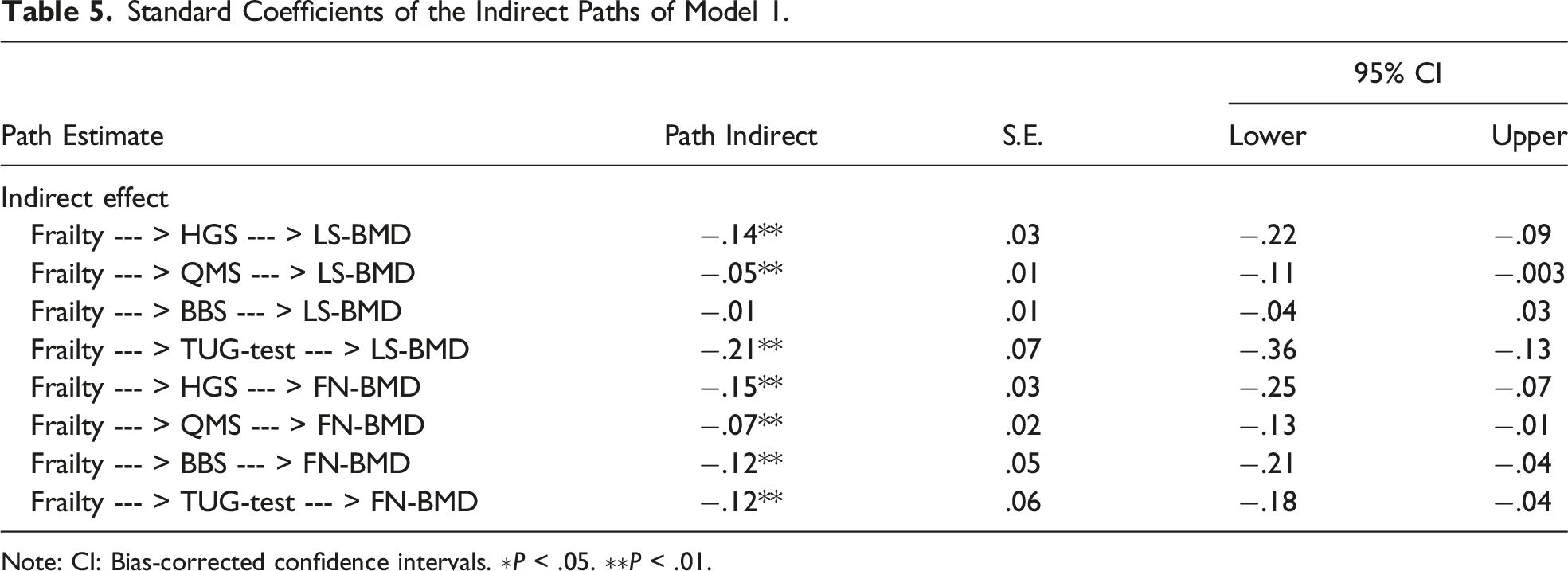
Note: CI: Bias-corrected confidence intervals. ∗P < .05. ∗∗P < .01.
Model 2
In model 2, the independent variable was defined as binary. The results demonstrated that the proposed model of the mediating role of muscle strength and balance in the relationship between FS (pre-frail = 1; non-frail = 0) and osteoporosis had acceptable goodness-of-fit (Table 3) (Figure 3). Model 2 with standardized estimates.
Standard Coefficients of Direct Paths of Model 2.

Standard Coefficients of the Indirect Paths of Model 2.

Model 3
In model 3, the independent variable was defined as binary. The results represented that the proposed model of the mediating role of muscle strength and balance in the relationship between FS (pre-frail = 1; non-frail = 0) and osteoporosis had acceptable goodness-of-fit (Table 3) (Figure 4). Model 3 with standardized estimates.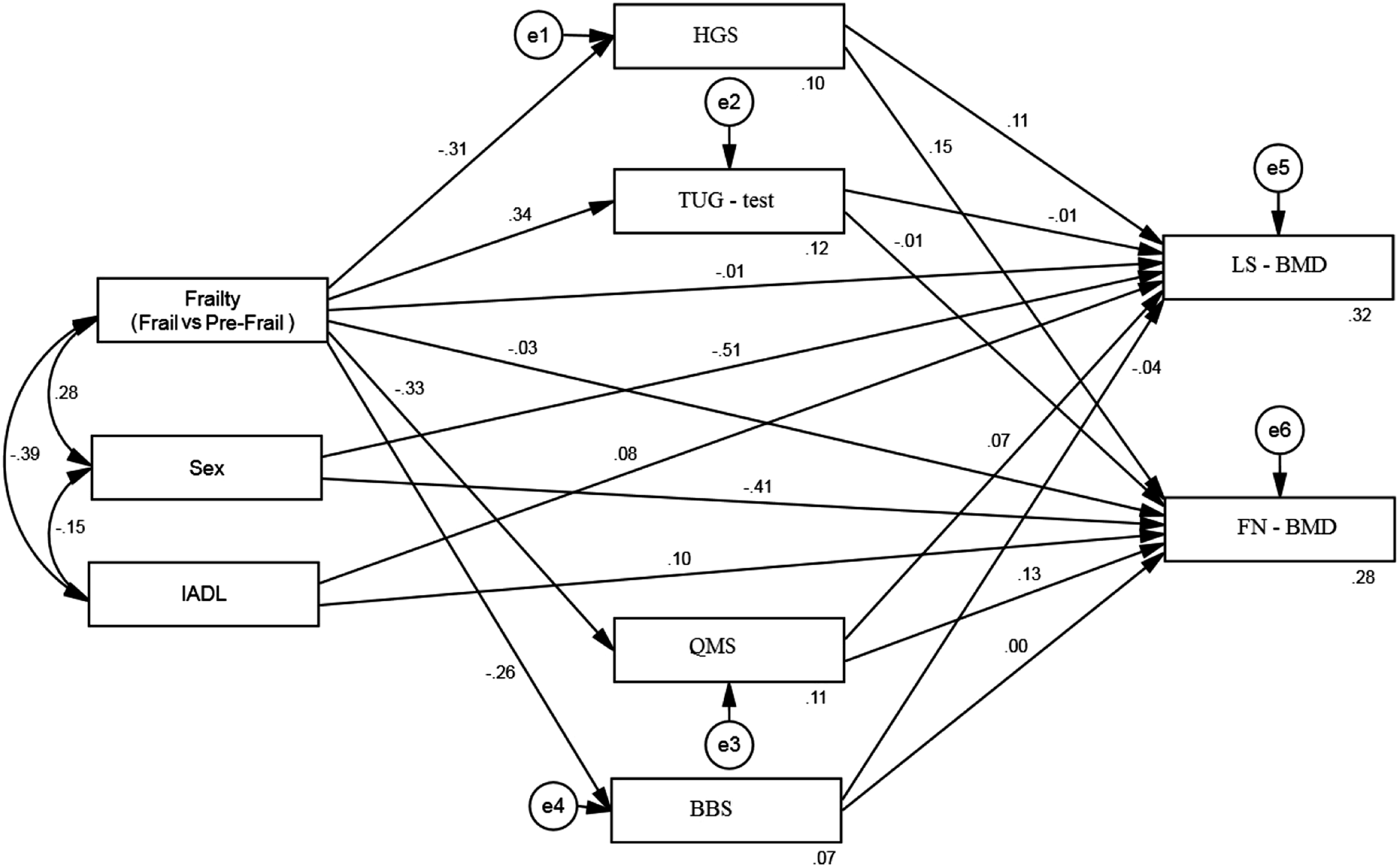
Standard Coefficients of the Direct Paths of Model 3.

Standard Coefficients of the Indirect Paths of Model 3.

Note: CI: Bias-corrected confidence intervals. ∗P < .05; ∗∗P < .01.
Discussion
This study aimed to examine whether muscle strength and balance mediate the relationship between frailty syndrome (FS) and osteoporosis in a large sample of community-dwelling older adults. The results of the current study have revealed that frailty is associated with osteoporosis. The results of Wang et al (2015) also suggested that osteoporosis occurred in 26.3% of males and 38.5% of females in the frail group, 13.6% of males and 16.2% of females in the pre-frail group, and 1.6% of males and 1.9% of females in the non-frail group (P < 0.05). There was a significant relationship between age ≥80 years (OR 4.8; 95% CI, 3.05-10.76; P = 0.027), female gender (OR 2.6; 95% CI, 1.18-2.76; P = 0.036), and the presence of underlying chronic diseases (OR 3.71; 95% CI, 1.61-10.43; P = 0.021) with the occurrence of osteoporosis. 51
In the present study, the BMD of the older adults was calculated to diagnose osteoporosis. The results of this study indicated that the LSBMD and FNBMD values in g/cm2 were lower in both the frail and pre-frail groups than in the non-frail group. However, no significant difference was found between LSBMD and FNBMD when comparing the two frail and pre-frail groups.
In the present study, even after adjusting for gender and IADL, a strong relationship was found between frailty and low BMD in the lumbar spine and femoral neck of the older adults. This finding is consistent with the results of previous studies.52–54 In addition, an independent negative and significant relationship was observed between frailty (in both the pre-frail and frail groups) and BMD (β = −0.074 and −0.092, respectively, P < 0.05) in the study of Wysham et al 53 (2020).
In the study by Kenny et al (2006), the FNBMD T-score decreased significantly with increasing frailty level (P = 0.014). The results of Kenny’s study showed that two components of the frailty model, including HGS and walking speed, were independently associated with a low FNBMD score. 52
In the study by De Sousa e Silva Araujo et al (2017) after adjusting for age and BMI, lower BMD values were significantly associated with frailty (P = 0.033) and pre-frailty in females (P = 0.037). 55
Consistent with the results of the present study, the results of the study by Lee et al (2015) also illustrated that there was a statistically significant relationship between osteoporosis (OR: 7.73, 95% CI: 5.01-11.90, P < 0.001), hip fracture (OR: 8.66, 95% CI: 2.47-30.40, P = 0.001) and falls (O.R: 2.53, 95% CI: 1.35-4.76, P = 0.004) with frailty.
The odds ratio (OR) for the occurrence of osteoporosis in females was 2.62 and 8.25 in the pre-frail and frail groups, respectively. In the current study, frailty was found to be closely associated with a reduction in BMD and low muscle strength. 14
However, in a Swedish study of elderly females >75 years of age, no association between BMD and frailty was found. 56 This discrepancy may be attributed to the complexity of the development of frailty as a multifactorial syndrome associated with neuromuscular and neuroendocrine changes, which in turn cause changes in body composition. 57
The results of the ongoing study showed that LSBMD and FNBMD were lower in females than in males in all three groups (frail, pre-frail, and non-frail), with the largest effect in the pre-frail group.
To explain this result, it can be said that the process of bone regeneration is influenced by the changes in estrogen production that occur after menopause. Estrogen deficiency leads to decreased bone metabolic activity, increased bone resorption, and consequently progressive loss of trabecular bone. 58
The decline in sex hormones with age is one of the factors that affect the decrease in BMD in males and females. In females, the decrease in estrogen, caused by menopause is the main reason for the decrease in BMD, as this hormone is responsible for stabilizing calcium in bones and maintaining bone density. 59
The results of previous studies have indicated that low BMD with osteoporosis and fractures is more common in females.55,59,60 The results of the study by Lee et al (2015) demonstrated that the OR for osteoporosis in females was 2.62 and 8.25 in the pre-frail and frail groups, respectively. 14 However, in sex-stratified analyses, there was no significant relationship between frailty and BMD in the females studied by Wysham et al 53 whereas a strong independent negative relationship between frailty and BMD was found in males (β = −0.247, P = 0.001).
The results represented that HGS and QMS were lower in the pre-frail group compared with non-frail group, in frail group the pre-frail group, and in the frail group compared with non-frail group. This result can be explained by the pathophysiological feature of FS, in which older adults with FS have lower muscle strength. 61 In the study by Lenardt et al 61 (2016), the mean HGS of pre-frail and frail older adults was 28.4 ± 9.8 kg and 22.5 ± 6.1 kg, respectively, and the mean HGS of frail older adults was significantly lower than that of pre-frail older adults (P = 0.000).
Additionally, the results of the current study have shown that muscle strength (HGS and QMS) is associated with osteoporosis and plays a mediating role in the relationship between frailty and osteoporosis. This finding is consistent with the results of other studies.16,62–66
The results of Choi et al’s study (2023) illustrated that after adjusting for age, people with osteoporosis had a 1.684-fold higher prevalence of low muscle strength (LMS) than people without osteoporosis (OR, 1.684; 95% confidence interval (CI), 1.500-1.890). 62
Mcgrath et al 66 (2017) also reported that in the US general population aged ≥40 years, the odds of developing osteoporosis increased by approximately 6% in men and 10% in females for every 0.1 kg decrease in HGS.
To explain the results of the present study, it can be said that higher HGS levels probably increase BMD and ultimately decrease the prevalence of osteoporosis. These results may be explained by the effect of myokines (eg, irisin) released by muscles on bone health. 67 Irisin levels are positively related to bone density status, and low irisin levels may lead to an increase in the incidence of hip fractures. 68
The findings of the current study illustrated that BBS had no statistically significant difference in the pre-frail group compared with non-frail group. However, it was lower in the frail group compared with pre-frail group, and lower in the frail group compared with non-frail group.
Furthermore, the TUG test was higher in the pre-frail group than in the non-frail group, higher in the frail group than in the pre-frail group, and higher in the frail group than in the non-frail group. Eagles et al (2017) expressed that TUG test and BBS outcomes can predict declines in performance and frailty in the older adults. Therefore, the calculation of BBS and TUG test scores can be readily incorporated into planned functional assessments of the older adults. 69
It can also be used as part of a frailty screening tool to prevent a variety of outcomes such as falls and fractures in the elderly. 70 Although in terms of BBS scoring, 1.3% of the older adults in the ongoing study had severe balance impairment and 7.8% had moderate balance impairment, the low BBS scores and high TUG test scores in the frail group indicated that these older adults were at risk for falls. Some subgroup analyses suggest that TUG test may play a role in predicting falls in older people with impaired function.71,72 Large et al 73 (2006) revealed that individuals with high TUG test levels had an 11% increase in the rate of falls compared with individuals with normal TUG test. Abreu et al 74 (2009) expressed that there was no correlation between a positive history of falls and TUG test or BBS levels.
In summary, balance (BBS and TUG test) and muscle strength (HGS and QMS) are associated with osteoporosis, and these variables play a mediating role in the relationship between frailty and osteoporosis. Abreu et al 74 (2009) also reported that there was a significant difference in BBS and TUG test scores between older adults with and without osteoporosis, confirming the role of balance and muscle strength in the occurrence of osteoporosis.
To explain this finding, it can be said that older adults with imbalances or problems with muscle strength are people who are weak and have very low physical performance. 75 Hence, these people have FS symptoms and are prone to osteoporosis. Therefore, FS intervention programs that take an integrated approach to building muscle strength and maintaining balance are recommended. 14
As a secondary finding of the present study, the results indicated that frailty was related to the intake of vitamin D3 by the older adults studied. However, the results of the study by Cai et al (2022) suggested that taking a high dose of vitamin D3 did not prevent frailty. This result may be due to the frequency of type 1 errors in Cai’s study.
They also reported that there was no significant relationship between vitamin D3 dose and frailty status in stratified analyses based on serum (OH) D25 level. 76 During the 8-year follow-up by Bolzetta et al 77 (2018), no difference in the incidence of frailty was observed with the status of taking vitamin D3 supplementation, even after adjusting for 13 baseline confounders (HR = 0.95; 95% CI: 0.72-1.25).
According to the results of the ongoing study and other results reported by other investigators, future studies that consider frailty as a consequence of vitamin D3 intake are essential to evaluate whether vitamin D3 supplementation can be associated with a lower incidence of frailty over time.
The strengths of this study are numerous and interconnected, enhancing its validity and relevance in the field of geriatric health. Firstly, the utilization of population-based data significantly bolsters the representativeness and generalizability of our findings. These results underscore the crucial need for timely interventions aimed at preventing or delaying FS and its detrimental consequences, including osteoporosis. Furthermore, the degree of FS was assessed using a validated and reliable instrument, ensuring the accuracy of our data and aiding in the precise identification and management of elderly conditions. This is crucial for informed decision-making at both clinical and policy levels.
Additionally, the determination of participants’ osteoporosis status through DEXA scans, such as BMD measurements, provided a precise reflection of bone health, anchoring our results in reliable diagnostic standards. The employment of structural equation modeling allowed for an in-depth evaluation of the complex relationships between variables, a methodological approach that stands out given its extensive use across various disciplines within the humanities and behavioral sciences. Finally, this study marks a pioneering effort in the comprehensive analysis of factors influencing osteoporosis among the Iranian older adults, offering novel insights into the mediating variables that are critical for planning effective health interventions and understanding the intricate dynamics between frailty and bone health. The present study has limitations that should be considered when interpreting the results. Sampling in one city makes it difficult to generalize the study to the total population of the elderly in Iran due to ethnic and cultural diversity. Besides, the cross-sectional design of the study may have limited the ability to explore the causal relationship between FS and osteoporosis. Serum levels of vitamin D were evaluated in the AHAP cohort study, but because of the large amount of missing data, data on serum levels of vitamin D were not included in the present study, which is another limitation of the current study. This study did not examine the duration of calcium and vitamin D supplement use.
Conclusion
The results of the current study have indicated that balance (BBS and TUG test) and muscle strength (HGS and QMS) are associated with osteoporosis, and these variables play a mediating role in the relationship between frailty and osteoporosis. Therefore, FS intervention program to reduce osteoporosis incidence should provide an integrated approach to muscle strength and balance maintenance.
Supplemental Material
Supplemental Material - Effects of Frailty Syndrome on Osteoporosis, Focusing on the Mediating Effect of Muscle Strength and Balance in Community-Dwelling Older Adults (≥60 years) in Iran: Results From the Amirkola Health and Aging Project Cohort Study
Supplemental Material for Effects of Frailty Syndrome on Osteoporosis, Focusing on the Mediating Effect of Muscle Strength and Balance in Community-Dwelling Older Adults (≥60 years) in Iran: Results From the Amirkola Health and Aging Project Cohort Study by Abbas Shamsalinia, Seyed Reza Hosseini, Ali Bijani, Reza Ghadimi, Mohammad Reza Kordbageri, Kiyana Saadati, and Fatemeh Ghaffari in Geriatric Orthopaedic Surgery & Rehabilitation
Footnotes
Acknowledgements
The authors would like to thank the Deputy of Research and Technology of Babol University of Medical Sciences, the Social Determinants of Health Research Center, and all individuals who contributed to the AHAP cohort study.
Author’s Contributions
SH. SRH. A.B. R.G and F.G. contributed to the conception and design of the study and the study protocol. SRH. AB. RG. and F.G. managed the running of the study. MR.K conducted data analysis, prepared Figures 1-![]() and all authors helped with data interpretation. F.G. A. SH. and K.S. wrote this manuscript with input from all co-authors. All authors critically reviewed, provided feedback, and approved the final version of the manuscript.
and all authors helped with data interpretation. F.G. A. SH. and K.S. wrote this manuscript with input from all co-authors. All authors critically reviewed, provided feedback, and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

