Abstract
Aims
To explore clinical characteristics, perioperative management and outcomes of Hip Fracture patients with advanced Chronic Kidney Disease (HF-aCKD) compared to the general Hip Fracture population without aCKD (HF-G) within a large volume tertiary hospital in Western Australia.
Methods
Retrospective chart review of patients admitted with hip fracture (HF) to a single large volume tertiary hospital registered on Australian and New Zealand Hip Fracture Registry (ANZHFR). We compared baseline demographic and clinical frailty scale (CFS) of HF-aCKD (n = 74), defined as eGFR < 30 mls/min/1.73 m2, with HF-G (n = 452) and determined their outcomes at 120 days.
Results
We identified 74 (6.97%) HF patients with aCKD. General demographics were similar in HF-aCKD and HF-G populations. 120-days mortality for HF-aCKD was double that of HF-G population (34% vs 17%, P = .001). For dialysis patients, 120-days mortality was triple that of HF-G population (57%). Except for the fit category of HF-aCKD group, higher CFS was associated with higher 120-days mortality in both groups.
Of all HF-aCKD patients, 96% had operative intervention and 48% received blood transfusion. There were no new starts to dialysis peri-operatively. Each point reduction in eGFR below 12 mL/min/1.73 m2 was associated with 3% increased probability of death in hospital.
Conclusions
120-days mortality was double in HF-aCKD and triple in HF-dialysis that of the HF-G within our institution. Clinical frailty scale can be useful in predicting mortality after HF in frail aCKD patients. High rate of blood transfusions was observed in HF-aCKD group. Further studies with larger HF-aCKD numbers are required to explore these associations in detail.
Introduction
There are more than 25 000 presentations of Hip Fracture (HF) across Australia and New Zealand each year associated with significant morbidity, mortality and an annual economic cost of approximately 1 billion dollars. 1 Among HF patients, those suffering from chronic kidney disease (CKD) are of interest due to impaired bone and mineral metabolism predisposing to fractures. 2 The global burden of CKD is rising, with a recent systematic review revealing a prevalence of 13.4%. 3 Dialysis patients have a higher incidence of HFs (1.23%) compared to the general population (.30%). 4 In addition, dialysis patients have an increased 1-year mortality rate of 55-64%4,5 almost 2 and half times greater than the general population. 4 A higher incidence of HFs is also noted in patients with earlier stages of chronic kidney disease,6,7 which leads to greater in-hospital mortality, length of stay and costs of admission. 7
Hip fractures are a significant concern to Australia’s ageing population, with the average age of HF in Australia being 82, and a quarter of presentations affecting those aged 90 years and older. 1 These patients are often frail and present with multiple comorbidities, therefore optimising survival and regain of function requires multidisciplinary approach to perioperative management. Frailty is a predictor of increased mortality and post-operative complications in geriatric surgical populations.8,9 Clinical Frailty Scale (CFS) has been demonstrated to be a useful bedside tool for frailty assessment, allowing incorporation into routine clinical practice and assisting in key clinical decisions. 10
Given the increased incidence of HF in CKD and the additional morbidity and mortality associated with it, understanding the characteristics of this cohort and identifying factors associated with key clinical outcomes is important. Fiona Stanley Hospital (FSH) is a leading centre in HF management with over 500 fracture presentations each year. 1 In this study, we aimed to explore and compare characteristics of Hip Fracture patients with advanced Chronic Kidney Disease (HF-aCKD) and without CKD (HF-G), including their peri-operative management and outcomes within a tertiary hospital in Western Australia.
Materials and Methods
Study Design and Methods
In this retrospective audit, we reviewed medical records of patients admitted with fragility HF to FSH, as recorded on Australian and New Zealand Hip Fracture Registry (ANZHFR).
The HF-aCKD group was identified by reviewing the admission eGFR of all patients admitted over 2 years between January 1, 2017 to December 31, 2018 with a HF at FSH. Advanced CKD (aCKD) was defined as a history of known CKD prior to admission and defined as CKD stage 4 and 5 (an estimated glomerular filtration rate (eGFR) of ≤30 mL/min/1.73 m2). Patients with acute kidney injury (AKI) cases were excluded.
The HF-G group was also derived from the ANZHFR database over a 11-months period between November 1, 2017 and October 31, 2018. Advanced Chronic Kidney Disease patients in this group were excluded to factor out overlapping cases. Hip Fracture admission was defined as a period from when patients were admitted to hospital until discharge to usual place of residence, residential care or death. Where multiple HF admissions occurred, the most recent was selected. Ethics approval was obtained from our hospital’s office of ethics and research governance prior to commencement of the study.
Data Collection
Demographic information was extracted from registry data for both groups. In addition to baseline demographics, Clinical Frailty Scale (CFS) and 120-days mortality were collected for both populations. CFS was retrospectively assessed based on information written by medical and allied health teams in patients’ progress notes. CFS assessment was done by 2 independent clinicians (KD and CO) for HF-aCKD group and 1 clinician (SN) for HF-G group. Inconclusive cases were subsequently reviewed and resolved by a geriatrician (PK). CFS is further categorised into fit (CFS 1-3), vulnerable (CFS 4), frail (CFS 5-6) and severely frail (≥7).
In the aCKD group, additional details on co-morbidities were identified from discharge summaries and anaesthetic records and measured using the Charlson Comorbidity Index (CCI). Outcomes were categorised into biochemical, clinical and social. Biochemical information was obtained through iSOFT software. Anaemia was defined as a haemoglobin of < 115 g/L, hyperkalaemia as a potassium level of > 5.2 mmol/l and acidosis as having bicarbonate of < 22 mmol/l. Other clinical information such as transfusion requirement, death and discharge destination were extracted by reference to medical records and discharge summary.
Statistical Analysis
Statistical analyses were performed via “R statistical package” Comprehensive R Archive Network (CRAN) and SPSS. Chi-squared regression and independent variable t-test was used for categorical and numerical data respectively. Levene test was used to determine variance. For HF-aCKD group, baseline demographic variables were tested individually in a logistic regression model to explore characteristics which were predictive of mortality. 95% confidence intervals [CI] and the statistical significance level was set at .05 for each significance test.
Results
A total of 1061 HF cases were identified between January 1, 2017 and December 31, 2018. For HF-aCKD population, 82 cases had eGFR ≤ 30 on admission, 8 were excluded due to AKI, giving rise to 74 patients (6.97%) in the study group. For the HF-G group, 509 HF patients were identified between November 1, 2017 to October 31, 2018. 57 CKD patients were excluded to factor out overlapping cases. (Figure 1) Sample selection.
Demographics
Baseline Demographics.

Patients in HF-aCKD and HF-G group were mainly frail (64.9% vs 34.8%) according to CFS category. There was higher proportion of severely frail patients (CFS ≥ 7) in HF-G compared to HF-aCKD group (30.1% vs 9.5%). (Figure 2). Clinical frailty scale (CFS).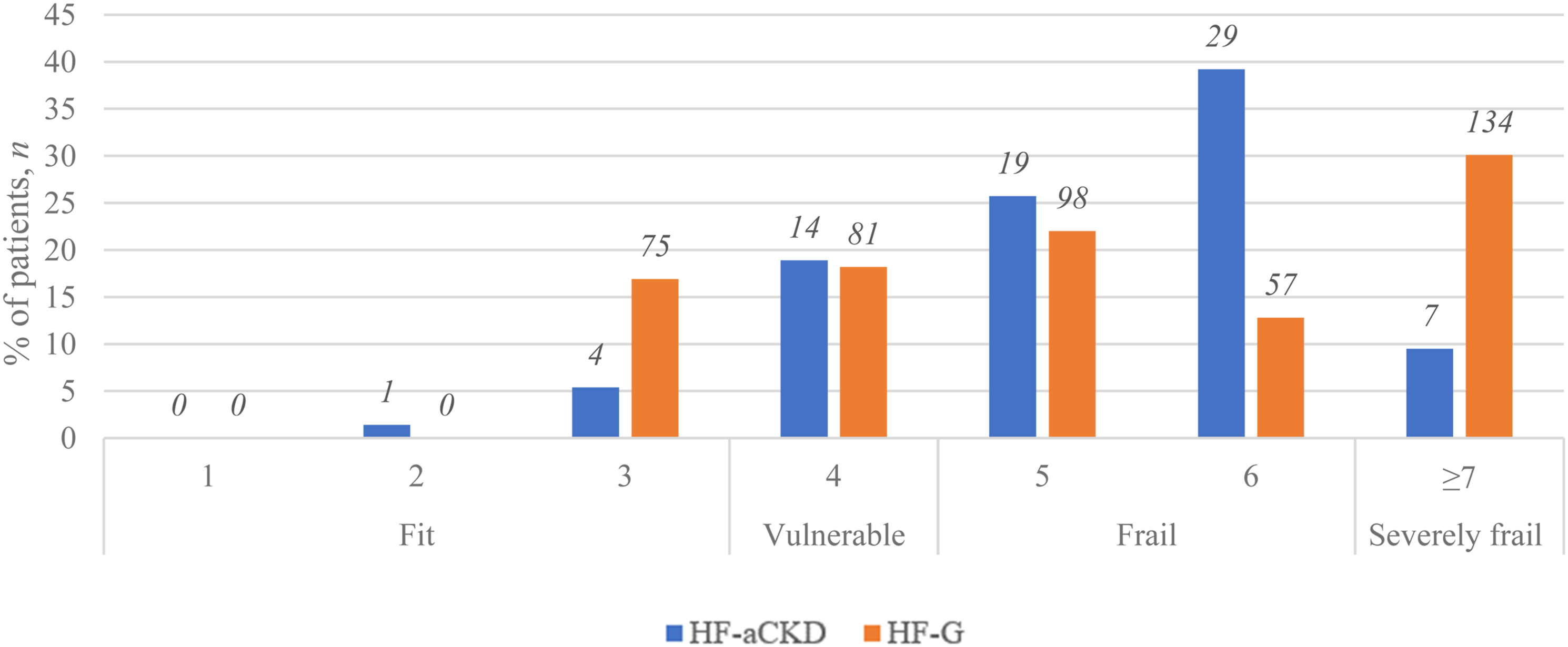
Clinical Management
Majority (95.9%) of HF-aCKD patients underwent operative intervention. Almost half (48%) of HF-aCKD cohort received transfusion during admission. Majority (93.2%) did not require critical care admission postoperatively. There were no patients who became newly dialysis dependent peri-operatively.
Post-Operative Outcomes
Biochemical
Perioperative Laboratory and Clinical Outcomes in HF-aCKD Patients.

Clinical
Mortality rate for HF-aCKD was 9.5% in hospital and 33.8% at 120 days. For the 7 patients on dialysis pre-operatively, the in-hospital mortality rate was 14.3% and 120-days mortality at 57.1%. 120-days mortality rate was significantly higher among HF-aCKD compared to HF-G (17%) (P = .001). Higher CFS scores were associated with higher 120-days mortality for both HF-aCKD (P = .032) and HF-G, except for the fit category of HF-aCKD group (Figure 3). All patients that were managed non-operatively died in hospital following a shared decision for palliation. 120 days mortality according to CFS category HF-aCKD vs HF-G.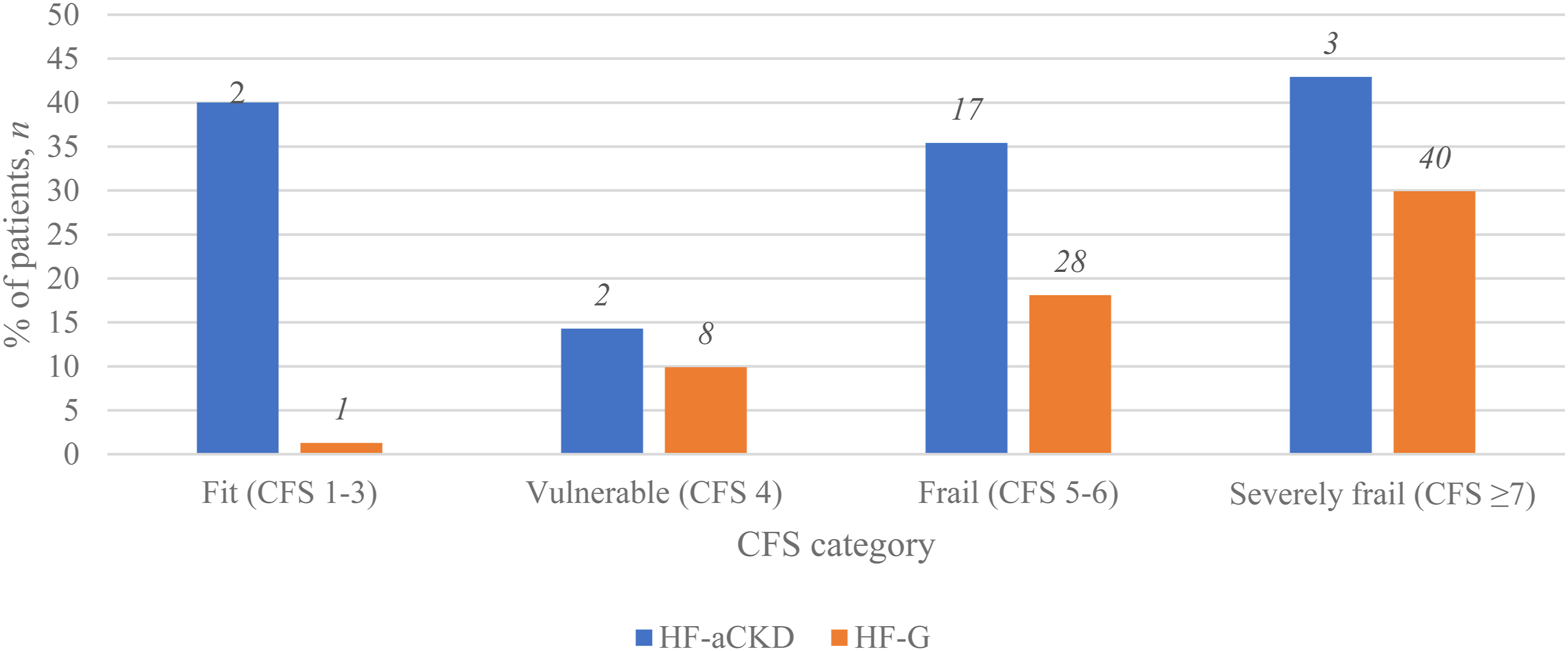
Social
Out of the 56 HF-aCKD patients who were originally from home, only half (51.8%) returned home even after a period of inpatient rehabilitation. One-third (30.4%) went into nursing home, about one-tenth (10.7%) died, and the remaining (7.14%) had unknown discharge destination. Most patients that returned home were independent with instrumental activities of daily living (ADL) (86%) and lived with other family members (71%) prior to HF. Majority (94.4%) of HF-aCKD patients from residential care returned to nursing home post HF with 1 died in hospital. 120 days mortality was 26.8% for patients who lived at home and doubled to 55.6% for nursing home residents.
Possible Predictive Factors for Outcome of Death in HF-aCKD
By using logistic regression, we identified 3 pre-fracture variables which were independently associated with 120-days mortality in HF-aCKD: nursing home residence, walking with a frame and increasing CFS. Patients who were from nursing home at the time of HF had a higher 120-days mortality compared to patients who lived at home (P = .028). Patients who walked with a frame had higher 120-days mortality than those who walked unaided (P = .047). There was no statistically significant difference in 120-days mortality between patients who walked unaided and those who walked with a stick or cane. Increasing CFS was associated with higher 120-days mortality (P = .032). Patient with a CFS of 7 has 55% risk of death at 120 days compared to 17% in CFS 4.
Association of pre-admission eGFR with 120-days mortality was not statistically significant (P = .058). However, where admission eGFR was less than 12, each point reduction in eGFR was associated with 3% increased probability of death in hospital.
Discussion
This observational study provides insight into characteristics of HF patients with aCKD, their management within a tertiary hospital and clinically relevant outcomes.
Demographics in HF-aCKD population were similar to the HF-G group overall, representing an older adult population with fragility HFs that were mostly community dwelling older adults at presentation. Average age was above 80 years in both groups and of female predominance. The proportion of patients walking unaided in the HF-aCKD group was 10% less than HF-G, likely reflecting the negative impact of aCKD related pathology on mobility. Similarly, the higher proportion of “frail” CFS 5-6 patients may be due to the impact of aCKD related sarcopenia, immobility and comorbidity. 11 Interestingly, there was a lower proportion of severely frail patients in the HF-aCKD compared with HF-G. This is likely related to the higher mortality rate in aCKD group which reduces the survival of severely frail patients. 6
Our study demonstrated that increasing CFS was associated with increasing 120-days mortality. The probability of death at 120-days for HF-aCKD increases from 17% for CFS 4 to 55% for CFS ≥7. CFS scores have been found to be predictive of mortality in various literature.9,11 The unusually high mortality rate in the “fit” sub-group in our study may have been skewed due to low sample number (n = 5) however it may also reflect the high degree of medical comorbidities in aCKD not usually present in general HF patients who are robust. One patient with aCKD was too unwell to undergo surgery for HF and died in hospital from severe acute on chronic renal failure. One died from intracranial bleed and midline shift following a fall post discharge.
The in-hospital mortality of 10% for our aCKD group is similar to the HF-G population from registry data. This may reflect early recognition and management of this vulnerable group of patients at a high-volume tertiary centre with a mature orthogeriatric model of care and long-term participation in the ANZHFR. In particular, no patient was newly commenced on dialysis perioperatively which reflects pre-operative discussions clarifying the goals of care for each patient and multidisciplinary involvement of surgeons, anaesthetists and geriatricians to optimise care.
120-days mortality for all HF patients is approximately 13% in Australia. 1 Our study showed that the 120-days mortality for HF-aCKD group (34%) was double that of HF-G group (17%), and triple in dialysis patients (57%). These findings were similar to other studies that demonstrated high mortality rates of 44% among HF-CKD patients 12 and up to 70% among dialysis patients 13 at 1 year follow-up. One UK study has demonstrated 13.7 times greater risk of death for dialysis patients at 1 year post HF compared to non-dialysis patients. 13 This may be attributed to complications such as wound haematoma, infection, electrolyte imbalance and mechanical failure which were more common among CKD patients with HF. 14
Pre-fracture mobility is considered to reflect overall health status and therefore serves as a strong predictor for mortality.1,15 It is worth noting that patients in HF-aCKD group had statistically higher usage of walking aids and higher 120-days mortality rate as compared to HF-G. In the HF-aCKD population, patients who walked with frames had higher probability of death at 120 days than those who ambulate without assistance (P = .047). This observation is in keeping with findings from a prospective 10-years study which documented a 28% increased risk of mortality in patients who walked with assistive device prior to hospitalisation. 15 Our study did not demonstrate increased mortality associated with walking stick or cane usage. The ability to walk unaided and with a stick are often regarded as having similar level of independence by various literature and scoring system.16,17
In our study, only 51.8% of HF-aCKD patients who lived at home prior to their fracture managed to return home. However, in 2019 ANZHFR data, 1 73% of all HF patients returned to private residence at 120 days. This may be attributed to the higher rates of postoperative complications, greater comorbidities and frailty burden associated with aCKD, limiting return to independent community living. 14 In addition, patients on dialysis might not be able to participate in the standard rehabilitation due to additional time required for dialysis. 14 These factors heavily impact on functional recovery post-fracture. The majority of HF-aCKD patient that managed to return to private residence had protective factors including being previously independent with ADLs and living with other family members. 18
Our results showed high mortality rates among nursing home patients with aCKD, consistent with data from prior systematic reviews and meta-analysis. At 120 days, more than half of nursing home residents had died. A large retrospective cohort study of 60 111 patients with HF also found high mortality rate of 30% among nursing home residents at 180 days. 12
Similar to previous studies,19-21 our findings suggest an inverse relationship between eGFR and postoperative mortality in patients with eGFR ≤ 12 on admission. A systematic review and meta-analysis on cardiac and vascular surgeries demonstrated an elevated relative risk of mortality of 2.85 and 3.75 in patients with eGFR of < 30 mL/min/1.73 m2 and < 15 mL/min/1.73 m2 respectively compared to normal eGFR of > 90 mL/min/1.73 m2. 19 The impact of eGFR on non-cardiac non-vascular surgery remains as an area requiring further research. Hence, our study provides insight into the prognostic value of pre-operative eGFR in non-cardiac, non-vascular surgeries.
Blood transfusion rates in HF vary greatly depending on severity of anaemia, hospital protocols and transfusion threshold.22,23 Chronic Kidney Disease patients are more likely to need blood transfusion than non-CKD counterparts due to lower baseline haemoglobin and greater bleeding risk. 24 Majority of our HF-aCKD population were anaemic postoperatively and close to half received blood transfusion. This is significantly higher than the postoperative transfusion rate in other large observational studies.25,26 However, both anaemia and blood transfusions are independently associated with increased risk in mortality. 27 In our centre, patients presenting with HFs are routinely given intravenous iron intraoperatively. Intra-operative intravenous iron is associated with lower rates of blood transfusion, reduction in length of stay and improved 30-days mortality.27,28 Further research is needed to determine the efficacy of intravenous iron to manage perioperative anaemia and reduce mortality in patients with aCKD.
To our knowledge this is the first Australian based population study exploring the demographics, management as well as clinical outcomes of HF patients with aCKD. We were also able to draw on a sub-group of HF-G patients managed at our centre within an overlapping 12 months period for comparison. We have analysed routinely collected and clinically relevant demographic, biochemical, clinical and outcome data. This information can be directly extrapolated into routine practice to further assist health practitioners and patients in making complex and challenging decisions in a highly vulnerable patient group. As there is now robust registry data on the HF-G population, the presentation of data on both HF-G and HF-aCKD subgroups in our study has laid the foundations for detailed direct comparison of the 2 subgroups in the future.
The main limitation of our study is the relatively small sample number size of aCKD patients. Despite reviewing 1061 records over 2 years, only 74 aCKD patients were identified. Comparison with the HF-G group, which was drawn from an overlapping 12 months period, identified 452 HF-G patients and allowed valuable insights into comparative baseline demographic and frailty data, as well as key clinical outcomes of the 2 groups. Clinical frailty scale was assessed by a different assessor for this subgroup. However, inter-rater accuracy of CFS has been established in other studies. 29 We did not explore admission eGFR, baseline CCI, biochemical outcomes, transfusion requirement, and discharge destination for HF-G group. This limited direct comparison between the 2 groups in these domains. Our study focussed on clinically relevant outcomes in the aCKD group, but we did not explore for specific postoperative complications, duration of rehabilitation, and readmission rates. A longer duration of follow up and more detailed clinical notes review would provide further information for shared decision making as well as resource allocation and planning. In the future, a longitudinal cohort study would better evaluate and directly compare HF-aCKD patients with the HF-G population.
Conclusion
Our study has demonstrated higher 120-days mortality among aCKD patients with HF, especially for those on dialysis pre-operatively. 120-days mortality for HF-aCKD group was double that of HF-G group, and triple in dialysis patients. Clinical frailty scale can be useful to incorporate into clinical assessments to prognosticate frail older adults with aCKD who develop HF. High postoperative transfusion rate was observed in our HF-aCKD population. Each point reduction in eGFR below 12 mL/min/1.73 m2 was associated with 3% increased probability of death in hospital. Patients who resided in nursing home, walked with a frame and had high pre-morbid CFS had significantly higher probability of death at 120-days. These findings can guide clinicians when discussing about treatment expectations to patients and their families. Further studies with larger HF-aCKD patient numbers are required to explore these associations in more detail.
Footnotes
Acknowledgments
The authors thank Lisa Welthy (Clinical Nurse Specialist) for her significant contribution to data extraction and collection for this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
