Abstract
Background
The aim of this study was to investigate the feasibility of using a novel neuromuscular electrical stimulation (NMES) device for enhanced recovery after total hip replacement surgery.
Methods
Sixty patients undergoing total hip replacement for osteoarthritis of the hip were randomized and divided into 2 groups: 1 group received postoperative treatment with the NMES device, and the other group did not receive NMES. The primary outcome measures were postoperative pain, lower limb swelling, and length of stay (LOS) postsurgery. Secondary outcomes included wound drainage at 24 hours and acceptability by the intervention of the device.
Results
Data from 60 participants were analyzed (NMES (n = 30), control group (n = 30)). Patients in the NMES group demonstrated a general trend of beneficial postoperative pain, calf swelling, and average length of stay from postoperative to discharge. However, wound drainage largely remained static for both groups. The overall comfort rate of the device was 93.3%.
Conclusions
The results of this study suggest that the gekoTM NMES device is partly useful for enhanced recovery after total hip replacement surgery. In addition, the device should be considered tolerable and safe. A larger study is required with this device in the future to determine its effectiveness on compelling data.
Introduction
Total hip replacement (THR) is a frequently performed and highly successful surgical intervention for treating osteoarthritis of the hip. 1 The increasing numbers of patients undergoing THR, combined with the rapidly growing progress of surgical techniques and evidence-based treatment options, have put considerable pressure on surgeons and other health care professionals to produce excellent postoperative recovery, leading to lower mortality and morbidity as well as high patient satisfaction.2,3 The current economic climate and the restricted health care budgets further necessitate a shorter hospital stay and lower costs on the health care system.4,5 Clinical pathways and protocols introduced to achieve these goals include a variety of peri-operative interventions to fulfill patient expectations and achieve the desired outcomes. 6 However, some postoperative issues remain, and the main clinical reasons for delayed discharge following THR are pain, swelling and general weakness. 7 Immediate postoperative decreased muscle function or weakness is partly due to the onset of postoperative pain and swelling, which is widely accepted to increase the risk for complications. 8 Efforts to enhance recovery and reduce the length of stay after THR should focus on postoperative pain, swelling, and rapid recovery of muscle function.
Early physical therapy intervention yielded faster achievement of short-term functional outcomes. Neuromuscular electrical stimulation (NMES) is a promising treatment method to counteract muscle impairment in hip osteoarthritis and improve function to expedite recovery from joint replacement surgery. 9 Several studies have demonstrated that NMES, which stimulates nerves to activate the muscle, significantly improves blood flow in the veins.10-13 Nonetheless, its application in clinical orthopedic practice is still limited, partly due to concerns regarding wound drainage and patient acceptability.13,14,9
Materials and Methods
In this study, we primarily investigated the use of NMES to reduce postoperative pain management, reduce limb swelling, and average hospital length of stay (LOS) postsurgery. The secondary investigation is to assess the NMES device, to test the effects of the wound drainage at 24 hours and acceptability by the intervention of the device.
Ethical standards: This was a single-center, randomized, open-label study assessing the feasibility of using an NMES device (geko™, FirstKind Limited, Buckinghamshire, UK) for enhanced recovery after THR. This study was approved by the Institutional Review Boards of our hospital and conducted according to the principles described in the Declaration of Helsinki. All participants or their family members signed the informed consent form.
Subject Selection
Details of Inclusion and Exclusion Criteria.

THR: Total Hip Replacement, DVT: Deep Vein Thrombosis; BMI: Body Mass Index; MI: Myocardial Infarction; CCF: Congestive Cardiac Failure; AF: Atrial Fibrillation; NMES: Neuromuscular Electrical Stimulation.
Baseline Demographic and Clinical Characteristics.

BMI: Body Mass Index; NMES: Neuromuscular Electrical Stimulation.
The gekoTM NMES device was used for the study, which is small disposable, internally powered, and can be applied externally to the leg. The device is manufactured by Firstkind Ltd, High Wycombe, United Kingdom (27 mA, 1 Hz, pulse width 70-560 ms).15,16 It is self-adhesive and applied to the outer/posterior aspect of the knee. This positioning enables integral electrodes to apply a stimulus to the common peroneal nerve, which branches from the sciatic nerve. These nerves control the contraction of several muscles in the lower leg so that discernible dorsiflexion of the foot can be observed. The stimulation of these nerves by the gekoTM device causes the muscles to contract isometrically and will not affect the normal movement of the limb or mobility of the patient. Contraction of the lower leg muscles increases blood flow from the lower limbs back to the heart, thus increasing venous return—such as the common femoral vein, superficial femoral vein, popliteal vein, gastrocnemius veins, soleal veins, posterior tibial veins and peroneal veins—increasing local blood circulation, and helping prevent venous thrombosis. Furthermore, contracting the calf muscles increases lower limb blood flow to meet the metabolic demands of exercising skeletal muscles.11,17,18 These devices were worn continually from postsurgery until Day 5. Following the manufacturer’s instructions for use, the NMES devices were changed each day.
Procedure and Data Collection
This study used the NMES device in a randomized, controlled study.
Data were collected prior to surgery (Day 0), the first postoperative day (Day 1), the third postoperative day (Day 3), and the fifth postoperative day (Day 5). At each time point, adverse events and device deficiencies were monitored.
Postoperative pain was quantified through the visual analog scale (VAS) pain score (0 = no pain; 10 = worst pain) while moving on Days 1-3-5. 19 The NMES device was placed on an area that should not have been painful to the subject. Each subject responded to electrical stimulation to varying degrees, so the electrical stimulation was reduced to a lower intensity level that caused significant muscle contractions. 20
Lower limb swelling was examined in the supine position by taking measures of the circumference of both the thigh (10 cm above the patella) and the calf (10 cm below the tibial tubercle) on Days 0-1-3-5. 21 The measurement position was marked with an indelible marker to ensure that measurements were continuously recorded on the same part of the lower limb, and the same staff member completed all measurements.
Length of stay (LOS) during postsurgery counts as the number of postoperative nights in the hospital until discharge. LOS was under the strictly functional discharge criteria (ability to get dressed independently, to get in and out of bed, to sit and rise from a chair/toilet, to be independent in personal care, to be mobile with a walker/crutches, and to walk >70 m with crutches under full weight-bearing). In addition, sufficient postoperative pain treatment (VAS <3 on activity) and discharge acceptance were defined as the days until a patient was ready for discharge. 7
Wound drainage was quantified through the negative pressure drainage device. According to the postoperative protocol, mobilization started as soon as peripheral sensory and motor skills were approved, usually 4-6 h after surgery. After removing the drains postoperatively at 24 h,22,23 full weight-bearing and range of motion were allowed. Starting on postoperative Day 1, the first walking exercises with crutches began under physiotherapeutic supervision, including mobilization of the hip and muscle strengthening.
Before discharge, the patients in the NMES group were asked to complete a short verbal questionnaire. They were asked to give a verbal categorical rating of the NMES treatment as ‘very comfortable’, ‘comfortable’, ‘bearable’ or ‘unbearable’. Acceptability by the intervention of the device was recorded on a completed short verbal interview detailing their experience with the NMES treatment.24,25
Statistical Analysis
All statistical analyses were performed with Prism 8 (GraphPad Software Inc). Descriptive statistics were used to describe the cohorts and their demographics. Categorical variables were analyzed using chi-square statistical analyses, while continuous data were analyzed using independent t test. The significance level was set at P < .05.
Results
Postoperative Pain
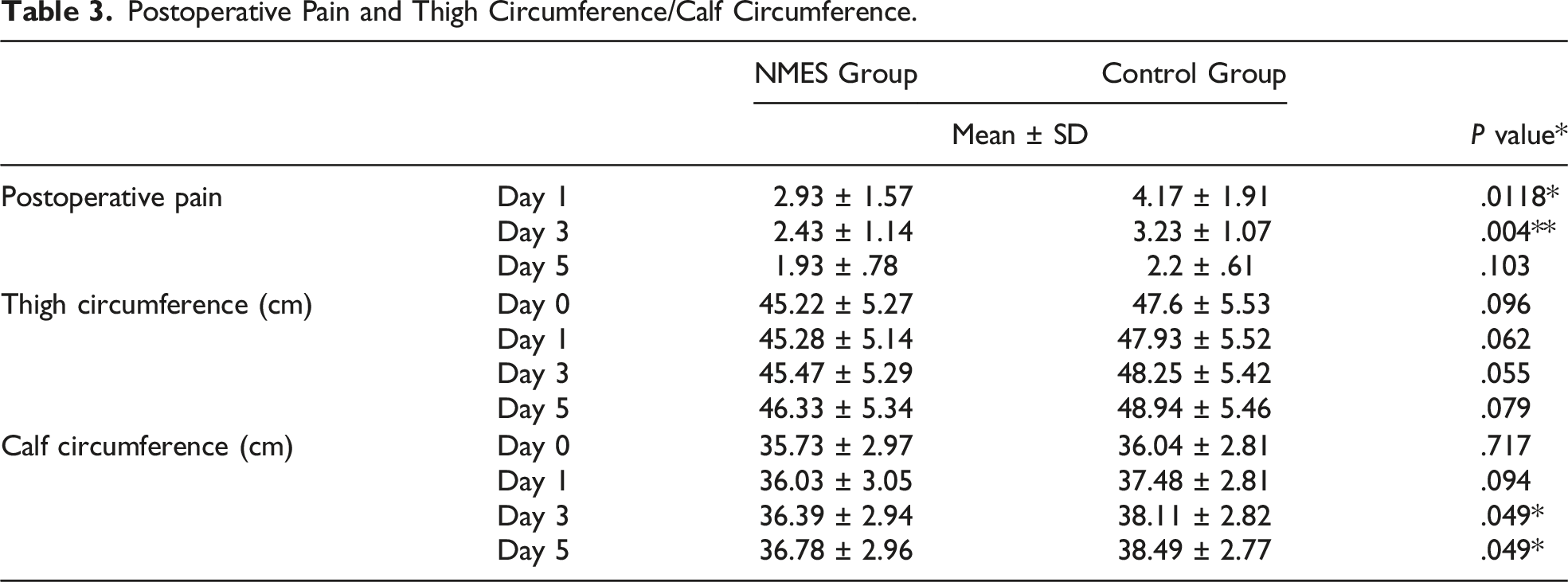
Postoperative Pain and Thigh Circumference/Calf Circumference.

Postoperative pain of 2 groups.
Lower Limb Swelling
Table 3 shows the swelling data. There was no significant difference in the mean swelling measured before THR surgery (Day 0) between the 2 groups in thigh circumference or calf circumference (P > .05). Between-group differences in the mean change in calf circumference were significant at all time points postoperatively (Days 3-5) (Figure 2). However, the change in thigh circumference in the 2 groups showed very little significance (Days 1-3-5) (Figure 3). The overall lower limb volume change showed a general increasing trend in both groups, but the NMES group showed a smaller increase. Change in calf circumference. Change in thigh circumference.

Length of Stay Postsurgery
There was a significant difference between the 2 LOS groups. The mean LOS was 6.9 ± 1.8 for the NMES group and 8.6 ± 2.0 for the control group (P = .002) (Figure 4). Length of stay at post-surgery.
Wound Drainage at 24 hours
There was no significant difference between the 2 groups. The mean wound drainage at 24 h was 339.8 ± 149.1 mL in the NMES group and 335.6 ± 161.7 mL in the control group (P = .711) (Figure 5). Wound drainage at 24 hours.
Acceptability
Acceptability.

Discussion
The aim of this study was to investigate the feasibility of using a novel neuromuscular electrical stimulation (NMES) device for enhanced recovery after total hip replacement surgery. While the sample size was small, the fact that this was a single surgeon series within the same hospital means that a high degree of pathway and treatment standardization was possible. This is an important factor when assessing outcomes such as LOS, which may have multifactorial contributing factors. A significant general risk factor for joint disease is age, as the prevalence of hip arthritis increases with age. 1 In the study by Schrader, 26 they found that 67.3% of total hip arthroplasty (THA) were implanted in patients aged 60-79 years. This is congruent with our results.
The NMES device provided a potential clinical benefit of helping to enhance recovery after surgery. THA is ranked 11th among the most painful surgical procedures. 27 The key factor for patient satisfaction is pain, measured using a visual or numerical scale. In a clinical situation, volumetric measurements are valuable for monitoring the severity of edema after surgery. 28 While the gold standard for volumetry is the water-displacement method, it is not always suitable for patients in the postoperative period. Here, we reported the circumference change (both the thigh and the calf) in measurements (conducted by 1 assessor) to ensure the reliability of the results. Pain and swelling delayed discharge following THR. Immediate postoperative decreased muscle function or weakness is partly due to the formation of postoperative pain and swelling, which is widely accepted to increase the risk for complications,8,29 The current economic climate and the restricted health care budgets further necessitate a shorter hospital stay and lower costs on the health care system.4,5 This may call for reconsideration regarding instant access to physiotherapy and further improvement of analgesia and detumescence. Therefore, early rehabilitation is important. 28 NMES can be better used for enhanced recovery in the postoperative period following THR surgery. The NMES theories of analgesia and detumescence may include 1. Stimulating the release of endogenous analgesic substances, such as enkephalins and endorphins (an opioid-like neurotransmitter) 30 and 2. Prolonging the period of capillary opening prior to occlusion, keeping the peripheral circulation open and promoting extracellular fluid exchange.31,32
In our experience, the early postoperative period is particularly challenging for patients for socioprofessional reintegration. Our results on postoperative pain showed improvement in the NMES group before 5 days. They showed no significant difference between the 2 groups regarding pain values at Day 5 postoperatively. The gekoTM NMES device provided a benefit for postoperative pain management to enhance recovery early after surgery. The reason for the 2 groups showing no significance after 5 days may be that patients improved in walking ease and diminution of joint motion pain during the intervention of gait training.
In the study by Wainwright, 33 they found that the NMES device significantly treated postoperative edema of the calf end of the thigh that occurs following THR. But in our study, our results on lower limb swelling show that the gekoTM NMES device is conducive to decreasing calf swelling but is ineffective in the thigh. However, considering the circumference changes reported and the steps to ensure reliability (same assessor and marked points of measurement), it is highly likely that the differences reported are accurate. The situation may be because the gekoTM NMES device applies a stimulus to the common peroneal nerve, which controls the contraction of several muscles in the calf without the contraction of muscles in the thigh.
We demonstrated that NMES enhances analgesia and detumescence. On the 1 hand, patients can get up, walk with walking aids indoors, get dressed, and do personal hygiene completely independently as early as possible. On the other hand, this relieves the burden on the nursing staff. Our results on LOS show that using NMES encourages mobilization and thus improves muscular strength and the achievement of discharge criteria early.
The augmentative effect of the gekoTM device on microcirculation, both in terms of flow and pulsatility. 34 Contraction of the lower leg muscles increases blood flow from the lower limbs back to the heart, thus increasing venous return and local blood circulation. 11 Previous studies have been concerned with increasing wound drainage.10,14 The present study shows no significant difference in wound drainage between the 2 groups. To our knowledge, this is the first study assessing wound drainage with NMES physiotherapy in THR patients.
In a previous study, the application of NMES had to be discontinued, as patients could not tolerate the ‘stinging’ sensation associated with the stimulus. 35 Discomfort has been identified as a limiting factor in the use of surface NMES. Lyons et al 36 concluded that smaller electrodes were found to be more comfortable. The GekoTM NMES device is light and small. Self-adherent integral electrodes are applied to the posterior-lateral aspect of the knee to apply a stimulus transdermally to the common peroneal nerve. The device, coupled with its disposable battery-operated characteristics, means that mobility and comfort are maintained throughout its usage period. According to our research, more than 93% of patients in the NMES group described the use of the device as ‘very comfortable’ or ‘comfortable’ during the application of NMES. No 1 described the use of the device as ‘unbearable.’ This may prove the acceptability of NMES to patients. The gekoTM NMES device is safe and robust enough to use in the clinical setting. No adverse events occurred during the protocol or were reported to us afterward.
A clear limitation of this study is its small sample size. Only primary cementless implants with THR under central neuraxial anesthesia were considered. Further studies should be conducted with a larger sample size and more implants under other anesthesia conditions to enable statistical analysis. At the same time, because the NMES device is easily visually distinguishable and the muscles contract significantly during electrical stimulation, it was not possible to blind and placebo this study.
Doheny and associates showed that 1 must increase electrode size on obese or overweight subjects to maintain the efficacy of electric stimulation. 37 As mentioned, all subjects in the study used the same size electrode, even with a wide range of BMIs, which would alter the efficacy of the NMES for each subject.
Furthermore, the long-term clinical effects of NMES have yet to be evaluated. The reported recovery course is restricted in the hospital. Therefore, possible external influencing factors on pain and swelling in the postoperative period, eg, family, daily routine, work and sports, could not be considered.
We also noticed that the original stimulation intensity gradually increased with the increase in the number of electrical stimulations. With NMES, the device needed to increase the stimulation intensity to achieve the same stimulation effect. The gekoTM NMES device is effective in reducing swelling and pain. Benefitting from its small size and ease of operation, the device could reduce dependency on medical professionals, as it can be used in a home environment. However, the maintenance and repair of the device also need to be considered. Randomized controlled group analysis to use the gekoTM NMES device for a long time after discharge should be performed in the future. To date, and to our knowledge, none has been published.
Conclusion
The results of this study suggest that the gekoTM NMES device is partly useful for enhanced recovery after total hip replacement surgery. In addition, the device should be considered tolerable and safe. A larger study is required with this device in the future to determine its effectiveness on compelling data.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
