Abstract
Atypical femoral fractures (AFF) are stress or insufficiency fractures induced by low energy trauma or no trauma, frequently correlated with prolonged bisphosphonate therapy. The diagnosis follows major and minor criteria, originally described by the Task Force of the American Society for Bone and Mineral Research in 2010 and updated in 2014. However, the definition of AFFs in the report excluded periprosthetic fractures. When atypical fractures occur close to a prosthetic implant the situation become critical, the surgical treatment is often demolitive and supported by medical treatment. Moreover, acute ORIF as a first line treatment is frequently burdened by a high failure rate , and often a stem revision is required as second line treatment. The healing process is long and difficult with poor functional results and impairing outcomes. We present a case treated at our institution of a 78 year old woman with a history of a femoral atypical periprosthetic fracture, complicated by multiple surgical revisions. Its arduous management reflects all the difficulties that these type of fractures could present to the surgeon, while its good final result may teach us how to approach them in a correct way.
Keywords
Introduction
2010 American Society for Bone and Mineral Research Task Force Case Definition of Atypical Femoral Fracturess.

Clinical Case
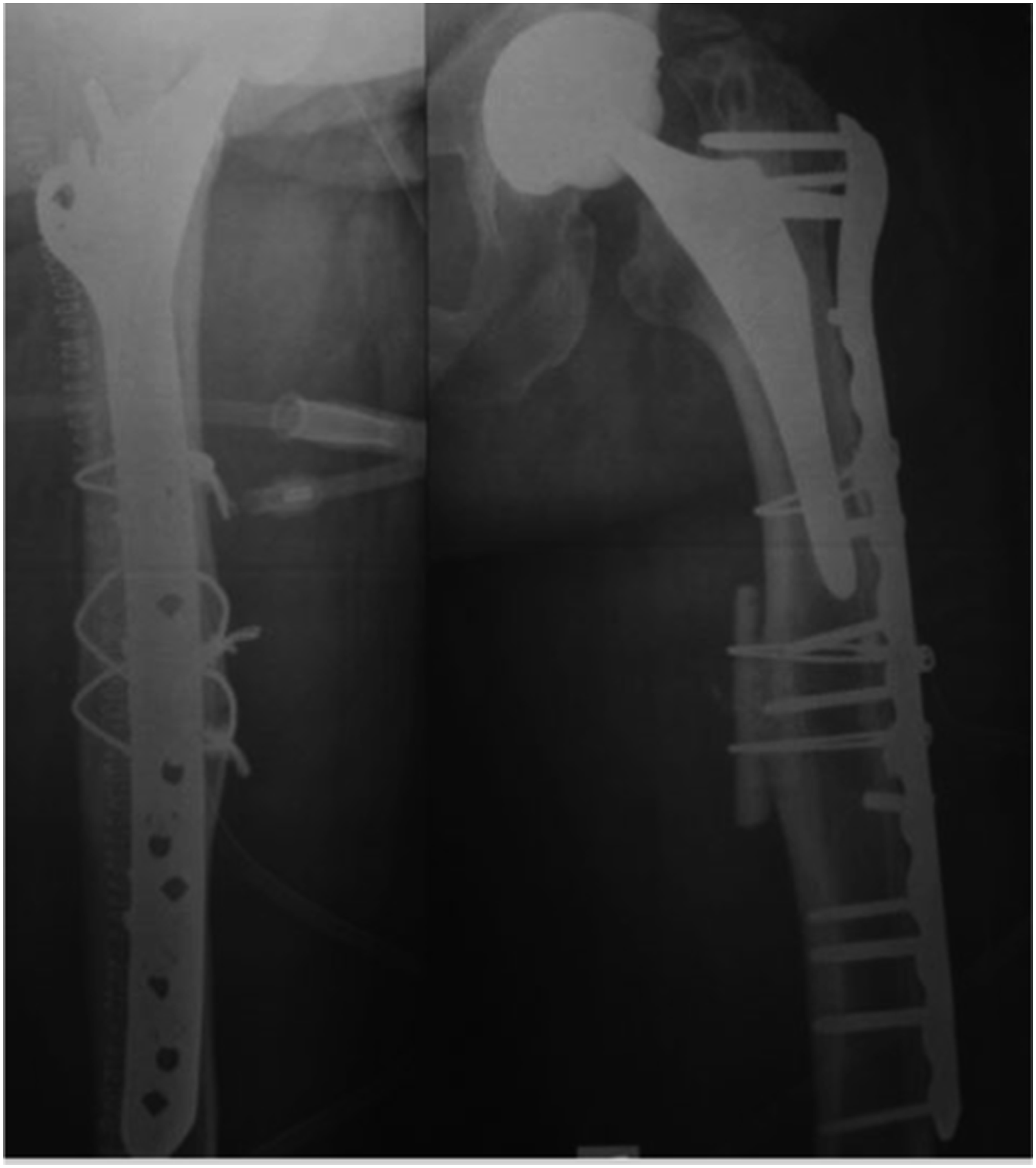
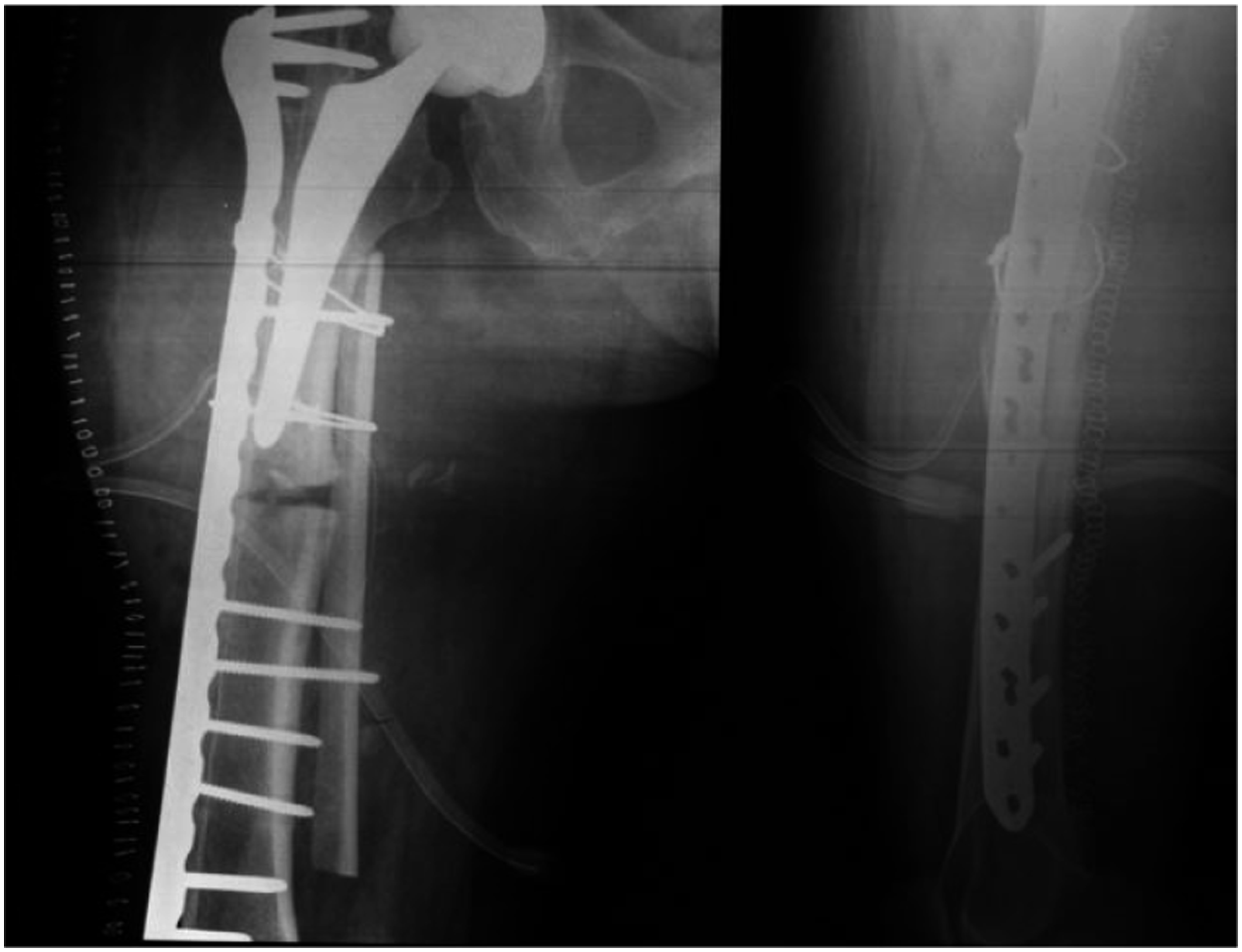
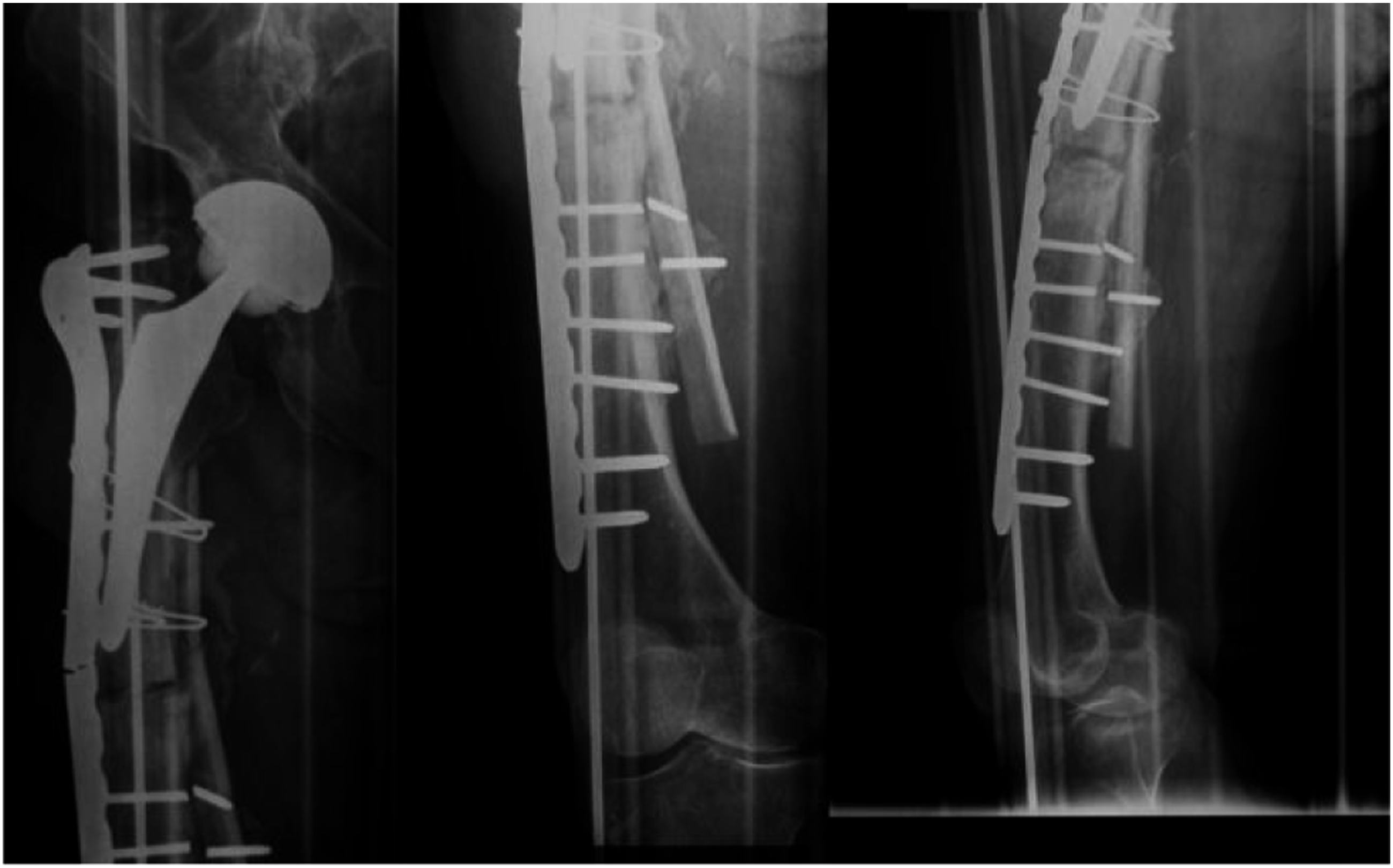
A 78-year-old woman with right cementless THA implanted following a femoral neck fracture in 2012, fell in August 2015. Radiographs revealed a periprosthetic non-comminuted transverse fracture with medial cortical thickening just distal to the tip of the stem (Figure 1). The patient also reported aggravating tight pain over the past 3 months. The patient comorbidities were heart disease, diabetes mellitus type 2 and osteoporosis, treated with Alendronic acid (70 mg/week) and cholecalciferol for 3 years. The patient underwent open reduction and internal fixation (ORIF) with NCB periprosthetic femur plate system (Zimmer Biomet, Warsaw), cerclages and medial cortical strut allograft (Figure 2). Intraoperatively the stem was found to be stable. Alendronic acid was suspended and substituted with Teriparatide. After the surgery, the patient was allowed to early active and passive mobilization with partial weight bearing with walker. However, only after 4 days she sustained a hardware failure (Figure 3). The patient underwent a new surgical open reduction and internal fixation with a longer plate augmented with a longer cortical strut on the medial side (Figure 4). Wheelchair use and non-weight bearing were advised for 30 days. After 6 months of partial weight bearing the patient returned to our hospital for the onset of right tight pain. Radiographic evaluation revealed breakage of the locking plate and a refracture at the same place (Figure 5). Implant instability and bone stock deficiency were found during surgery. The prosthesis was then replaced with an Arcos Modular Femoral Revision Stem Interlocking 20 mm × 250 mm (Zimmer Biomet, Warsaw) distally locked with 3 cortical screws. The ceramic head was replaced too with a 36 mm Biolox Delta (Figure 6). She was allowed to protected weight bearing walking for 6 weeks and then full weight bearing. At 6 month follow up the fracture was considered clinically healed but radiographic union was still incomplete. However, the patient was able to walk with a walker without pain. Transverse vancouver B1 periprosthetic fracture with cortical thickening. Open reduction and internal fixation with NCB periprosthetic femur plate system (Zimmer Biomet, Warsaw), cerclages and medial cortical strut allograft. Hardware failure 4 days after the surgery. Reduction and fixation with a longer plate and a longer cortical strut on the medial side. Refracture at the same level after 6 months of partial weight bearing. Revision surgery with arcos modular femoral revision stem interlocking 20 mm × 250 mm (Zimmer Biomet, Warsaw) distally locked with 3 cortical screws.


Discussion
Reviewed articles.

BPs: bisphosphonate; F: female; ALN: alendronate; RIS: risedronate; N/S: not specified; RA: rheumatoid arthritis; GCs: glucocorticoids; CONS: conservative; ORIF: open reduction internal fixation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
